Abstract
Vinyl polymerization is an important path for in-situ polymerization of thermoplastic resins. BPO (Benzoyl peroxide) and DMT (dimethyl-p-toluidine) were chosen to initiate the radical polymerization of MMA (methyl methacrylate) in this work. Optimization of initiator concentration was studied through reaction rate of polymerization progress and properties of PMMA (polymethyl methacrylate). At first, the catalytic of DMT in MMA polymerization was studied through polymerization experiments and rheological experiments. Results showed that DMT could catalyze more than one molecular BPO in the whole reaction period. It can catalyze the decomposition of BPO after being connected to PMMA chains. Second, the BPO: DMT ratio was set to 2:1 to study the ideal content by comparing the mechanical properties of PMMA. Results showed that with the increase of initiator contents, T g , molecular weight, and residual monomer were all declining. Meanwhile, the mechanical properties of PMMA matrix first increased and then decreased. This phenomenon was analyzed by 1H-NMR and SEM. And the optimal BPO-DMT concentration was 1.0 mol%–0.5 mol%.

Introduction
Recently, an increasing interest in in-situ polymerization of fiber-reinforced thermoplastic composites arose in composite industry.1–3 According to previous research,4–6 PMMA (polymethyl methacrylate) resin has excellent potential in this area. Its high mechanical properties, low cost, rapid molding, and low energy consumption characteristics are urgently needed in composites industry. 7 Therefore, the applied research of in-situ polymerization of PMMA composites plays an essential role in this field. 8
The high level of polymerization heat and auto-accelerate effect makes the polymerization of MMA hard to control.9,10 Therefore, a proper initiator and initiator content should be studied in detail to lessen the risk of thermal runaway and reach a high property of matrix at the same time. However, this has been less studied in previous literature. It should be mentioned that there are two differences between in-situ polymerization research and bulk polymerization of MMA. It is important to study the relationship between the properties of PMMA, PMMA molding process time, and initiator content to manufacture fiber-reinforced PMMA composites.
Peroxide-tertiary amine is a widely used redox initiation, initiating vinyl monomer polymerization at room temperature. Ostu and others 11 confirm the existence of two free radical active species in BPO-DMA redox initiation, benzoyl radical from BPO and aminomethyl radical from DMA. Feng and Qiu12–16 systematically study the effects of connecting different groups on the para-position of benzene ring of the aromatic tertiary amine. Researchers reach a consensus that a lone pair electron of tertiary amine attacks the oxygen atom of peroxide and forms an amide cation and a benzoyl anion. Then, further redox reaction forms a molecule of benzoic acid, a molecule of aminomethyl radical, and a molecule of benzoyl radical. The latter two molecules can initiate monomers polymerization independently and retain at the end of the macromolecular chain.17–24 Therefore, most studies set the ratio of initiators to be 1:1. However, peroxide and tertiary amine have different activities. Peroxide and tertiary amine are consumed equivalent in the initiator period but may not consume equivalent in the whole reaction period.
On-line analytical methods, including immersion rheometer test and on-line infrared test, help researchers to study deeper in mechanisms of resin polymerization processes. Furthermore, these methods promoted studies of polymerization kinetics of acrylates as well, such as the influence of side reactions 25 of tertiary amine, rheological behavior,26,27 and simulation.28–32 The ratio of reactants, reaction rate, and properties of products could establish a more direct relationship through studies in the whole reaction.
This study first determines the optimal ratio of two components of initiator in a low content by studying the polymerization rate of reaction. 29 After that, a series of initiator contents are set to study the optimum content of initiators by comparing the properties of PMMA. The mechanism of connection of mechanical properties with initiator content is figured out by SEM and 1H-NMR. This work intended to obtain an optimum content of PMMA resin and pave the way for the in-situ polymerization of fiber-reinforced PMMA composites.
Experimental
Materials
MMA: AR, Adamas Reagent, Ltd BPO: 75%, remainder water, Aladdin Industrial Corporation. DMT: CP, Sinopharm Chemical Reagent Co, Ltd Methylbenzene: AR, Sinopharm Chemical Reagent Co, Ltd Petroleum ether 60–90°C: AR, Sinopharm Chemical Reagent Co, Ltd.
