Abstract
In this work, novel potassium methyl siliconate (PMS) was employed as a new nano-type antioxidant for acrylonitrile-butadiene rubber (NBR). NBR/PMS composites were produced by incorporating various contents of the prepared compound as anti-ageing agents (PMS) to improve the heat resistance of these composites. Potassium methyl siliconate (PMS) was prepared, and its chemical structure was confirmed by different elemental analyses. The curing properties, filler dispersion, thermo-oxidative ageing test, mechanical properties, FTIR, and TGA of the NBR/PMS composites were evaluated. It was detected by TEM images of PMS. The particles had a rhombic shape, and the particle size ranged from 1.23 nm to 7.84 nm. PMS could successfully promote the interfacial interaction between rubber and filler and the uniform dispersion of silica particles into the NBR matrix. As a result, the mechanical characteristics of the NBR/PMS composites were significantly improved, and they were superior to those of the NBR/TMQ composites with similar filler contents. Furthermore, the crosslinking density of the NBR composites was reduced after adding PMS (an antioxidant), which had a great effect on the mechanical properties. The results exhibited that PMS dramatically enhanced the solvent extraction resistance and the thermo-oxidative ageing resistance of NBR/silica composites more efficiently than TMQ. Overall, this work extended the application scope of PMS to develop novel and effective antioxidants for elastomers.
Introduction
Rubber possesses a unique viscoelastic property that has been widely applied in electrical, aerospace, and automobile applications for vibration and noise reduction. 1 Unfortunately, rubber molecule chains frequently suffer from uncontrolled oxidation attributed to the impacts of ozone, heat, mechanical stress and oxygen during storage, processing, and applications. 2 The phenomenon is commonly referred to as the “oxidative ageing” of rubber. Oxidative ageing typically causes a considerable deterioration in chemical or physical characteristics, such as loss of tensile properties or cracking. Consequently, the service life is shortened, and the use of rubber products is limited. As a result, increasing the oxidative ageing resistance of rubber products is both feasible and cost-effective. Antioxidants have the aim of preventing the rubber goods from being damaged (preventing oxidation).1,2 To trap free radicals, these antioxidants are typically added to the elastomer. Rubber without antioxidants will easily oxidize to become sticky and soft, then eventually hard and cracked (ageing).
Nitrile rubber (NBR) is widely known to possess mechanical flexibility, strong polarization, and chemical stability. NBR has been extensively used in a wide range of industrial commodities, particularly, footwear and automotive rubber products. Nevertheless, because of the large number of double bonds on the backbone of rubber, NBR is highly sensitive to ageing due to thermo-oxidative ageing and ozone attack. 3 At present, the most convenient way of delaying the thermo-oxidative of rubber is to incorporate commercial antioxidants like derivatives of aromatic amine or phenol.4,5 Regrettably, most traditional rubber antioxidants have toxicity problems, which could lead to environmental pollution and serious risks. In addition, the industrial processing techniques used to produce most antioxidants are complicated and unfavorable to the environment. 3 Apart from commercial antioxidants, macromolecular antioxidants have been explored recently. But it was difficult to increase the production of these antioxidants, and the pollution caused by their complicated synthesis processes was much greater. It is extremely desirable to create innovative antioxidants with green synthesis techniques and high anti-oxidative performance for rubbers, as environmental sustainability is becoming a growing problem. 4–6
Silicon (Si) has a strong affinity with oxygen; therefore, it usually exists as silica (SiO2) under natural conditions. 7 Silica (SiO2) is a significant non-carbon reinforcing filler for the manufacturing of lighter-coloured rubber products or “green tire”. Silica surfaces contain a large number of silanol groups, and as a result, some polar rubber additives will readily absorb onto the surface of silica, which impacts the manufacturing of rubber products. 6 Typically, silica is modified with bis[3-triethoxysilylpropyl] tetrasulfane (TESPT), which increases the dispersion of the silica into the matrix and improves the reinforcing capability of the silica by encouraging filler/polymer interaction and decreasing filler/filler interaction, improving the crosslinking network properties. 8 For instance, fatty acids such as stearic acid, oleic acid, and including cationic salt, namely cetyltrimethylammonium bromide, were used for their treatment to increase the dispersion as well as the interaction involving the silica fillers and polymer. This improved the dispersity and physical interaction of silica nanoparticles among the polymer and silica filler. When added to elastomers, silicon and its derivatives enhance the anti-oxidant characteristics as well as the interfacial adhesion, which enhances the mechanical properties.9,10
Although there are several industrial antioxidants available today on the market, TMQ (2, 2, 4-trimethyl-1, 2-dihydroquinoline) is the most common antioxidant utilized in the rubber industry. Potassium methyl siliconate (PMS) is used as a surface modifier to generate hydrophobicity and is suitable for binding, barrier coatings, surface treatments, and other applications. Siliconates provide several benefits, including UV resistance, low production costs, and adequate protection for natural building materials. PMS is used to modify the surface of a wide range of materials, and it can be combined with water glass, minerals and other fillers.
The present work aims to prepare potassium methyl siliconate (PMS) to apply as an antioxidant for nitrile rubber. PMS was incorporated through the ordinary mixing process of rubber. The rheological characteristics of the investigated compounded rubber were estimated. The physico-mechanical properties have been systematically evaluated. Additionally, the effects of various PMS contents on the ageing characteristics of the composites at various times were examined. The surface morphology of the composites as well as their dielectric properties had been discussed.
