Abstract
This study modified hydrogels prepared from silk fibroinand graphene oxideby injecting plasma into the silk. The SF-GO hydrogels, modified by plasma jet with different discharge time (30, 90, and 150 s) and working gases (argon, air, helium), undergo electrochemical AC impedance spectrum tests. After hydrogel modification with the plasma of any working gas, the impedance in the very low frequency (10−2∼100 Hz) decreased with a longer plasma discharge time. In the very low frequency, the sequence of impedance change is Ar group <air group ≈ He group <empty. The electric capacity of the hydrogel showed an increasing trend with those modified by argon plasma and a decreasing trend for those hydrogels modified by air and helium plasma. The sequence of electric capacity change is Ar group >empty >air group >He group. Fourier transform infrared spectrum, and X-ray diffraction spectrum showed a reduction of β-fold structure and graphene oxide content in SF-GO hydrogels modified by plasma.
Introduction
Bioelectronic devices are receiving wide interest from researchers, leading to various bioelectronic device inventions, such as bioelectronic skin,1,2 bioelectronic gloves, 3 biological tissue brakes, 4 filter dampers, 5 and ion diode. 6 These bioelectronic devices require an energy source and storage. Common biological power sources include flexible biological batteries 7 and triboelectric nanogenerators. 8 In terms of energy storage, supercapacitor 9 can store sufficient electricity and supply it under certain conditions. Conventional supercapacitor 10 is unsuitable for energy storage in bioelectronic devices due to its rigid, bio-incompatible nature. The biological supercapacitor has been widely used in bioelectronic devices due to its comfort, softness, and biocompatibility. The biological supercapacitor comprises two active electrodes and a hydrogel electrolyte. Active electrode materials include metal compounds, such as titanium dioxide, 11 manganese vanadium compounds, 12 carbon-based materials, such as carbon nanotubes,13,14 graphene, 15 transition metal carbides, 16 carbon nanofibers, 17 conductive polymers, such as polyaniline, 18 polypyrrole. 19 The hydrogel electrolyte, located between two active electrodes, serves as an ion transport channel. Hydrogel biomaterials are divided into two categories: artificial synthetic materials, mainly polyethylene glycol, 20 polyvinyl alcohol, 21 polyacrylamide,22,23 natural materials, such as chitosan,24,25 gelatin, 26 sodium alginate, 27 silk fibroin.28,29 Silk fibroin hydrogels 30 have bright prospects in bio-hydrogels for human applications due to their excellent biocompatibility, biodegradability, mechanical properties, and low immunogenicity. The electrical conductivity can be achieved by injecting some conductive substances into the silk fibroin hydrogel.
Conventional methods to improve the electrochemical performance of hydrogels include changing the number of electroactive substances 31 and the internal network structure of hydrogels. 32 However, these methods can damage the biocompatibility and mechanical properties of the bio-hydrogels. Currently, the application of plasma jets as an indirect means of the electrochemical performance of hydrogels is rarely reported. Atmospheric pressure low-temperature plasma jet33,34 is environmentally friendly, easy to operate, does not require a vacuum device, and can generate a large amount of high-energy charged particles and active substances, 35 such as HO˙, O˙, HO2˙, H2O2, O3 and so on. Besides preserving the material, the plasma can greatly affect the biological material, and it has huge application potential in material modification.36,37 This study aims to investigate the effect of plasma on the electrochemical properties of hydrogels. A SF-GO hydrogel is modified by injecting plasma into the silk fibroin solution, using sodium dodecyl sulfate (SDS) as the surfactant 38 and graphene oxide as the conductive material. The effects of different plasma discharge time and working gas types on hydrogels’ structure and electrochemical properties are investigated. The outcome of this study can provide a benchmark for applying plasma to the future optimization and design of biological supercapacitors.
Experimental section
Materials and equipment
Silkworm cocoons were obtained from the official store of Xintourou, deionized water was acquired from the official store of Honghuangzhili, and anhydrous sodium carbonate (analytical grade, molecular weight is 105.99) was bought from Sinopharm Chemical Reagent Co., Ltd. Anhydrous ethanol (analytical grade, molecular weight is 46.07) was obtained from Sinopharm Chemical Reagent Co., Ltd. Anhydrous calcium chloride (purity ≥96.0%, molecular weight is 110.98) was obtained from Xilong Science Co., Ltd. 8000–14,000 Da dialysis bags were bought from Jintai Xingda Biological Enterprise, graphene oxide (the thickness is about 1 nm, and the diameter of the single-layer sheet is 0.2–10 μm) was acquired from Hongdachang Evolution Technology Co., Ltd., and sodium dodecyl sulfate (chemically pure grade, molecular weight is 288.38) was bought from Sinopharm Chemical Reagent Co., Ltd. Liquid nitrogen.
Balance (1000, Wante), Magnetic Stirrer (ControlECO, Four E’s Scientific), Digital Thermometer (YH-101, Yuanhao), oven (FCD-3000 Serials, Shanghai kuntian Co., Ltd), Centrifuge (LC-LX-H165 A, Lichen), Ultrasonic Cleaner (G-100S, Shenzhen Geneng Cleaning Equipment Co., Ltd), Flowmeter (LZB-6WB, Shuanghuan), Air compressor (JB12, Jieba), Plasma Generator (CTP-2000K, Nanjing Suman Plasma Technology Co., Ltd), Electrochemical Workstation (CHI660 E, Shanghai Chenhua Instrument Co. Ltd), 4°C Refrigerator (BC-43, Ronshen), Freeze Dryer (YTLG-10A, Yetuo), Fourier Transform Infrared Spectrometer (Nicolet 8700, Thermoelectric Nicolet Instruments Inc, US), X-ray Diffractometer (X'Pert MPD, PHILIPS, Netherlands).
