Abstract
We used, in this work, a potentially effective adsorbent for removing nickel (Ni2+) from wastewater, employing modified polystyrene; the di-acylated polystyrene with iminoether (DIPS-N-Ac-Imi). The product was utilized for the adsorption of Ni(II) from waste water at ambient temperature. The di-acylated polystyrene with iminoether DIPS-N-Ac-Imi and DIPS-N-Ac-Imi/Ni2+ were characterized using infrared spectroscopy (FT-IR), thermogravimetry, and X-ray diffraction (XRD). Subsequently, the percentage of adsorption of DIPS-N-Ac-Imi/Ni2+ was determined via atomic absorption spectroscopy. An electrical study of DIPS-N-Ac-Imi/Ni2+ was performed using Impedance Spectroscopy, revealing a stimulated thermal conduction mechanism. Based on conductivity analyses, the activation energy was deduced from the Arrhenius diagram and corresponded to Ea = 1.07 eV, which suggests that DIPS-N-Ac-Imi/Ni2+ is a proton-type semiconductor.
Introduction
Ni(II) is one of the most toxic metals, it belongs to a broad category of persistent environmental pollutants like Hg(II), Pb(II), and Cr(VI). It’s is a primary water pollutant in aquatic environments due to its harmful effects on living organisms and its inability to degrade naturally.1,2 Once individuals are confronted with elevated amounts of nickel contaminating the environment, many diseases can affect humans, including pulmonary fibrosis, cardiovascular problems, and cancer. 3 Water polluted by heavy metals, such as Ni2+, poses a significant problem due to its toxicity. Numerous methods have been used to remove Ni(II), including ion exchange, membrane separation, flotation, osmos inverse, electrocoagulation from wastewater.4–9 A wide range of modified polystyrene have been utilized for heavy metal removal.10–14
In fact, incorporating transition metals to organic ligand improves their electrical properties. From where more attention has been devoted to studying the conductivity and electrical properties of modified polystyrene.14,15
The chemical modification of the polystyrene structure involves reactions on the benzene ring nucleus. Consequently, this functionalization gives rise to new derivatives, which serve to widen the fields of use of polystyrene (PS). The purpose of functionalized polystyrene is to enhance its properties and to further expand its possible uses, particularly by altering the type and quantity of functional groups present in the modified polystyrene. For example, functionalizing polystyrene increases its resistance to metal corrosion. 16 Furthermore, polystyrene nanocomposites can be obtained with desired properties. For example, they can improve thermal stability and flammability. 17 They can be used to synthesize silver nanoparticles in aminated polystyrene-functionalized cells; the resulting compound is a highly reactive nanocomposite, whose catalytic activity has been demonstrated through methylene blue reduction. 18 The PS-metformin/CuCl2 complex exhibits antibacterial activity and simultaneously acts as a recyclable nanocatalyst in indole and aniline N-arylation reactions. These special properties make it increasingly valuable in various fields of application.19,20
However, according to our research, no previous studies mention the use of N-acylated substituted by iminoesther modified polystyrene for the Ni(II) elimination from wastewater, and the electrical properties of DIPS-NH-Ac-Imi/Ni2+, have not been reported yet.
The principal goal of this work is to use an inexpensive method for removing Ni(II) ions from waste water by utilizing our modified polystyrene as an adsorbent, knowing that the proposed method has been used successfully in order to remove Ni2+ from wastewater. Subsequently, we will report the impedance and conductivity of the obtained DIPS-NH-Ac-Imi/Ni2+ derived from polystyrene complexes.
Experimental
General considerations
The following materials were used for the synthesis and analysis of this study. The polystyrene utilized in this investigation is a commercially available linear with a molecular weight of MW of DIPS = 350,000 g.mol−1. Orthoester (EtC(OEt)3), 2-aminobenzimidazole (1H-benzimidazol-2-amine) and Stannous chloride (SnCl2, 99.99%), were obtained from Fluka (FLUktuierendeKAskade, France) and used without any further purification. Nitric acid (HNO3, 86%), chloroform (CHCl3, 99%), absolute ethanol (EtOH), hydrochloric acid (HCl, 37%), methanol (MeOH) sulfuric acid (H2SO4, 95%–98%), chloroform (CHCl3, 99%) and were purchased from Sigma Aldrich (formally Millipore Sigma, France) and used as-received.
