Abstract
Graphene oxide and polyaniline have some unique properties and are considered as new generation materials for various applications. One major application of these materials is the adsorption of pollutants. In this study, Graphene oxide was synthesized with different levels of oxidation by using the modified Hummers method. Polyaniline/graphene oxide composite was synthesized by in situ polymerization with GO loading at 10% and 50% and further the composite is casted inside a disposable syringe by using N-methyl-2-pyrrolidone (NMP) as a dispersing agent. An adsorption experiment was carried out for water containing pesticide chlorpyrifos. The produced materials were characterized by FTIR spectroscopy & and XRD. Morphology was studied by SEM images. The FTIR spectra shows the formation of functional groups like -OH, -COOH on graphene, which may increase the hydrophilicity and adsorption properties. FTIR spectra of the composite portrayed the formation of bonds between polyaniline and graphene oxide. XRD peaks were obtained corresponding to crystals of polyaniline and graphite sheets. The adsorption efficiency was determined by analysis of water samples before and after filtration by using GC-MS. The composite produced with 50% amount of GO showed optimum results for the removal of chlorpyrifos. However, the composite with 10% GO loading is showing very low efficiency for the removal of chlorpyrifos. The PANI/GO composite is a possible candidate for the treatment of river water polluted by agricultural pollutants.
Introduction
Since the 1970s with the ‘Green Revolution’, there has been a significant jump in the use of chemical fertilizers and pesticides for the increase of agricultural yield. Pesticides are commonly used to prevent crop damage and increase production. In 2015-16, the total consumption of pesticides was 54,121 tonnes in India according to the Ministry of Chemicals and fertilizer 1 Most common pesticides are organo-phosphates, organo-chlorines, acetanilides, triazines etc. Some common pesticides found in wastewater such as Breakdown products of pesticides like Malaoxon, Organochlorines like Lindane, Triazines like Atrazines, Cyclodienes like Heptachlor, Acetanilide like Alachlor, Butachlor, Organophosphates like Malathion, Parathion methyl, Diazinon and Chlorpyrifos and Dichlorodiphenyltrichloroethane (DDT).
Chlorpyrifos is a common Organo-phosphate pesticide used for the killing of pests. According to the survey of IIT Kharagpur, it is also found in river water more frequently especially in the lower Gangetic area. 2 The detrimental effects of chlorpyrifos on the environment and on the living beings has sparked the concerns w.r.t. hazards to health, pollution of the environment, remains as a food residue, labour safety at work field, and regulatory issues. 3 The presence of this pesticide in water causes problems for aquatic animals and also human health. 4 Removal of this pesticide from water sources is necessary to reduce the impact on living beings.
Water-treatment has been studied by various researchers to solve the problem of water pollution. Significant research has been done on adsorption, membrane filtration, nanofiltration, photocatalytic degradation, oxidation, electrochemical degradation for treatment of water. 4 Among these, the adsorption process of water treatment is the most promising process due to high efficiency, simplicity and cost-effective.5–8
Graphene has sp2 hybridized carbon in six-membered rings arranged as planar sheets first synthesized by Boehm et al. in 1986 9 due to its unique properties has received attention by various researchers as a potential adsorbent. Graphene has a large surface to volume ratio and large theoretical specific surface area 10 which make it a suitable candidate for adsorbent. 11
However, graphene is difficult to process, poorly soluble and forms agglomeration in solution due to van der Waals interaction which makes it difficult to use along with polymers. To benefit from the unique properties of graphene while using polymers, Graphene Oxide (GO) has been proposed which has a similar structure like graphene. GO has some extra functional groups like -OH, C = O, -COOH and C-O-C. 12 These functional groups improve dispersibility, wettability with polymer matrices while improving hydrophilicity. Oxidation of followed by sonication is an easy top-down synthesis method for producing GO in comparison to difficult bottom-up synthesis method for graphene makes GO more attractive for an adsorbent material. For easy synthesis, functionalization, chemical versatility, elevated active surface area and redox properties make conducting polymers like polyaniline make good candidates for formation for composites with Graphene Oxide for adsorbent application.13–21
In this experiment, Graphene Oxide was synthesized by the modified Hummers method in two batches. In one batch, oxidation was done for several hours. In the other batch, oxidation was done overnight. PANI/GO composite was synthesized by in situ polymerization, by using both the samples of GO. Characterization study was done by FTIR, XRD, and SEM. Filtration of water containing chlorpyrifos was carried out and change in the amount of chlorpyrifos after filtration was studied by Gas chromatography-mass spectrometry.