Experimental
Raw materials
- Acrylonitrile-butadiene rubber (NBR) with 34% acrylonitrile content was purchased from Bayer AG (Leverkusen, Germany). Other chemical compounds and additives, including stearic acid, sulfur, CBS (N-cyclohexyl-2-benzothiazole sulfenamide) and zinc oxide (ZnO), from supplied by by Sigma-Aldrich Chemie GmbH (Munich, Germany). Antioxidant 2,4-trimethyl-1,2-dihydroquinoline (TMQ) was provided from commercial suppliers. Silica (Hi-Sil 233D) white powder was supplied from PPG Industries Inc (Delfzijl, Netherlands). All Commercial-grade rubber additives were all utilized exactly as received. - Methyl trichloro silane 98% (GC) with molecular weight 149.48 was obtained from Sigma Aldrich-Germany. Methanol (purity: >99.5%) supplied by Piochem laboratories chemicals -Egypt. KOH pellets were supplied by MERK. All of the above reagents were used as received.
Methods
Preparation of potassium methyl siliconate
1. 97.6 parts methanol 24.4 parts demineralized water were added to 400 parts methyl tri chloro silane in a-1-liter flask over the course of 30 min with vigorous stirring. 2. Heating the mixture up to 50°C to remove acid. 3. Cooling the mixture at 0°C and separating out the alcohol- acid phase from the siloxane phase. 4. The siloxane phase was heated to reflux for 15 min at 73°C after adding 40 parts of methanol, the mixture stir for 5 min. 5. Adding the siloxane phase to water-KOH (0.6 M) to form potassium methyl siliconate.
Preparation of NBR composites
Composition of NBR vulcanizates.
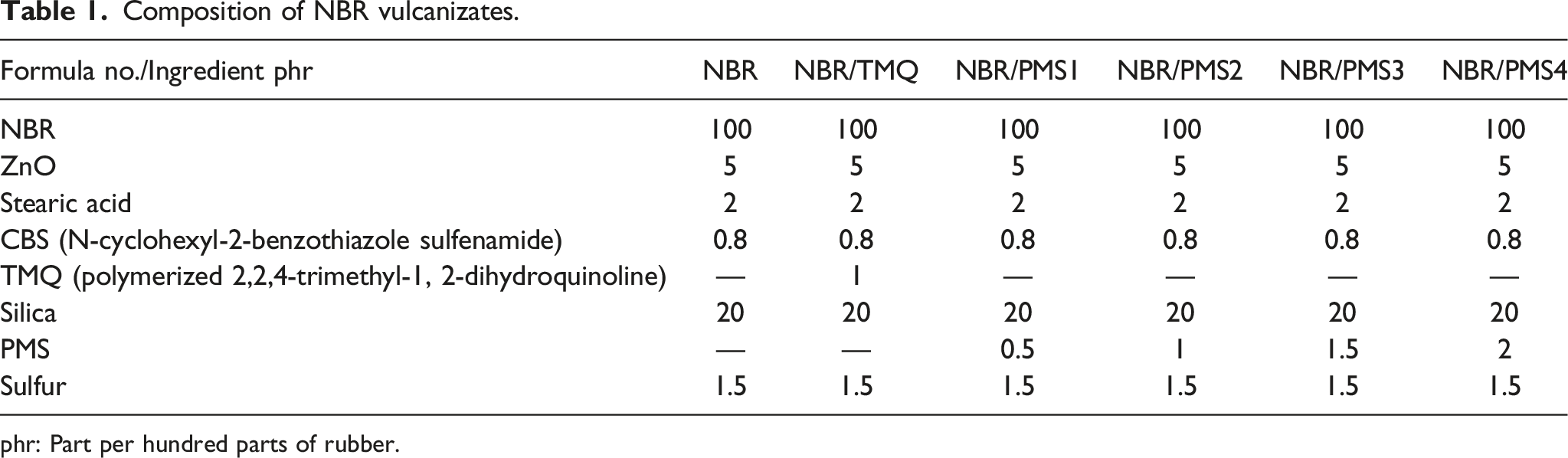
phr: Part per hundred parts of rubber.

Synthesis route of Potassium methyl siliconate (PMS).
Measurements and characterization
Fourier transform infrared spectroscopy (FT-IR) of the synthesized antioxidant (PMS) was performed using a FTIR spectrophotometer (FTIR-8400 Shimadzu, Japan) and KBr pellets with wave numbers between 4000 and 400 cm−1.
Scanning electron microscope (SEM) was performed with a Quanta instrument, model FEG250. Gold was used to cover the samples, and the electron microscope was worked at 104 V of excitation potential.
Transmission electron microscope (TEM) was carried out using electron microscope (JEOL, Japan) model a JEM-3010 with 300 kV acceleration voltages.
Thermo-gravimetric analysis (TGA) were performed with Perkin Elmer analyzer equipment, (USA) (Switzerland) under a nitrogen atmosphere with a temperature range of 50 to 1000°C and a heating rate of 10 °C/min10°.
Differential scanning calorimetry (DSC) tests were performed using Perkin-Elmar DSC-7 under static air atmosphere. The samples were heated at a rate of 20°C/min from 25 to 500°C and weighed between 6 and 8 mg. For each sample, the average value of three measurements was taken into account.
