Abstract
A novel adsorbent material for the removal of Copper and chromium metal ions from aqueous solution was prepared by modifying chitosan. Cross linked chitosan material was prepared using vanillin as a crosslinking agent and further it was incorporated with graphitic-C3N4 to obtain the polymeric matrix. The composite material thus obtained was characterized using various characterization techniques such as FTIR, TGA, SEM and XRD. A peak at 1658 cm−1 indicates the formation of Schiff base and the peaks at 2θ = 9°, 15°, 21° and 24° indicates the incorporation of g-C3N4 and the increased crytallinity of the composite material. Adsorption studies were done in order to obtain the maximum capacity of the adsorbent for the Copper and Chromium ions which were found to be 166.66 and 250 mgg−1 respectively. The adsorption isotherm best fitted a Langmuir model and the adsorptive process was found to have pseudo second-order kinetics showing the chemical sorption type. Thermodynamic studies indicated that the adsorption process was spontaneous and endothermic in nature.
Introduction
The presence of heavy metal ions in the wastewater causes water pollution and thus increases water toxicity. The cause of pollution is due to the ongoing development of human activities and the increase in the development of industries, industrial activities have resulted in the release of effluents which causes pollution. Industries such as electroplating industries, Rayon industries, metal rinse process, brass manufacturing, mining, metal smelting, and electrolysis applications are one of the major producer of contaminants.1,2 These industries release a significant amount of water during their physical, chemical, and biological processing stage which results in the release of organic and inorganic pollutants into the water bodies and also can cause serious environmental issues. 3 These metal ions cause serious health issues such as hepatic and renal damage, mucosal irritation, gastrointestinal attraction, and problems in the nervous system. Heavy metals such as Copper, Chromium, Lead, Arsenic, Cadmium, chromium, etc. are non-degradable and are carcinogenic and affect human and animals by entering the body through the food chain.4,5
Heavy metal ion such as copper is essential and is required for enzyme synthesis as well as tissue and bone development only in significant doses. When it is consumed more than the limit it will be poisonous and is carcinogenic and can cause respiratory issues, liver and kidney failure, headache, vomiting, nausea, and stomach pain. Chromium is another carcinogenic materials and it exists in two different states one is Cr(VI) and another is Cr(III). In nature, chromium (VI) is an oxidizing agent that is extremely toxic and can be hazardous to plants and animals and can result in lung and gastrointestinal cancer, severe diarrhea, and hemorrhage. Due to its mutagenic and carcinogenic characteristics, the hexavalent form has been deemed more dangerous to general public health. The World Health Organization (WHO) suggested a 2.0 mg/L limit for Cu(II) in drinking water as the maximum acceptable level. So the metal ion containing wastewater must be treated before being released into the water bodies. 5
There are several methods for eliminating these effluents. Heavy metal ions have been removed from numerous industrial effluents via chemical precipitation, ion exchange, membrane separation, evaporation, electrolysis, and other conventional processes. But these techniques are frequently costly or inefficient, particularly when attempting to remove heavy metal ions from diluted solutions. 5 Adsorption is one such technique that is cost-effective and relies on the use of solid adsorbents and seems to be one of the most economically advantageous and technically simple when compared to these. So the researchers are in search of these inexpensive adsorbents which have the potential for metal-binding abilities and have become more serious in recent years.6-13
Nowadays researchers are focused on the use of a biopolymer material chitosan which has unique properties such as biocompatibility, and bio-degradability. Lei Zhang et al. 14 Focused on the removal of heavy metal ions using chitosan and modified chitosan for the removal of various heavy metal ions from the aqueous phase. Studies on the functionalization of chitosan are available. The pseudo-second-order kinetic model and the Langmuir adsorption isotherm model were followed for the adsorption process.
