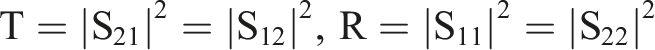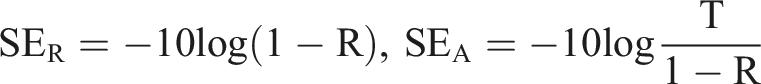Abstract
Despite the significant progress made in polymer/graphene nanocomposites, the development of high-performance polymer/graphene nanocomposites, combined with excellent mechanical and electromagnetic shielding properties, remains a priority and a challenge. The lanthanum doping large-size graphene oxide (La-LsG) was obtained via static oxidation and thermal treatment. The effects of static oxidation and lanthanum doping on the property and morphologies of graphene and composites were investigated. The physicochemical property of considerable graphene oxide (GO), large-size graphene oxide (LsGO), lanthanum-doped graphene oxide (La-GO), La-LsG was analyzed by transmission electron microscopy (TEM), scanning electron microscopy (SEM), Raman spectroscopy, and X-ray energy spectroscopy (EDS). SEM and EDS characterized the microstructure of the composite material, and the mechanical properties and electromagnetic shielding performance of the composite material were studied. La-GO and La-LsG exhibited good dispersion and structural integrity, as well as enhancing the strength of the composites. The La-LsG/poly (methyl methacrylate) (PMMA) composites exhibited excellent mechanical properties (is ca. 73.6 MPa) and electromagnetic shielding effectiveness (is ca. 28 dB) in X-band frequencies (8.2–12.4 GHz), which may be assumed to be a promising integrated material of structure and function.
Keywords
Introduction
In the present day, various types of electronic and electrical equipment are being used more extensively by the rapid advancements in science and technology. There is an increasing concern that the 1–20 GHz range of electromagnetic interference and electromagnetic radiation is becoming a major issue in society as a whole.1–3 A great deal of research has been done on electromagnetic shielding materials in order to effectively control harmful electromagnetic interference.4–9 In general, low-resistance conductor materials are used to shield electromagnetic waves by reflecting and absorbing electromagnetic waves on the material's surface and by absorbing loss during transmission. 10
At present, in terms of electromagnetic shielding materials, although metal materials have good electrical conductivity and electromagnetic wave reflection ability, metal materials have high density, difficult processing, easy corrosion, and poor mechanical properties. Therefore, nanocomposites that are easy to process, have good mechanical properties, and are light-weight have attracted the attention of a large number of researchers. Graphene,11–13 carbon nanotubes,14,15 rare metal,
16
carbon foam, etc., have been proved to be filled as nanofiller in the polymer to prepare nanocomposite materials. Among the reinforcements of composite materials, graphene, a monolayer 2D planar carbon material, has attracted much attention due to its unique electron transport, excellent thermal conductivity, and excellent mechanical properties.17–19 In the meantime, many potential applications have been explored, such as electromagnetic scattering and reinforcing agents in nanocomposites.20–25 By adding nanofiller, the mechanical properties of composites are degraded.26–32 As electromagnetic shielding material, it does not need to be a high-strength material. However, some particular application still requires high strength capability and electromagnetic shielding capability in one material, such as the aerospace industry.20,26 Electromagnetic shielding is the process of stopping electromagnetic waves from propagating through a transmission path. The shielding effectiveness (SE) measures the degree of attenuation of electromagnetic waves. SET, SER, SEA, and SEM indicate total, reflection, absorption, and multiple internal reflection effectiveness, respectively, and are measured in dB. Generally, SEM is often neglected when SER ≥ 10 dB, so the shielding effectiveness is given by
In addition, the types of shielding materials are determined by the S parameters (S11, S21, S22, S12) obtained by the vector network analyzer. According to these S parameters, the reflectance rate (R), and transmittance rate (T) can be calculated in the equation.