X-ray diffraction was conducted using Pan Alytical Emprean diffractometer with Cu-Kα radiation (λ = 1.5406 Å). Within the scattering range of 2 θ, the diffraction curves were obtained.
Cure characteristic was determined using an oscillating disc Monsanto rheometer model 100 USA, as per ASTM D-2084.
Zwick Roell Z010 electronic tensile testing equipment (Ulm, Germany) performed the mechanical test in accordance with ASTM D412-06a (2013) ISO 37: 2011. The average values of tensile strength (TS), stress at 100%, 200% strains, and elongation at break (EB) were calculated for each composite using three parallel samples.
Determination of crosslink density
The following equation was utilized to measure the equilibrium swelling (Q%) in accordance with the standard procedure (ASTM D3616-95) (2019):
Crosslink density (ν), mol/cm,
3
was determined by equilibrium swelling method. After 24 hours, samples were removed from toluene and quickly weighed on an analytical balance after the toluene on surface was wiped off with tissue paper. In order to completely remove the toluene, they were then dried in a vacuum oven before reweighed. The value of crosslink density was determined using Flory-Rehner equation.11,12

Thermal oxidative aging of the NBR vulcanizates containing different content of antioxidant (PMS) was performed in a laboratory oven C for 7 days. Before tests of their mechanical properties, the aged specimens were kept at room temperature for 24 h to achieve thermal equilibrium. The following equations were used to estimate ageing coefficient (k), elongation at break retention, and tensile strength retention: 


Dielectric properties
Dielectric relaxation spectroscopy
This technique was used for obtaining the dielectric properties where the sample was subjected to an a.c electric field. The permittivity ε’, loss factor tan δ and a.c conductivity σa.c measurements were carried out by a computer-controlled broadband impedance analyzer (SolartronTM 1260- Schlumberger Ltd, UK). The range of frequency was from 0.1 Hz to 1 MHz and the temperature range was from −20°C to +50°C. For diminishing noise at low frequencies, good electromagnetic shielding was implemented for the whole sample. For automating the measurements, the impedance analyzer was interfaced through a GPIB cable IEE488 with a personal computer. The software LabVIEW was used for the acquisition of data. The error in permittivity and loss factor was 1 and 3%, respectively. The temperature of the samples was controlled by a temperature regulator with Pt 100 sensor. The temperature error was about 0.5°C. The samples were stored in desiccators in the presence of silica gel to void the moisture. Then, the sample was transferred to the measuring cell and left with P2O5 until the measurements were carried out.
Results and discussion
Characterizations of PMS
Figure 1 shows the FTIR spectra of PMS. The intensive adsorption peaks at 3456 and 3464 cm−1 were due to the bending vibration of O-H in free water.
13
The broad peak between 2800 and 3750 cm−1 was due to silanol OH groups and adsorbed water. Comparatively, new peaks at 2922 cm−1 and 2851 cm−1 were associated with the asymmetric stretching vibration of C–H. Peaks at 1644 cm−1 were responded to the stretching vibration of O-H in bound water of products. The absorbance peaks at 1271, 1104, 1019, and 768 cm−1 correspond to the molecular vibrations of siloxane components on the PMS.14–16 The peak at 1271 cm−1 of the PMS resulted from stretching vibrations of the Si-CH3 bonds, whereas the peak at 768 cm−1 corresponds to the vibrations of a collection of bonds, including Si-O, Si-C, and/or Si-O-C.15,16 The peak at 1019 cm−1 is typically attributed to stretching of Si-O-Si bonds developed among the silanols.
17
The vicinity of 977 cm−1 peaks represented the asymmetric stretching vibration of Si-O-Si, which originated from gel structure. Peaks of 455 cm−1 were assigned to Si-O tetrahedral of PMS. FTIR spectrum of Potassium methyl siliconate (PMS).
Transmission electron microscopy showed the structural formation of potassium methyl siliconate. As can be seen in Figure 2, PMS was a rhombic-shaped particle without any deformation, and the diameters of most particles were between 20 and 170 nm with a narrow distribution Several larger PMS particles are usually assembled into blocks floated on the surfaces. Besides the larger particles and blocks of particles were smaller particles (less than 20 nm in size). During PMS synthesis, some PMS molecules permeated into amorphous regions, where they grew into nanoparticles. It would be easier for PMS molecules to give (more void spaces). Therefore, the PMS polymer surface coverage would be higher on rubber. This partially explains why PMS-treated rubber composites absorbed less water and swelled less. It was clear from the image that all particles have a regular geometric surface. TEM images of PMS.
Thermogravimetric analysis (TGA (has proved to be a suitable method to investigate the thermal stability of PMS. The knowledge of degradation and mode of decomposition under the influence of heat was highly recommended for the optimization of process parameters. The decomposition temperature indicates the highest processing temperature that can be used.
