Abstract
Piezoelectric polymer Poly (vinylidene fluoride) (PVDF) is extensively used as sensor and actuator devices because of their excellent piezoelectric and pyroelectric properties. The melting point of PVDF, however is relatively low and the service temperature of PVDF is up to 100oC. This restricts the use of PVDF in high temperature applications. This can be improved by adding appropriate fillers to the PVDF. In the present study, a composite of PVDF-Onium salt has been developed and characterized to improve the thermal efficiency of PVDF for high temperature applications. Poly (vinylidene fluoride)-Onium salt composite films have been developed using the solvent cast method and characterized for structural, surface and electrical properties to investigate the presence of β-phase (required for sensor applications) through X-ray diffraction (XRD), scanning electron microscopy (SEM), differential scanning calorimeter, Raman and Infrared spectra, and dielectric properties. The melting point of PVDF-Onium salt composite was found to be higher (175°C) as compared to the PVDF polymer alone (168.2°C) which has been discussed in detail. The PVDF-Onium salt composite sensors were further tested for dynamic strain sensing application for the first time. The performance of these sensors was evaluated by recording the vibration modes of the cantilever beam and resulting from impact loading.
Keywords
Introduction
Poly (vinylidene fluoride) (PVDF) has received a lot of attention because of its excellent mechanical properties, chemical stability, high dielectric permittivity, and distinct pyroelectric and piezoelectric features.1,2 Compared to other fluoropolymers, PVDF has an easier melt process because of its relatively low melting point. The service temperature of PVDF is 100o C. 3 A lower service temperature would restrict the use of PVDF in more demanding, higher temperature applications. Fillers can be used to improve the temperature stability of PVDF.4,5 It has been shown that the addition of onium salt, or clay to PVDF followed by annealing has improved its melting temperature. 6 According to Lawrence et al., annealing of PVDF-Onium salt or PVDF nano clay compositions involve heating for at least 5 min within the polymer’s melting area. Furthermore, it was revealed that annealing a PVDF-Onium salt composition for 1 min at 170°C resulted in a greater melting point of the composition. This technique can raise the melting temperature of PVDF by at least 15°C above its normal melting point. Short conditioning durations above 180°C followed by moderate cooling, can also result in a high melting point material. Annealing at 170°C raises the melting peak temperature above 176°C, and melting peak temperatures of 185°C have been observed depending on annealing time.
Most commonly used onium compounds include tetrabutyl ammonium perchlorate (TAP), tetramethylammonium hydroxide, tetra-n-butyl ammonium chloride (TBAC) and trimethylsulfonium chloride, etc. Different onium salts can be used in a variety of applications. Phase-transfer catalysts include salts of ammonium, phosphonium, and sulfonium. All the onium salts and especially diazonium and iodonium salts are very useful reagents in organic synthesis. The PVDF composite with either an onium salt, or a modified clay exhibits phase changes. Without stretching the material, this composite has a significantly greater concentration of the highly piezoelectric crystalline phase. 7 Electrospinning of PVDF/TBAC blend nanofibers has been done and it is observed that the dielectric and piezoelectric properties of the PVDF nanofibers were improved by the addition of TBAC. 8
The effect of onium salts such as benzyl triphenyl phosphonium chloride (BTPC) and tetrabutyl ammonium perchlorate (TAP) on PVDF was investigated by Vijayakumar et al. 9 and found that a composite containing about 0.5% onium salts produced a high fraction of β-phase crystals in PVDF/BTPC and PVDF/TAP compression moulded films. Onium salts increased the melting temperature of the composites by 7°C. Using polarising microscopy, they reported that the onium salt acts as nucleating agents, resulting in a significant reduction in spherulitic size. Benzyl triphenyl phosphonium chloride is thus an additive of practical importance for PVDF to improve piezoelectric and dielectric properties. The addition of onium salts also increases the toughness of PVDF significantly, according to the study of mechanical properties.
The present study focuses on the development and characterisation of PVDF–BTPC composite films containing adequate concentrations of onium salt to increase thermal stability of PVDF. Further, The PVDF-Onium salt sensors were developed out of these films and these sensors are tested for dynamic strains sensing application for the first time. The performance of these sensors are further assessed by recording the vibration modes of the cantilever beam and monitoring dynamic strains caused by impact loads. These sensors, therefore, are useful for various applications where precision due to vibration and impact load plays an important role.