Methods
MMA bulk polymerization experiment
Three components were added into test tubes by a series of molar fractions in regular sequence (BPO, MMA, and DMT). Samples were marked as BPO: DMT: MMA = x mol%: y mol%: z mol%. Then, these tubes were covered with lids to prevent volatilization, shaken and mixed well, and placed in calorstat at 30°C. Characteristics of the polymerization reaction from beginning to end were recorded.
Rheological measurement
Rheological properties were assessed on a BROOKFIELD’s DV-III Ultra rheometer by measuring the torsional force of the sc4-31 rotor (USA). Since the initial viscosity of MMA resin is 0.58 mPas (25°C), rheological behavior in the viscosity range of resin mixture from 1 mPas ∼ 5000 mPas were measured and were recorded.
Purification of PMMA samples
2 g PMMA was ground into powder and was dissolved in 98 g toluene (40°C). Then 100 g petroleum ether was added into the solution, and a few white sediments were precipitated from the solution. The sediments were filtered and dried in a 40°C oven for 1 h to remove the residual solvent.
Polymerization temperature history experiment
The reaction solution was poured into a 20 mL glass bottle. The bottle was plugged by a cork and put in a water bath at 30°C. A thermometer was used to record the temperature of the solution.
Glass transition temperature
T g of PMMA samples were evaluated by DMA testing machine(DMA 242E-NETZSCH) based on ASTM D7028-07. The vibrational mode was bending, and the frequency was 1 Hz; the heating rate was 2 K/min; specimen size was 58 mm × 10 mm × 3 mm; E′ was chosen for analysis.
Molecular weight distribution
Molecular weight distribution of PMMA matrix was evaluated by GPC, tetrahydrofuran as elution solvent.
Mechanical properties
Tensile and bending properties were tested in Instron universal testing machine according to standard ASTM D638-14 and ASTM D790-17, respectively.
Residual monomer content
Residual monomer content was tested by 1H-NMR in CDCl3.
Scanning electron microscope
The specimens for the morphological observation were prepared via fracturing the blends under cryogenic conditions using liquid nitrogen. The fracture surfaces of specimens were treated by spray-gold(20 mA, 60 s), and were evaluated by SEM (EVO15-Carl Zeiss, Germany).
Results and discussion
Optimal initiator ratio
MMA bulk polymerization experiment
A series of molar fractions of BPO and DMT are set for experiments. The molar fraction and molding quality evaluation diagrams are formed in Figure 1. Polymerizing capability of MMA as a function of DMT and BPO contents at 30°C.
Based on the internal quality and polymerization time of the PMMA, these samples are classified into two grades. According to the internal quality of PMMA resin and polymerization time, the polymerization ability of PMMA is divided into two grades. Polymerizable: the resin polymerization time is short, and the internal bubble is few; Fail to polymerize: fail to polymerize for more than 100 h or too many bubbles in the resin.
Results show that the polymerizable initiator contents are concentrated on the bottom right part of the diagram. These data show that the polymerization of MMA monomer is difficult at lower content (<0.1 mol%–0.1 mol% of BPO-DMT). When the molar fraction of BPO-DMT exceeds 0.5 mol%–0.5 mol%, the polymerization rate is too fast, the internal bubbles increase.
Some works conclude14–16 that the effect of BPO content on polymerization rate is slightly higher than that of aromatic amine at low conversion stage (<4%). The influences of the DMT and BPO contents on MMA bulk polymerization are explored, respectively. Results show that adding BPO to MMA alone can initiate polymerization in a long time (>30 h), but adding DMT alone cannot initiate polymerization. Compared with adding DMT content, the polymerization rate increases more with adding BPO content. Besides, results show that MMA is hard to polymerize when the molar fraction of DMT exceeds than that of BPO. Side reactions increase a lot when many DMT fail to react with BPO. 25 This side reaction could cause a significant lessening of the main polymerization reaction. According to the study of Zoller and others, 29 aminomethyl radicals are connected more than benzoyl radicals to the polymer chain in the low conversion stage, which can be evidence. Based on the above conclusions, the molar fraction is carefully set for further experiments within the polymerizable range.