Experimental section
Materials
Graphite powder (90%, 105 µm), aniline (C6H5NH2, 99%), ammonium persulfate (K2S2O8, 98%), hydrogen peroxide (H2O2, 30%–32%) were purchased from Nice Chemicals P. Ltd (Kerala, India). Sulphuric acid (H2SO4, 98%), hydrochloric acid (HCl, 35%–38%), sodium nitrate (NaNO3, 99.5%) were purchased from Research Lab. Fine chemical industries (Mumbai, India). Potassium permanganate (KMnO4, 99%) was purchased from SRL chem. Ethanol (99.9%) was purchased from Changshu Hongsheng Fine chemicals industries (China). De-ionized water was purchased from Merck (Darmstadt, Germany). All the materials were used as received.
Synthesis of graphene oxide
Graphene oxide (GO) was synthesized by oxidation of graphite powder using modified Hummers’ method in two batches followed by sonication to exfoliate graphite oxide to graphene oxide like the work done by Syed Shahabuddin et al.
11
For the first batch, the oxidation reaction was done for 4 hours and in the second batch, oxidation reaction was done overnight ∼18 h. The prepared Graphene oxide in the first batch and second batch were designated as GO-1 and GO-2 respectively. The reaction process is same in both the batches, which is illustrated in Figure 1. Schematic representation of synthesis of graphene oxide from graphite.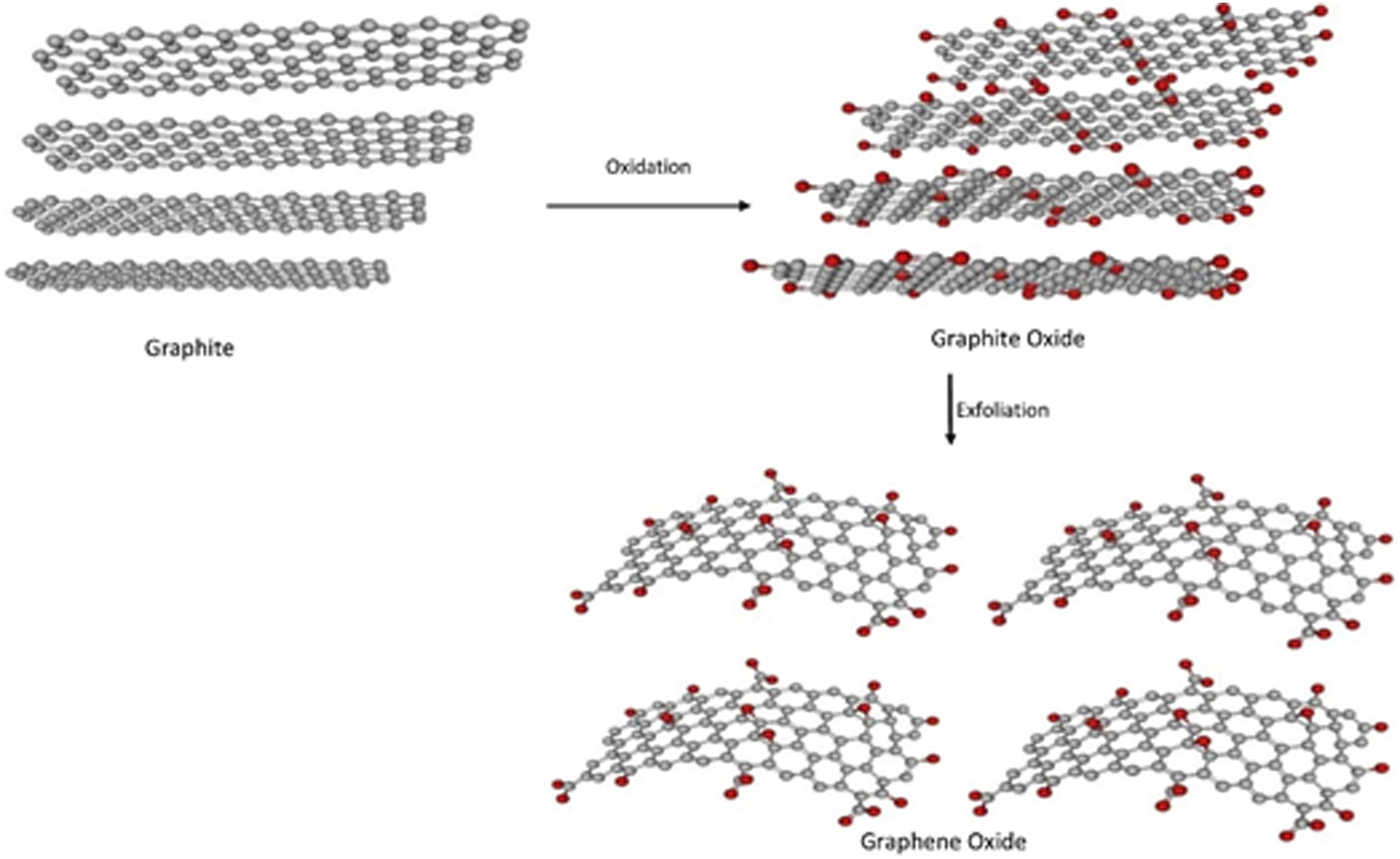