Materials and experimental methodology
Materials: PVDF granules were procured from M/s Reliance Corporation (Ahmedabad, India). Benzyl triphenylphosphonium chloride was used as onium compound material and this material was procured from M/s Ultra Lab Products (Bangalore, India).
Methodology: A measured quantity 6 g of PVDF was dissolved in DMF by stirring and heating using a magnetic stirrer. Once PVDF was completely dissolved, the onium salt (10% weight) was added to the PVDF solution. The solution of PVDF-Onium salt was mixed in the magnetic stirrer until the onium salt was completely dissolved with the PVDF solution. The blend of PVDF-Onium salt was then poured in a clean glass mould and kept in the furnace by maintaining the temperature at 150oC. The PVDF-Onium film (Figure 1) thus obtained was flexible. PVDF-Onium salt film.
Characterisation: These films were characterized for structural, surface and mechanical properties by X-ray diffraction (XRD), scanning electron microscopy (SEM), Fourier Transform infrared spectroscopy (FTIR) measurements, Raman measurements, differential scanning calorimetry (DSC), and dielectric test. β-phase conformation with high crystallinity was obtained by XRD technique. X-ray diffraction patterns of the PVDF-Onium salt film were recorded using Bruker 2200 X-ray diffractometer. The X - ray source was CuKα radiation, which was operated at 40 kV and 40 mA. The samples were scanned in the 2θ range of 10–60o and the sample was rotated at the scan speed of 1o/minute. Scanning electron micrographs were recorded using a EVO-80 scanning electron Microscopy (SEM) to evaluate microstructure of composite and uniform dispersion of onium ions in polymer matrix. All the specimens are coated with a conductive layer of sputtered gold. Infrared spectrum is recorded with a Bruker Vector spectrometer.
Thermal Characterisation: Differential scanning calorimetric measurements are conducted with a Perkin Elmer DSC-7 to evaluate the degree of crystallinity. In the DSC experiment, the sample was subjected to temperature ranging from 130–190oC and dielectric properties of PVDF-Onium salt film were studied using dielectric spectroscopy as a function of frequency ranging from 1 Hz to 107 Hz at room temperature.
Poling of PVDF-Onium salt film and the electroding on both sides of the film were done by using conductive silver ink as discussed in detail in previous papers from our laboratory.10,11
To check the sensitivity, the sensor is attached to the root of an aluminium cantilever beam and was connected to the oscilloscope. The sampling rate was set to 1 kHz. The response of the PVDF-Onium sensor bonded to a beam has been obtained by subjecting the beam to free mechanical vibration. The sensor response to impact loading is measured by dropping a steel ball of diameter (d) = 0.0095 m, mass (m) = 3.5 g from a known height h = 0.4 m onto the cantilever beam’s tip.
Results and discussions
The PVDF-Onium film prepared by solvent cast method is shown in Figure 1. Poly (vinylidene fluoride) and onium salt are entirely dissolved in dimethyl formamide, resulting in a clear, transparent liquid solution. The composite film thus obtained is flexible and the β-phase confirmation has been done using XRD technique.
Scanning electron microscopy
Figure 2 presents the scanning electron micrographs of the PVDF-Onium composite film. The existence of patterned structure is visible in solvent casted PVDF-Onium composite film. Similar observations are made by other researchers also.12–13 Surfaces of film with onium salt addition at 10% concentration was rough, porous, and void of spherulites. There is increase in pore densities or change in topography for composite film. Onium salt is thus dispersed at a molecular scale in PVDF. SEM image of PVDF-Onium film micro-structural morphology.
X-ray diffraction Measurements
Figure 3 presents the X-ray pattern of the PVDF-Onium composite film. Here, peak positions of reflections corresponding to PVDF-Onium are marked in the pattern. The occurrence of most intense diffraction peak at 2θ value of 20.38o corresponding to (110) planes, confirms the presence of the β-phase of PVDF-Onium film.
