Abstract
The objective of the present study is to formulate added value polymer granite composite (PGC) based on granite (G) waste scraps as filler and unsaturated polyester (UP) obtained from chemically recycled post-consumed poly (ethyleneterephthalate) wastes (PET) as a matrix to be used in different candidate applications. Both wastes were collected from local dumping sites. Factors affecting the properties of the acquired PGC products e.g. polymer constitutions, granite/styrenated polyester ratios were studied systematically. The reached polymer granite composite has many advantages e.g. low density, very small water absorption percentage, short curing and hardening time, acceptable compressive strength values and significant thermal stability. Moreover, the environmental reward due to the utilisation of accumulation of the two problematic solid wastes is certainly a gain.

Keywords
Introduction
According to the literature, Egypt is ranked among the top eight world producers of natural stone sectors e.g. marble and granite. Egypt’s quarry production was nearly 3.2 million tons comprising over 25 different types of Egyptian marble and granite. The contribution of the natural stone industry to the Egyptian economy has grown very fast over the last three decades, and about 70% of those activities are found in Shaq Al-Thu`ban area, east western near Cairo. This industrial cluster possesses big hazard to residents in near communities lying nearby this area. 1
Due to defects and fractures in the granite rock mass, only about 20–30% and rarely more than 50% of the granite quarried can be marked as commercial blocks. Quarrying waste includes poor quality stone, unshaped lumps, and finger fragments that are disposed in the dump found nearby, which causes severe impacts to the environment and health of humans. In addition, about 60% of the volume of original blocks is wasted due to sawing, cutting and finishing operations. Those wastes are disposed in landfills after the tailing ponds, and accounted for ecosystem damages. 2
Also, another fatal problem which is encountered as one of the most disadvantageous results of anthropogenic activities is the accumulation of plastic waste. More than 400 various types of domestic and industrial plastic wastes are known and many of those are specified as mixed plastic waste, which is not recycling friendly. 3
Poly (ethyleneterephthalate) (PET) is non-biodegradable thermoplastic polyester. In the purest form PET is an amorphous glass like material, which is characterized by appropriate chemical resistance, and an acceptable thermal stability. 4
Poly (ethyleneterephthalate) is mainly applied into many products; the most important one is water and soft-drink bottles. In 2016, about 485 billion bottles PET were produced, and it is forecast that in 2021, 583.3 billion of these plastic bottles will be produced. 5
The increased consumption of PET leads to serious ecological problems as rapid waste stream accumulates yearly due to its short life duration. Disposing of this waste to the landfill is becoming undesirable due to the legislation pressures, rising costs and poor biodegradability of such polymers. Therefore, other ways for PET waste treatment is highly desired such as recycling.6–9
Post consumed PET waste is recycled by different techniques and can be reused in various fields. This waste is a potential source for the production of new chemical reagents such as unsaturated polyester (UP) resins, polyurethane foams...etc.7,8,10
Mechanical and chemical recycling are the most widely applied methods all over the world for processing PET wastes.11–13
Poly (ethyleneterephthalate) wastes are chemically recycled either by complete depolymerization into monomers or through partial depolymerization into low molecular weight products and/or oligomers. The monomers can be re-polymerized and the generated recycled polymer can be applied for new articles production.7,8,14
Aminolysis and glycolysis are the most common applied techniques for chemical processing of PET wastes.15–22
Very few or may be none published works discussed formulation of polymer granite composite (PGC) based on polyester obtained from chemical recycled the post consumer PET waste, as a matrix and granite scraps, as an inorganic filler. Paul et al. 23 used the virgin polyester resin incorporating particulate ceramic granite to prepare composite while Piratelli-Filho and Shimabukuro 24 applied an epoxy polymeric matrix reinforced with particulate ceramic granite. Ramakrishna and Rai, also, produced polymer composite from granite scraps and virgin poly (butyleneterepthalate) toughened epoxy resin. 25
This study presented a method for a newly developed polymer-granite composite formulation based on granite waste scraps and post-consumer PET waste bottles. The UP, based on glycolyzed product of PET, was used after mixing with styrene as a matrix for the ground granite powder filler. For evaluating the performance of the generated polymeric granite composite in different candidate applications, various parameters that can affect the physical, thermal and mechanical properties of PGC were studied systematically.
Methodology and preliminary characterization of constituents
Granite powder
Granite cutting scraps were collected from local markets containing mud and debris. Those scraps were cleaned with water then dried under sun with precautions. The dried scraps were ground in a high speed mortar mill for 10 min for every 50–60 g of sample, then sieved to separate the different particle sizes and the lowest particle size powder (mesh size ≈100-150 micron) was used in the present work, assuming that the finest particle size ensures better mechanical properties. The ground sieved granite powder was dried again in an oven at 60°C for 24 h then subjected to X-Ray Fluorescence (XRF) analysis.
X-Ray fluorescence analysis
The main oxides (mass, %) in the granite powder based on X-RF analysis.

X-Ray diffraction
X-Ray powder diffraction (XRD) patterns were obtained at room temperature using a Philips diffractometer (Model PW 1390) Japan, employing Ni-filtered Cu Kα radiation (λ = 1.5404 A°). The diffraction angle (2θ) was scanned at a rate of 2°/min. Based on XRD the crystal architecture of G powder comprises mainly; Quartz (SiO2), Anorthite (combination of: Na2O, CaO, Al2O3 & SiO2), Microcline (K2O.Al2O3. 3SiO2) in addition to Albeit (due to the presence of CaO); Anorthite (oxides of Na; Ca; Al; & Si) and Biotite where Fe2O3 is its main constituent. The great stiffness of granite stone is attributed to its high quartz content, moreover the great proportions of silica and alumina in G are coexisting with the abundant aluminosilicate phases in the XRD pattern, Figure 1.