Renu et al. 2 . Has reported the review on the elimination of heavy metals from wastewater using chitosan based adsorbent materials. The data provided for the elimination of chromium, cadmium, and copper using those commercially available and natural adsorbents. Anush et al., 5 developed graphene based chitosan Schiff bases a new adsorbent material for the removal of Cu (II) and Cr (VI) ions from aqueous medium and studied for its metal ion adsorption properties. The converted chitosan performed well in terms of increased adsorption capacity towards Cu (II) and Cr (VI) of 111.11 and 76.92 mg g−1, respectively. Both the pseudo-second-order kinetic model and the Langmuir isotherm model suit the adsorption data well. Cu (II) in acidic solution and Cr (VI) in basic solution both had >80% desorption, indicating successful adsorbate species recovery. Jian long Wang 15 published review on the elimination of numerous contaminants from wastewater using modified chitosan adsorbents for use as an adsorbent in the removal of both organic pollutants and inorganic pollutants (such as dyes, PPCPs, PFOS, and humus) (e.g., heavy metal ions, nitrate, phosphate, borate, and fluoride).
The most effective way to remove metal contaminants in drinking water, according to Qasim Zia’s assessment of the subject, is to use chitosan for the adsorption of heavy metal ions. Several studies were reviewed, including the process of heavy metal ions adsorption on chitosan and the negative effects of heavy metal ions, as well as the adsorptive removal of metal ions from cross linked chitosan, chitosan nano fibers, chitosan nanoparticles, chitosan composites, modified/pure chitosan, and highly permeable chitosan. 1
Carbon nitrogen materials have received increased interest in recent years due to their low coefficient of friction, electrical characteristics, mechanical qualities, and super hardness. 6 According to theoretical calculations, there are five crystalline structures of carbon nitride, with g-C3N4 garnering special interest due to its stronger stability than the others. Due to its highly ordered tri-s-triazine units, g-C3N4 can stack via a hydrophobic effect and π–π interaction which helps in the adsorption process effectively and can adsorb even aromatic compounds and toxic metal ions.17–19
So considering the above advantage in the present work chitosan Schiff base bionanocomposite was prepared using vanillin as the precursor and was further incorporated with g-C3N4 to form the composite material and was characterized using FTIR, TGA, XRD and SEM techniques. As the incorporation of g-C3N4 provides enhanced adsorptive sites for the adsorbent material and was evaluated for the evaluation of adsorption isotherm and kinetics. Thermodynamics entities such as Gibbs free energy, entropy and enthalpy were studied. Meanwhile reusability of the material was also assessed.
Experimental
Materials
Chitosan (CS) was purchased from Sigma Aldrich (India). Acetic acid, copper sulphate and potassium dichromate, thiourea, ethanol and vanillin was purchased from spectrochem (India).
Methods
Synthesis of g-C3N4
Thiourea (10 g) was placed in a crucible and was kept in a muffle furnace and heated to up to 550°C for 4 h with a heating rate of 15° per minute in order to obtain the fine yellowish powder. 20
Preparation of 2D g-C3N4
5 g of g-C3N4 was finely powdered, placed in a crucible and further heated in a muffle furnace with a cover at 550°C for 4 h at a heating rate of 15°C per minute to get a yellowish fine powder of 2D g-C3N4. 20
Synthesis of chitosan Schiff base incorporated g-C3N4
0.5 g of chitosan was dissolved in 50 mL of 1% acetic acid solution with constant stirring until a clear solution was obtained. Further 0.3 g of vanillin dissolved in 20 mL of ethanol was added slowly to the solution and was stirred to obtain a uniform dispersion. A mass of 40 mg of g-C3N4 was added slowly to the resulting solution and was sonicated for 1 h and stirred overnight to obtain the final composite solution. The homogenized solution was transferred to the Petri dish and was kept in vacuum oven at 60°C. Thin films were obtained after 48 h and were used for the further studies Scheme 1. Synthesis of Biocomposite film.
Characterization
The FTIR spectra of CS, g-C3N4 and CSV were recorded on a IRSPIRIT- FTIR spectrometer (Shimadzu) the samples were analyzed using K-Br disc method in the range of 400–4,000 cm−1.