9
X-Ray diffraction pattern of PVDF-Onium salt.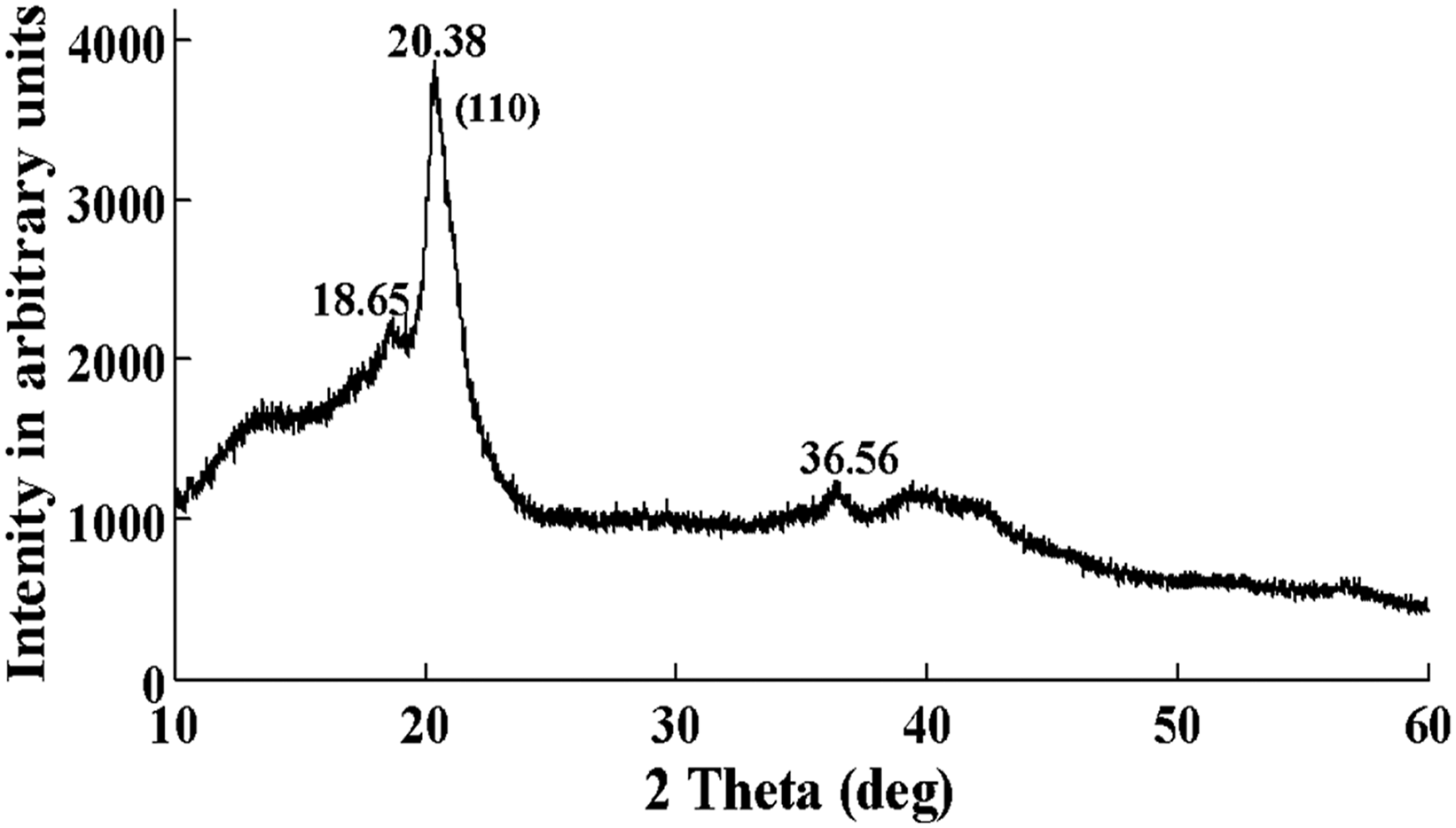
FTIR Measurements
The intensity gained from this measurement can be used to clarify the relative amount of crystalline phases. Figure 4 shows the IR Spectra of PVDF-Onium composite film. The spectrum was taken in the range of 400–1500 cm−1. The peaks at 757 and 883 cm−1 in Figure 4 correspond to the α and β phases, respectively. Tiwari et al.