18
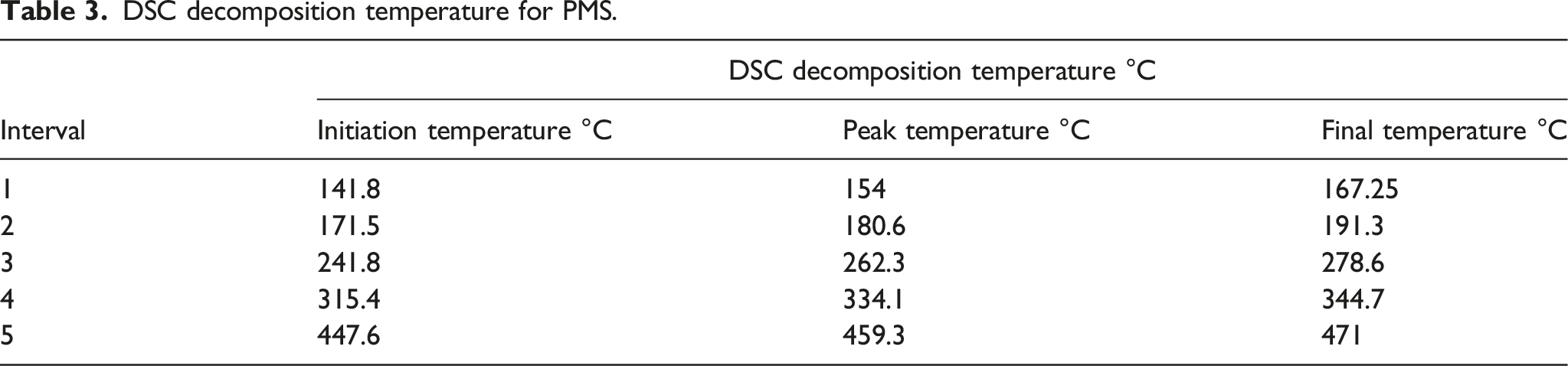
It was evident that the thermal degradation process for PMS proceeded in five steps. The initial and final degradation temperatures were ∼978°C. It can be observed that the residue obtained at the degradation amount is 75%. Usually, the analysis of TGA data is not straightforward. The rough results cannot be directly converted into absolute features of the studied material. They may depend on different factors, such as sample mass, heating rate or gas environment. The results reported in this work were specific to the experimental conditions used. The TGA curves of the polymer samples were shown in Figure 3(a) and the TGA data were given in Table 2. It was seen from this figure that the weight of the sample decreases continuously as the temperature increases. TGA data relating to the temperatures corresponding to 15% weight loss (T15), 50% weight loss (T50), 90% weight loss (T90), and maximum weight loss (Tmax) were the main criteria used to indicate the thermal stability of the synthesized polymers. The relative thermal stability of the synthesized polymers had been evaluated by comparing the decomposition temperatures at different weight loss percentages, as shown in Table 2. The higher values of T15, T50, T90 and Tmax, gave the higher thermal stability of the investigated systems. The PMS underwent single-step decomposition over the temperature range from 350.7°C to 978.85°C. It was also observed that the synthesized PMS was stable up to 1000°C. The data obtained from the DSC curves were in good agreement with that obtained from the TGA thermo grams. TGA detects the thermal stability and the thermal degradation stages of the tested sample. (a) TGA decomposition temperatures of PMS. (b) DSC curve for PMS (Weight loss vs. temperature). TGA decomposition temperatures of PSM.

DSC decomposition temperature for PMS.
X-ray diffraction (XRD) was used to investigate unknown crystalline materials (e.g., minerals, inorganic compounds). Figure 4 shows the XRD spectra of PMS. The main products C-k-S-H/C-S-H between a broad peak of 25–35° attributed to the characteristic gel structure of PMS, originating from the alkali dissolution and the subsequent condensation of k-O, Si-O results indicated that the amorphous gel structure of PMS was prepared well. The microstructure morphology of PMS is shown in Figure 4. As seen, the sample exhibited a successive gel and compacted microstructure. X-ray diffraction of PMS.
SEM technology was utilized to identify the inter-phase morphology of each polymer/filler composite. Figure 5 shows the fracture surface morphology of composites A, B, C, D, and E. Aggregation of silica with cracks and minor flow lines was observed in composite A. However, for composite B, silica particles were irregularly dispersed and formed enormous agglomerations in the NBR/PMS1 matrix due to the strong hydrogen interactions that formed between the hydroxyl groups on the surfaces of PMS and the silica particles. It was observed that rhombic-shaped PMS particles were dispersed in the NBR matrix, as exhibited in Figure 5(c)–(e). PMS affected the surface morphology of the NBR and appeared in the SEM image for composite C. For the NBR/PMS3 and NBR/PMS4 composites, it was shown that the size of agglomerates was distinctly reduced and the silica particles were distributed uniformly in the NBR matrix, as obtained in Figure 5(d) and (e), respectively. The superior dispersion of silica particles through the NBR chain in the NBR/PMS3 and NBR/PMS4 composites could be owing to the chemical reactions that occurred between PMS and silica (SiO2), the strong polarity of the SiO2 particles was significantly decreased, improving the compatibility of the silica particles with the NBR matrix.
19
SEM photographs of NBR composites with antioxidant (A) NBR/TMQ, (B) NBR/PMS1, (C) NBR/PMS2, (D) NBR/PMS3 and (E) NBR/PMS4.
Cure characteristics
Curing characteristics of NBR compounds.