27
And the shielding effectiveness and scattering parameters (S parameters) relation can be described as
At present, various methods have been used to prepare or modify graphene.28,29 Due to the agglomeration and stacking created by uneven dispersion, graphene-reinforced composites have difficulty exerting their high mechanical properties and conductive properties as nanofillers in composites. 30 Although graphene oxide can improve its compatibility in the polymer matrix, the structure of graphene itself is destroyed and its performance cannot be well exerted. Therefore, it is necessary to modify the graphene structure to meet the specific application requirements of composite materials. Rare earth elements have attracted extensive attention because of their special electromagnetic properties. Using doping, it is possible to alter the electromagnetic properties of carbon materials in an anisotropic way, thereby improving the material's electromagnetic shielding and wave absorption properties.31,32 Among them, lanthanide metals are paramagnetic and active, and can react with water. At high temperature, they can also react with non-metals such as C and O. At the same time, high temperature can also reduce some oxygen-containing functional groups in graphene oxide, thereby improving the integrity of graphene. So, using lanthanum doping in graphene oxide to enhance its electromagnetic shielding properties and mechanical properties in a polymer matrix is possible.
In this work, we used a mild static oxidation method to prepare Ls-GO with a more complete graphene sheet structure. The Ls-GO and GO are immersed in a lanthanum nitrate solution, followed by thermal reduction to form La-LsG and La-GO. The different properties and morphologies of graphite, GO, Ls-GO and La-LsG were compared. Then, the nanocomposite materials with PMMA as the polymer matrix were prepared by in-situ polymerization, and the prepared graphene nanofillers were added to the matrix through a special bonding process, and different nanocomposite materials were prepared respectively. Subsequently, the electromagnetic shielding properties and mechanical properties of different nanocomposite materials were tested and analyzed. Finally, the effects of nanofillers and preparation methods on the structure and properties of composites are discussed.
Experimental
Raw materials
Our works have studied the effect of lanthanum doping on large-size graphene sheets prepared by the modified Hummers method and thermal treatment. The commercial natural graphite flake was provided by Sinopharm Chemical Reagent Co. Ltd. (Shanghai, China) with a primary size of 30 μm, and the purity is greater than 95%. Sulfuric acid (H2SO4, 98%), potassium permanganate (KMnO4, 98%), hydrogen peroxide (H2O2, 30%), lanthanum nitrate (La(NO3)3, 99%), benzoyl peroxide (BPO, 99%), and methyl methacrylate (MMA, 99%) were procured from Hongyan Chemical Reagent Co. (Tianjin, China) and used.
Preparation of graphene oxide (GO) and large-size graphene oxide (Ls-GO)
In this paper, the preparation method of Ls-GO was based on the works we have reported 33 and inspired by the preparation method of ultra-large graphene oxide of Hongbin Lu 34 with some modifications. Before the reaction, the water between layers of graphite flake was removed by concentrated sulfuric acid (100 mL, 98%), and then the sulfuric acid was removed by filtration.
1 g graphite and 1 g sodium nitrate were dispersed respectively in sulfuric acid (50 mL, 98%) in a round bottom flask. Subsequently, 5 g potassium permanganate is slowly added to the mixture in batches to avoid the risk of explosion. Throughout the addition process, the system temperature was kept at 0°C or so. Continue to stir slowly for 2 h to ensure a uniform system. The hybrid system was kept at 35–40°C for 24 h without any mechanical agitation (static conditions). After the reaction, the mixture was poured into ice water (500 mL), and 10 mL of hydrogen peroxide was used to decompose the insoluble manganese salts. The reaction mixture was purified via repeated cleaning, high-speed centrifugation, and freeze-drying to obtain LsGO, the experimental process is shown in Figure 1. Schematic illustration for the preparation of Ls-GO.
Preparation of lanthanum-doped graphene oxide (La-GO) and lanthanum-doped large-size graphene oxide (La-LsG)
The La-GO and La-LsG was prepared via a thermal method. To recapitulate briefly, GO (0.05 g) and LsGO (0.05 g) was added to a saturated lanthanum nitrate solution respectively and stirred evenly to form a suspended solution. Then the impregnated powder was obtained by filtering and drying. After that, the resulting powder was heated to 450°C in Ar ambiance for 6 h, and then maintained in H2 ambiance for 6 h. Finally, the La-GO and La-LsG powder is obtained by cooling.