14
has also reported the presence of such beta phase peaks. IR spectra of PVDF-Onium film.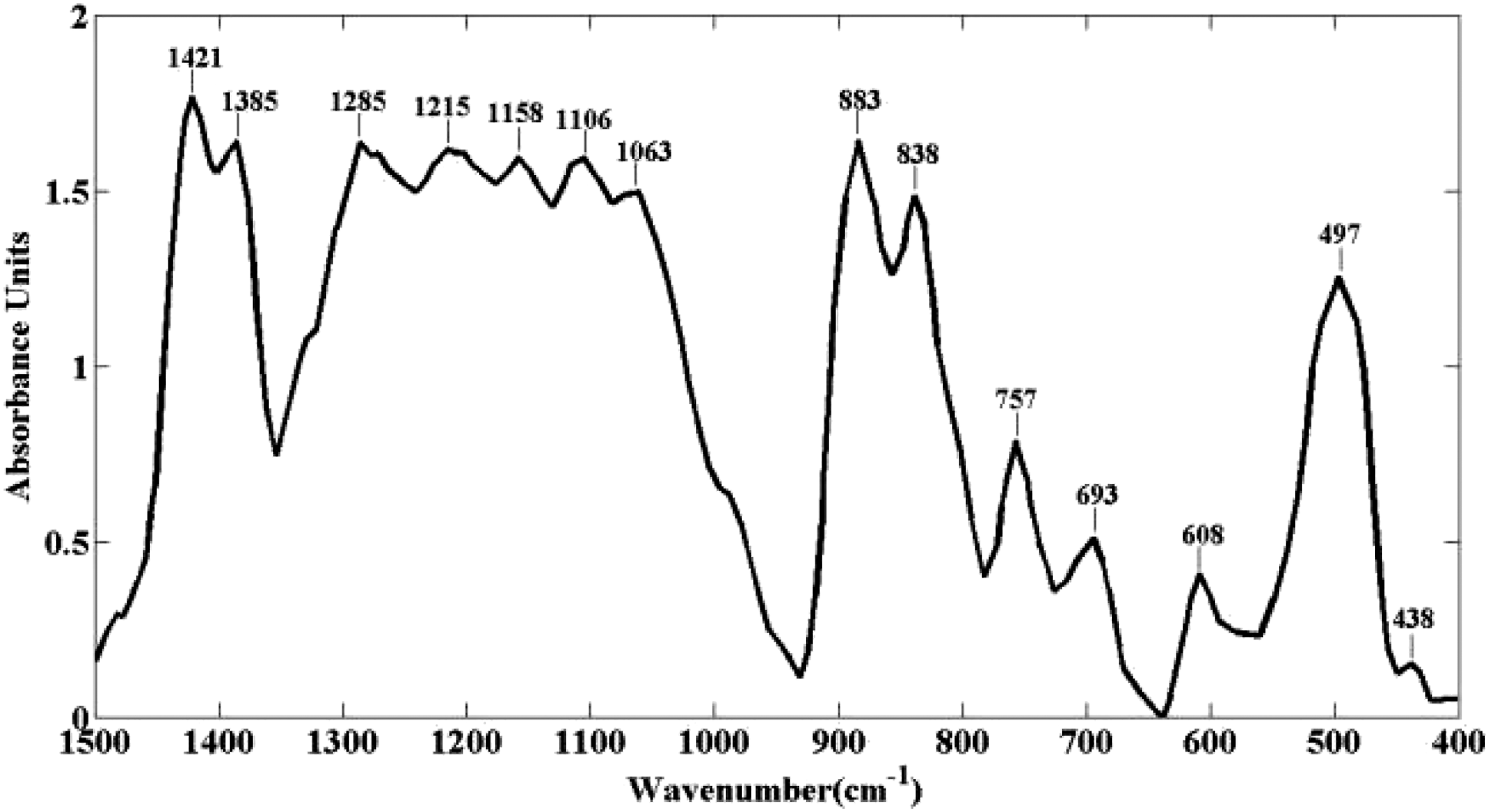
Raman measurements
Figure 5 shows Raman spectra of PVDF-Onium salt film ranging from 500 cm−1 to 1400 cm−1. The major Raman bands observed in the case of PVDF-Onium salts were 794.8, 999.9, and 1429 cm−1 that further confirm the presence of β-phase. Raman spectrum of PVDF-Onium film.