Rheological behavior of polymerization
Through on-line measurements of resin polymerization viscosity, the viscosity curves of MMA bulk polymerization in the early stage (before gel) are obtained. According to the mechanism of gel effect in free radical polymerization, the time of early stage and middle stage could be characterized by the time when the viscosity of the reaction system increases rapidly. Moreover, the reaction times of resin with different initiator contents are characterized.
As Figure 2, when the molar fraction of DMT is more than 0.2 mol%, the increase of DMT content hardly affects the reaction time. This phenomenon can be explained by the reaction kinetics of the diffusion-controlled mechanism. When the proportion of DMT in this reaction system is high, more DMT are participating in the reaction of capturing free radicals on polymer chains, as shown in Figure 3. Because the friction of large molecules in solution is higher than that of small molecules. The diffusion rate of DMT is faster than that of the large molecule (DMT at the end of PMMA chain).7,10 According to the theoretical model, the rate constant of the double radical termination is very high, and the limiting factor of double radical termination rate is the diffusion rate of radical. DMT radicals end parts polymerize in advance. Therefore, the increasing of DMT content could not increase the reaction rate but could decrease the molecular weight of polymers. Viscosities of MMA polymerization systems as functions of DMT contents at 30°C. DMT radical’s reaction progress.
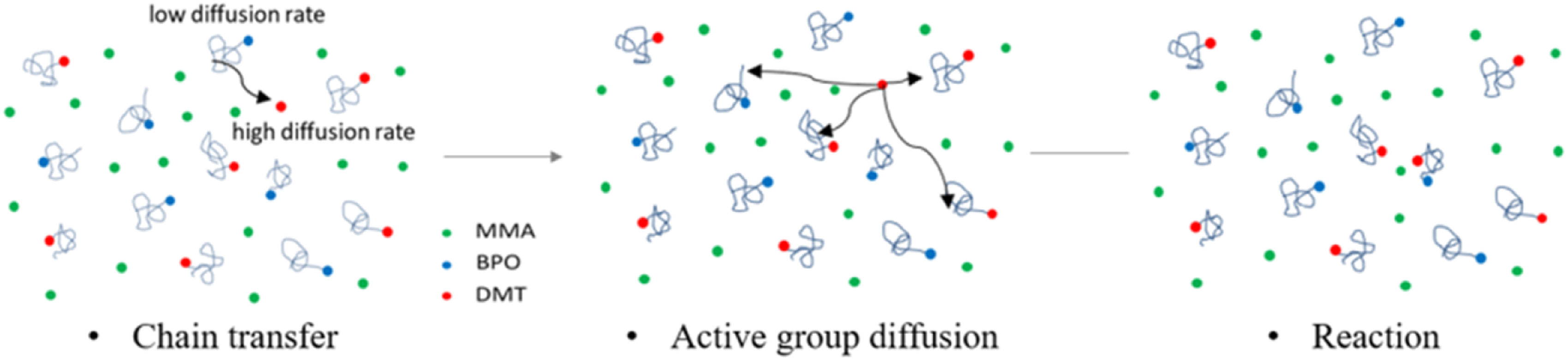
When the molar fraction of DMT is less than 0.2 mol%, reaction time declines with DMT content increasing. These results show that one molecule DMT may catalyze more than one molecule of BPO decomposition. To further study the mechanism of this reaction, a conjecture is put forward: a molar tertiary amine first initiates a BPO decomposition and forms a PMMA macromolecule; Then the catalytic activity of PMMA polymers which connect the end-group DMT remains; Furthermore, it can further participate in redox reactions with BPO; At the same time, two new free radicals are generated to initiate other MMA monomers.