Preparation of polymer matrix composites
To obtain uniformly dispersed polymer matrix composites, the polymer monomer (MMA) was blended with the reinforcement (2 wt.%) at room temperature and treated by stirring and ultrasonic for 2 h. Then the system was initiated by BPO to form a preliminary composite system. BPO as a trigger to add to the mixing solution in a heated water bath of 80°C. Then, the prepolymer in N2 heating temperature of 260°C, in the autoclave to form a composite material. Graphene, GO, La-GO, and La-LsG are four reinforcement materials. Here, GO is prepared by using the modified Hummer's method, while graphene is prepared by low temperature hot pressing reduction. The preparation method was described in detail in our previous work. 35
Characterization of materials
Transmission electron microscopy (TEM) was used to investigate the produced graphene oxide, LsGO, and La-LsG. A Tecnai G2 F30 FEI transmission electron microscope was used to analyze the samples. The morphologies of cross-sectional surfaces of the pieces were observed using a JSM-6700, JEOL Field-emission scanning electron microscope (SEM). X-ray diffractometer (X' Pert PROMPD, PANalytal) using Cu K radiation (=0.15406 nm) was employed to analyze the structure of samples. Raman spectroscopy was performed on a Renishaw in via Laser-Raman Spectrometer using a 532 nm laser. Moreover, the bending strength test was performed on a universal testing machine (Instron 3382) at room temperature, following the national standard of the People's Republic of China (GB/T 9341-2008) for the determination of the bending properties of plastics. These samples (80 mm long, 10 mm wide, 4 mm high) is loaded at the center point on a stainless steel platen with an indenter speed of 2 mm/min. And the flexural strength (fM) was calculated by
In addition, an Agilent technologies E8362 B network analyzer measured the electromagnetic shielding SE, and the sample size is 22.86 × 10.16 × 2 mm Figure 2. Schematic diagram of the synthesis route of La-LsG/PMMA composites.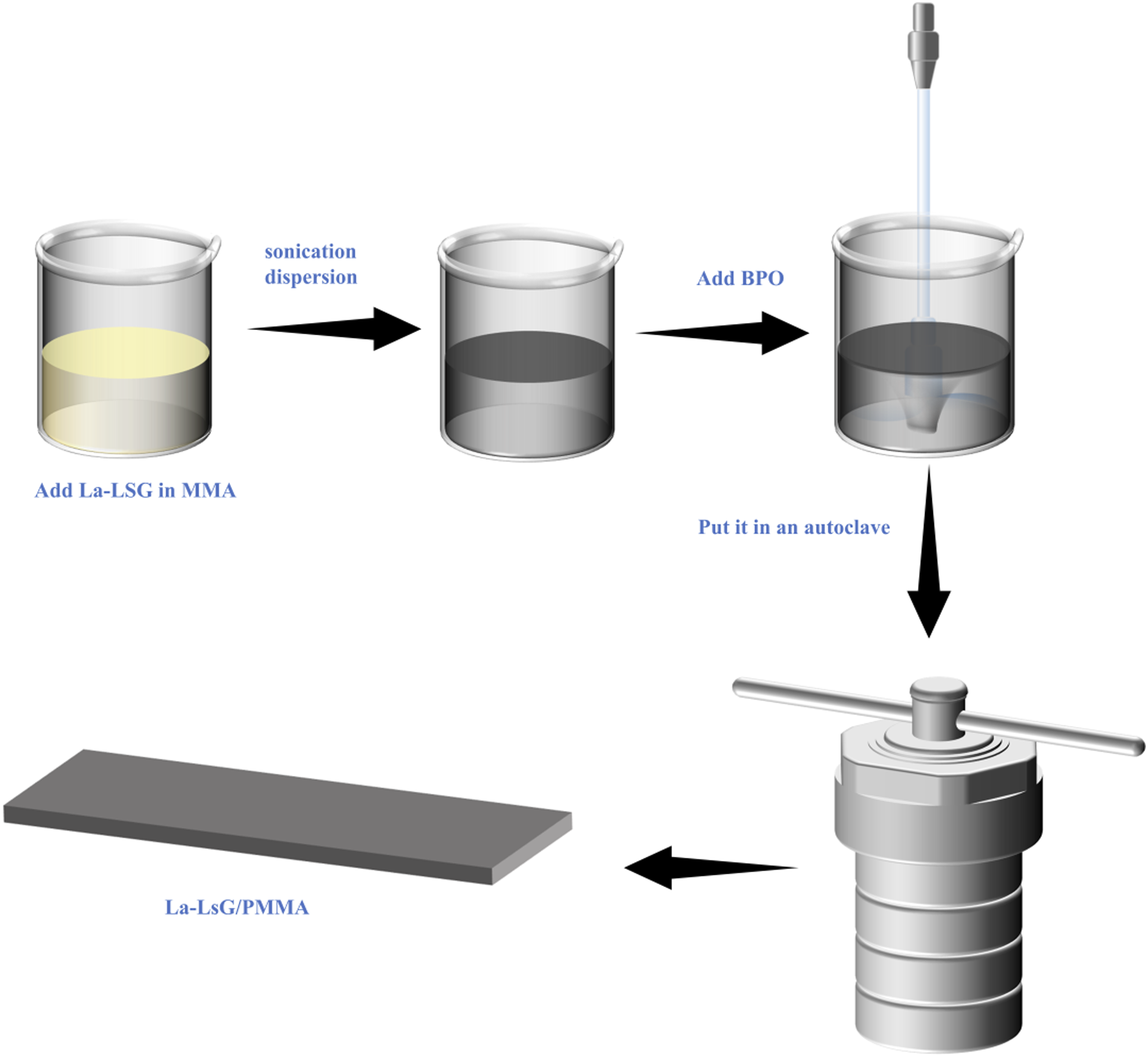
Results and discussion
The structure of graphene oxide and lanthanum-doped graphene oxide