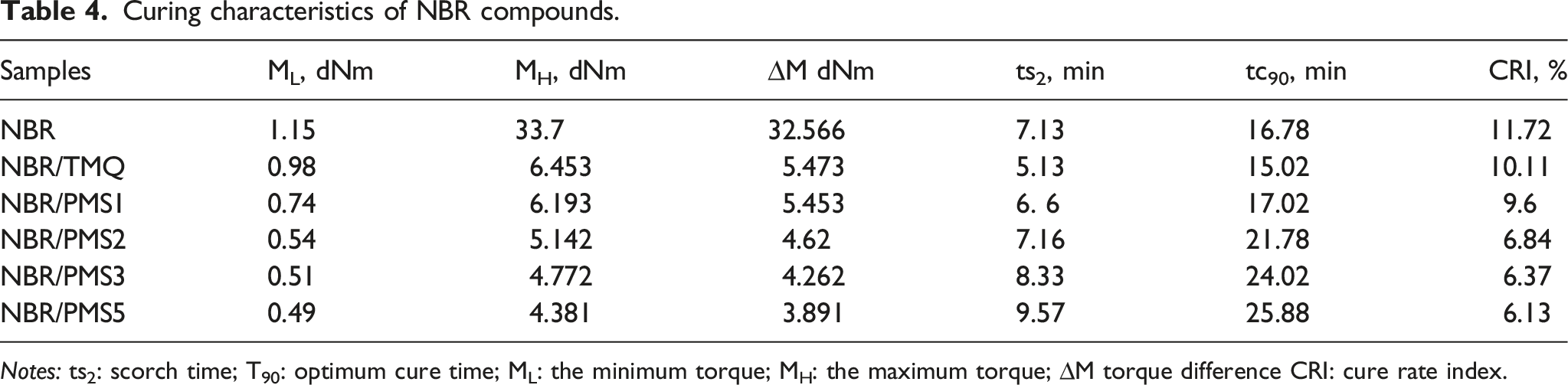
Notes: ts2: scorch time; T90: optimum cure time; ML: the minimum torque; MH: the maximum torque; ∆M torque difference CRI: cure rate index.
Mechanical properties of NBR/PMS composites
Changes in mechanical properties before aging
The composites were vulcanized for the optimum cure time, and it was essential to test the mechanical properties after the addition of PMS to the NBR matrix. As displayed in Figure 6(a), the tensile strength values of the NBR composites reached maximums with further increases in PMS contents. Observably, the tensile strength of the NBR/PMS composites was improved by 67.8% (from 5.88 to 9.5 MPa). Such substantial improvements could be more clearly demonstrated by the arrangement of rubber chains throughout the rubber/filler interfacial region after stretching. The enhanced cure properties of the antioxidants mentioned above may be related in some way to their high tensile strength. Interestingly, with increases in the content of PMS, the elongation at break values of composites showed opposite behavior in contrast to tensile strength.
21
Tensile properties of the NBR and NBR/PMS composites with different amounts of PMS.
For modulus at 100% and 300% elongation, composite NBR/PMS had a higher value than NBR/TMQ, while composite NBR/PMS4 had the highest one as shown in Figure 6(b). It suggests that the addition of PMS significantly improves the silica-rubber interaction of the composites and that PMS is also more effective than the widely available antioxidant TMQ. The fact that the PMS may covalently connect to the NBR matrix throughout vulcanization results in a stronger bond between the NBR matrix and PMS, which increases the stress transfer efficiency of the composite.
The swelling characteristic of NBR/silica composites antioxidant (TMQ and prepared PMS).

As demonstrated in Figure 7, the crosslinking density of NBR/PMS composites with various content of antioxidants was significantly decreased compared with NBR/TMQ. It was noteworthy that the presence of PMS resulted in the lowest crosslinking density for the NBR. Due to the different radicals that PMS antioxidants may consume throughout the vulcanization process, leading to a discriminating effect on the crosslinking density. This result was also in accordance with the curing properties mentioned above. The decrease in the crosslinking density increased the elongation at break. However, when the high amount of PMS was added, the nanoparticles began to agglomerate into larger-sized granules, which decreased its influence on the crosslinking density of the NBR/PMS vulcanizates.
23
Crosslinking density of NBR composites with antioxidant (TMQ and different contents PMS).
Thermo-oxidative aging resistance of NBR composites
The mechanical properties of NBR composites might be significantly reduced by thermo-oxidative ageing. The most important performance criteria for rubber materials in industrial applications were their mechanical properties; therefore, it’s crucial to observe changes in these characteristics as the ageing time of composites increases. The tensile strength, elongation at break retention and ageing coefficient were used to evaluate the ageing properties of the samples.