After that, the concentration of DMT is fixed to 0.1 mol%, and the concentration of BPO is increased from 0.1 mol% to 0.4 mol%, as shown in Figure 4. Viscosities of MMA polymerization systems as functions of BPO contents at 30°C.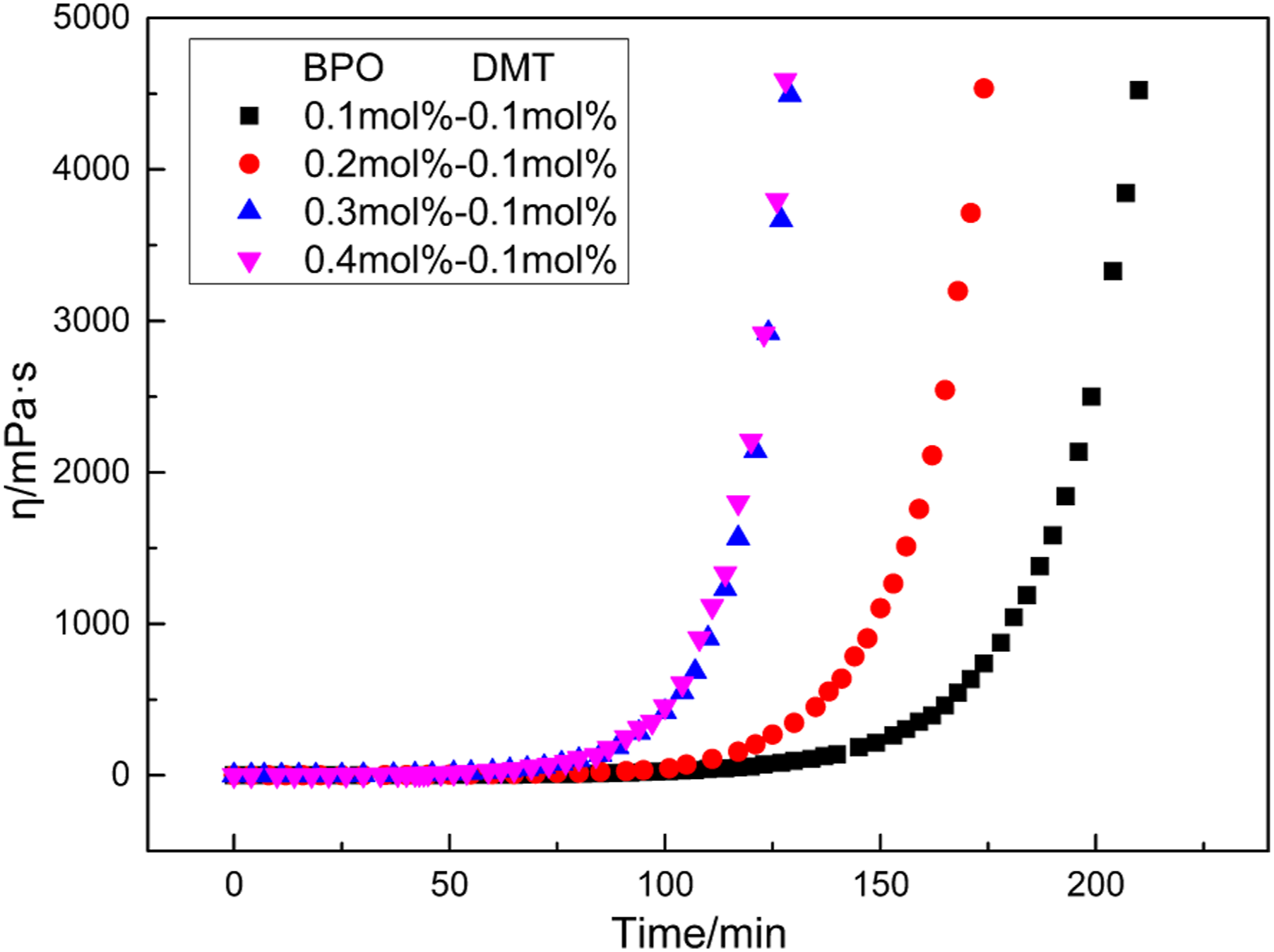
The trends in Figure 4 indicate that when the content of BPO is between 0.1 mol% to 0.3 mol%, increasing the molar fraction of BPO has speeded up the reaction rate. Moreover, when the content of BPO exceeds 0.3 mol%, increasing the molar fraction of BPO has little effect on the reaction rate.
Structure of PMMA polymer analysis
In order to verify the previous conjecture, the following experiments are conducted. Figure 5(a) (left) shows the molecular weight (MW) distribution of polymers with different DMT additions. With the ratio of DMT to BPO decreasing from 1:1 to 1:4, the molecular weight peaks are changing to higher molecular weight, molecular weight distribution widens, and a new peak appears at a higher molecular weight position. MW (a) and UV spectrum (b) of PMMA samples.