DSC Measurements
DSC traces of PVDF-Onium salt films are shown in Figure 6. Poly (vinylidene fluoride)-Onium salt has a melting point of 175°C., which is greater than PVDF polymer (168°C). Thus, the melting temperature of PVDF can be increased up to 7°C above its typical melting temperature by mixing it with onium salts. DSC curves of PVDF-Onium salt.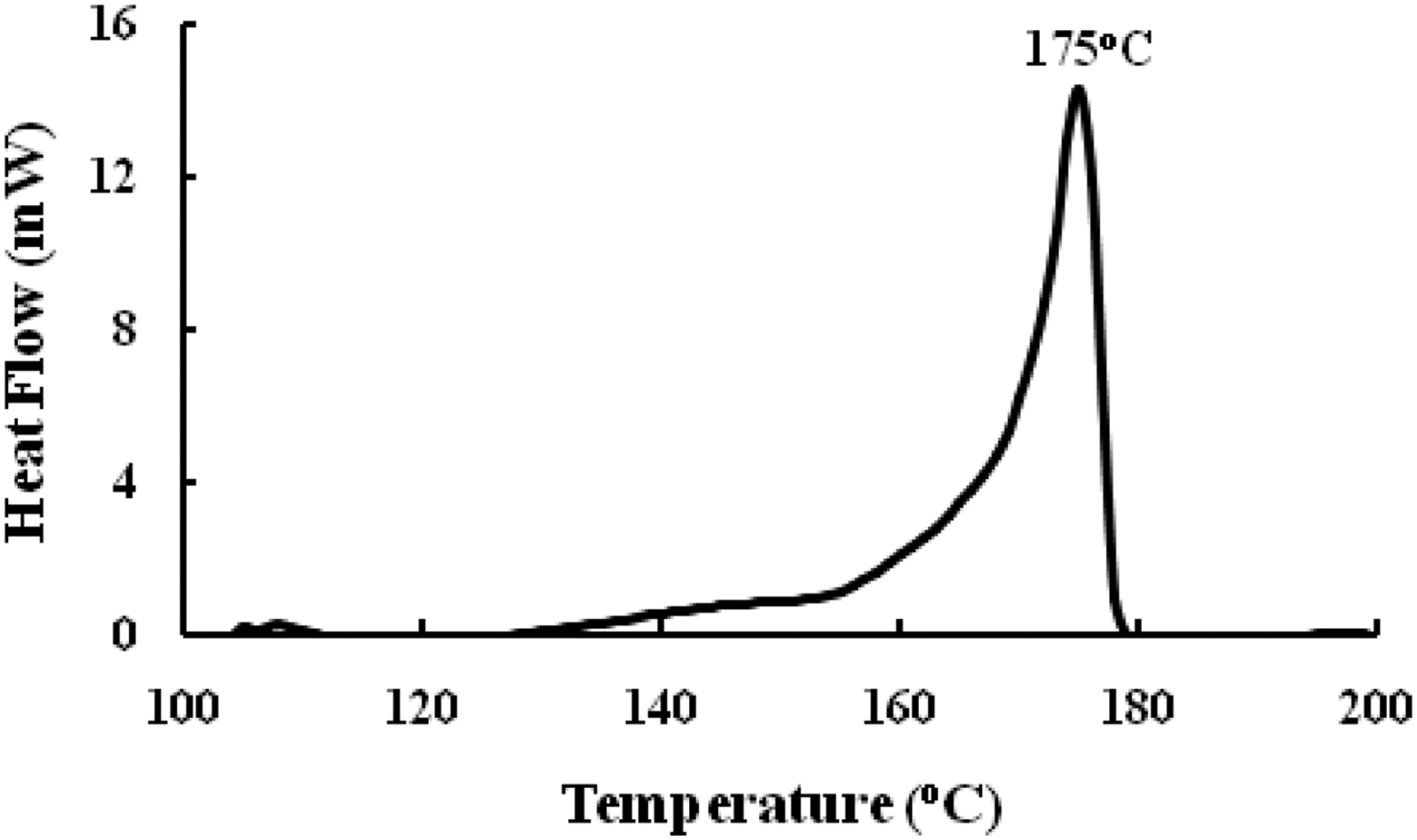
Dielectric Properties
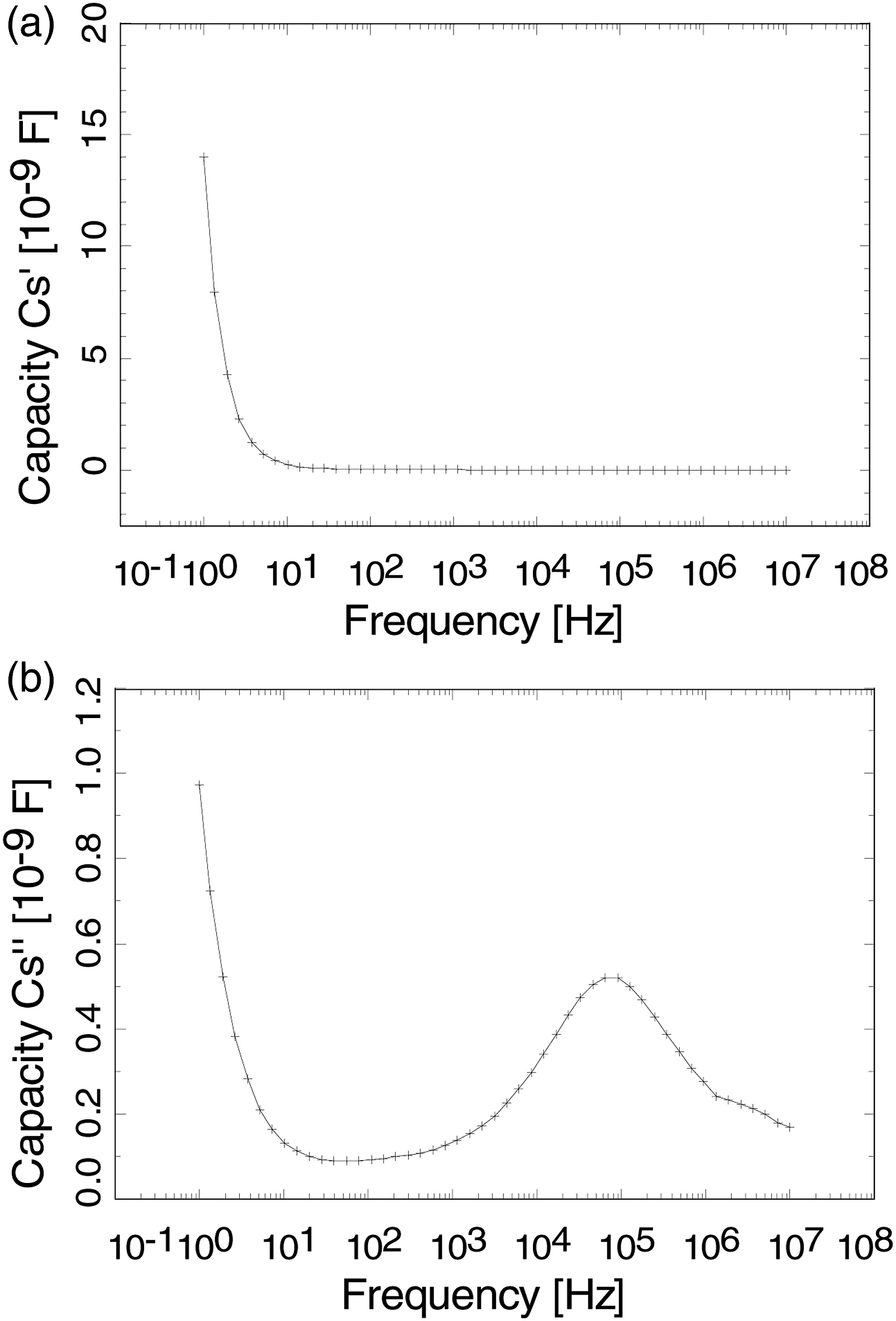
Figures 7(a) and (b) show the relative dielectric permittivity and electrical conductivity of PVDF-Onium film. The results indicate that the dielectric permittivity is higher at lower frequency and decreases as the frequency increases, whereas the electrical conductivity was found to increase as a function of frequency at 106 Hz. Figure 8(a) and (b) represent the frequency dependence of real and imaginary part of capacitance of PVDF-Onium film. It is found that at a lower frequency, both the real and imaginary part of the capacitance is high and as frequency is increased, the capacity is decreased. The imaginary part capacitance is maximum for frequency around 100 kHz and decreases for both the other sides of frequency spectra. (a) Relative permittivity and (b) Conductivity of PVDF-Onium salt. (a) Real part capacitance and (b) Imaginary part capacitance of PVDF-Onium salt.