Thermal analysis of the CS, g-C3N4 and CSV were recorded using Shimadzu DTG-60 thermo gravimetric analyzer. The samples were screened in the heating range of 0–600°C at a constant rate of 10°C/min under inert atmosphere using nitrogen as the carrier gas.
X-Ray diffraction patterns of CS, g-C3N4 and CSV were recorded by means of Rigaku miniflex 600-XRD instrument using Cu-Kα radiation (λ = 1.5406 Å) with an differential angle of 2θ.
SEM micrographs were recorded for CS, g-C3N4 and CSV using JEOL_JSM5800LV, USA) with different magnifications using field emission scanning electron microscopy.
The total metal ion concentration in the solutions was measured in mg L−1 using a GB 932 plus atomic absorption spectrophotometer calibrated with standard metal ion solutions. Thermodynamic properties were screened to examine the adsorption process at various temperatures, including 30, 40, 45, and 50°C.
Metal adsorption studies
Adsorption studies
Adsorption potential of the CSV against Cu(II) and Cr (VI) was carried out by batch experiments in duplicate with the variable concentrations of the metal ions from 20 to 100 mgL−1. To run the batch experiments, known concentrations of the metal ions solutions were taken in the beaker and the 25 mg of the prepared CSV material was suspended and stirred slowly. The amount of the metal ions remaining in the solution after the treatment with CSV was estimated using the atomic absorption spectrophotometer (AAS) at fixed time intervals. The adsorption ability of the metal ions was deduced using equation (1).
Desorption studies
Desorption studies were carried out to analyze the amount of adsorbed metal ions on the CSV. The CSV material, taken after the adsorption of the ions was washed with water and dried in oven to complete dryness. The oven-dried and metal ion-adsorbed CSV was suspended in stripping solutions of HCl (25 mL of pH 1.2) and NaOH (25 mL of 0.5 M) for 3 h at room temperature with slow stirring. The desorbed Cu(II) and Cr (VI) ions from the materials was analyzed using AAS to determine the amount of metal ions present in the adsorbent.
The percentage of desorption was calculated using the following equation
Infrared spectral characterization
In the FTIR spectra of g-C3N4 (Figure 1(a)) shows a prominent characteristics peak in the range of 1,640, 1,410 and 750 cm−1 is assigned for the C = N, C-N and Triazine moiety.
20
The FTIR spectrum of the parent moiety CS (Figure 1(b)) shows a broad peak in the range 3,200–3,400 cm−1 is due to the –NH and -OH stretching. A weak band is observed at 2,900 cm−1 is due to the aliphatic stretching. Peaks observed at 1,597 and 1,323 cm−1 is due to the amide linkages of CS. Strong band at 1,080 cm−1 is due to the glycosidic linkages.
5
After the modification of chitosan with vanillin a new peak appeared at 1,658 cm−1 shown in Figure 1(c) due to the imine bond formation which has been formed due to the cross linking of chitosan with vanillin and the additional peaks of g-C3N4 are observed in the range 1,640, 1,410 and 750 cm−1 of C = N, C-N and Triazine moiety.
15
FTIR spectrum of g-a) C3N4, b) CS and c) CSV.
Thermogravimetric Analysis
CS shows three step degradation (Figure 2), in the first step an initial weight loss of 20% takes place in the temperature zone of 80–100°C takes place due to the evaporation of the adsorbed water molecule. Second step degradation takes place with a weight loss of 40% in the temperature range of 200–350°C due to the breakage of polymeric chains.
5
The final weight loss of 40% takes place in the range of 350–600°C resulting in the complete weight loss of the product. TGA of CSV shows a two step degradation process (Figure 2), in the first step a weight loss of 10% was observed in the temperature zone of 30–200°C is due to the entrapped water molecule which gets evaporated of at particular temperature. Second stage degradation took place in the range 250°C and ended at 600°C with a final weight loss of 40% which is due to the degradation of polymeric entities. At last about 40% of the residual matter remained as such which shows the presence of graphitic chain in the polymeric matrix.