During the oxidation process, graphite's structure was altered dramatically. Oxygen-containing functional groups are embedded between graphite layers, combining with other functional groups or elements as an active group. With the continuous action of many oxidants, the graphene layer also began to show a trend of fragmentation with the increase in layer spacing (Figure 3(a)). And these small layers were not only spread out but also tightly stacked. Accompanied by intense agitation at high temperatures, excessive oxidants attacked the graphite sheets indiscriminately. These led to more breakages and defects in the generated GO and were more likely to be further oxidized and destroyed by induced effects under severe external conditions.36,37 As these breakages continued to tear, the GO fragments became more fragmented and stacked on top of each other. Thus, La-GO also exhibited the characteristics of easy aggregation, as shown in Figure 3(b). TEM of GO (a), La-GO (b), LsGO (c), and La-LsG (d, e), respectively. SEM (f) and the EDS analysis (g) of La-LsG. XRD (h) and Raman spectra (i) of samples.
Nevertheless, the LsGO had relatively noticeable improvement in dimensional integrity compared with the traditional oxidation method and was continuously dispersed, similar to cloud shape as shown in Figure 3(c). The relatively moderate static reaction conditions were more conducive to forming a sizeable overall layer, which depended on the synergistic effect of multiple effects. Compared with SP2 of C atoms in the crystal, those atoms (breakages or defects) located at the outer edge of the crystal were more vulnerable to oxidizer attacks due to their higher reactivity. 36 Especially with the help of heating and shear stress produced by stirring, all this becomes more intense. This indicated that the oxidizer tends to act more uniformly on the whole graphite layer over time when the reaction conditions are relaxed. This phenomenon is due to the combined action of swelling and defect adsorption. Moreover, the ductility and integrity of La-LsG are much higher than La-GO, as shown in Figure 3(b), (d), and (e). As shown in Figures 3(g) and (h), the lanthanum effectively doped LsGO with the active group's assistance, and the rest of the functional group could prompt La-LsG to disperse effectively in the system. These results were the same as EDS and XRD analysis results.
