Abstract
Epoxy resin (EP) is widely used in electronics due to its excellent properties and cost efficiency. In this study, to enhance both the mechanical performance and fire safety of EP for engineering applications, a novel phosphorus–nitrogen composite flame-retardant epoxy resin was developed for electronic packaging. The material was synthesized by incorporating the synthesized compounds HPM-EGDGE (A) and DOPO-EGDGE (B) into DGEBA bisphenol A-type epoxy resin. The results demonstrated that the addition of A and B significantly improved the thermal stability and flame retardancy of the composite resin. When 10 wt% of A and B (relative to DGEBA) was added, the resin achieved a UL-94 V-0 rating and a limiting oxygen index (LOI) of 33.1%. Furthermore, the tensile strain and bending strain of EP-3 and EP-4 increased by 42% and 102%, respectively, along with corresponding improvements in tensile and bending strength. Scanning electron microscopy analysis revealed notable differences in the char residue morphology of the composite resins compared to EP-0 after combustion. The presence of A and B promotes char formation and reduces the evolution of flammable gases, thereby enhancing flame retardancy. These results suggest that this material holds great promise for use in advanced engineering applications.
Keywords
Introduction
In the 21st century, the electronic industry has rapidly developed due to the high demand for mobile communication technologies and various electronic products. As a result, epoxy resin (EP), as the main material of circuit basic plates and electronic packaging, has demonstrated significant market potential.1,2 However, pure EP (EP-0) has several limitations, such as easy fatigue, low heat resistance, poor impact resistance, and a low ultimate oxygen index (LOI) of 19.8%, making it highly flammable and prone to releasing large amounts of heat and toxic flue gas during combustion, posing significant risks to human safety.3,4 Notably, miniaturized electronic products often require higher fire protection performance to prevent overheating and burning. Therefore, improving the flame retardant performance of EP is crucial. The toughness, heat resistance, flame retardant, and other properties of EP can be improved through modifications, thereby ensuring the reliability of the packaging device.5,6 Due to the susceptibility of EP to combustion, flame retardant is usually added to EP during formulation to enhance its fire resistance.7,8 The most commonly used flame retardants preparing EP include halogen derivatives or antimony-containing flame retardants. In 2003, the European Union banned the use of halogen-containing lead flame retardants due to their negative environmental impact. As a result, the production of the “green” flame retardant of EP has become a research focus.9,10
Halogen-free flame retardant can be introduced into EP through the addition method and reaction method. In the addition method, flame retardant additives are introduced into EP. This method is economical and convenient. However, flame retardant does not participate in the curing reaction. Additionally, the compatibility of flame retardant with EP is often poor. EP modified with the through addition method has poor fire resistance, which reduces the mechanical properties of EP.11,12 A reactive flame retardant is a polymer material with flame retardant synthesized through molecular design. It has a long-lasting flame retardant effect and can retain the original heat resistance and mechanical properties of the resin. Therefore, reactive flame retardants represent a key development direction.13,14 Reactive flame retardant EPs can be categorized into different groups based on the types of flame retardant elements incorporated into their structure. They include nitrogen-containing, silicon-containing, and silicon-containing curing agents; phosphorus-containing and phosphorus-containing curing agents; phosphorus-silicon synergistic, and phosphorus-nitrogen synergistic flame retardant systems.15,16 Li et al. 17 prepared PPXSPB from penttritol, trichlorooxide, and paradenziline to investigate flame retardant curing of EP. Chen et al. 18 used DOPO, APTS, and phenylglycol to prepare EP flame retardant curing agent PTDOB and mixed it with DDM for flame retardant curing of EP. Yan et al. 19 used melamine and DOPO as raw materials to synthesize the DOPO derivative with three amino groups to obtain the expected flame retardant and thermal stability. Han et al. 20 prepared a DPO-MA flame retardant curing agent using diphenylphosphoic oxygen (DPO) and maleic anhydride (MA) to modify EP. Satdive et al. 21 synthesized an epoxy curing agent (PSA) containing phosphorus and silicon from phenylphosphonyl dichloride, ethylenediamide, and GPTMS. Fang et al. 22 used DOPO, TGIC, and APTES as raw materials to prepare flame retardant curing agent TDA to modify EP. Huo et al. 23 prepared 4-flame retardant methanol, TEMP, and DOPO and mixed them with DDM to modify EP.