The wastewater collection was carried out in the first week of March 2023. It’s collected from waste water of stainless-steel manufacturing, and storage battery industries, its color is yellow with turbidity. Wastewater sampling was carried out by placing the 1L flask capacity in the basin at a depth of 10 cm. Before treatment with polymers, the sample was kept and filtered at 25°C.
Synthesis of amino polystyrene
The method used for the preparation of amino polystyrene was adopted from a previous study. 14
Preparation of N-acylated polystyrene
Synthesis of di-polystyrene acylated: (DIPS-NH-Ac)
To 1.0 g of aminated polystyrene dissolved in toluene, we add 1.0 mL of adipoylchloride (C6H8Cl2O2) in THF dropwise. The mixture is then heated under reflux for 24 h. The resulting product is filtered and washed with ether.
Synthesis of di-polystyrene acylated with iminoether: (DIPS-N-Ac-Imi)
N-2-benzimidazoyl Iminoether was prepared using a method from previous work.
14
0.5 g of N-acylated polystyrene was dissolved in a small amount of ethanol, then 1.0 g of iminoether was added, and the mixture was heated using a microwave for 1 h. The product obtained was filtered and dried. The colors of different compounds are presented in Figure 1. (a) DIPS-NH2; (b) DIPS-NH-Ac and (c) DIPS-N-Ac-Imi.
Extraction of Ni2+ located in wastewater
0.1 g of DIPS-N-Ac-Imi polymer was incubated with 20.0 mL of wastewater and left at room temperature for 24 h. The resulting mixture was filtered and washed with demineralized water (3 × 50.0 mL), the solid phase was washed with 30.0 mL of distillated water at T = 50°C during 20 mn for desorption of Ni2+ retained by studied polymers. The obtained aqueous phase was analyzed by atomic absorption spectroscopy for dosing the amount of removal Ni2+.
We have repeated the extraction 3 times we have obtain the same dosage.
Characterization methods
FTIR spectroscopy, X-ray diffractometer, TGA, DTA, atomic absorption were employed for comprehensive characterization
FTIR
They were enregistred using A Nicolet Infrared Spectrometer IR–200 FT-IR (Thermo Fisher Scientific, USA) was employed to determine the different functional groups present in the polystyrene derivatives. The attenuated total reflectance (ATR) mode was utilized in the range of 4000 and 400 cm−1, with a measurement accuracy of approximately 4 cm−1.
XRD
We used X-ray diffraction patterns which were carried out using a Bruker D8-Advance diffractometer furnished with a vertical goniometer type accelerator and operating with (CuKα) = 1.54,059 Å) to obtain X-ray diffraction (XRD) scans. Typically, slow measurements were performed in angular ranges from 5 to 120° with a step size of 0.01°2θ and average acquisition times of 2 h.
Atomic absorption
Atomic absorption spectroscopy uses the property of certain chemical elements to capture or emit light (UV and/or visible) when heated to a very high temperature in a flame or in an oven. The quantity of metal ions attached to the synthesized polymers was determined through atomic absorption spectroscopy analysis using an analysis Perkin–Elmer AAnalyst 200, with calibration performed using solutions of the studied metal ranging from 0.5 to 5 mg/dm3.
TGA and DTA
TGA and DTA were conducted using a thermal analyzer (Perkin Elmer STA 6000, USA) with N2 circulation and a gradual heating rate of 5°C per minute from room temperature up to 450°C. This technique allowed us to observe changes in product stability at elevated temperatures.
Impedance
The impedance analysis was performed using a Hewlett-Packard 4192A impedance analyzer, covering the frequency range from 5 Hz to 13 MHz.