First, the decrease of reactive points number of DMT with the DMT contents decrease, which leads to an increase in PMMA molecular weight. The wider molecular weight distribution indicates that the active point of the reaction is different, and this kind of active substance is more involved in polymerization. It can be speculated that another reactive active substance in the system is more involved in the polymerization, resulting in wider molecular weight distribution. This weight distribution indicates that a group of higher molecular weight polymers is formed during the reaction when DMT concentration is less than BPO.
Then, the PMMA polymer (DMT: BPO = 1.0 mol%: 1.0 mol%) is purified. Moreover, the UV absorption spectra of purified PMMA samples are acquired in Figure 5(b) (right).
The peak of methylbenzene (206.5261) shift to 257.5 and 312 in theory due to the redshift effect of -NR2. It can be seen that a strong peak appears on 261.8 nm and a weak peak appears on 307.8 nm. Compared with the experiment results, it can be confirmed that DMT is connected at the end of PMMA main chain. Besides, this purified PMMA could catalyze BPO decomposing as before conformed by new polymerization experiments.
In conclusion, DMT as an end group of PMMA could catalyze BPO decomposing and form a polymer with a higher molecular weight. At room temperature, DMT mainly has two effects in BPO- DMT redox initiation system: 1. DMT has the capacity to capture free radicals in the polymerization process of MMA. When the molar fraction of DMT is higher than that of BPO, the side reaction is greatly accelerated, causing polymerization of MMA monomer hard to proceed. 2. DMT can catalyze BPO to decompose as an initiating agent to produce primary free radical active species. The aromatic tertiary amine connecting to the PMMA chain maintains partial activities and could continue catalyzing another BPO decomposition, as shown in Figure 6. DMT catalyzing another BPO decomposition.

The secondary catalytic mechanism of BPO decomposition by end-group DMT is the same as that of the primary catalytic mechanism described by the former work. It should be noted here that the higher molecular weight polymers are decreasing with the ratio of DMT increasing. When the molecular ratio is close to 1:1, this effect is masked. On one hand, the reaction points of DMT connected to macromolecular chain are less than DMT molecular. On the other hand, the DMT connected to macromolecular chain is affected by steric hindrance effect. Therefore, end-group DMT is in a weak position in the competition with DMT molecular in catalyzing BPO decomposition. The higher molecular weight polymers could appear only when the proportion of DMT is significantly lower than BPO. Considering these factors above, a suitable ratio in the BPO-DMT redox system in manufacturing exists. In this study, the optimum ratio of BPO: DMT is 2:1.
Optimal initiator content
The ratio of BPO: DMT is set to 2:1 to study the optimal initiator content of PMMA in-situ polymerization.
MMA polymerization rate
The relationship between reaction progress, peak temperature, and the initiator ratio is determined through polymerization temperature history experiments in a 30°C water bath, as shown in Figure 7. Temperature history of PMMA polymerization at 30°C.
Results show that polymerization time is effectively declining with the initiator content increasing. When the initiator content is 0.2 mol%–0.1 mol%, total polymerization time is 267 min; When the initiator content reaches 2.0 mol%–1.0 mol%, total polymerization time is 50 min.
Peak temperatures of polymerization increase with the initiator content increasing from 0.2 mol%–0.1 mol% to 1.0 mol%–0.5 mol%. However, peak temperatures decrease with initiator content increasing from 1.0 mol%–0.5 mol% to 2.0 mol%–1.0 mol%. It can be noticed that a little peak arises at the start of the reaction when initiator content reaches 2.0 mol%–1.0 mol%. This rising temperature at the start of the reaction could cause an accelerating of many side reactions and weaken the polymerization of monomers. This phenomenon has few connections with this work. So, the phenomenon is not delved in detail. Nevertheless, three important information are gained through this experiment: (1) Total reaction time at all kinds of initiator content (We consider that the reaction is terminated when the center temperature of PMMA resin is reduced to the ambient temperature. This is to meet requirements for molding PMMA resin matrix). (2) A cooling device may need to be prepared in PMMA resin polymerization progress (The actual forming PMMA parts are large in size. When the thickness of the sample used in the experiment is exceeded, the peak temperature of PMMA parts will be higher, so cooling equipment may be needed to avoid forming failure due to overtemperature. When forming larger PMMA parts, water cooling facilities are added to the mold.). (3) Two dominant factors may control the properties of PMMA resin, and an optimal initiator content exists.