The Raman spectra of samples are shown in Figure 3(i). Two distinct peaks could be observed at ca. 1350 cm-1 (D peak) and 1590 cm-1 (G-peak), respectively. The D-peak described the disorder and defect degree of carbon materials, and the G-peak is the characteristic peak of C atoms due to in-plane vibration at the graphite phase. As graphite-derived materials, the C atom has prominent distinct peaks on several curves, and the intensity is the same. However, the intensity of D-peak showed different defects and disorders with oxidation degree and lanthanum doping. The ratio of D-peak strength to G-peak strength (Id/Ig, R) was calculated to study and measure the variation of samples disorder during oxidation and doping. With the process-taking place, the oxidation of graphene always occurred preferentially at the edge, grain boundary, or defect site of graphene. Under the action of shear stress (as the starting point), the tiny graphene sheets (breakages or defects) were continuously ripped away from the central part of the system with the heating and continuous intense stirring. Thus, the defect was further aggravated, the R of 0.9268. This trend was further amplified after doping because the lanthanum doping makes some SP2 carbon atoms transform to SP3, the R of 1.0259. In contrast, the trend had specific changes in La-lsG. In the static oxidation system, the graphite sheets were oxidized and continuously swelled by solution without shear stress, so the tendency of miniaturized graphene was inhibited, and the R of La-LsG is 0.9181, as opposed to that of those traditional oxidation and doping processes. This result is consistent with other test results, as shown in Figure 3(b)–(e) and (h).
The effects of La-LsG on the structure and properties of polymer matrix composites
Figure 4 shows SEM of composite's cross-sectional surfaces. Due to the excellent compatibility and interfacial binding interactions between GO and matrix. Compared with the traditional oxidation and doping method, the crazing of La-LsG/PMMA uniformly dispersed on the cross-sectional surfaces of composites. In addition, it is also observed in Figure 4(b) that the La-LsG was pulled out of the matrix under the action of external forces, the size is ca. 50 μm. According to the theory of hole(38), when the separation happened between the nanofiller with polymer matrix under external force, resulting in the formation of crazing or cavities. It is indicated that distribution patterns of La-LsG can be homo-disperse, because of the well dispersion and interfacial binding interactions between La-LsG with polymer. SEM images of composites: La-GO/PMMA (a), La-LsG/PMMA (b) (c), EDS of La-LsG/PMMA (d).
Figure 5 shows the flexural stress curves of the composites. As the matrix material, PMMA shows the flexural strength (σfM) of ca. 59.6 MPa as shown in curve a. It is evident that the flexural strength increased to ca. 62.1 MPa after the filling of graphene oxide. This shows that the functional groups can provide compatibility and effectively interfacial binding interactions with matrix. When the composite is bent by external force, the flexural strength of the composite can be enhanced due to the higher elastic modulus of the matrix of GO. At the same time, because GO forms stress points in the matrix, under the action of external force, GO/PMMA is easier to fracture than PMMA, resulting in a slight decrease in the flexural modulus. Flexural stress curves of the samples. a: PMMA b: GO/PMMA c: La-GO/PMMA d: La-LsG/PMMA.
After the thermal treatment process of lanthanum doping, the oxygen-containing groups are removed and changed to CO2 gas from GO. Thus, the defects and disorder degree of composites are declined, and its primary performance in the flexural strength of La-GO/PMMA increase to ca. 68.5 MPa as shown in curve c.
Flexural property data of PMMA, GO/PMMA, La-GO/PMMA and La-LsG/PMMA.