Plasma jet device design
The atmospheric pressure low-temperature plasma jet was generated in a copper tube (inner diameter = 3 mm). A layer of the glass tube (inner diameter = 4 mm, outer diameter = 8 mm) was covered on the copper tube. The outlet of the glass tube was a tiny glass tube (inner diameter = 2 mm, outer diameter = 4 mm). The exposed top of the copper tube was connected to the gas source, which was sealed with silicone. The gas sources included argon cylinder, air compressor, and helium cylinder. A flowmeter controlled the flow rate of gas, which was kept constant at 2 L/min. The bare copper tube above the glass tube was connected to the high-voltage terminal of the CTP-2000K plasma generator with copper clips. The gas flowing through the copper tube was ionized by adjusting the voltage regulator and the output frequency adjustment knob. The entire copper tube was used as the positive electrode.
Plasma ionization parameter settings.


Plasma jet device.
Experimental method
The regenerated silk fibroin solution was prepared based on the traditional method of degumming-dissolution-dialysis. 39 The solution of ternary mixed system of CaCl2:C2H5OH:H2O (molar ratio is 1:2:8) was used to dissolve silk. In this study, the concentration of the silk fibroin solution used was 4.1658 %. The silk fibroin samples were divided into ten groups. A plasma jet with variable working gases (argon, air, helium) and discharge time (30, 90, and 150 s) was injected into the silk fibroin (SF) solution, these modified SF solution was labeled as Ar30s, Ar90s, Ar150s, air30s, air90s, air150s, He30s, He90s, He150s. The unmodified silk fibroin solution was labeled as the empty group. A 50 mg/mL sodium dodecyl sulfate (SDS) was prepared. Weighed 173.8 mg graphene oxide powder, dissolved it in 86.9 mL deionized water in a beaker, and used the 600 W ultrasonic cleaner to shake the sealed beaker for 45 min to obtain a stable and homogeneous graphene oxide solution with a concentration of 2 mg/mL. Ultrasonication can make graphene oxide powder disperse uniformly in deionized water. 3 mL SF solution from each silk fibroin sample was mixed with 300 μL SDS solution and 3.75 mL GO solution, stirred for 5 min to obtain a homogeneous SF-GO solution. The SDS was used as a gel accelerator and GO as a conductive dopant. The mixed solution was put into an oven at 37°C quietly. With the help of SDS, the cross-linking between silk fibroin chains accelerated, the SF-GO gel would formed naturally in less than 35 min. Afterward, the SF-GO gel samples undergo electrochemical testing and structural characterization.
Characterization
(1) The electrochemical behavior of the hydrogel capacitor was measured by the electrochemical workstation. The electrodes used for the electrochemical test were platinum with a side length of 5 mm and were 17 mm apart. (i) The impedance of the hydrogel capacitor was measured by AC impedance spectroscopy
40
(EIS). The initial voltage was set to 0 V, the frequency range was 0.01 Hz∼1 MHz, and the scanning speed was 0.1 V/s. The impedance Z of the equivalent circuit in the AC impedance test of the hydrogel can be calculated as shown in equation (1).

where RΩ is the internal resistance of gel in Ω, C
d
is the double layer electric capacity in F, R
ct
is the charge transfer resistance in Ω, and Z
w
is the Warburg impedance in Ω.
Impedance Z is a complex, which can be expressed as real part Z′ and imaginary part Z″: Modulus of impedance Z: The phase angle φ of the impedance is: (ii) The electric capacity C of the hydrogel capacitor was calculated from equation (5)
41
according to the cyclic voltammetry (CV) of the electrochemical workstation:



where, (2) The hydrogels were rinsed with liquid nitrogen and immediately put into a freeze dryer to vacuum for 4 days. The structure of the dried sample powder was examined using Fourier transform infrared spectroscopy (FTIR) and X-ray diffractometer (XRD).
Results and discussion
The effects of plasma on the electrochemical properties and structure of hydrogels, including impedance and electric capacity, were investigated to promote the application of plasma technology in bio-capacitors. Among the plasma parameters that affect the electrochemical properties and structure of hydrogels, discharge time and working gases are very important. Therefore, the electrochemical properties and structure of hydrogels treated with different discharge time (30, 90, and 150 s) and working gases (Ar, air, He) of plasma were investigated. The unmodified silk fibroin gel was used as the control group. Other parameters remained constant, as described in Section 2.2.
Effect of discharge time (30, 90, and 150 s) on the electrochemical properties and structure of hydrogels with argon as a working medium
AC impedance spectrum (EIS) is used to observe the impedance of the hydrogel with frequency (0.01 Hz∼1 MHz). Figure 2 demonstrates the impedance of the hydrogels of argon plasma treated with different discharge time. It can be seen, the argon plasma jet has no effect on the hydrogel impedance in the mid-high frequency (102∼105 Hz) range. The impedance at the highest frequency (105 Hz) represents the internal resistance of the hydrogel. Since the argon plasma jet does not affect the hydrogel’s internal resistance, the impedance curve is a horizontal line at high frequency. The capacitance characteristics are detected in the low frequency (100∼102 Hz) range. The double-layer electric capacity effect is produced between the hydrogel electrolyte and the electrode, and the impedance curve slope becomes negative. Nevertheless, the argon plasma jet still has no effect in this range. Normalized impedance modulus |Z| of hydrogels modified by Ar plasma with different discharge time (30, 90, and 150 s).
The ion diffusion effect in the hydrogel electrolyte appears in the low-frequency range (10−2∼100 Hz). The slope of the impedance curve becomes negative. The impedance of the hydrogel modified by argon plasma jet is greatly reduced at the low-frequency range compared to the unmodified hydrogel. A rising argon plasma discharge time shrinks the hydrogel impedance. At the argon plasma discharge time of more than 90 s, about half of the hydrogel impedance was reduced in the low-frequency range compared with the control group. The argon plasma jet contains many charged particles and active substances, increasing the ion concentration when injected into the hydrogel. The ion diffusion effect is greatly enhanced, reducing the hydrogel impedance. The longer the argon plasma discharge time generates more charged particles, reducing the impedance.
Electric capacity of hydrogels modified by argon plasma jet for 30, 90, and 150 s.

Fourier transform infrared spectrum (FTIR) analysis of the secondary structure of each group of hydrogel is conducted, and the FTIR curves are analyzed in the range of 500–4000 cm−1. Figure 3 shows the FTIR curve of the hydrogels modified by argon plasma with different discharge time. The FTIR curves of all groups have strong infrared absorption peaks at 1234, 1525, and 1628 cm−1, which are in the amide III, II, and I bands of silk fibroin, respectively, belonging to the β-fold structure of silk fibroin.