PMMA matrix properties
Properties of PMMA matrix.

Results show that T g and MW of PMMA both decreasing with the initiator content increasing. PMMA strength is increasing with initiator content increasing from 0.2 mol%–0.1 mol% to 1.0 mol%–0.5 mol%, and is decreasing with initiator content increasing from 1.0 mol%–0.5 mol% to 2.0 mol%–1.0 mol%. The latter one is easy to figure out. When the MW of resin is lower than critical molecular weight, the reduction of MW could cause a significant reduction of mechanical properties as initiator content increases.
However, the low mechanical properties of the resin at high molecular weight indicate that another factor affects the mechanical properties of the resin. 1H-NMR and SEM are used to figure out the reason for the former phenomenon. 1H-NMR is used to determine the monomer content in PMMA resin, as shown in Figure 8. The signals with chemical shift 3.76δ and 3.60δ can be assigned to the O-CH3 groups of methyl methacrylate monomer and methacrylate units incorporated into the polymeric chains. Those at 5.56δ and 6.10δ to the acrylic methylene group, = CH2. 1H-NMR of PMMA resin (a) initiator content 0.2 mol%–0.1 mol%, (b) initiator content 1.0 mol%–0.5 mol%.
Residual monomer of resin at different initiator content.

The resin’s fracture surfaces in two initiator content are shown in Figures 9 and 10. The fracture surface of sample presents brittle fracture morphology when initiator content is at a high level. The fracture section contains fracture sources (red circle), specular areas, foggy areas, and rough areas. Fracture surfaces of resin at 1.0 mol%–0.5 mol% initiator content. Fracture surfaces of resin at 0.2 mol%–0.1 mol% initiator content.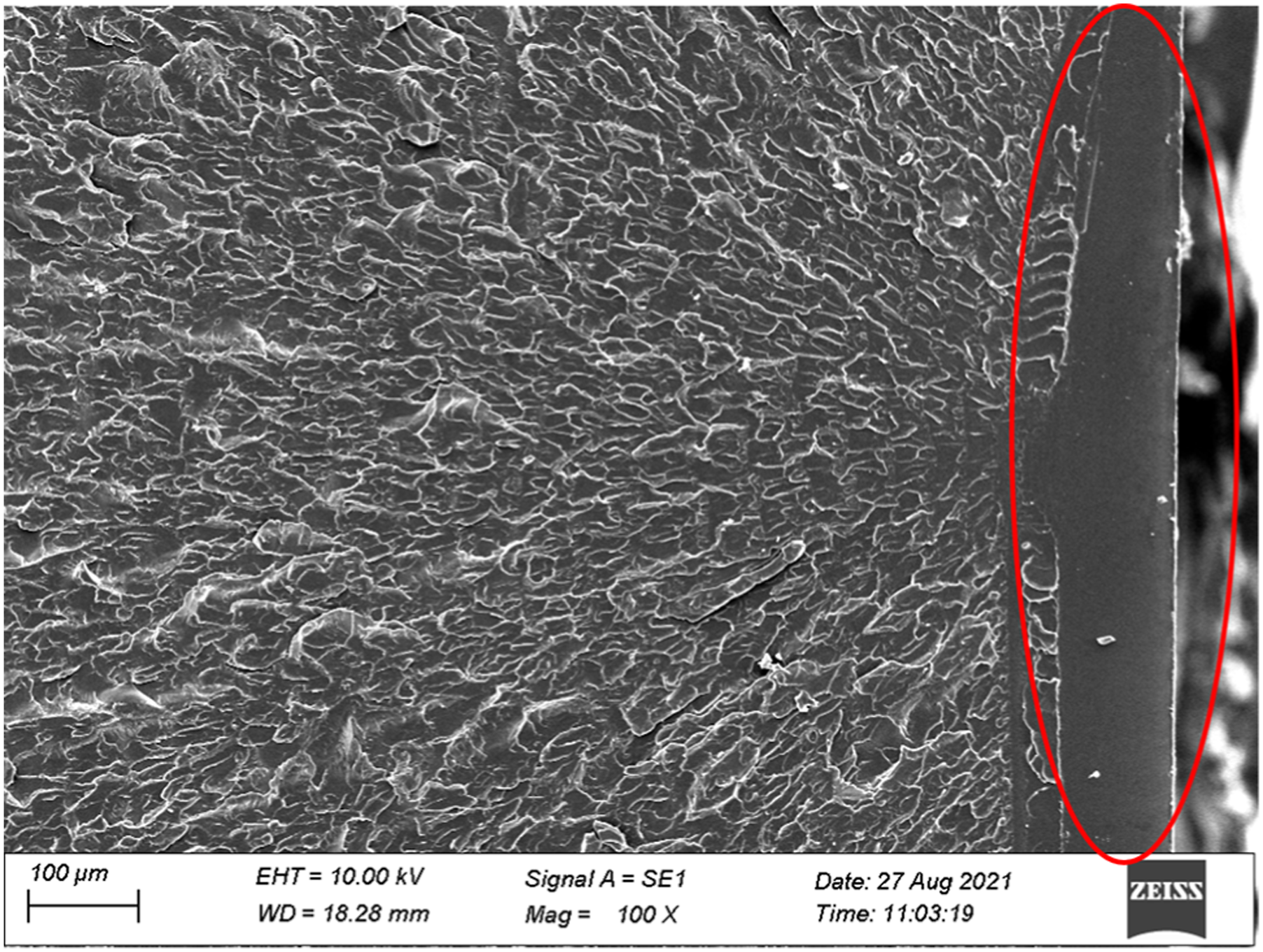

The fracture morphology of PMMA resin presents a distinct difference when initiator content is at a low level. Several pits appeared on the fracture surface of sample and are embedded in resin matrix. It can be determined from the shape of these pits' boundary caused by residual monomers in the resin matrix.
Combined with the 1H-NMR spectrum, it can be concluded that more unpolymerized monomers are retained in PMMA resin due to the decrease of initiator content. These monomers in resin lessen the binding energy of PMMA matrix. When the resin is under load, the cracks initiation is started from those monomers’ gathered areas. The cohesion of polymer is reduced, which is macroscopically manifested as the strength and modulus of resin matrix are reduced.