27
X-Ray Diffraction (X-RD) of granite waste powder.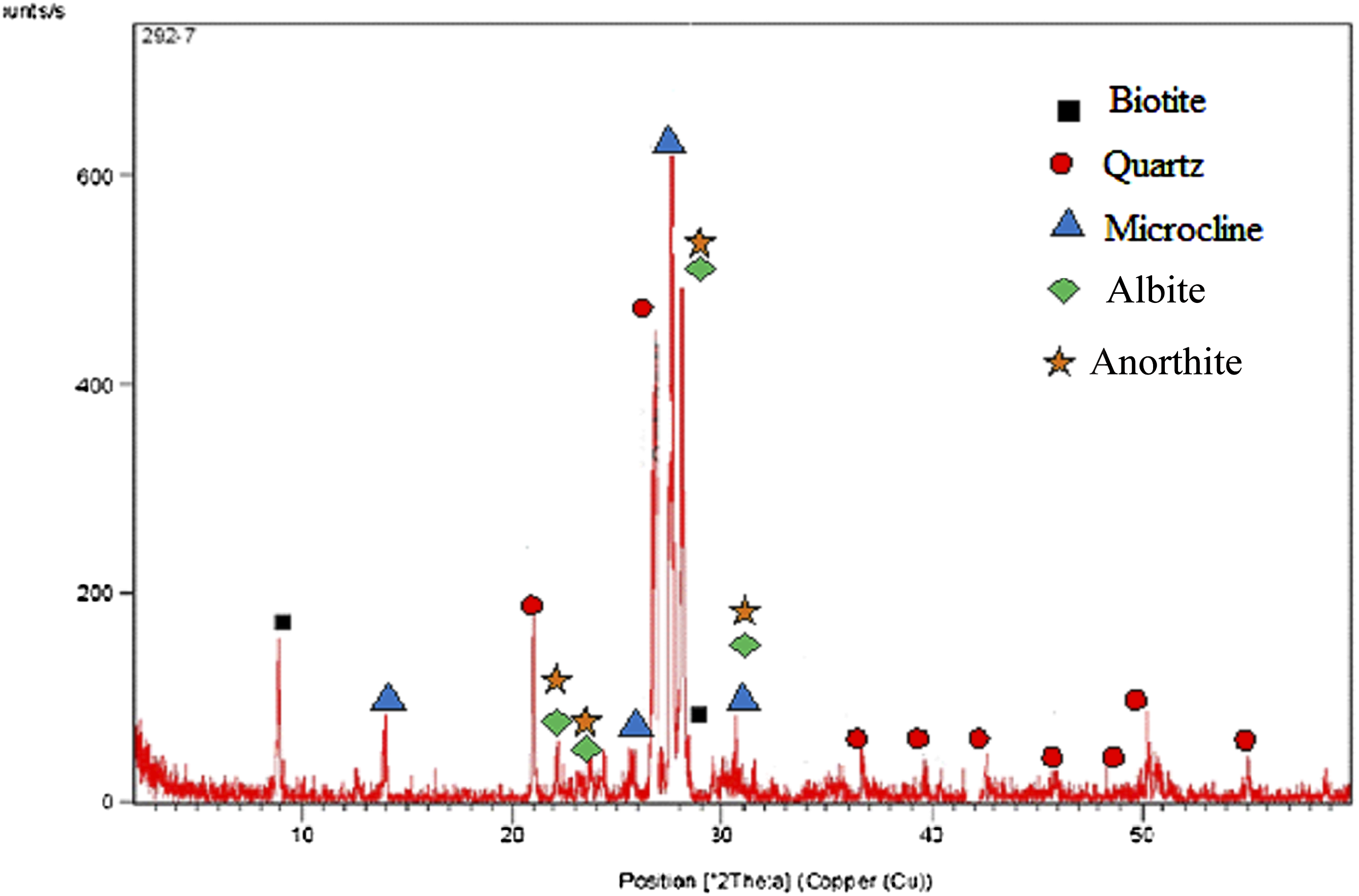
Fourier transform-infrared examination
Alongside to XRD analysis, the Fourier Transform-Infrared (FT-IR) examination for G powder was performed at room temperature. The FT-IR spectra of the G, the prepared UP and the formulated PGC were performed using a JASCO FTIR-6100 E, Fourier Transform Infrared Spectrometer (Japan). The acquired data of G is represented in Figure 2, and the spectrograph is characterized by group of peaks translating the configurations analyzed through X-RD. Fourier Transform-Infrared examination of granite powder (G).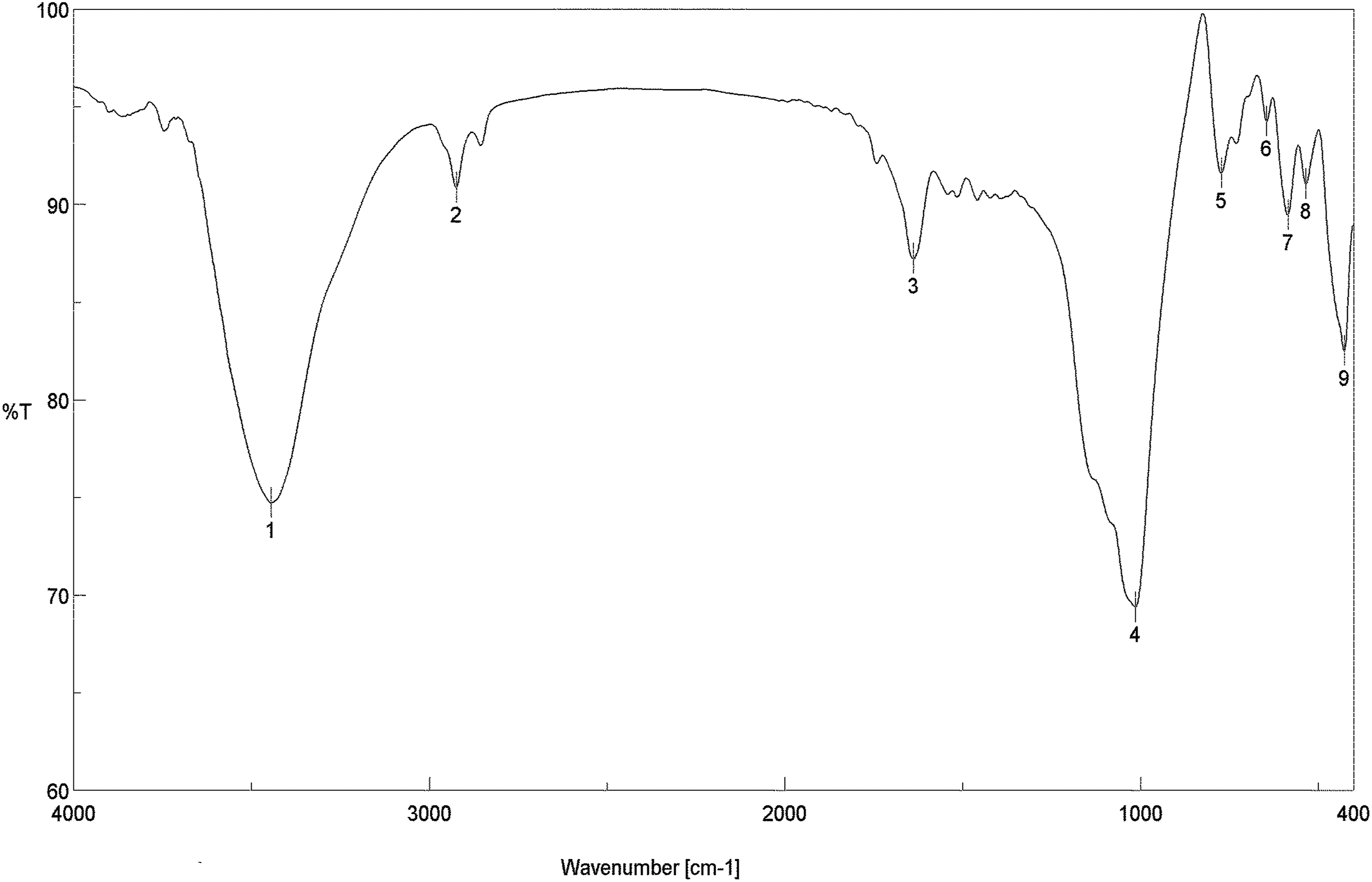
The most characteristic peaks can be detailed as follows: broad strong peak centered at 3445 cm−1 which can refer to O-H stretching vibration in Al-OH and Si-OH. The broad strong peak centered at 1014 cm−1 can be assigned to oxygen bridge in Si-O-Si asymmetric vibration and also to non-oxygen bridge Si-O symmetric vibration. Peaks near 720 cm−1 can be attributed to Al-O symmetric vibration, while the peak at 645 cm−1 can be an indication to quartz fragment in the granite powder. The two sharp peaks at 585 cm−1 and near 530 cm−1 are indicative to the iron oxide.
Poly (ethyleneterephthalate) waste bottles
Clear postconsumer soft drink water PET bottles were collected from a nearby dump site. They were first cleaned thoroughly by conditioning in 1% aqueous solution of sodium hydroxide for about 1 h to remove any surface impurities and then washed with distilled water many times. The clean PET waste bottles were chopped into small flakes (∼5 × 5 mm2), oven dried at 80°C, then subjected to glycolysis degradation treatment.
Glycolysis of PET wastes
Glycolysis process of PET wastes is based on recycling the shredded PET flakes with diethylene glycol/propylene glycol mixture at the ratio 1:0.5:0.5 (by masses) and dibutyl tin oxide (DBT) as a trans-esterification catalyst. Dibutyl tin oxide was added at 0.5% relative to PET mass. The mixture was charged into a four necks round-bottom flask that is fitted with stirrer, reflux condenser, nitrogen inlet, and thermometer. The flask was immersed in an oil bath and the temperature was raised to 200°C under reflux for 8 h under a nitrogen atmosphere. At the end of the reaction period (8 h), the mixture was left to cool at ambient temperature. Depolymerization process of PET was followed by determining the hydroxyl value in reaction mixture according to the method described previously. 28 The glycolyzed product (GP) recorded hydroxyl number ≈647.3 mg KOH/g.