Synthesis of GO-1
5g Graphite powder, 2.5 g sodium nitrate and 120 mL sulphuric acid were mixed in a beaker immersed in an ice-bath under constant stirring at 500∼1000 r/min for 1 hour. Sodium nitrate acted as a catalyst here. 15g KMnO4 was added gradually for the oxidation of graphite. After 2 hours of stirring, reaction beaker was removed from ice bath and 150 mL of de-ionized water was added gradually. The temperature of the vessel was increased to 90°C and stirred for another 2 hours. 50 mL of 30% H2O2 were added to the reaction mixture for neutralization of excess KMnO4 and stirred for 30 min 700 mL de-ionized water was used to dilute the reaction mixture which was kept overnight in room temperature. Formed graphite oxide settled in the lower part of the beaker, which was washed with 1M HCl followed by washing with ethanol and water. Washing was stopped after the filtrate turned neutral. Graphite oxide was recovered by centrifugation at 4000 r/min. 100 mL DI water dispersion of graphite oxide was sonicated for 30min at 50–60 kHz to exfoliate graphite oxide into graphene oxide sheets.
The step-wise representation of the experiment carried out at the laboratory is represented in a schematic diagram as Figure 2. Pictorial representation of the instruments, set-up and various stages of reaction and end products are represented in Figure 3 Schematic representation of Synthesis method of GO-1 through Hummers method. Photograph of various stages during GO-1 synthesis.