According to the tensile properties in Figure 8, the unfilled NBR composites (without antioxidants) showed a sharp decrease in tensile strength or elongation at break after ageing 3 days, whereas the NBR composites that contained antioxidant (TMQ and PMS) exhibited a relatively slow change. (a) -The tensile strength and (b) the elongation at break of NR composites after different aging times.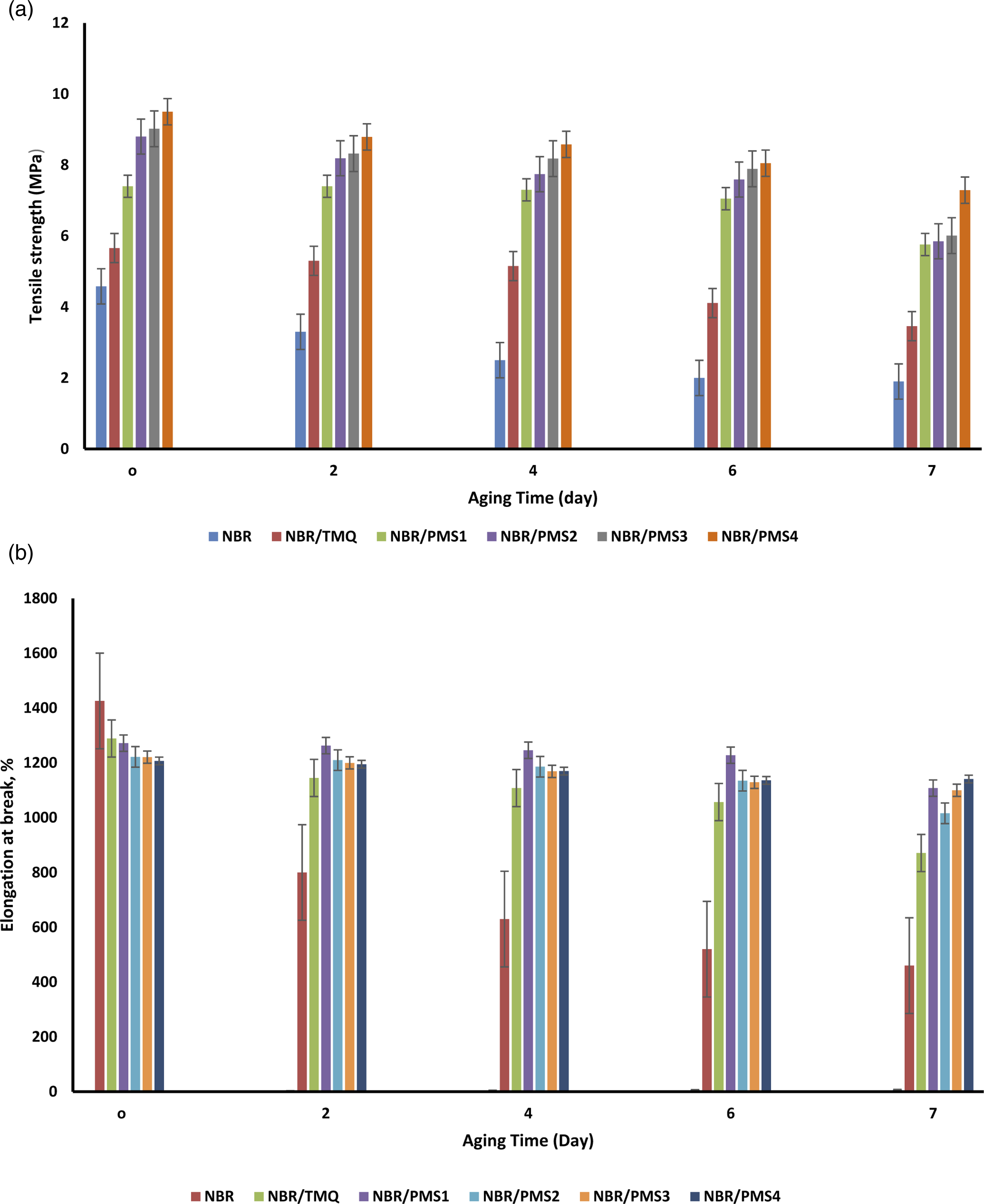
Figure 8 showed the tensile properties of NBR/PMS composites after ageing 7 days that displayed a relatively slow change. Figure 8(a) demonstrates the tensile strength of NBR composites after varying ageing times. It was clear that NBR with TMQ caused an observed decrement in tensile strength. Although the tensile strength of NBR vulcanizates with various PMS contents gradually increased, the best values were found in NBR/PMS3 and NBR/PMS4 vulcanizates. Furthermore, when composites aged, their tensile strength gradually declined. For NBR vulcanizates, including PMS, the rate of tensile strength reduction during auto-oxidation was high. The elongation at break of the resultant vulcanizates was shown in Figure 8(b) and maintains the same trend as the tensile strength. The NBR/TMQ composite showed a sharp decline in elongation at break with increasing ageing time; for example, the NBR sample with TMQ lost most of its elongation at break after 6 days. Furthermore, PMS has a much greater anti-oxidative ability than the widely used antioxidant TMQ. Notably, compared with other NBR composites, NBR/PMS3 and NBR/PMS4 composites showed superior tensile performance over ageing time. 24
The changes in retention values of tensile strength and the elongation at break for NBR composites aged for different times were illustrated in Figure 9(a) and (b), respectively. The NBR/PMS tensile strength displayed good retention whenever the ageing time was not more than 4 days. Unfortunately, their tensile strength retentions drastically dropped after 4 days.
3
The retentions of tensile strength and elongation at break for the NBR/TMQ, NBR/PMS1 and NBR/PMS2 were not much different. Comparing these results with that of NBR/PMS3 and NBR/PMS4 gave better retention values of tensile strength and elongation at break, the appropriate concentrations of PMS were 1.5 and 2 phr. Therefore, it is recommended to apply PMS as a good antioxidant for NBR, so its results after ageing were better than the conventional TMQ. The performances of the NBR composites before ageing, and ageing (a) The tensile strength retention, (b) The elongation at break strength retention, (c) The aging coefficient and (d, e) the representative stress-strain curves of NBR composites.