Formulation of unsaturated polyester resin (UP)
The UP resin was acquired from the obtained GP hydroxy-terminated oligomers at a ratio of 1.1/1 (mol/mol) hydroxyl/carboxyl. The hydroxyl number of the glycolyzed products was used to determine the amount of acid required. The GP oligomers, end product, were charged in a reaction vessel fed with both maleic anhydride (MA) and adipic acid (AA) in a molar ratio of GP: MA: AA as 1.1: 0.7: 0.3. The mixture was heated gradually from 80oC up to 140oC for an hour (h). Then the temperature of the mixture was elevated at a rate of 10oC/h until reaching 210oC. A Dean–Stark trap was applied for monitoring the amount of water removed throughout the reaction. The total reaction time was persisted for about 8 hours and left to continue until reaching the determined acid number, then the acid number was determined by titration of a sample solution in chloroform with standard 0.1 N alcoholic KOH (ASTM-D 3644).
Fourier Transform-Infrared spectrum of the GP and its UP
Fourier Transform-Infrared spectrum of GP is shown in Figure 3, showing a strong broad band between 3200 and 3600 cm−1 which is characteristic for the stretching frequency of the OH group. The sharp band at 1680-1740 cm−1 is due to the stretching frequency of the ester carbonyl group, while the bands at 2880, 2940, 2960 cm−1 may stand for the stretching frequency of the CH, CH2 and CH3 groups, respectively. The band at 1610 cm−1 is characteristic of the aromatic C=C stretching, and bands at 730 and 880 cm−1 are due to the bending frequency of aromatic CH. Figure 3 represents the FT-IR spectrum of the UP based on glycolysis of PET. It is quite similar to glycoside product showing similar bands characteristic of the stretching frequencies of the ester carbonyl and hydroxyl groups. The band at 1640 cm−1 is characteristic for HC=CH indicating the participation of MA in the polymer chain. The bending frequencies at 775, 745, 705, and 670 cm−1 strongly suggest the presence of cis olefinic and aromatic residues in the polyester resin. The band at 980 cm−1 is characteristic for the trans-olefinic C-H bending, and can be considered as an indication that an isomerization of maleic acid to fumaric acid took place during the polymerization process.
29
Fourier Transform-Infrared of glycolysis products (GP) and unsaturated polyester (UP).
Nuclear magnetic resonance of the UP
Nuclear magnetic resonance analysis for the UP based on glycoside PET was performed using a Mercury-300BB spectrometer in presence of dimethyl sulfoxide as solvent and the data obtained is represented in Figure 4. From the spectrum, it can be addressed that the peak near 8.04ppm can be accounted for the four phenyl protons of terpehthalic acid (4H, C6H4). Asymmetrical doublets, appeared in the vicinity of 6.75–6.74 ppm that can be referred to the olefinic protons in the CO-CH=CH=CO chromospheres of maleic and fumaric acid residues. Peaks disclosed at 4.15–4.42ppm are due to methane proton of PG residue. A group of signals between 3.7-4.7ppm is most attributed to the methylene proton in DEG. Peaks located at 3.38–4.13ppm indicated the proton in -O-CH2-CH2-O & -O-C-CH2-O fragments in the UP. Two sharp signals at 2.28 and 2.4 ppm are characteristic for the methylene protons in adipic acid residue, while peaks at 1.06–1.50ppm described methyl groups in propylene glycol residue. Nuclear magnetic resonance spectrum of UP based on glycolysis of PET waste. PET: Poly (ethyleneterephthalate); UP: unsaturated polyester.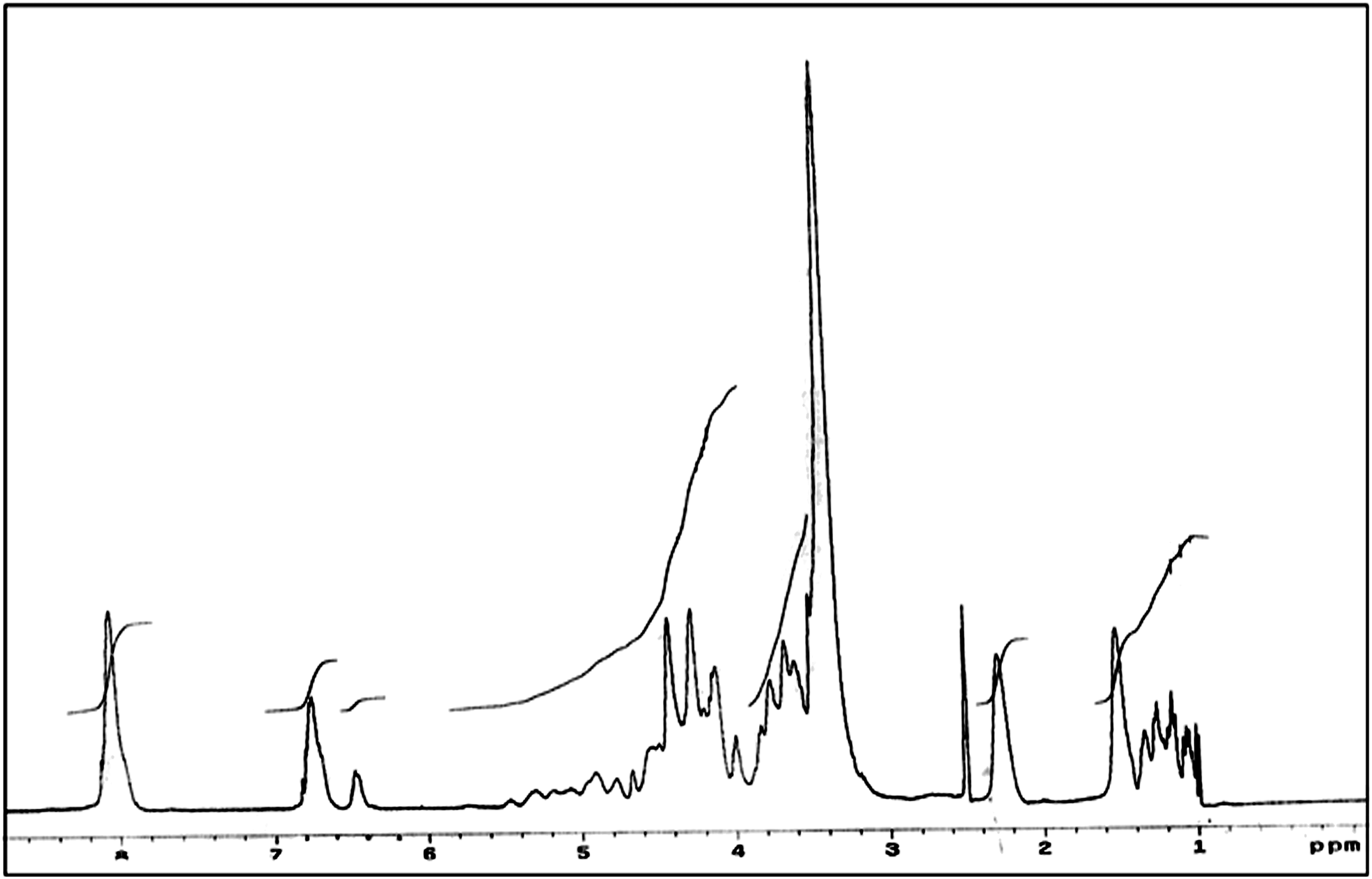
Polymer-granite composites formulation