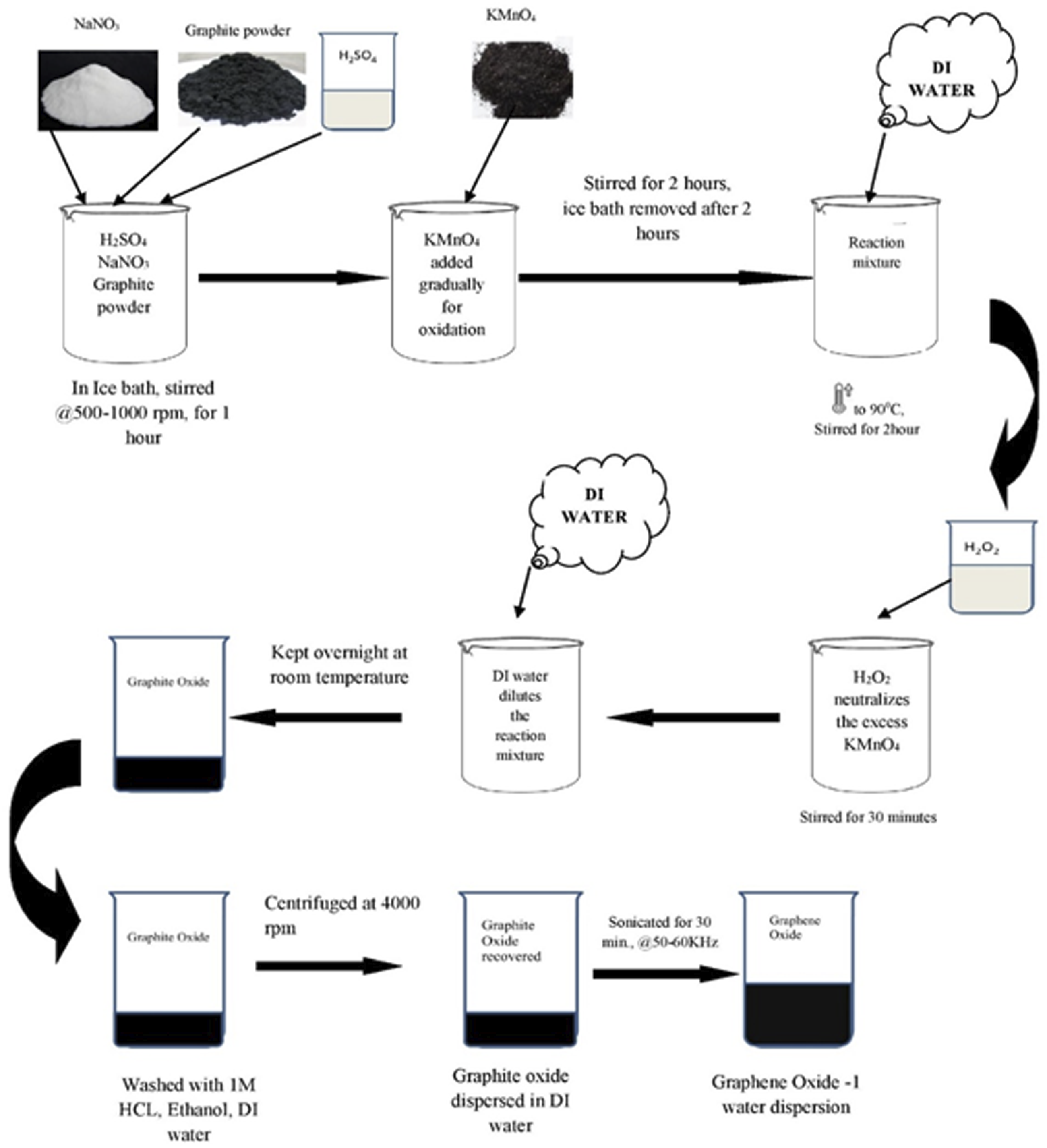

Synthesis of GO-2
GO-2 was synthesized just like the first batch with the same amounts of chemicals with some changes to the process. The only difference being the oxidation reaction was done for overnight. That means after addition of KMnO4 and stirring of the reaction mixture for 2 h in ice-bath, the reaction mixture was kept for overnight at room temperature for a higher level of oxidation. After oxidation reaction, 150 mL water was added for dilution followed by an increase in temperature up to 90°C, stirred for 2 hours followed by 50 mL of 30% H2O2 addition to the reaction mixture for neutralization of excess KMnO4 and stirred for 30 min 700 mL de-ionized water was used to dilute the reaction mixture which was kept for 40 h at room temperature. Settled graphite oxide was washed with 1M HCl followed by ethanol and DI water. Produced graphite oxide was recovered by centrifugation at 4000 r/min. We observed the produced graphite oxide to be slightly lighter grey in comparison to the first batch. Graphite oxide DI water dispersion of 100 mL was sonicated at 50–60 kHz for exfoliation to graphene oxide.