The anti-ageing capabilities of the elastomers are properly determined using the ageing coefficient (k) Figure 9(c). The values of the K coefficient, which compares the mechanical properties before and after the ageing process. Therefore, according to the data obtained in Figure 6, the positive effect of antioxidant PMS applied to NBR valcanizetes which able to prevent the composite’s performance drop during the ageing processes. Compared to the widely used commercial antioxidant in the rubber industry, PMS can protect NBR composites more efficiently. 3 The stress (σ)-strain (ɛ) curves in Figure 9(d) and (e) demonstrate that after 7 days of age, the tensile strength of the NBR/PMS4 composite may still be higher than that of other composites. As more PMS is incorporated into the NBR vulcanizates, the tensile strength gradually increases while the strain at break reduces, as seen in the figure. Generally, investigations on accelerated thermal-oxidative ageing indicated that PMS might work well as antioxidants for NBR elastomers. This is because the PMS was able to properly remove oxygen radicals.23,24 Thus, it was concluded that antioxidant PMS had a superior effect on NBR/PMS in terms of thermo-oxidative ageing protection, but TMQ antioxidant was ineffective for this rubber composite.
Dielectric properties
Dielectric constant and dielectric loss (ε′, ε″) of NBR/TMQ, NBR/PMS1, NBR/PMS3 and NBR/PMS4 were measured over a frequency range of 0.1 Hz up to 10 MHz and a temperature range from −20°C to +50°C. All investigated samples have the same behavior. Figure 10 illustrates the obtained data of ε′ and ε″ for NBR/TMQ and NBR/PMS3 as a function of frequency at different temperatures, From Figure 10(a), ε′ decreased as the applied frequency increased, showing an anomalous dispersion. At this range of frequency, the permittivity had a contribution from orientation polarization.
25
Also, at low frequencies, ε′ increased with the increase in temperature. This significant increment in ε′ was a result of the increment of a number of dipoles as temperature increases.
26
Variation of the permittivity ε′ and dielectric loss ε″ versus. Applied frequency (f) for NBR/TMQ and NBR/PMS3 at different temperatures (from-20 to +30°C).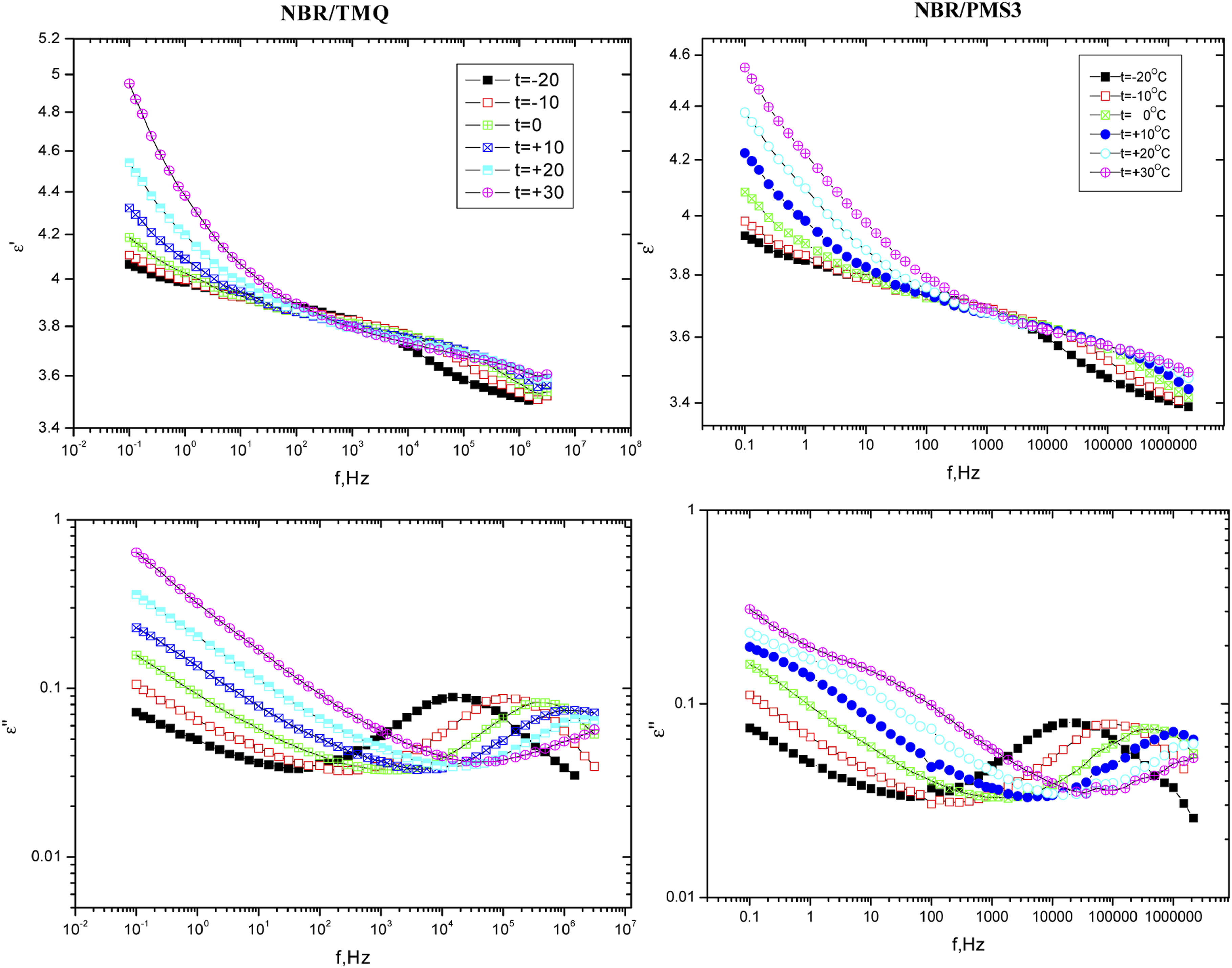
From Figure 11(b) It was obvious that the curves relating ε″ broader than the Debye curve indicating more than one relaxation process.