Many researchers have conducted numerous studies on flame retardant EP. However, the study on composite flame retardant electronic packaging EP materials synthesized using HPM-EGDGE and DOPO-EGDGE intermediates has not been reported. In this study, a new phosphorus and nitrogen composite flame retardant electronic packaging EP material was prepared by adding synthetic HPM-EGDGE (A) and DOPO-EGDGE (B) to bisphenol type A epoxy resin. The P element in the DOPO-EGDGE intermediate and the N element in HPM-EGDGE intermediate improved the flame retardant performance of EP. The open ring reaction of the C = C double bond on the maleimide in the HPM-EGDGE intermediate sealed the epoxy resin chain, thus improving the thermal stability of the EP. The O-C-C-O-flexible ether bond in the two intermediates improved the brittleness of EP and its mechanical properties.
Experimental
Materials and methods
N-(4-Hydroxyphenyl)maleimide (HPM), ethylene glycol diglycidyl ether (EGDGE), benzyl triethylammonium chloride (BTEAC), 9,10-Dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO), 4,4’-Diaminodiphenylmethane (DDM) were sourced as analytical pure reagents from manufacturers in China. Bisphenol A diglycidyl ether epoxy resin (DGEBA) of industrial grade was supplied by Hangzhou Wujingang Adhesive Limited Company, China.
Infrared spectroscopy (FT-IR) was conducted using a Nicolet 67 Fourier infrared spectrum analyzer (Thermo Nicolet, USA). The samples were prepared using the KBr compression system and determined within the spectral range of 4000–500 cm-1. The IR test was conducted at an ambient temperature of 25°C and air relative humidity of 39% using air as a background. DSC was performed using an 821e/400 differential scanning calorimeter (Swiss METTLER) under the N2 atmosphere at a flow rate of 50 mL/min, a heating rate of 10°C/min, and a temperature range of 50–350°C. Thermal weight analysis (TGA) was conducted using STA449F3 synchronous thermal analyzer (Germany) under an N2 atmosphere at a flow rate of 50 mL/min, a heating rate of 10°C/min, and a temperature range of 80–800°C. The tensile tests were conducted using an INSTRON Universal test machine (Instron Company, USA) following the GB/T2567-2021 “Resin Casting Performance test method.” A Zeiss Sigma 300 (Zeiss, Germany) scanning electron microscope (SEM) was used to analyze gold injection treatment on the surface.
Preparation of HPM-EGDGE (A)
Precisely, 1 mol of HPM, 2 mol of ethylene glycol diglycidyl ether (EGDGE), and 0.001 mol of benzyl triethyl ammonium chloride were added to the three-neck flask equipped with a thermometer and reflux condensate tube and stirred. Then, the mixture was reacted at 100°C for 4 h under nitrogen gas protection, linking the epoxy group on the EGDGE side to HPM to obtain the yellow sticky product HPM-EGDGE (A). The synthesis route is shown in Figure 1. The synthetic roadmap of HPM-EGDGE (A) and DOPO-EGDGE (B).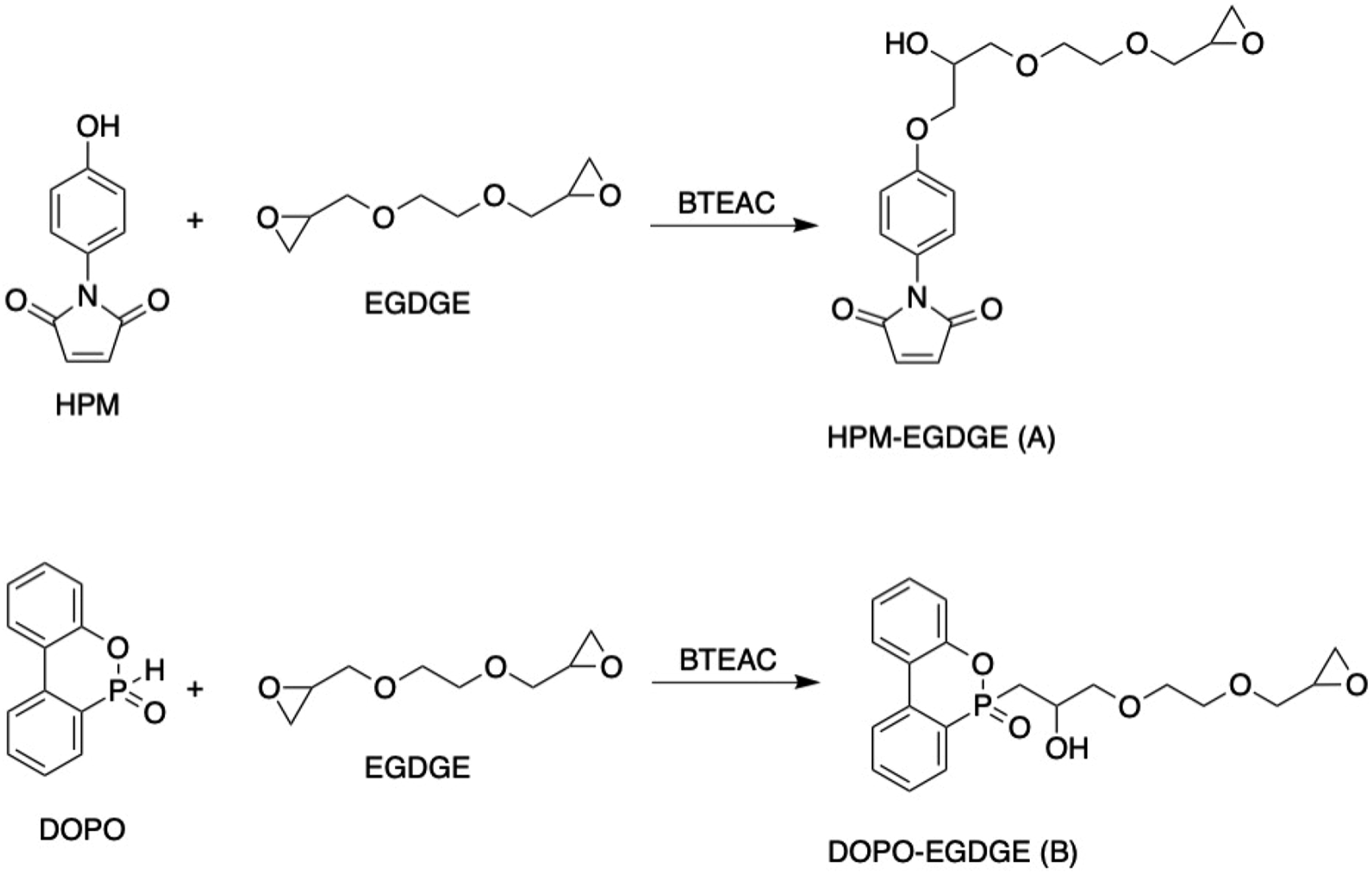
Preparation of DOPO-EGDGE (B)
First, 0.05 mol of DOPO, 0.2 mol of EGDGE, and 0.006 mol of BTEAC were mixed in the three-neck flask equipped with a thermometer and reflux condensate tube. The mixture was stirred and reacted at 110°C for 3 h. After the mixture was cooled at 60°C through rotary pressure evaporation, a light yellow sticky product DOPO-EGDGE (B) was obtained. The synthesis route is shown in Figure 1.
Preparation of HPM-EGDGE/DOPO-EGDGE/EP composite resin
HPM-EGDGE/DOPO-EGDGE/EP composite resin system ratio data table.