42
The result indicates that the secondary structure of silk fibroin of the hydrogel modified by argon plasma jet remained unchanged, mainly β-fold. However, the infrared absorption peaks of the Ar group at the wavelengths of 1234, 1525, and 1628 cm−1 are reduced compared with that of the empty group, indicating that the β-fold content in the hydrogel is reduced after modification by argon plasma. Besides the three infrared absorption peaks corresponding to the β-fold structure of silk fibroin, the FTIR curves of all groups showed a strong infrared absorption peak at 3292 cm−1, corresponding to the O-H stretching vibration region of graphene oxide,
43
indicating the presence of graphene oxide in the hydrogel. After the modifications of the hydrogels by argon plasma jet, the infrared absorption peaks of the FTIR curves at 3292 cm−1 are slightly weakened, indicating that the content of graphene oxide is also reduced during the modification. The active substances produced by the argon plasma jet react with graphene oxide, decomposing the graphene oxide in the hydrogel. The peak at 618 cm−1 corresponds to the benzene ring substitution region, the peak at 1061 cm−1 corresponds to aliphatic ether, the peaks at 1410 and 1441 cm−1 correspond to alkyl groups, and the peaks at 2925 and 3078 cm−1 correspond to C-H stretching vibration zone. The peaks at 1410 and 1441 cm−1 indicate the presence of alkyl groups, specifically sodium dodecyl sulfate, in the hydrogels. In SF-GO hydrogel, the silk fibroin is mainly β-fold structure, and O-H group of GO combines well with silk fibroin. Argon plasma can reduce the β-fold and GO content. Fourier transform infrared spectra of hydrogels modified by Ar plasma with different discharge time (30, 90, and 150 s).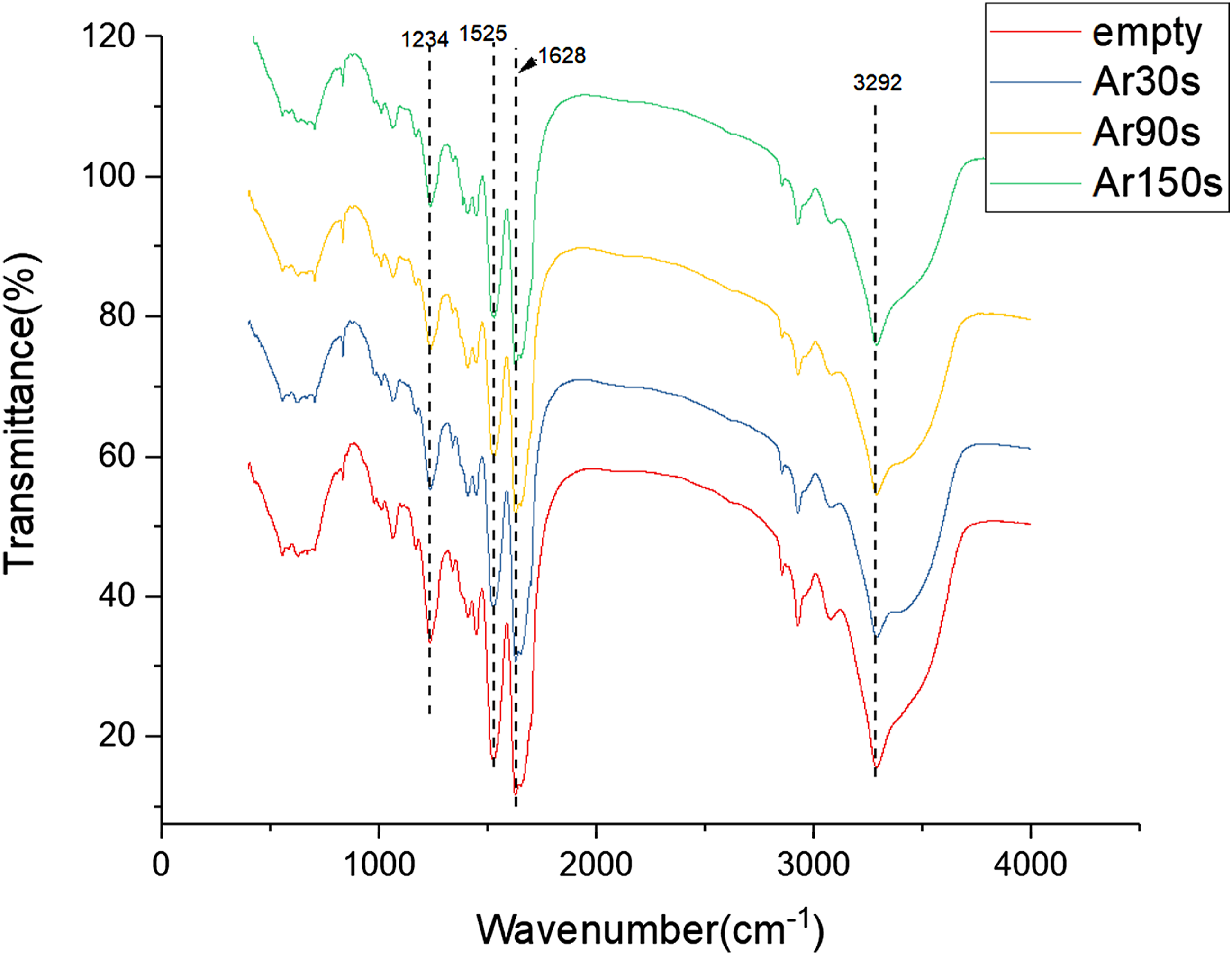
Figure 4 displays the X-ray diffraction spectra (XRD) of the hydrogels modified by argon plasma with different discharge time. The X-ray diffraction curves of all groups show significant diffraction peaks at 2θ = 20.45°, belonging to the Bragg angle of the β-fold structure
44
of silk fibroin. The result indicates that the secondary structure of the silk fibroin in the hydrogels modified by argon plasma jet remained unchanged, mainly the β-fold structure. After argon plasma modification, the diffraction peaks at 2θ = 20.45° become slightly flat, indicating that argon plasma modification reduces the content of the β-fold structure and crystallinity of the hydrogels. In addition, the X-ray diffraction curves of all groups also have a diffraction peak at 2θ = 9.7°, which corresponds to the Bragg angle of graphene oxide,
45
indicating the presence of graphene oxide in the hydrogels. The X-ray diffraction spectra results are aligned with those of FTIR spectra. X-ray diffraction spectra of hydrogels modified by Ar plasma with different discharge time (30, 90, and 150 s).