15
TGA curves of CS and CSV.
XRD analysis
X Ray diffractograms of g-C3N4, CS and CSV is shown in Figures 3(a)–3(c) respectively. g-C3N4 showed in Figure 3(a) shows a prominent peak at about 2θ = 25° and 14° which reflects partial crystalline nature.
20
In Figure 3(b) the diffractogram of CS, peaks were observed at 2θ = 9 and 21° which shows the presence of partial crytallinity of the sample.
15
In Figure 3(c) it is clearly visible that after the modification of CS with vanillin and g-C3N4 peaks were observed at 2θ = 9°, 15°, 21° and 24° indicates the incorporation of g-C3N4 and slight increase in the crytallinity. X-ray diffractograms of a) g-C3N4 b) CS and b) CSV.
SEM analysis
SEM micrographs of CSV and Cu(II) and Cr(VI) adsorbed CSV are shown in Figures 4(a)–4(c) respectively. It is clear from the image that the surface of the CSV shown in Figure 4(a) appears to be rough and with irregularity. After the adsorption it is evident that the interpenetrated leaves like structure of Cu(II) and Cr(VI) ions have been accumulated on the CSV indicating interaction between the adsorbent and the Adsorbate.
15
Microscopic images of a) CSV, b) Cu(II) and c) Cr(VI) adsorbed on CSV.
Adsorption
Adsorption process of a material is due to the chemical or the physical interactions which takes place between the adsorbent and the adsorbate. In the present work chitosan acts as cationic polysaccharides which contain nitrogen and oxygen as hetero atoms which are the main governing process for the increased adsorption capacity and also the complex formation between the active functional groupand the metal ion present in the bulk. In the present study the adsorption was studied for both the metal ions, for the adsorption process of Cu(II) pH 7 was used in this case the adsorption is due to the electrostatic interaction between the metal ions and the lone pair of electron which results in the formation of complex via coordinate bond. And for the hexavalent Cr(VI) species the adsorption was studied at pH 3 as per the stability diagram of Cr(VI) H2O system. From the literature it is noticeable that pH lesser then 4, HCrO4 is the active species and is prominent and pH greater than 7.5 chromium exists as Cr2O72- and is predominant in that range. For the adsorption process of chromium on CSV it was studied at lower pH as it posses positive charge which results in the effective binding of the Chromate ions. Thus pH 7 and pH 3 was used for the adsorption of Cu(II) and Cr (VI) ions for the studies material and due to the solubility issues at lower pH adsorption of Cr(VI) ions cannot be performed for CS. 21
Effect of initial metal ion concentration