Results and discussion
Infrared spectroscopy of HPM-EGDGE and DOPO-EGDGE structures
HPM-EGDGE, DOPO- EGDGE, and EGDGE infrared test maps are shown in Figure 2. According to the infrared absorption spectrum curve of HPM-EGDGE in Figure 2, 3488 cm-1 was the resonance absorption peak of O-H, 1705 cm-1 was the C = O bond resonance peak, 1608 cm-1 was the C = C bond resonance peak, 1519 cm-1 corresponded to the benzene-ring skeleton, 1204 cm-1 was the peak of C-N, 1100 cm-1 was the C, and 911 cm-1 was the resonance peak of the epoxy group. From the infrared absorption spectrum curve of DOPO-EGDGE in Figure 2, 3417 cm-1 was the resonance absorption peak of O-H, 2875 cm-1 was the stretching vibration of aliphatic C-H, 1235 cm-1 was the P = O resonance absorption peak on phosphate, 1108 cm-1 was the resonant absorption peak of C-O-C, and 858 cm-1 corresponded to the resonant absorption peak of the epoxy group. In the infrared absorption spectrum curve of EGDGE (Figure 2), the peak at 1106 cm-1 corresponded to the resonant absorption of C-O-C, and the peak at 913 cm-1 corresponded to the resonant absorption of the epoxy group. HPM- EGDGE, DOPO-EGDGE, and EGDGE infrared spectrogram.
These characteristic absorption peaks were consistent with those in the literature,24,25 indicating that HPM-EGDGE and DOPO-EGDGE were synthesized.
Mechanical properties of HPM-EGDGE / DOPO-EGDGE / EP composite resin
The bending stress–strain curves of the HPM-EGDGE / DOPO-EGDGE / EP composite resin are shown in Figure 3. As shown in Figure 3, the addition of HPM-EGDGE and DOPO-EGDGE to EP initially increased the bending strain of composite resin and then decreases. The bending strain value of 9.37 was the largest for EP-4. Compared with the bending strain value of pure epoxy resin 4.65, the bending strain value of EP-4 increased by 102%. Except for EP-1, the bending strain values of EP-2, EP-3, and EP-5 exceeded EP-0, indicating that the bending toughness of composite resin successively increased. EP-4 also had the largest values of bending modulus and bending maximum stress, but the values of other added quantities were high and low, respectively, compared with EP-0. Overall, EP-4 had the best bending performance. Bending stress–strain curve of composite resin.
The tensile stress–strain curves of HPM-EGDGE / DOPO-EGDGE / EP composite resin are shown in Figure 4. As HPM-EGDGE and DOPO-EGDGE content increased, the tensile strain of composite resin was first increased and then decreased. The tensile strain value of EP-3 was 9.34, which increased by 42% compared with the tensile strain value of EP-0 (6.57). The tensile strain values of other composite resins were higher than that of EP-0, indicating that the tensile toughness of composite resin increased. In terms of tensile modulus and tensile maximum stress, the value of EP-3 was also the largest. The value of other added quantities was also increased compared with EP-0. Overall, EP-3 had the best tensile properties. Tensile stress–strain curve of composite resin.