Results and discussion
FTIR
The FTIR spectra of the compounds are presented in Figure 2. FT-IR spectra of (a) PS-NH2; (b) DIPS-NH-Ac; (c) DIPS-N-Ac-Imi and (d) DIPS-N-Ac-Imi/Ni2+.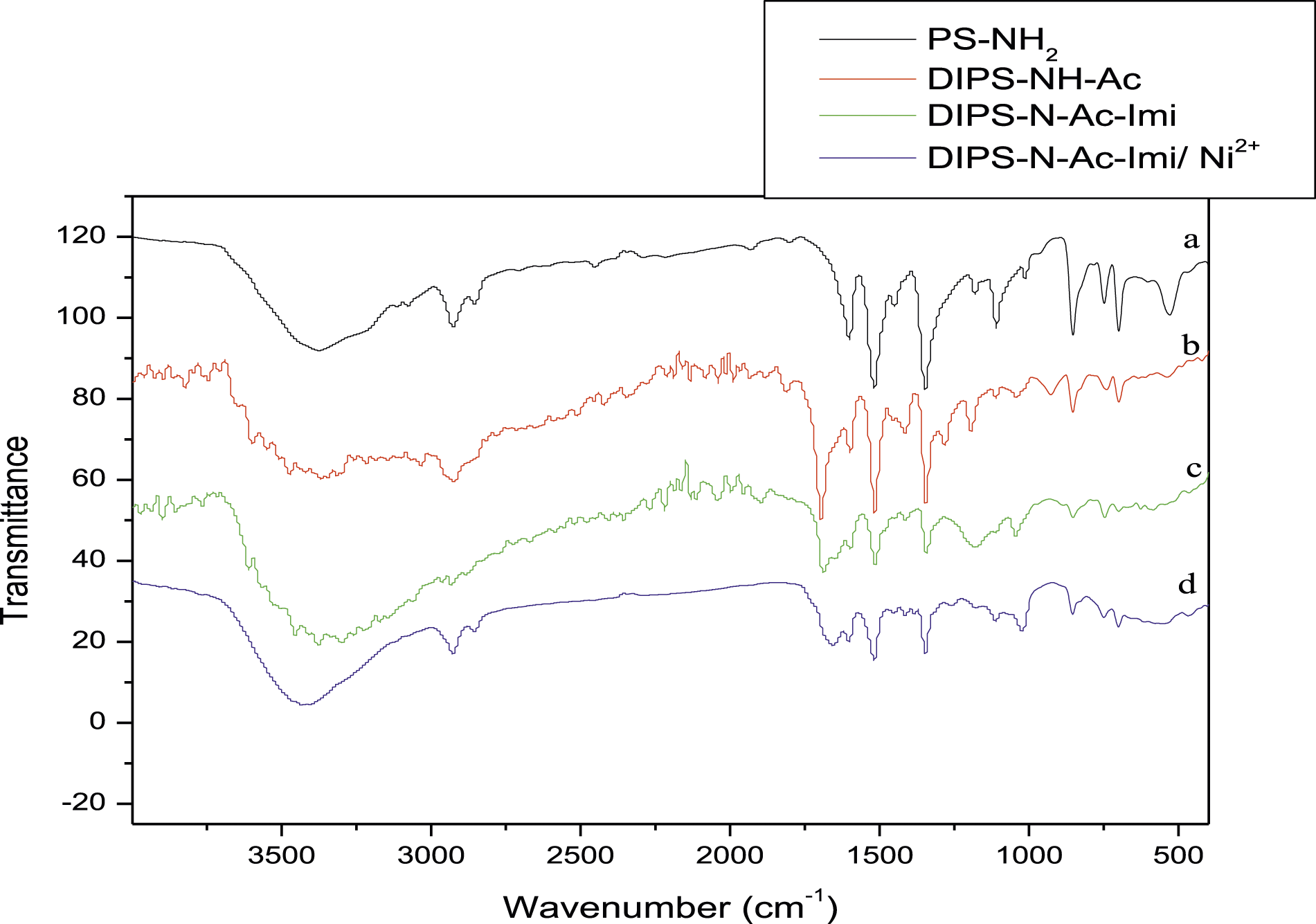
The characteristic bands corresponding to the various functional groups DIPS in DIPS-NH-Ac, DIPS-N-Ac-Imi, and the complex DIPS-N-Ac-Imi/Ni2+.In curve (a), the spectrum of DIPS-NH-Ac shows the appearance at 1644 cm−1 a band that corresponded to C=N of iminoether which proves the insertion of N-2-iminoether. Comparing the FTIR spectrum of DIPS-N-Ac-Imi (curve c) and the spectrum of DIPS-N-Ac-Imi/Ni2+ (curve d), we notice that some peaks intensities have changed due to the adsorption of Ni2+. The adsorption primarily influenced the vibrations of (C=O) and (C=N) bands, as well as the nitrogen atoms’ free doublets. We conclude then the Ni2+ displays excellent interaction into the doublets of N atoms in iminoether and the NH of the functionalized amine polystyrene derivatives.14,18
X-ray diffraction
The pattern resulting from X-ray diffraction of PS-NH2, DIPS-NH-Ac(dipolystyrene-NH-acylated), DIPS-N-Ac-Imi, DIPS-N-Ac-Imi/Ni2+ and iminoether are presented in Figure 3. The X-ray exhibits sharp and diffuse peaks. Sharp peaks corresponded to crystalline structure of iminoether, while the X-ray pattern of DIPS-NH-Ac, DIPS-N-Ac-Imi, DIPS-N-Ac-Imi/Ni2+ presents diffuse peaks, indicating that the modified polymer have an amorphous structure. X-ray diffraction patterns of (a) PS-NH2; (b) DIPS-NH-Ac; (c) DIPS-N-Ac-Imi; d) DIPS-N-Ac-Imi/Ni2+ and (e) iminoether.