Application of PVDF-onium salt composite sensor
Sensitivity to mechanical vibration
The poled β-phase PVDF-Onium sensor was tested for static and dynamic strain sensing properties. To investigate the sensitivity of the fabricated PVDF-Onium salt sensors under dynamic strain, the response of the sensor bonded to a beam has been obtained by subjecting the beam to free mechanical vibration (Figure 9(a)). The cantilever beam specimen used was aluminum, with dimensions of length, L = 0.33 m, width, w = 0.03 m, and thickness, h = 0.0015 m. The beam is induced to vibrate freely by causing initial displacement and then allowing it to oscillate on its own. An oscilloscope was used to record the voltage response from the PVDF-Onium sensor due to free vibration (Figure 9(b)). (a) Bonding of PVDF onium sensor on cantilever beam for free vibration and impact testing (b) Voltage response (c) Frequency response.
The beam is induced to vibrate freely by causing initial displacement and then allowing it to oscillate on its own. An oscilloscope was used to record the voltage response from the PVDF-Onium sensor due to free vibration (Figure 9(b)). As illustrated in Figure 9(c), the frequency response was derived using the Fast Fourier Transform (FFT). In FFT, the sampling points used were 2 × 1012. The peaks reveal the resonance frequencies of the cantilever beam’s different modes. Using Euler Bernoulli beam theory, the first and second natural frequencies (F1 and F2) of the beam can be calculated:
4.2 Sensitivity to Impact load
The PVDF-Onium salt sensor’s response to impact loading is measured by dropping a steel ball from a known height onto the cantilever beam’s tip. At the tip of the cantilever beam, a ball with a diameter of d = 0.0095 m was dropped from a height of h = 0.4 m. The volume of the sphere is given by V = 4πr 3 /3. For the density of a steel ball ρ = 7796 Kg/m3, the mass is found to be m = Vρ = 3.5 g. In terms of energy, E = mgh = 0.0137 N.m can be used to estimate the magnitude of the impact.
The steel ball hits at the beam tip and rebounds at a height of 1–2 mm. Figure 10(a) shows the response of the sensor mounted at the cantilever’s root for impact loading. Figure 10(b) shows the frequency response obtained by performing an FFT of the sensor signals. The peaks in the frequency response of the PVDF-Onium salt sensor effectively represent numerous resonant frequencies of the beam. Frequencies F1 = 10 Hz and F2 = 64 Hz are the natural frequencies from the graph (Figure 10(b)) which matches well with the calculated values. As a result, the developed PVDF-Onium thin films can detect dynamic stresses caused by impact loads. The response of PVDF-Onium salt bonded on a cantilever beam subjected to impact of magnitude 0.0137 N.m at the cantilever tip (a) Voltage response (b) Frequency response.
From the results obtained, it can be concluded that the developed PVDF-Onium sensors can capture multiple modes of vibrations and are capable of sensing the dynamic strains resulting from impact load. These features are very useful for various commercial and aerospace applications, where the precision due to vibrations and impact load is one of the vital parameters.
Conclusions
Poly (vinylidene fluoride)-Onium salt composite films were developed by using a solvent cast method. The films were developed with a view to improve the thermal stability of PVDF sensors. Those composite films had the advantage that addition of 10% weight of Onium salt had transformed the film directly into the required β-phase conformation eliminating elaborate stretching process required for PVDF.
The film was characterised for its phase using XRD technique. IR and Raman spectra further confirmed the presence of β-phase in PVDF-Onium salt composite film. Scanning electron micrographs features show the fibrillar features of the composite with dispersed particles of Onium salt in PVDF. The electrical conductivity increased as the frequency increased, whereas the dielectric permittivity was stronger at lower frequencies and decreased as the frequency increased. DSC confirmed that the melting point of PVDF-Onium salt film was higher (175°C) as compared to the PVDF polymer alone. Thus, PVDF-Onium salt sensors can be used for higher temperature applications than PVDF sensors. Further, the sensitivity of the composite was evaluated by subjecting it to free vibration modes and impact loading. The calculated natural frequencies at F1 = 10.48 Hz and F2 = 65.73 Hz matched well with the observed values. The developed PVDF-Onium sensors can capture multiple modes of vibrations and are capable of sensing the dynamic strains resulting from impact load. These films, therefore, are useful for various commercial and aerospace applications where precision due to vibration and impact load plays an important role.
Footnotes
Acknowledgements
The authors wish to thank the financial support provided by CSIR and AR&DB, New Delhi to carry out this project. The authors also thank Director, CSIR-NAL, Bangalore and Head, MT for the support. We also thank Dr Harish Barshilia, Ms A Vanaja, Ms Kalavathi, Dr K Venkataramaiah and Ms Jesmary for their help during experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Aeronautics Research and Development Board.