Thermal performance analysis of HPM-EGDGE / DOPO-EGDGE / EP composite resin
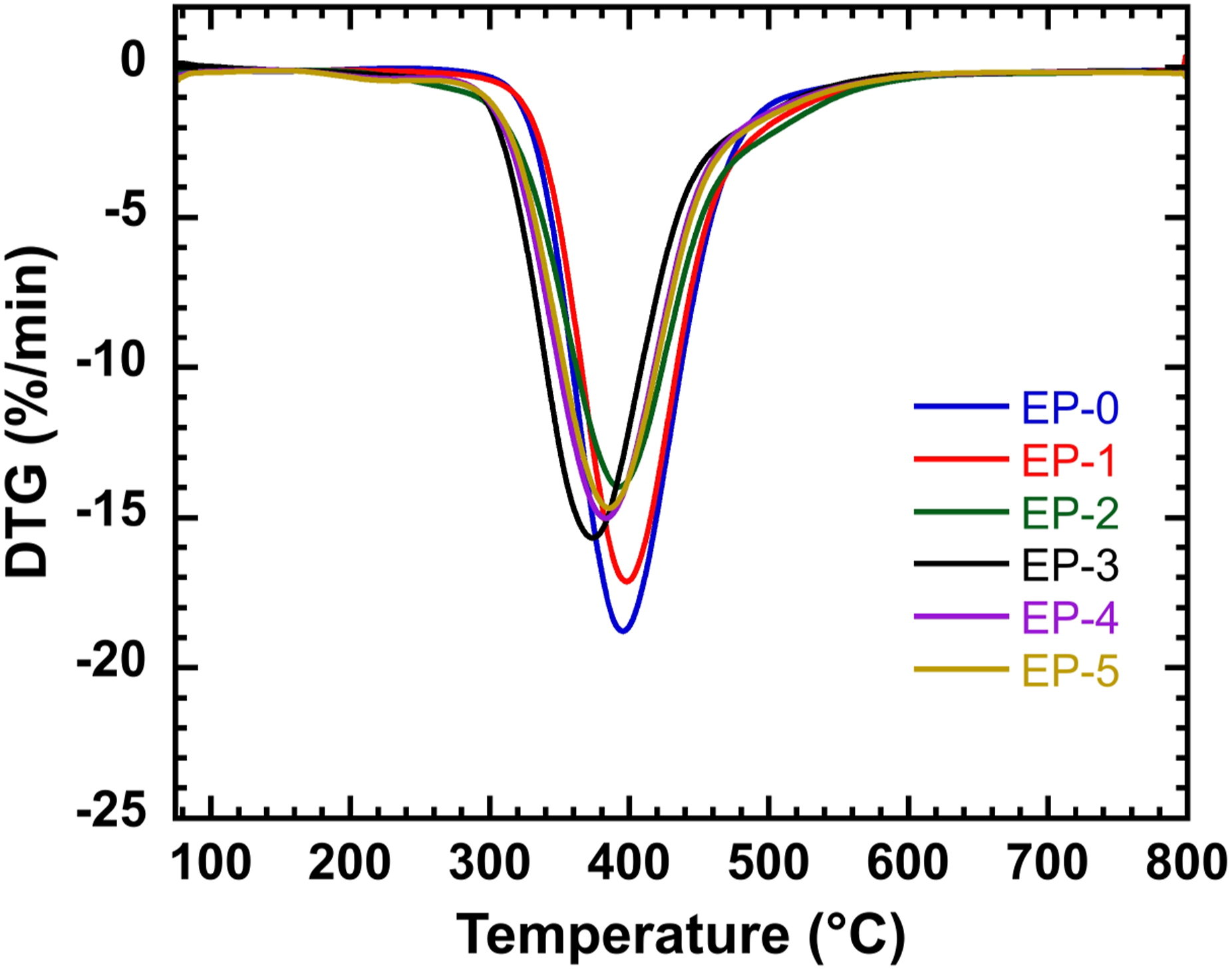
The initial degradation temperature (T5%), the temperature at 50% weight loss (T50%), the maximum thermal weight loss rate (Rpeak) and its corresponding temperature (Tpeak), and the residual carbon amount (Char Residue) at high temperatures are commonly used as key parameters to characterize the thermal stability energy of the material. According to the TG curve in Figure 5 and Table 2, the initial decomposition temperature of composite resin fluctuated with the addition of HPM-EGDGE and DOPO-EGDGE. Only T5% of EP-3 exceeded that of EP-0, while those of all other resins were lower. The T50% value of EP-0 was similar to that of composite resin. However, the T50% values of EP-2 and EP-3 were greater than that of EP-0. At 800°C, the residual carbon content of EP-3 increased by 57% compared with that of EP-0. Other composite resins also exhibited higher char residues, indicating an enhanced char-forming ability. The increased residual carbon suggests the formation of a more continuous and compact char layer, which contributes to improved flame retardant performance of the epoxy resin in the condensed phase. As shown in the DTG curve in Figure 6 and the data in Table 2, the thermal weight loss rates of EP-1 to EP-5 are lower than that of EP-0. This suggests that the incorporation of HPM-EGDGE and DOPO-EGDGE modifies the thermal degradation pathway of the epoxy resin system, favoring char formation and reducing the evolution of flammable volatiles. These effects contribute