Effect of discharge time (30, 90, and 150 s) on the electrochemical properties and structure of hydrogels with air as a working medium
Figure 5 illustrates the influence of air plasma’s discharge time (30, 90, and 150 s) on the hydrogel impedance. From the impedance plot and at the mid-high frequency (102∼105 Hz) range, the air plasma jet does not affect the impedance of the hydrogels. At the highest frequency (105 Hz), the impedance curves indicate that the air plasma jet does not affect the internal resistance of the hydrogel. The capacitance characteristics appear in the low frequency (100–102 Hz) range. The double-layer electric capacity effect is generated between the hydrogel electrolyte and the electrode, and the slope of the impedance curve begins to become negative. Nevertheless, the air plasma jet still has no effect in this range. Normalized impedance modulus |Z| of hydrogels modified by air plasma with different discharge time (30, 90, and 150 s).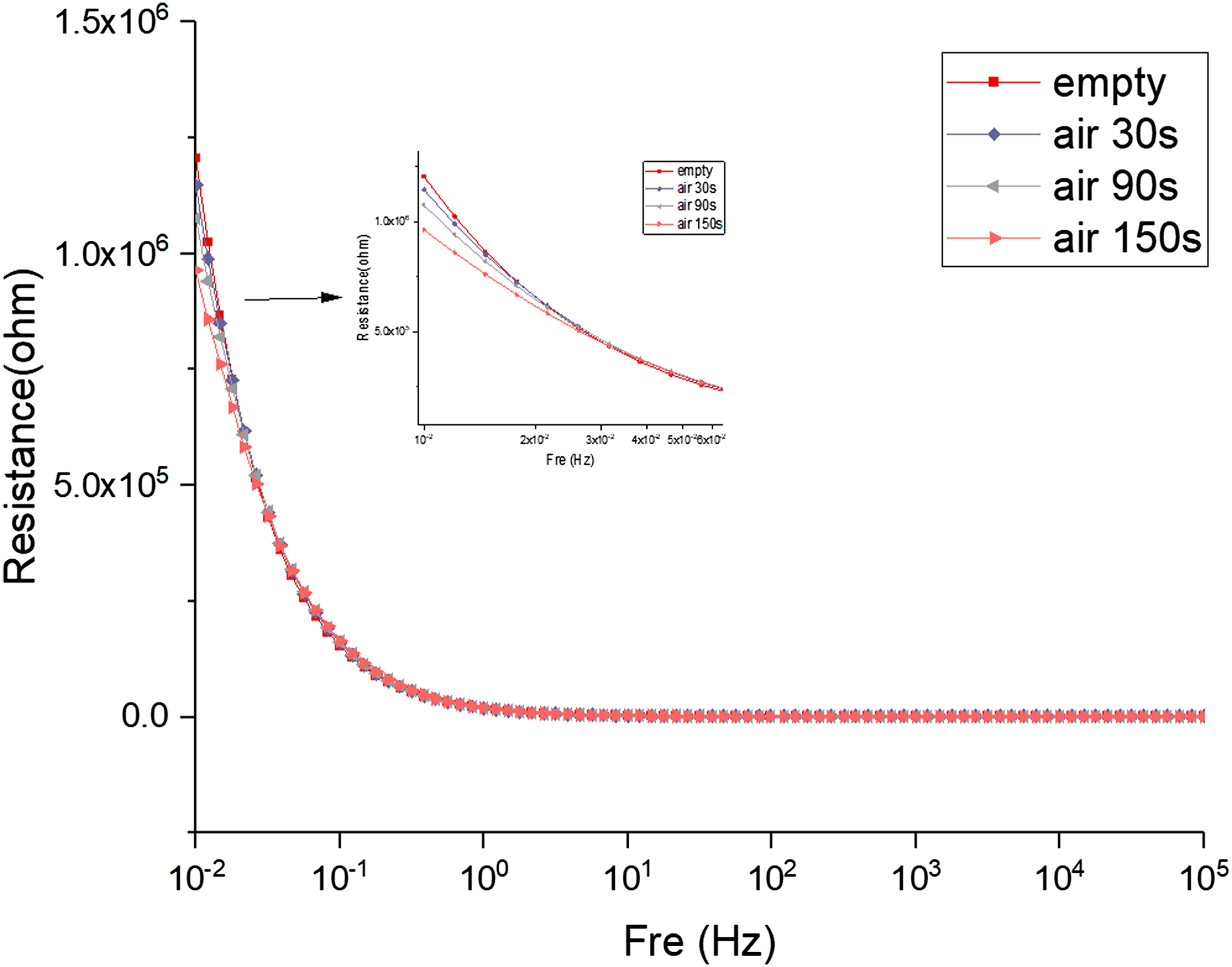
In the very low-frequency range (10−2∼100 Hz), the ion diffusion effect in the hydrogel electrolyte begins to appear, and the slope of the impedance curve begins to become negative. The impedance of the hydrogel modified by the air plasma jet decreases at a very low frequency compared to the hydrogel without plasma modification. A longer discharge time of air plasma further decreased the impedance of the hydrogel, which is more obvious in the range of 0.01–0.06 Hz. The air plasma jet contains many charged particles and active substances, increasing ion concentration when injected into the hydrogel. Therefore, the ion diffusion effect is greatly enhanced, reducing the impedance of hydrogel. The longer the discharge time of air plasma produces more charged particles, increasing the ion concentration in the hydrogel and reducing the impedance.
Electric capacity of hydrogels modified by air plasma jet for 30, 90, and 150 s.