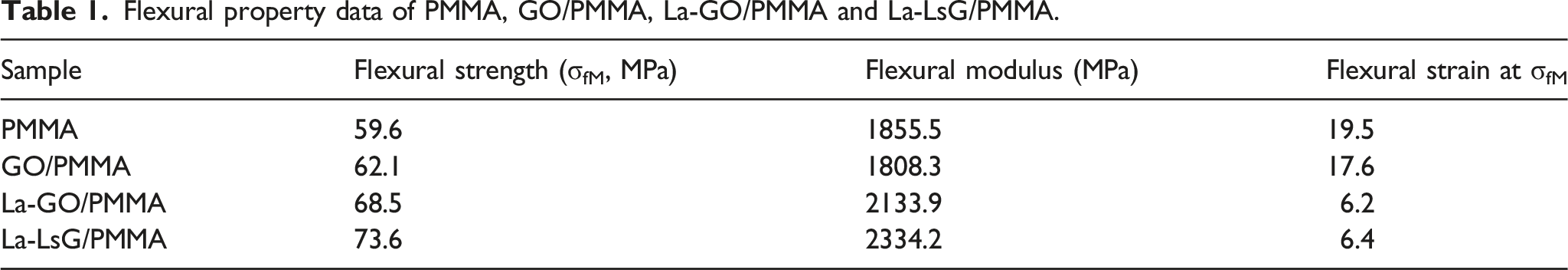
Figure 6 shows the variation of electromagnetic shielding effectiveness of materials in the X-band (8.2–12.4 GHz). The materials used in the measurement are different fillers added in the same amount. As a matrix and pure polymer, PMMA (a) exhibits extremely low shielding properties. Similarly, the GO/PMMA (b) also exhibit lower shielding effectiveness, which is because the conjugated network structure of graphene is heavily functionalized, resulting in more defects, making the graphene sheets with insulating properties, and its inherent low conductivity and large number of defects limit electromagnetic shielding effectiveness. When GO is thermally reduced to graphene, graphene has a more complete lamellar structure, which can form a relatively complete conductive network in the polymer matrix, and its electromagnetic shielding efficiency can be significantly increased, and the peak value can reach 23 dB, as shown by the curve d. In addition, curves c,e demonstrate the electromagnetic shielding performance of the two lanthanum-doped composites. Compared with graphene/PMMA, the electromagnetic shielding effectiveness of La-GO and La-LsG is reduced due to the destruction of lanthanum doping. Due to the doping of lanthanum, graphene becomes damaged, and the lamellar structure is weakened, which makes constructing a conductive strip more difficult, thus reducing the reflective properties of graphene. It is worth noting that both curves c and e have peaks at high frequencies and the shielding effect of La-LsG/PMMA is better than that of La-GO/PMMA. As shown in Figure 6, the curves c, e exhibits a peak near 12.2 GHz, and the peak can reach 28 dB. There are two reasons for this phenomenon. First, compared with La-GO, La-LsG has larger size and more complete structure, which can effectively suppress the generation of defects. Therefore, the conductive network formed by La-LsG is relatively complete and has better electromagnetic shielding effect. Second, lanthanides are paramagnetic and can absorb electromagnetic waves, especially high-frequency electromagnetic waves. At the same time, lanthanides are also reactive and react with non-metals such as C and O. The diamagnetism of lanthanum itself increases the magnetic anisotropy of the material, thereby increasing the tape loss of the material, and finally improving the absorption shielding effectiveness of the material. Electromagnetic shielding effectiveness of samples. a: PMMA b: GO/PMMA c: La-GO/PMMA d: graphene/PMMA e: La-LsG/PMMA.
Conclusions
In this study, La-LsG was fabricated by static oxidation and thermal treatment, which provides better dimensional integrity and a lower level of defect than GO and La-GO. Both La-GO and La-LsG demonstrated excellent dispersion and integrity, and effectively reinforced the composites. The La-LsG/PMMA composite exhibits excellent mechanical properties (σfM is ca. 73.6 MPa) and electromagnetic shielding effectiveness up to 28 dB in the X-band (8.2–12.4 GHz) frequency range, which makes it a promising material for structural integration.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shaanxi Province (No. 2019JQ847 and 2021JM451), the Special Scientific Research Program Funded of Shaanxi Provincial Education Department (No. 19JK0368), and the PhD Start-up Fund of XPU.