The patterns of DIPS-NH-Ac (b) presented many sharp peak, at 10, 11, 15, 17, 21, 23 and 27 (2θ), and the patterns of DIPS-N-Ac-Imi (c) compared to DIPS-NH-Ac showed the disappearance of peaks at 10, 17 and 21 (2θ) and exhibits new peaks at 19, 34, 51. Upon complexing the modified PS with Ni2+, the X-Ray diffraction pattern showed the disappearance of peaks at 19, 23, 27 and 51 compared to the pattern of DIPS-N-Ac-Imi.
TGA and DTA
TGA analysis of all compounds is performed to identify changes in weight and temperature. The thermograms of TGA and DTA of DIPS-NH-Ac and DIPS-N-Ac-Imi show an exothermic undergo a two-step decomposition process. At around 350°C and 375°C the first step for DIPS-NH-Ac and DIPS-N-Ac-Imi, respectively, and the second step of decomposition process starts at around 475°C for DIPS-NH-DIAC and 520°C and DIPS-N-Ac-Imi. The decomposition temperature has increased going from DIPS-NH-Ac to DIPS-N-Ac-Imi so we can note that the polymer has become more stable and its thermal decomposition properties have been improved. Whereas when the DIPS-N-Ac-Imi is complexed by Ni2+, we note the appearance of new peaks around 240, as well as the decrease in intensity of the second decomposition peak which is explained by the effect of Ni2+ on the product. The thermograms of DTA/TGA of DIPS-N-Ac-Imi/Ni2+ present at around 200 and 300°C, an endothermic peak with mass loss, that can be attributed to the pyrolysis of DIPS-N-Ac-Imi/Ni2+. (Figure 4) TGA/DTA of (a) DIPS-NH-Ac, (b) DIPS-N-Ac-Imi and (c) DIPS-N-Ac-Imi/Ni2+.
Atomic absorption
Atomic absorption spectrometry (AAS) is a technique used to determine the concentration of certain metals in a sample.
The atomic absorption technique was used in order to evaluate the extraction rate of metals. The results were presented as percentages of metal extraction, based on its initial concentration. The assessment of metal removal percentage was performed using the subsequent equation:
The extraction of Ni2+ using and DIPS-N-Ac-Imi, was carried out in the waste waters of Ni.
We studied the influence of the concentration of the extraction agent on the extraction of Ni(II) ions to see the maximum extraction percentage. We varied the concentration of the modified polymer in the range of 1.0 to 10% (v/v) in water as a diluent. Maximum extraction (98%) was achieved with 7.0% modified polymer in water. Therefore, the percentage of metal extracted can be considered a response to the percentage of active extractant in the diluent.
Figure 5 illustrates the structure of the DIPS-N-Ac-Imi/Ni 2+. Scheme of complexation of the modified polystyrene by Ni2+.