to enhanced thermal stability and flame retardancy. The TG curves of composite resin. The TGA data for the composite resin. Note: T5%, T50%, and Tmax are the temperatures corresponding to the material loss mass 5%, 50%, and maximum weight loss rate, respectively, °C; YC800 is the remaining residual carbon mass fraction at 800°C,%. The DTG curves of composite resin.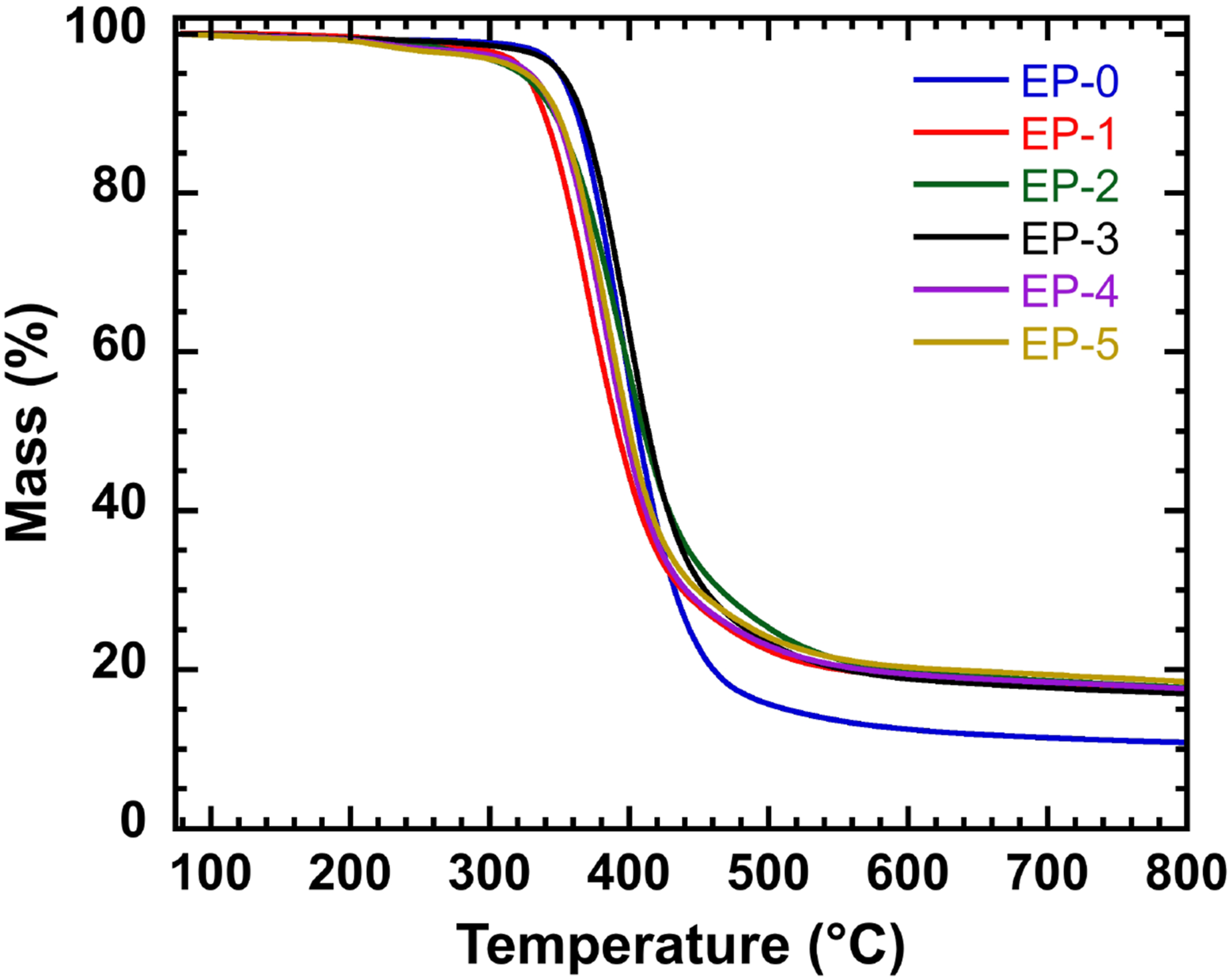

As shown in Figure 7, the Tg values of EP-3, EP-4, and EP-5 were 150, 208, and 212°C, respectively, which were all higher than the Tg value at 136°C of pure epoxy EP-0, but the Tg values of EP-1 and EP-2 were lower than the value of EP-0. The Tg of composite epoxy resin increased as the contents of HPM-EGDGE and DOPO-EGDGE increased, indicating that sufficient amounts of HPM-EGDGE and DOPO-EGDGE could improve the thermal stability of epoxy resin. The reason was that HPM-EGDGE and DOPO-EGDGE reacted with epoxy resin to provide more active sites for the opening reaction of the epoxy group, thus grafting more molecular chains on the epoxy resin molecular chain and increasing the epoxy resin crosslinking density and movement. The introduction of HPM and DOPO with certain stable structures increased the stability of the three-dimensional structure of epoxy resin macromolecular chain to a certain extent, thus reducing the flexibility between molecular chains. Therefore, as the contents of HPM-EGDGE and DOPO-EGDGE increased, the Tg of the composite epoxy resin gradually increased. The DSC curves of composite resin.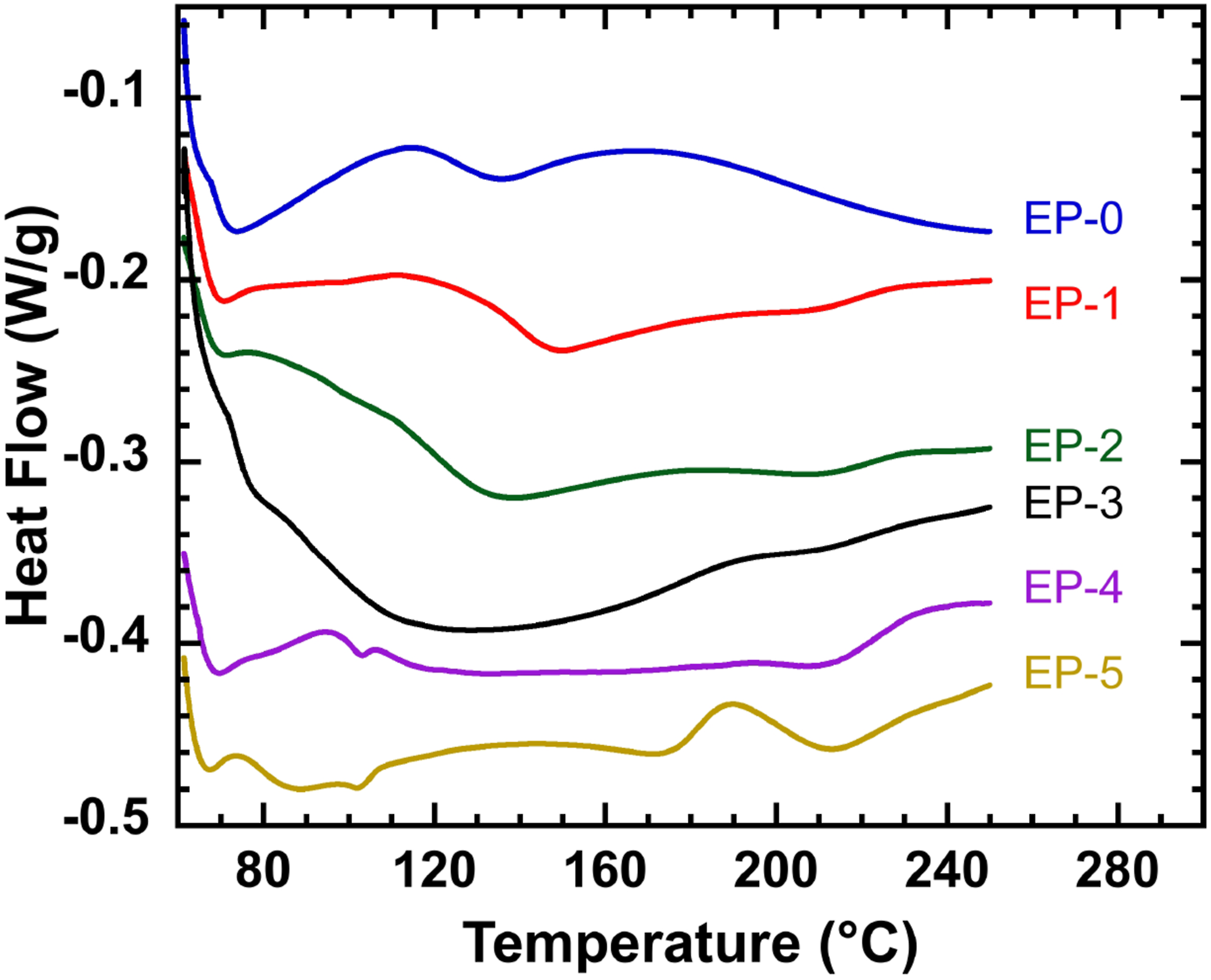