The effect of initial metal ion concentration on CSV is shown in Figure 5. It can be seen that in the initial metal ion concentration the extent of uptake was comparatively higher due to the concentration gradient developed between the Adsorbate and the binding capacity of the adsorbent material. As the concentration of the metal ions increased from 20 mg/L to 40, 60, 80 and 100 mg/L the change in qe value was observed. As the active sites on the adsorbent get saturated and further there was no change in the qe value indicating the attainment of the equilibrium situation. Further the mechanism involved in the adsorption was by the electrostatic interaction or by the formation of coordinate between the metal ions and the lone pair of electrons and increase in the adsorption capacity is due to the incorporation of the g-C3N4. Effect of metal ion concentration of Cu(II) and Cr(VI) on CSV.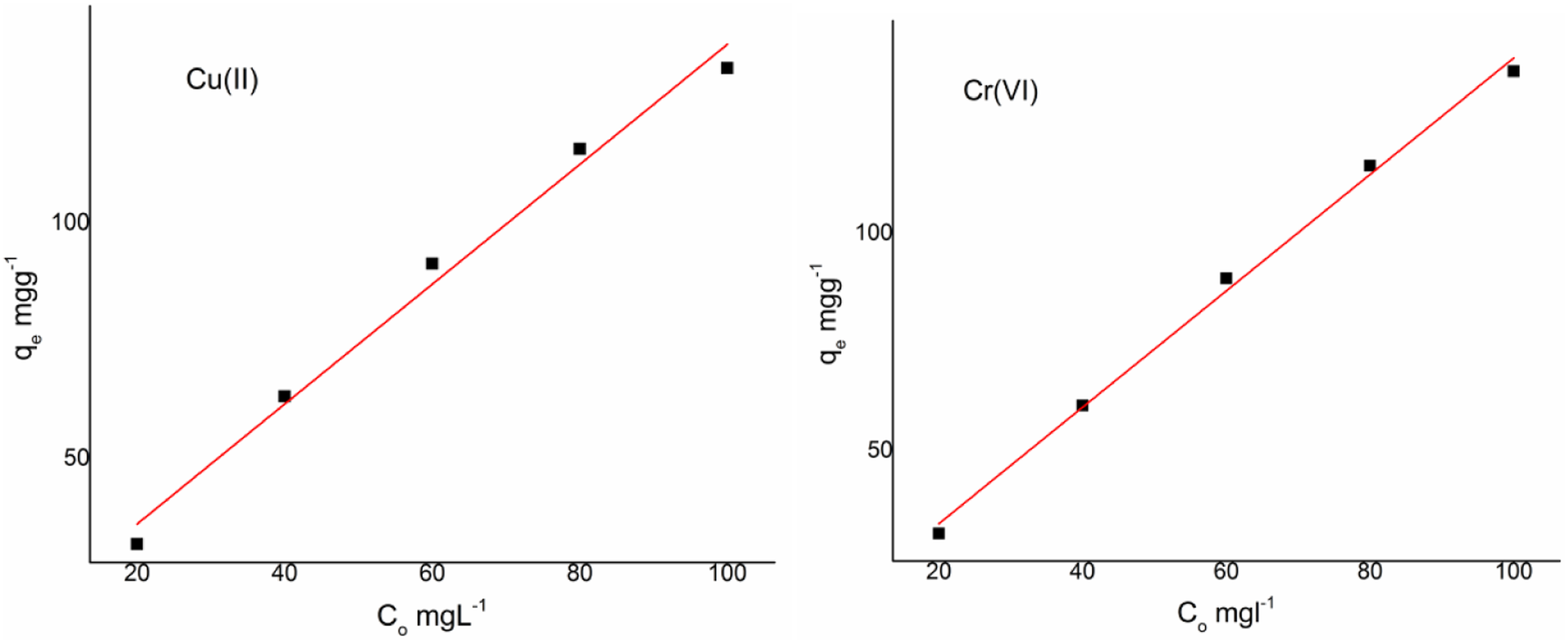
Effect of contact time
The contact time illustrates the effect of time on the adsorption process (Figure 6). Initially the uptake capacity was higher for the adsorbent due to the abundant availability of the available active sites and the metal ions. As the adsorbent site gets saturated after reaching a time zone of 105 min where there was a deviation in the path it indicates the uptake capacity goes on decreasing and at the end it will attain equilibrium indicating no more availability of the active sites. It was noted that the adsorption of metal ions was a complete fast process as the metal ions get diffused on the surface of the adsorbent faster with lesser time. The faster adsorption of the metal ions is due to the smaller size of ions which easily gets binded strongly with the adsorbent material. Effect of contact time on the adsorption of Cu(II) and Cr(VI) ions.
Adsorption Kinetics
Adsorption kinetics was used to study the time dependent adsorption process. The change in Cu(II) and Cr(VI) ion uptake as a function of timed epicts the effect of contact time and initial concentration of the metal ions. For each starting concentration of Cr(VI), the adsorption equilibrium time of (Cs-vanillin-gC3N4) composites reveals that the adsorption process is reasonably short due to sufficient attraction equation (3).
The kinetics of adsorption for Lagergren’s equation was used to calculate the removal of Cu (II) and Cr (VI) using Kinetics of pseudo-first-order.
22
The rate of change in uptake of adsorbate species over time is studied on the Lagergren pseudo first order model by considering the rate of change in saturation concentration uptake of adsorbate with respect to time. This was shown in the following mathematical equation.