Figure 6 depicts the FTIR curve of the hydrogels modified by air plasma with different discharge time. The FTIR curves of all groups have strong infrared absorption peaks at wavelengths of 1234, 1525, and 1628 cm−1, which are in the amide III, II, and I bands of silk fibroin and belong to the β-fold structure of silk fibroin. The result indicates that the secondary structure of the silk fibroin of the hydrogel modified by air plasma jet remained unchanged, especially the β-fold. However, the infrared absorption peaks of the air group at the wavelengths of 1234, 1525, and 1628 cm−1 are reduced compared with the empty group. Following the air plasma modification, the β-fold content in the hydrogels is reduced. Besides the three infrared absorption peaks corresponding to the β-fold structure of silk fibroin, the FTIR curves of all groups showed a strong infrared absorption peak at 3292 cm−1, corresponding to the O-H stretching vibration region of graphene oxide, indicating the presence of graphene oxide in the hydrogels. Following the modifications of the hydrogels by the air plasma jet, the infrared absorption peaks of the FTIR curves at 3292 cm−1 are weakened, indicating that the content of graphene oxide is also reduced. The generated active substances from air plasma react with graphene oxide, decomposing the graphene oxide. Similar to the argon group, the peak at 618 cm−1 corresponds to the benzene ring substitution region, the peak at 1061 cm−1 corresponds to aliphatic ether, and the peaks at 1410 and 1441 cm−1 correspond to alkyl groups, and the peaks at 2925 and 3078 cm−1 correspond to C-H stretching vibration zone. The peaks at 1410 and 1441 cm−1 prove the presence of alkyl groups, specifically sodium dodecyl sulfate, in the hydrogels. In SF-GO hydrogel, the β-fold structure in silk fibroin dominants, and O-H group of GO combines well with silk fibroin. Air plasma can reduce the content of β-fold and GO. Fourier transform infrared spectra of hydrogels modified by air plasma with different discharge time (30, 90, and 150 s).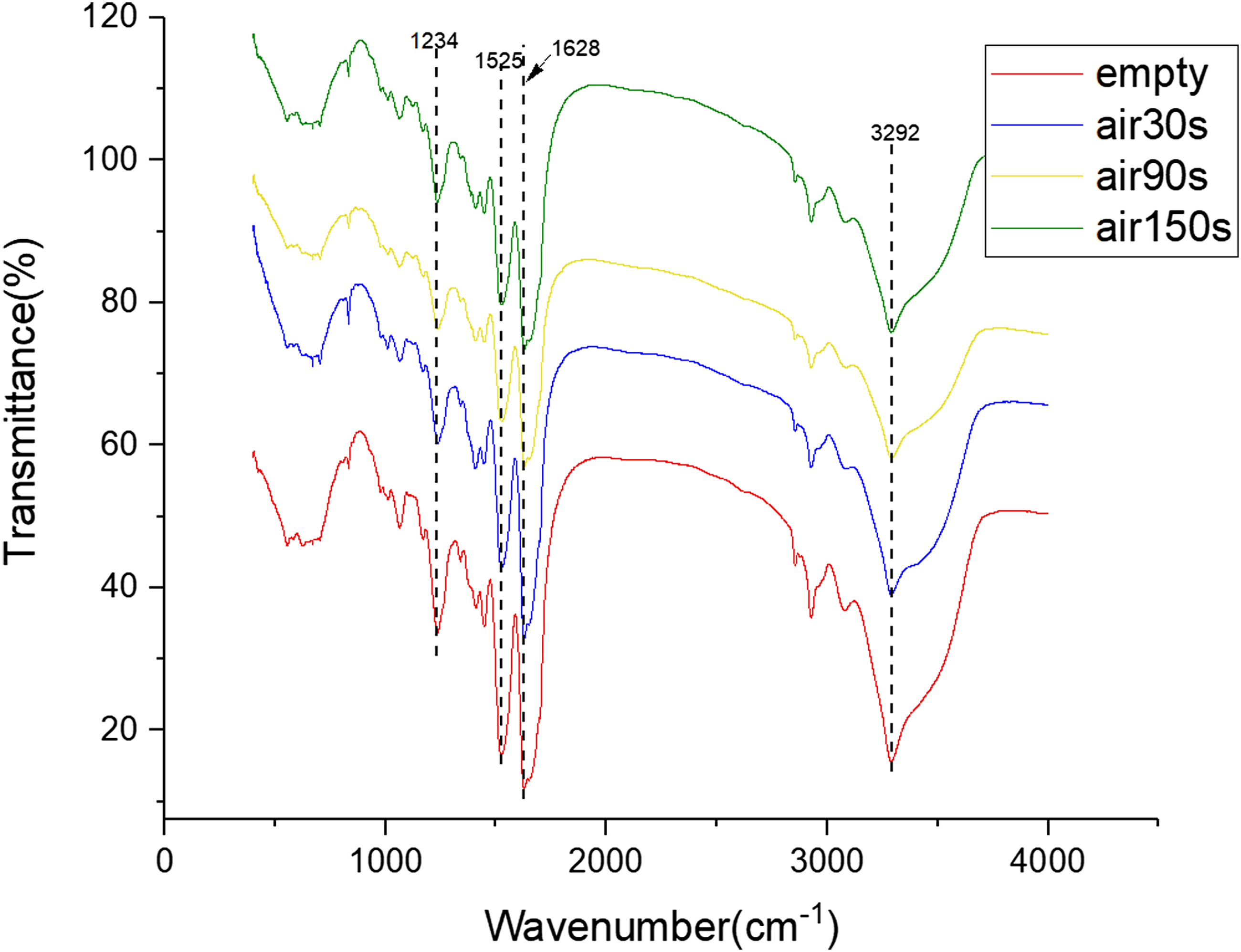