Analysis of the flame retardant performance of HPM-EGDGE / DOPO-EGDGE / EP composite resin
LOI values and UL-94 grade of composite resin.

SEM analysis of the surface morphology of residual carbon after vertical combustion of composite resin
As shown in Figure 8(a), the ash of EP-0 was relatively loose, featuring surface holes and insufficient connectivity between particles. These defects prevented the formation of a continuous dense structure, enabling further contact reaction between oxygen and the internal matrix, which facilitated heat transfer and sustained combustion. As the contents of HPM-EGDGE and DOPO-EGDGE increased, the residual carbon morphology of EP-1, EP-2, EP-3, EP-4, and EP-5 was significantly different from that of EP-0. In addition, the increased HPM-EGDGE and DOPO-EGDGE contents gradually decreased the surface holes, resulting in a denser residual carbon structure. This dense structure helped block heat and exhibited good smoke suppression. Surface SEM images of carbon residue after combustion of EP-0 (a), EP-1 (b), EP-2 (c), EP-3 (d), EP-4 (e), and EP-5 (f).
Conclusions
In this study, a novel phosphorus–nitrogen composite flame-retardant epoxy resin (EP) material for electronic packaging was developed by incorporating synthetic HPM-EGDGE (A) and DOPO-EGDGE (B) into bisphenol A-type (DGEBA) epoxy resin, using DDM as the curing agent. The effects of A and B on the flame retardancy and thermal properties of EP were systematically investigated. The results demonstrated that the addition of A and B significantly enhanced the flame retardant and thermal performance of the resin. Specifically, A