Polymer-granite composites was acquired through mixing granite powder waste (as filler) with the prepared polyester/styrene mixture as a matrix. The reached UP was, at first, mixed thoroughly with styrene, methyl ethyl ketone peroxide (MEKP) as initiator, and cobalt naphthenate (Co) as accelerator. The initiator and accelerator were added to the resin immediately prior to granite powder addition. The acquired composite mixtures were then casted in the stainless steel moulds with specific dimensions required for testing and characterization. The casting process was performed at ambient temperature and atmospheric pressure. The PGC samples were left for curing and hardening for 7 days to assure complete curing. The hard specimens were demoulded and subjected to different characterizations, measurements and evaluations.
X-Ray diffraction of polymer granite composite
The X-Ray Diffractograph for the PGC’s crystalline architecture is represented in Figure 5. It is clear that the composite showed nearly peaks position similar to that of granite powder. The only differences are the detection of peaks at 2θ = 29.5o; 43o; 38.5o and 39.5o, which are related to calcite (CaCO3) and can be attributed to the reaction of CaO in granite with CO2 during processing of the product. X-Ray diffractogram of polymer granite composite.
Fourier Transform-Infrared examination for PGC
Fourier Transform-Infrared spectroscopy was carried out to complete the characterization of the polymer granite composite under consideration. According to FT-IR analysis, the following most abundant mineral components were detected; quartz (bands at the vicinities 1180 cm−1, 1081 cm−1, characteristic doublet 799 cm−1 and 778 cm−1) and Biotite (near 1005 cm−1) Figure 6. These in addition to the peaks representing the organic moiety of the composite, i.e. the polymer including peaks near 1640 cm−1 (olefinic double bonds), in the range 775cm−1-650 cm−1 (cis olefinic and aromatic residues in the polyester resin). Fourier Transform-Infrared for polymer granite composite.
Compressive strength measurements for PGC
The compressive strength of the cured composite samples PGC were conducted according to ASTM D 695 on three to five cubical replicated samples with dimensions 3.0x3.0x3.0 cm3 using computer-controlled, hydraulic-servo, Shimadzu Universal Testing Machine, UH-I Series 1000 KN Model.
Results & discussion
In the present work an innovative formulation of polymer granite composite was obtained from incorporating ground granite scraps waste powder, as inorganic filler, into a styrenated-polyester resin based on glycolysis of PET waste as matrix. In addition to the considerable environmental reward of recycling PET & G wastes as an industry, its driving force is directed toward decreasing the elevated cost through decreasing the use of virgin PET as raw materials. Also, beside the ecology profits due to use and incorporation of inorganic waste filler; it produces long-span engineering products.
Physical characterizations of PGC
The most interesting physical properties of polymer granite composite (PGC)*.

*PGC was formulated of styrenated-polyester (SP) at the ratio (P: S 65/35 mass: mass) and granite waste powder as filling material at 75% mass relative to SP matrix.
Moreover, the distinguished low porosity of PGC reflects the acceptable high resistance to chloride ion and oxygen penetration that significantly reduced reinforcement product corrosion. Those two characters besides the acceptable others ones viz: exterior volume, volume of impervious portion, volume of open pores, apparent porosity, water absorption, apparent specific gravity and bulk density are essential to candidate the composite under consideration for some exterior applications. Therefore PGC, in addition to its various mentioned applications, can be properly used as anticorrosive linings of effluent drains, pavements, bridges, chemical plant floors and septic tanks... etc.