Integrating the above equation (3) with respect to t = 0 and qt = 0 gives

Kinetic parameters for the adsorption of Cu (II) and Cr (VI) on CSV.

The integrated form of equation (5) when t = 0 and qt = 0 is
The resulting linear plots were analyzed to assess the data’s fitness by plotting a graph of t/qt vs time (Figure 7) and log (qe-qt) vs time (not shown). Considering the values of the regression coefficient (R2) in data reveals the rate constants and adsorption capacity values at equilibrium (qe) for both the models are shown in Table 1. As compared to the pseudo-first-order model, with higher R2 values pseudo second order kinetic model (0.999 and 0.999, respectively, for Cr(VI) and Cu(II)values was found be closer to 1 it indicates that this model is well fitted for the corresponding equation. Second order kinetics of Cu(II) and Cr(VI) on CSV.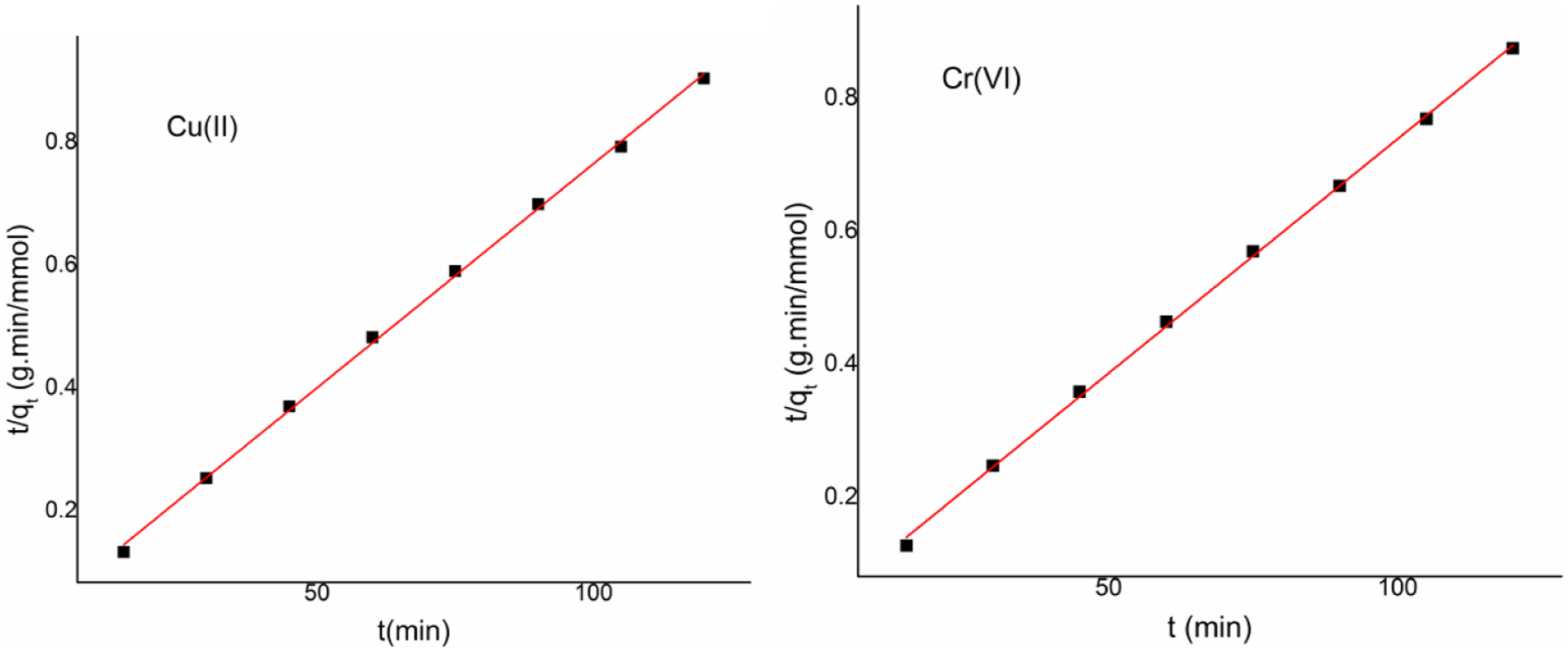
Furthermore, the anticipated adsorption capacities obtained from the fictional second-order equations were close to the actual values. The rate-limiting stage of the adsorption process was determined to be chemisorption, and the pseudo second-order model offered the best match for the chemical bonding between the functional groups of the Nano composite and the adsorbed, Cr(VI) and Cu(II) metal ions.