and B increased the thermal stability of the material, promoted char formation, and suppressed the release of flammable gases, thereby improving the flame retardancy of the EP matrix. When 10 wt% of A and B (relative to DGEBA) was added, the composite achieved a UL-94 V-0 rating and a limiting oxygen index (LOI) of 33.1%. Furthermore, the tensile strain of EP-3 increased by 42%, and the bending strain of EP-4 increased by 102%, along with corresponding improvements in tensile and flexural strength. Thermogravimetric analysis (TGA) showed that the T5% of EP-3 exceeded that of the unmodified EP-0, and the residual carbon content at 800°C was also higher. Differential scanning calorimetry (DSC) indicated that the glass transition temperatures (Tg) of EP-3, EP-4, and EP-5 were all higher than that of EP-0, which was measured at 136°C. Scanning electron microscopy (SEM) revealed that the char residue morphology of the modified resins differed significantly from that of EP-0. As the content of HPM-EGDGE and DOPO-EGDGE increased, the number of surface pores in the char decreased, resulting in a denser carbonaceous structure. This compact structure provided effective thermal insulation and contributed to smoke suppression. Future research is recommended to explore the specific industrial applications of these engineered flame-retardant epoxy systems.
Footnotes
Acknowledgments
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was financially supported by the Sichuan Province Key Research and Development Projects (2023YFG0237).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