Mechanical characterization
Impact of various granite waste powder: styrenated polyester* ratios (mass to mass) on PGC compressive strength values.

*based on glycolysis product of PET.
**P: S kept at 65:35% mass to mass ratios.
Increasing the added granite filler powder beyond the ratio of 75 mass % is accompanied with great diminish in the compression integrity of the formulated composite. The decrease in the compressive strength values as the granite contents increase can be attributed to the non-wetting of a part of G particles in the polymer matrix leading to discontinuance in homogeneity of the composite topography as set down by Ramakrishna et al. 25
The influence of polyester/styrene ratios on the mechanical properties of the formulated hard PGC was studied and the data obtained is illustrated in Figure (7-P/S). It is clear that the compressive strength values increased as the styrene contents increased, on the expanse of recycled polyester to a maximum at the S/P ratio of 65:35% (mass ratios). Raising the styrene ratio up to 40%, resulted in a slight loss in the compressive strength by 2.4% compared to the PGC having 35% styrene. Effect of, a: Styrene content, b: methyl ethyl ketone peroxide ratio, c: Co ratio on the compressive strength values of polymer granite composite.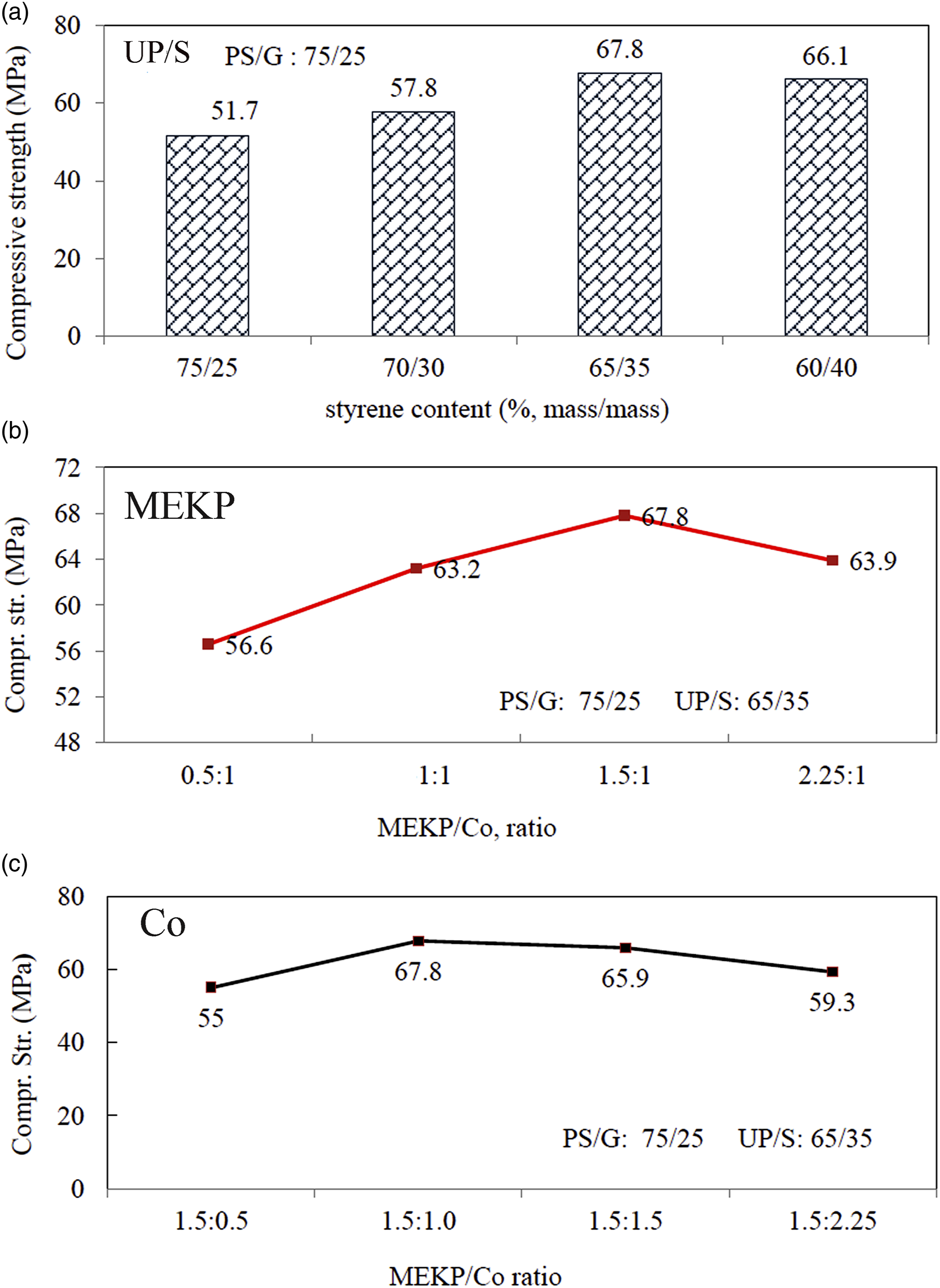
It is clear that, there was an optimum content of styrene monomer in the composite, but further than this value the important properties of the cured PGC product including the compressive strength can be deteriorated.