Figure 7 shows the X-ray diffraction spectra (XRD) of the hydrogels modified by air plasma with different discharge time. The X-ray diffraction curves of all groups display significant diffraction peaks at 2θ = 20.45°, 2θ = 20.45° belongs to the Bragg angle of the β-fold structure of silk fibroin. The result indicates that the secondary structure of the silk fibroin in the hydrogel modified by air plasma jet remained unchanged, mainly the β-fold structure. After hydrogel modification by air plasma, the diffraction peaks at 2θ = 20.45° become slightly flat, indicating a reduction of β-fold structure content and crystallinity of the hydrogels. In addition, the X-ray diffraction curves of all groups also have a diffraction peak at 2θ = 9.7°, which corresponds to the Bragg angle of graphene oxide, indicating the presence of graphene oxide in the hydrogels. The X-ray diffraction spectra results are aligned with those of FTIR spectra. X-ray diffraction spectra of hydrogels modified by air plasma with different discharge time (30, 90, and 150 s).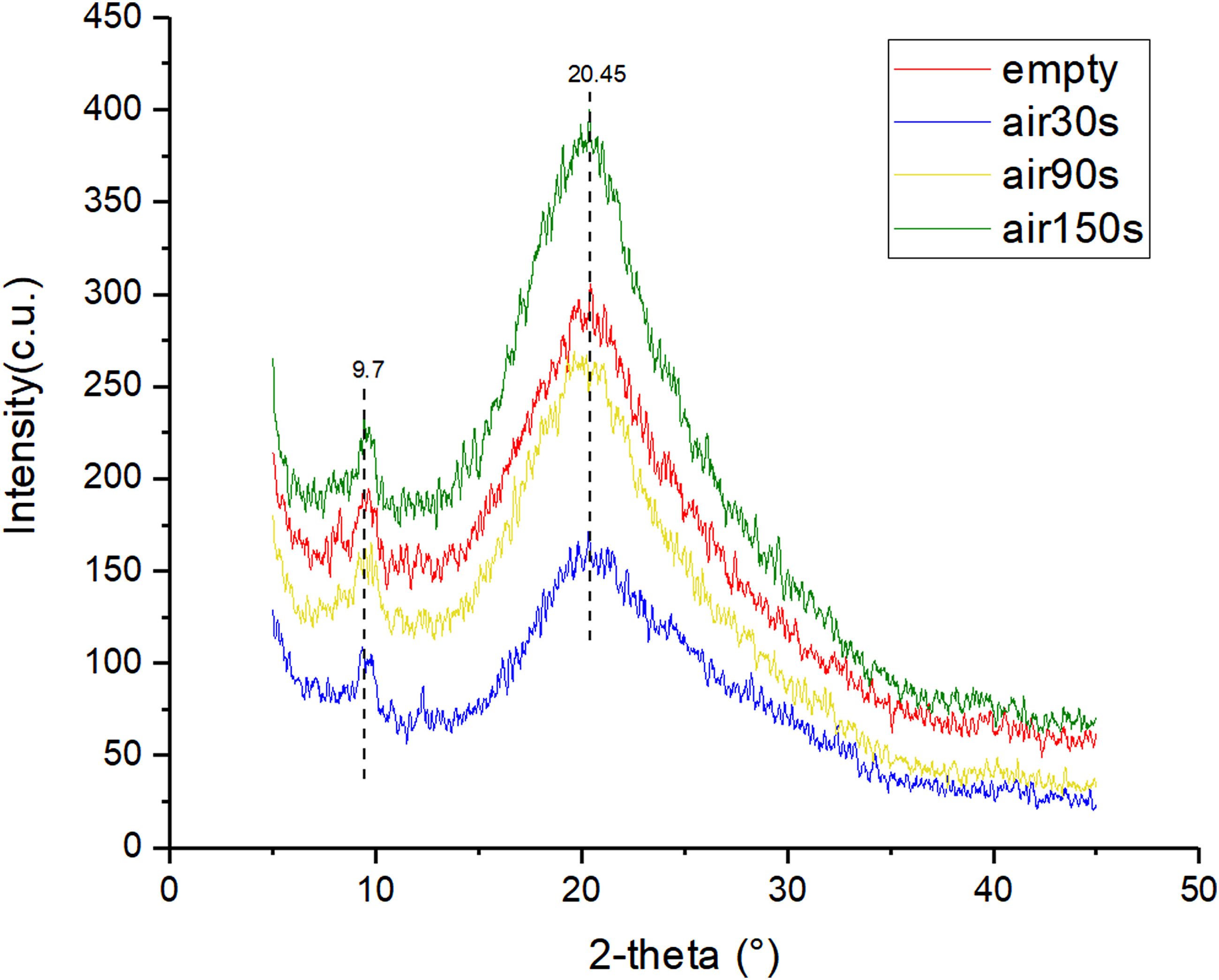
Effect of discharge time (30, 90, and 150 s) on the electrochemical properties and structure of hydrogels when the working medium is helium
Figure 8 shows the effect of the discharge time of the helium plasma on the impedance of the hydrogel. The impedance plot depicts that the helium plasma jet does not affect the impedance of the hydrogel in the mid-high frequency (102∼105 Hz) range. At the highest frequency (105 Hz), the impedance curves indicate that the helium plasma jet does not affect the internal resistance of the hydrogel. The capacitance characteristics begin to appear in the low frequency (100∼102 Hz) range, the double layer electric capacity effect is generated between the hydrogel electrolyte and the electrode, and the slope of the impedance curve begins to become negative. Nevertheless, the helium plasma jet still has no effect in this range. Normalized impedance modulus |Z| of hydrogels modified by helium plasma with different discharge time (30, 90, and 150 s).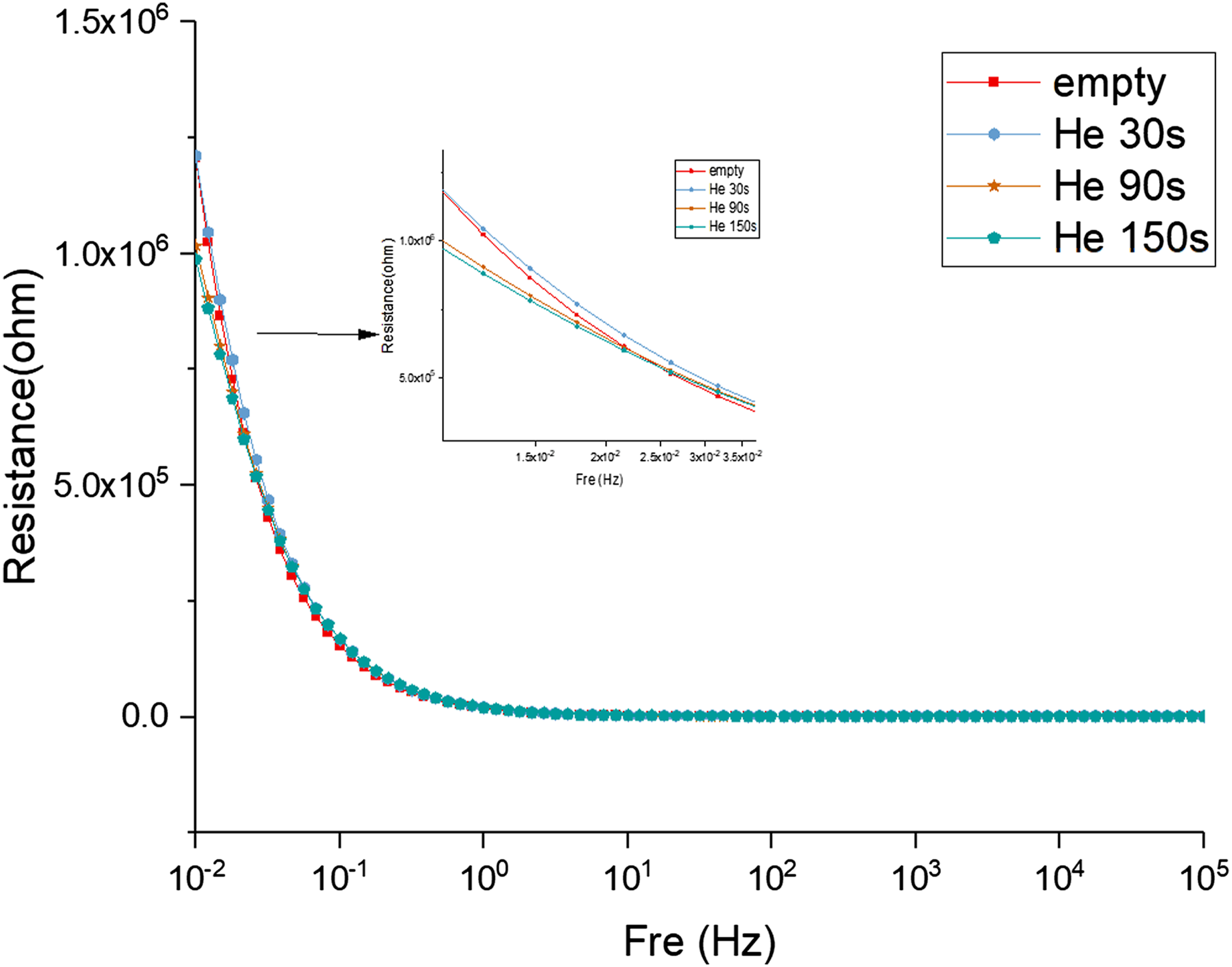
In the very low frequency (10−2∼100 Hz) range, the ion diffusion effect in the hydrogel electrolyte begins to appear, and the slope of the impedance curve becomes negative. At the same frequency, the impedance of the hydrogel modified by helium plasma jet tends to decrease compared to the unmodified hydrogel. However, the reduced impedance of the modified hydrogel is not obvious when the discharge time of the helium plasma is in the 30 s. Longer helium plasma discharge time further decreased the hydrogel impedance, which is more obvious in the range of 0.01–0.025 Hz. The helium plasma jet contains many charged particles and active substances, increasing the ion concentration when injected into the hydrogel, enhancing the ion diffusion effect, thereby reducing the impedance of the hydrogel. The longer discharge time of the helium plasma produced more charged particles, increasing the ion concentration in the hydrogel and reducing the impedance.