Elementary analysis
The chemical composition of the synthesized polymers was determined at each stage of synthesis by means of comprehensive elemental analyses, enabling the constituent elements of the polymers to be identified and quantified at different stages of their formation. Each stage of the synthesis was meticulously studied to characterize the changes occurring in the chemical composition of the polymers during their formation. These analyses provided crucial data on the elements present in the samples at different stages of the synthesis process. The results are presented as N/C ratios in Figure 6. The N/C value of aminated polystyrene (PS-NH2) is around 0.09. The degree of substitution of PS by amine groups was calculated as above 90% from the following equation, by considering the generic formula: (C8H10)n-(NH2)n Nitrogen/carbon atomic ratios as given by elemental analysis for PS-NH2, DIPS-NH-Ac and DIPS-N-Ac-Imi.

In summary, the determination of the chemical composition of synthesized polymers at each stage of synthesis has enabled a thorough understanding of the molecular evolution and chemical transformations that occur throughout the manufacturing process. These results provide a solid basis for characterizing and optimizing the properties of the polymers obtained.
All these results reveal the success of the chemical modification of polystyrene and the obtention of new polymeric derivatives with potential chelating properties towards metal ions, including Nickel.
Impedance
Complex impedance spectroscopy is a potent, non-destructive analysis methodology; extensively employed across various research domains such as microelectronics, electrical engineering and material physics. The complex impedance is expressed as: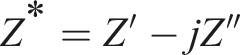
The complex impedance of DIPS-N-Ac-Imi/Ni2+ was studied using an analyzer Hewlett-Packard 4192A impedance, within the frequency span from 5 Hz to 13 MHz. Figure 7 represents the Nyquist graphs of DIPS-N-Ac-Im/Ni2+ at temperatures going from 582°K to 603°K. The resulting curves take the form of semicircles, corresponding to an equivalent circuit made up of a combination of a constant phase element, CPE, and a bulk resistance, Rb. Nyquist plot of DIPS-N-DIAC-Im/Ni2+ depending on temperature.
As the temperature rises, the diameters of the semicircles decrease, showing the presence of an activated thermal conduction mechanism. 21 The bulk resistance values at different temperatures are determined by extracting the intercept of the semicircular arc on the real axis (Z′. axis).