The type and concentration of initiator and accelerator employed in cross-linking reaction tend to play a significant role in achieving a product with optimal mechanical integrity.
Figure (7-MEKP) described the effect of various concentrations of MEKP as initiator, on the compressive strength values of polymeric composite. It is clear from the data obtained that, the compressive strength increased with increasing MEKP content keeping to Co added constant, until its mean value is equal to 67.8 MPa, and any further increase in the content of MEKP led to a decrease in the compressive strength values.
The impact of Co-naphthenate concentration as accelerator, on the compressive strength values of the PGC at constant MEKP initiator content of 1.5mass% was evaluated. The compressive strength values measured for each formulation increased with increasing Co-naphthenate concentration up to 1.5mass %, where maximum compressive strength value was recorded, as indicated in Figure (7-Co). Further increase in the concentration of accelerator beyond this value was accompanied with a decrease in the compressive strength values of PGC.
Thermal properties
TG analyses of the granite (G), styrenated polyester resin (SP) and polymer granite composite (PGC).

Note: TG: thermo-gravimetric.
P: S = 65/35mass: mass %.
SP: G = 25/75 mass: mass %.

Thermal analyses (TG & DTG) of granite waste filler (G), the styrenated-polyester resin matrix (SP) based on recycled PET waste, and their composite (PGC). PET: Poly (ethyleneterephthalate); TG: thermo-gravimetric; DTG: differential thermo-gravimetric.
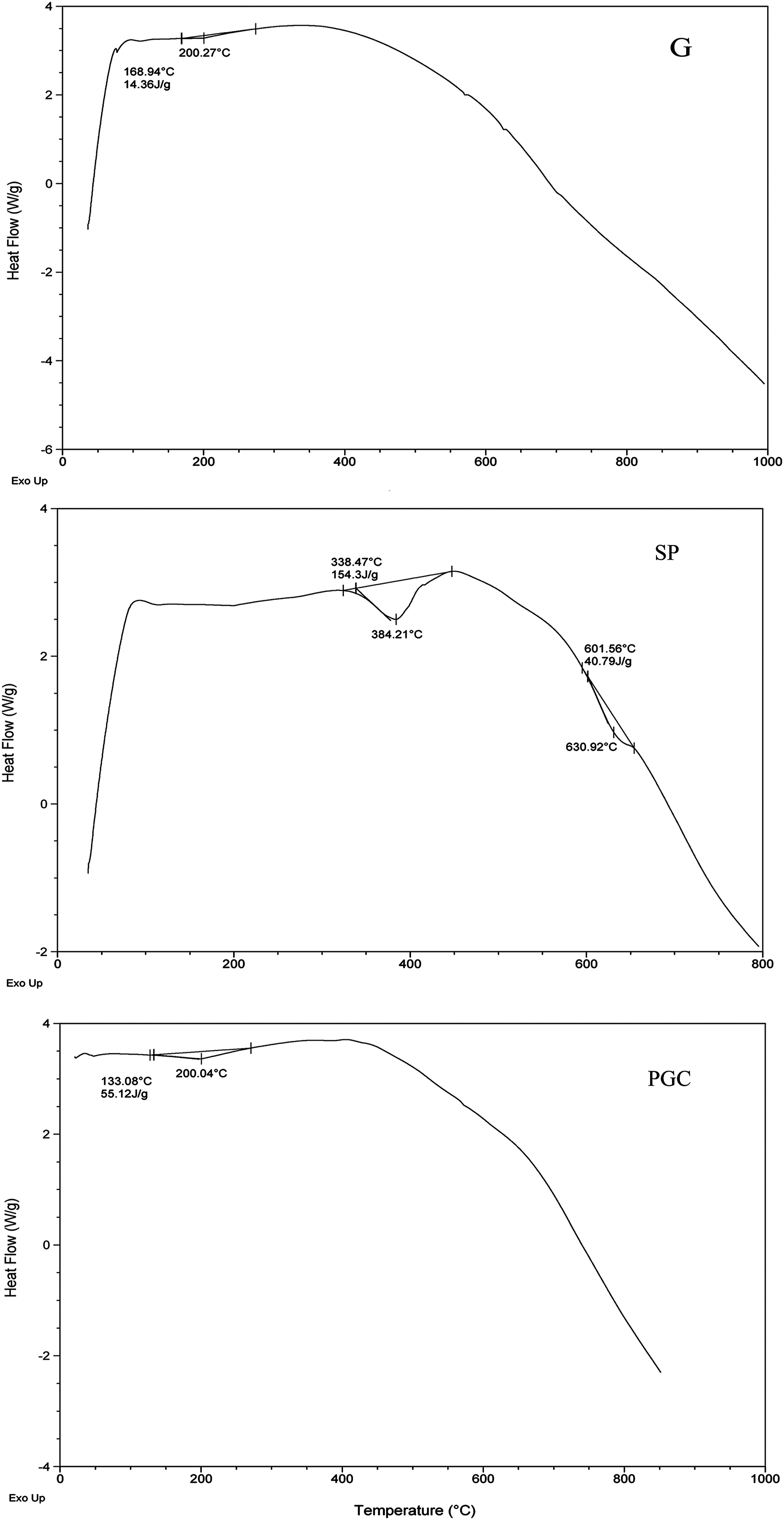
Differential scanning calorimetry diagram of granite waste filler (G), the styrenated-polyester resin matrix (SP) based on recycled PET waste, and their composite (PGC). PET: Poly (ethyleneterephthalate).