Electric capacity of hydrogels modified by helium plasma jet for 30, 90, and 150 s.

Figure 9 shows the Fourier transform infrared spectra of the hydrogels modified by helium plasma with different discharge time. The FTIR curves of all groups exhibit strong infrared absorption peaks at 1234, 1525, and 1628 cm−1, which are in the amide III, II, and I bands of silk fibroin and belong to the β-fold structure of silk fibroin. The result indicates that the secondary structure of silk fibroin of the hydrogel modified by helium plasma jet remained unchanged, especially the β-fold structure. However, at the wavelengths of 1234, 1525, and 1628 cm−1, the infrared absorption peaks of he group are reduced compared with that of the empty group, indicating that the β-fold content in the hydrogels is reduced. Besides the three infrared absorption peaks corresponding to the β-fold structure of silk fibroin, the FTIR curves of all groups have a strong infrared absorption peak at 3292 cm−1, corresponding to the O-H stretching vibration region of graphene oxide, proving the presence of graphene oxide in the hydrogels. After the hydrogels were modified by the helium plasma jet, the infrared absorption peaks of the FTIR curves at 3292 cm−1 were weakened, indicating the reduction of graphene oxide content. The generated active substances from helium plasma react with graphene oxide, decomposing the graphene oxide. The peak at 618 cm−1 corresponds to the benzene ring substitution region, the peak at 1061 cm−1 corresponds to aliphatic ether, the peaks at 1410 and 1441 cm−1 correspond to alkyl groups, and the peaks at 2925 and 3078 cm−1 correspond to C-H stretching vibration zone. The peaks at 1410 and 1441 cm−1 prove the presence of alkyl groups, specifically sodium dodecyl sulfate, in the hydrogels. In SF-GO hydrogel, the silk fibroin consists of β-fold mainly, and O-H group of GO combines well with silk fibroin. Helium plasma can make the β-fold and GO content decrease. Fourier transform infrared spectra of hydrogels modified by helium plasma with different discharge time (30, 90, and 150 s).