27
The data fitting is done by a computer program based on Havriliak-Negami and cole-cole and functions.
28
At low temperatures (until T = +10°C) the best fitting of the data was done by a superposition of two functions, Havriliak-Negami and cole-cole. The first absorption region that lies approximately around f = 0.1 Hz (Havriliak-Negami) could be attributed to the α-process,29,30 process related to the rotational motion of the segmental chain. The second absorption region that lies approximately around f = 104 Hz (cole-cole)
29
could be attributed to the γ-process, a process related to the orientational motion of cyan-side-groups.31–33 At higher temperatures (higher than +10°C), the normal mode relaxation has appeared (α’) (Havriliak-Negami), at about f = 100 Hz which is a signal of the fluctuational motion of the end-to-end dipole vector for the polymer chain and only observed in a Stockmayer type A polymer with a dipole moment vector parallel to the backbone chain.
34
As examples for analysis Figure 11(a) and (b) illustrate the analysis of dielectric spectrum of NBR/PMS3 at t = −10 and + 50°C respectively. Analyses of NBR/EPDM at different temperatures by decomposition of Havriliak-Negami, cole-cole and conductivity functions. (a). −10°C (b). +50°C.
Broadness, height and relaxation time of the γ -process of different composites at temperature T = −10°C.

The dielectric constant and loss of all composites increased by rising temperature, as illustrated in Figure 12. The activity of molecules improved with temperature, and the interference impact of the contact range decreased. As a result, the activity of polar groups increased, which further enhanced their capacity for transport in an electric field.
31
So the dielectric constant of composites increased with temperature. Also from this figure, the temperature increase had a relativity low effect until t = +10°C, after t = +10°C the dielectric constant had a sharp increase with temperature, this sharp increment is simultaneous with the appearance of α′ process (process related to motion end-to-end dipole vector for the polymer chain). So this sharp increment may be a result of increment in the number of polarized dipoles, which are end-to-end dipole vectors for the polymer chain. From Figure 12(b), the dielectric loss increased with temperature which was attributed to the increment of the relaxation polarization loss.
35
Also this figure showed that NBR/PMS3 composites had the highest value of (ε′) overall range of temperature which means that NBR/PMS3 composites have the best interaction between polymer chain and anti-oxidant(PMS).
36
Also The increment in ε″ values with PMS is lower than The increment in ε′ value So NBR/PMS3 composites have the most promising dielectric properties, which is agreed with the ageing performance and mechanical properties of the NBR composite. Variation of the permittivity ε′ and dielectric loss ε″ versus temperature for NBR/PMS1, NBR/PMS3 and NBR/PMS4 at certain frequency f = 100 Hz.
Conductivity of composites
Figure 13 shows that the conductivity of NBR/PMS increased with temperature which is the usual behavior for conductivity with temperature due to increment of conductive charges hence increment of conductive network and conductivity. Also from this figure, NBR/PMS3 composite has the highest values of conductivity overall range of temperatures which agrees with (ε′, ε″) values, ageing performance and mechanical properties. The conductivity values ranged from 10–15 to 10–14 S/cm at room temperature (30°C), which supports the use of such composites in insulation applications where the insulators range is [10-16-10-10 S/cm]. d.c conductivity σ as a function of temperature for NBR/PMS1, NBR/PMS3 and NBR/PMS4.
Conclusions
In conclusion, our work indicated the influences of nano antioxidant (PMS) on mechanical properties, crosslinking density, and ageing resistance of NBR/silica composites. PMS was successfully prepared, characterized in the Nano scale, and utilized in NBR compound. Antioxidant was added to NBR vulcanizates to delay vulcanization process and reduce crosslinking density. As a result, the mechanical characteristics of the NBR composites containing antioxidants were impacted by the decrease in cross-linking density. With antioxidant PMS, the tensile strength of NBR composites increased while the elongation at break decreased. Notably, the NBR/silica composites loaded with PMS had better filler dispersion and tougher and stronger rubber-filler interaction than those loaded with commercial antioxidant TMQ. PMS (antioxidant) significantly improved the anti-ageing protection of NBR composites, which may reveal new insights into the production of high-efficiency rubber composites. Also, NBR/PMS3 composite had the highest values of conductivity overall range of temperature which supports the use of such composites in insulation applications where the insulators range was [10-16-10-10 S/cm].
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