It is obvious from Table 4 that G powder disclosed ≈3.74% mass loss up to 1000°C. The TG thermogram for G, Figure (8, G), displayed three distinct mass loss steps. The first one started at room temperature and ended near 430°C with mass loss percent ≈0.811%. This loss can be referred to the water adhered to the surface of ground granite particles. The second step has onset temperature at 430°C and the offset at 610°C with mass loss ≈1.8% and can be attributed to the dehydration of intra and inter layer crystalline water. The last step fell beyond 610°C with mass loss 1.1% is due to the destruction and oxidation of some granite components. It is obvious that the total mass loss percentage up to 1000°C was only 3.74% indicating high thermal stability of the granite. The chemical analysis of granite, according to XRF located in Table 2, is composed mainly of inorganic oxides as stated before. The dehydration and dehydroxylation of organosilica occurred between 250-625°C, 27 while the boehmite particles were decomposed into γ-Al2O3 particles at 300–500°C. 32 On the other hand, the DSC thermogram of granite demonstrated very weak and wide heat absorption peak with onset temperature 168.94°C that offset at 275°C and appeared at 200.27°C, Figure (9, G). 23
Thermo-gravimetric analysis for SP matrix based on recycled PET waste is represented in Figure (8-SP). It is clear that about 90% of the mass of SP resin was lost in the range of onset at room temperature that offset near 450°C. This happened through two distinct steps; the first one has onset temperature at room temperature, i.e. ∼30°C and ended at 339.21°C with mass loss percent 8.7%. The highest heat absorption peak appeared at 384.21°C, Figure (9-SP), with mass loss 89.11%. The maximum degradation of SP was occurred at this step. After 450°C no changes in the residue mass was detected and about 10% out of the resin starting mass remained.
Figure (8- PGC) described the thermal degradation behavior of the PGC composite formulated of SP matrix and G filler at the ratio 25:75 (mass: mass %). The TG curve showed two mass loss steps; the first with the value 3.631% starting from room temperature till near 250°C, while the second step with mass loss of 21.55% was at temperature onset 250°C offset at about 460°C. It is clear also from the TG curve that, no significant mass loss (less than 3%) before 460°C was recorded which can be referred to the oxidation and crystallization that happened to the granite filling ingredients.
It is obvious from PGC thermogram in Figure (9-PGC), that the DTG represented detectable reduction in the degradation rate (1.44%.min/°C) at the temperature peak 323.22°C compared to that of the SP resin matrix with degradation rate 6.77% min/°C at the temperature peak 339.28°C, Figure (9-SP). This again confirmed that the composite formulated of styrenated polyester, based on recycled PET as matrix and granite waste powder as filler at the ratio 25:75 (mass/mass, %) can withstand high temperature up to 1000°C efficiently.
The results of thermal analyses indicated that polymer-granite composites possess good thermal stability; in addition the thermal processing of the granite waste materials should not provoke any problem to air pollution. The thermal reactions that occurred with firing, generates basically water vapour and possibly a non-significant release of carbon oxides. 26
Therefore and according to the thermal analyses data, it can be concluded that the polymer -granite composite under consideration can be candidate for many potential applications, specifically in construction fields when special thermal characters are issued.
Scanning electron microscope examination
Scanning Electron Microscope examination for the G was performed and the photos are represented in Figure 10(a) which showed the particle sizes of granite waste to vary from nanometres to micrometres and the micrograph manifested that G powder particles crop up in aggregates/agglomerates structures and the particles are mainly angular in shape with slightly sharp edges. On the other hand, photos for the PGC with the ratio 75:25 mass% G: SP dedicated good interfacial adhesion between the matrix and the filler. The angular fine shape of granite particles enhanced the proper bonding between G and SP leading to dense composite final product. Similar conclusion was reported by Passaretti et al.
33
This confirmed the data obtained for very low water absorption percent, low apparent porosity and the elevated compressive strength values of the PGC specimens with a homogeneous and denser structure. The presence of dark voids can be attributed to the filler particles pull-out from the polymer matrix, Figure 10(b). (a) The Scanning Electron micrographs of Granite (G) and (b) polymer granite composite (PGC).
Conclusions
The main aim of our long term projects is to share solutions for problems associated with solid wastes management. This laboratory scale study sustained in development of artificial granite-polyester styrene composite from two wastes, namely, granite scraps and post-consumer PET waste, with comparable mechanical integrity and complemented thermal stability. The product can be safe candidate to a lot of various applications like interior and exterior construction fields, some parts in automotive fabrication; insulation and anticorrosive purposes, aerospace industries, an inert matrix for some radioactive wastes containment and many others. These applications require acceptable compressive strength values, high temperature resistance and enhanced flammability performances that are decisive. The application of granite waste as an inorganic filler, for enforcement of the polymer matrix reduced the cost of the final composite and can be counted as a route for disposal of such wastes.
The prepared PGC composite possesses all these favourable requirements, thus it can be suggested as a new ecological and economically feasible candidate.
Agreeably to the authors' experiences, upgrading of the polymer granite composite production under consideration can be developed smoothly, for semi-industrial and industrial scales for its economical advantages, where the two base components were wastes, as well as being a safe solution for environmental pollution due to the management of the two problematic solid wastes accumulation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Science and Technology Development Fund (STDF) under project number 25745.