Figure 10 demonstrates the X-ray diffraction spectra (XRD) of the hydrogels modified by helium plasma with different discharge time. The X-ray diffraction curves of all groups exhibit significant diffraction peaks at 2θ = 20.45°, which belongs to the Bragg angle of the β-fold structure of silk fibroin. The result indicates that the secondary structure of the silk fibroin of the hydrogels modified by helium plasma jet has remained unchanged, mainly β-fold. After helium plasma modification, the diffraction peaks at 2θ = 20.45° become slightly flat, indicating a reduction of the hydrogels’ β-fold structure content and crystallinity. In addition, the X-ray diffraction curves of all groups also have a diffraction peak at 2θ = 9.7°, corresponding to the Bragg angle of graphene oxide, indicating the existence of graphene oxide in the hydrogels. The X-ray diffraction spectra results are aligned with those of FTIR spectra. X-ray diffraction spectra of hydrogels modified by helium plasma with different discharge time (30, 90, and 150 s).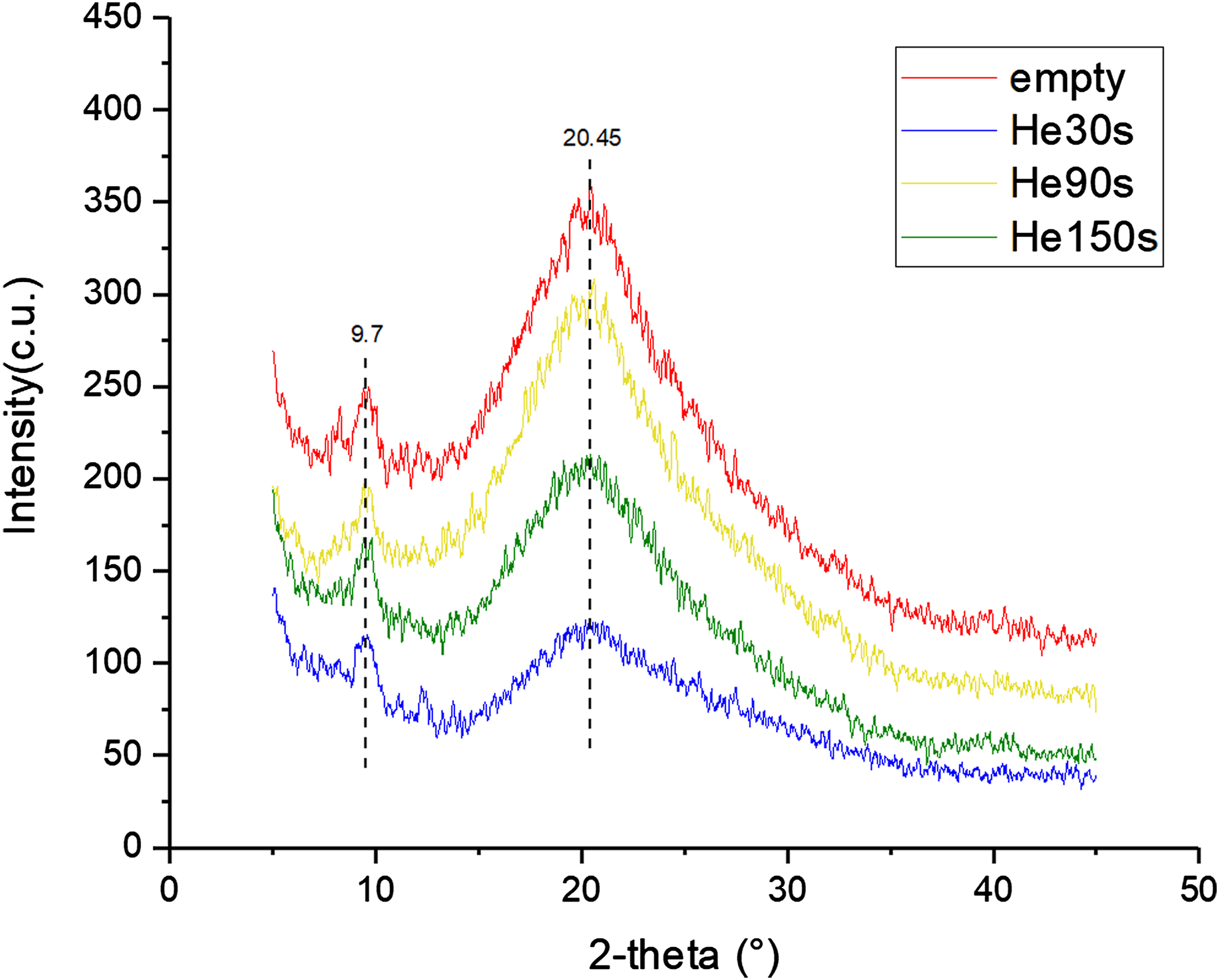
Conclusions
This study investigates the effect of the low-pressure plasma jet using different working gas (argon, air, helium) and discharges time (30, 90, and 150 s) on the electrochemical properties and structure of SF-GO hydrogel. Any working gas’s plasma can reduce the hydrogels' impedance in the very low frequency (10−2∼100 Hz) range, and the longer the plasma discharge time further decreases the impedance. The plasma jet generates a large number of charged particles and active substances, increasing the ion concentration when injected into the hydrogel and enhancing the ion diffusion, thereby reducing the impedance of the hydrogels. In the very low frequency (10−2∼100 Hz) range, the sequence of impedance change is Ar group <air group ≈ He group <empty. Interestingly, argon plasma jet modification can increase the electric capacity of SF-GO hydrogel, while air and helium plasma jet reduce the electric capacity of the hydrogel. However, the electric capacity of the hydrogels modified by air plasma jet is larger than that of the hydrogels modified by helium plasma jet. Overall, the sequence of electric capacity change is Ar group >empty >air group >He group, but plasma discharge time does not influence the electric capacity. The plasma jet can also reduce the content of β-fold structure and graphene oxide in the SF-GO hydrogel. This study proves that plasma jet can affect the electrochemical properties and structure of hydrogel, which can aid in applying plasma in biological supercapacitors. Further studies are needed to understand how plasma affects the electrochemical performance of hydrogels.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key R & D plan of Anhui Province (201904a07020013), Collaborative Innovation Program of Hefei Science Center (CAS, CX2140000018), and the Funding for Joint Lab of Applied Plasma Technology (JL06120001H).