In conclusion, the mechanical properties of PMMA resin are significantly affected by two factors, molecular weight, and residual monomer content. On one hand, molecular weight declines with the initiator content increases. The mechanical properties decrease a lot when molecular weight decrease exceed a line (1 × 105). On the other hand, residual monomer content declines with the initiator content increasing. The mechanical properties decline when residual monomer content reaches a high level. These two factors jointly determine the optimal content of BPO and DMT is 1.0 mol%–0.5 mol%.
Conclusions
Optimal DMT-BPO content is determined by studies of polymerization mechanism of MMA and analysis of properties of PMMA. First, the catalytic activity of DMT/BPO for PMMA polymerization is studied. The catalytic mechanism of DMT in DMT/BPO redox initiation system is investigated in detail. It is found that DMT is linked onto the end of PMMA chain, and this DMT-endcapped PMMA chain has the catalytic activity to initiate polymerization of MMA further. Optimal ratio range of BPO: DMT is determined to 2:1. Second, the mechanical properties of PMMA resin are significantly affected by molecular weight and residual monomer content. On one hand, mechanical properties decline a lot when molecular weight is lower than 1 × 105. On the other hand, mechanical properties are decreasing with residual monomer content increasing to a high level. These two factors (reaction characteristics of DMT with BPO and polymerization state of PMMA) determine the optimal content of BPO and DMT is 1.0 mol%–0.5 mol%. These results might be helpful for the optimization of the DMT/BPO ratio for a high-quality polymerization of MMA.
Footnotes
Author’s note
The authors claim that none of the material in the paper has been published or is under consideration for publication elsewhere.
Author contributions
All authors have contributed to the creation of this manuscript for important intellectual content and read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank National Key Laboratory of Advanced Composite Materials for financial funding.