Figure 8 illustrates the frequency dependencies of the real parts of the impedance of DIPS-N-Ac-Imi/Ni2+, at various temperatures. For all the temperatures, Z′ remained almost constant at low frequencies and then decreased in magnitude for frequencies above 104 to nearly vanish at 105. The low Z′ values at high frequencies are due to the fact that the space charges are more energetic at high frequencies and can overcome the barrier height. Frequency dependence of the real part of Z′ the complex impedance of DIPS-N-DIAC-Im/Ni2+ at different temperatures.
As observed in Figure 9, it shows the frequency dependence of the imaginary part, Z″, of DIPS-N-Ac-Imi/Ni2+ at different temperatures. Each temperature showed a single peak (Z″max), confirming the single semicircle shape of the Nyquist plot.
22
Additionally, it was noted that as the temperature increased, the frequency of this peaks changed to higher values, which accurately represent a relaxation process
23
in the produced DIPS-NH-Ac-Imi/Ni2+. Frequency dependence of the imaginary part of Z″ the complex impedance of DIPS-N-DIAC-Im/Ni2+ at different temperatures.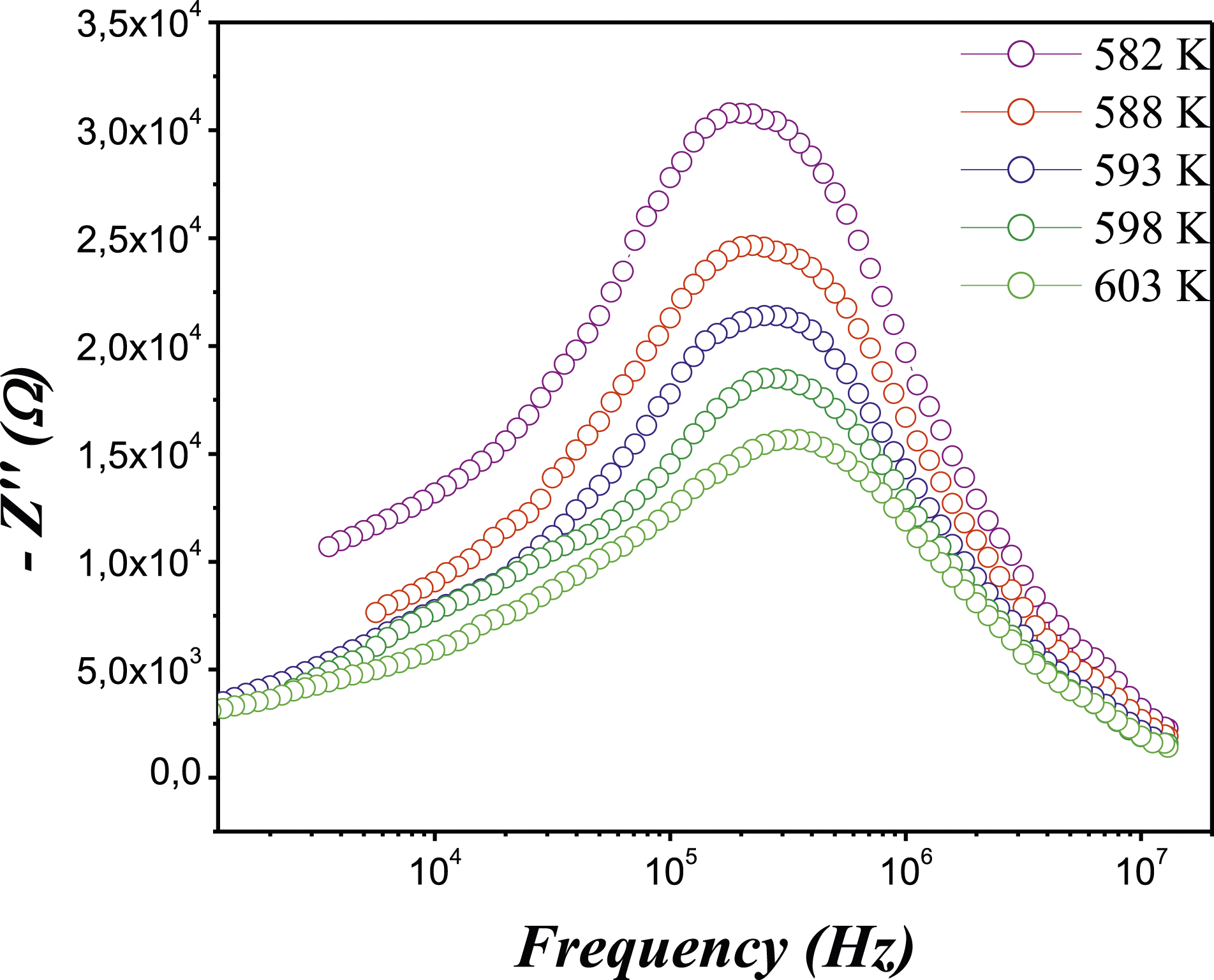
Conductivity analysis
The conductivity of DIPS-N-Ac-Imi/Ni2+ was performed by using a pellet with a geometric factor
Figure 10 displays an Arrhenius plot within the temperature range of 582 K to 603 K, demonstrating that the plot of ln(σT) relatives to 1000/T was linear, with an excellent correlation coefficient (R2 > 0.995), confirming the presence of an Arrhenius type behavior: Arrhenius plot of DIPS-N-Ac-Imi/Ni2+.

Conclusions
Aminated polystyrene was initially modified using adipoyl chloride. Subsequently, it was reacted with iminoesther to enhance its performance in removing Ni2+ions from contaminated water. The complexation of DIPS-N-Ac-Imi with nickel metal was investigated in order to eliminate Ni(II) ions out of the waste water.
The adsorption capacity and removal efficiency of the prepared adsorbent (DIPS-N-Ac-Imi) were confirmed by atomic adsorption. Infrared spectroscopy (FT-IR) was used to identify the different polymers obtained. X-ray diffractometry (XRD) analysis was conducted and shows that the structure of our polymer is amorphous. The differential thermal analysis (DTA) were utilized to investigate the thermal stability and structure of the modified polystyrene and the polystyrene with nickel metal, respectively.
Through the analysis of conductivity and impedance of DIPS-N-Ac-Imi/Ni2+, we noticed that this compound exhibits semiconductor properties as a functionalized polymer.
Footnotes
Acknowledgements
The authors would like to acknowledge the support from the Ministry of High Education of Tunisia for financial of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by University of Carthage.