Adsorption isotherm
The interaction between the solid phase and the metal solution in the bulk is obtained using the adsorption isotherm (Figure 8(a)). To examine the adsorption data, Langmuir and Freundlich adsorption isotherms were used. For a monolayer, homogenous site of the adsorbent surface, without transmigration in the plane, and uniform adsorption, the Langmuir isotherm is assumed. For a heterogeneous surface, the Freundlich isotherm is accurate. A better correlation coefficient for the Langmuir model compared to the Freundlich model shows that the adsorption was well suited to the Langmuir isotherm.
24
(a) Adsorption isotherm of a) Cu(II) and b) Cr(VI) on CSV. (b) Freundlich adsorption model for a) Cu(II) and b) Cr(VI) on CSV. (c) Langmuiradsorption model for a) Cu(II) and b) Cr(VI) on CSV.
Langmuir:
Freundlich:
KF and 1/n are the Freundlich equation’s adsorption equilibrium constant and adsorption strength constant, respectively equation (8).
Isotherm parameters for adsorption of Cu(II) and Cr(VI) on CS and CSV (I) CS.

Thermodynamics of adsorption process
Thermodynamic parameters provide the information of involvement of free energy during the adsorption process. The thermodynamic parameters such as Gibbs free energy, enthalpy and entropy are used to obtain the physico-chemical properties by using van’t Hoff equation.
38


The values of ΔG◦ were determined from the equation,
The plots of ln KC versus 1/T obtained for the uptake capacity of the metal ions is shown in Figure 9. Thermodynamic parameters such as Thermodynamic plot of lnKc versus 1/T for the adsorption of Cu(II) and Cr(VI) on CSV. Thermodynamic parameters for adsorption on CSV.

Comparison studies
Adsorption capacity of various chitosan modified adsorbents for the removal of Cu (II) and Cr (VI).

Desorption
The reusability of the synthesized materials was assessed in order to know the nature of repetitive adsorption and desorption. pH 1.2 and 0.5 N NaOH was used as the desorbing medium for Cu (II) and Cr (VI) ions. Due to the protonation of acid sites on the adsorbent surface, 85% of the copper ions were desorbed at pH 1.2 due to the lower affinity of the metal ions results in the stripping of ions in to the solution and then the adsorbent was regenerated. The Figure 10 shows that 88% of the chromium ions were desorbed at 0.5 N NaOH due to the lower affinity of the metal ions results in the stripping of ions takes place in to the solution and then the adsorbent was regenerated. Desorption of Cu (II) and Cr (VI) ions.
Conclusion
In this work a novel composite material has been synthesized by using chitosan, vanillin and g-C3N4. The increased adsorption is due to the presence of g-C3N4 which was incorporated within the polymer matrix. The composite material was characterized using FTIR, TGA, XRD and SEM techniques. Langmuir model showed the best fit for the adsorption process indicating the chemical interaction between the metal ions and CSV also it presents the formation of monolayer on the surface. Kinetic data represented the adsorptive process followed pseudo second order kinetics indicating the chemical reaction between the two reactants. Thermodynamic parameters indicated the adsorption process to be spontaneous and endothermic in nature. Therefore, it is evident from the study that synthesized CSV material can be used as a potential adsorbing material for the toxic metal ion contents in the waste water during the purification process and can be further recycled and used.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