The step-wise representation of the experiment carried out at the laboratory is represented in a schematic diagram as Figure 4. Pictorial representation of the instruments, set up of apparatus and various stages of reaction and end products are represented in Figure 5. Schematic representation of Synthesis method process of GO-2. Photograph of various stages during GO-2 synthesis.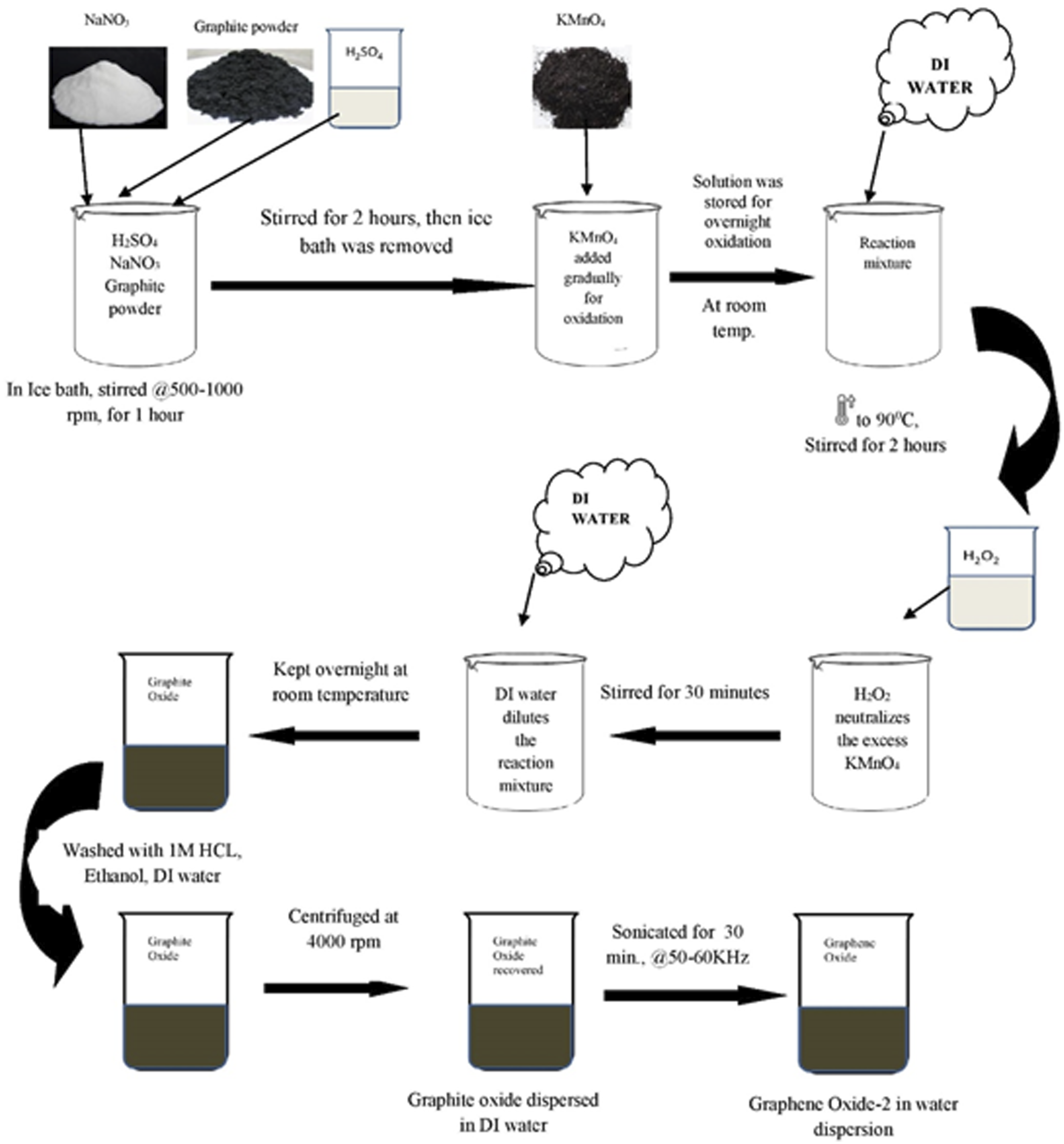

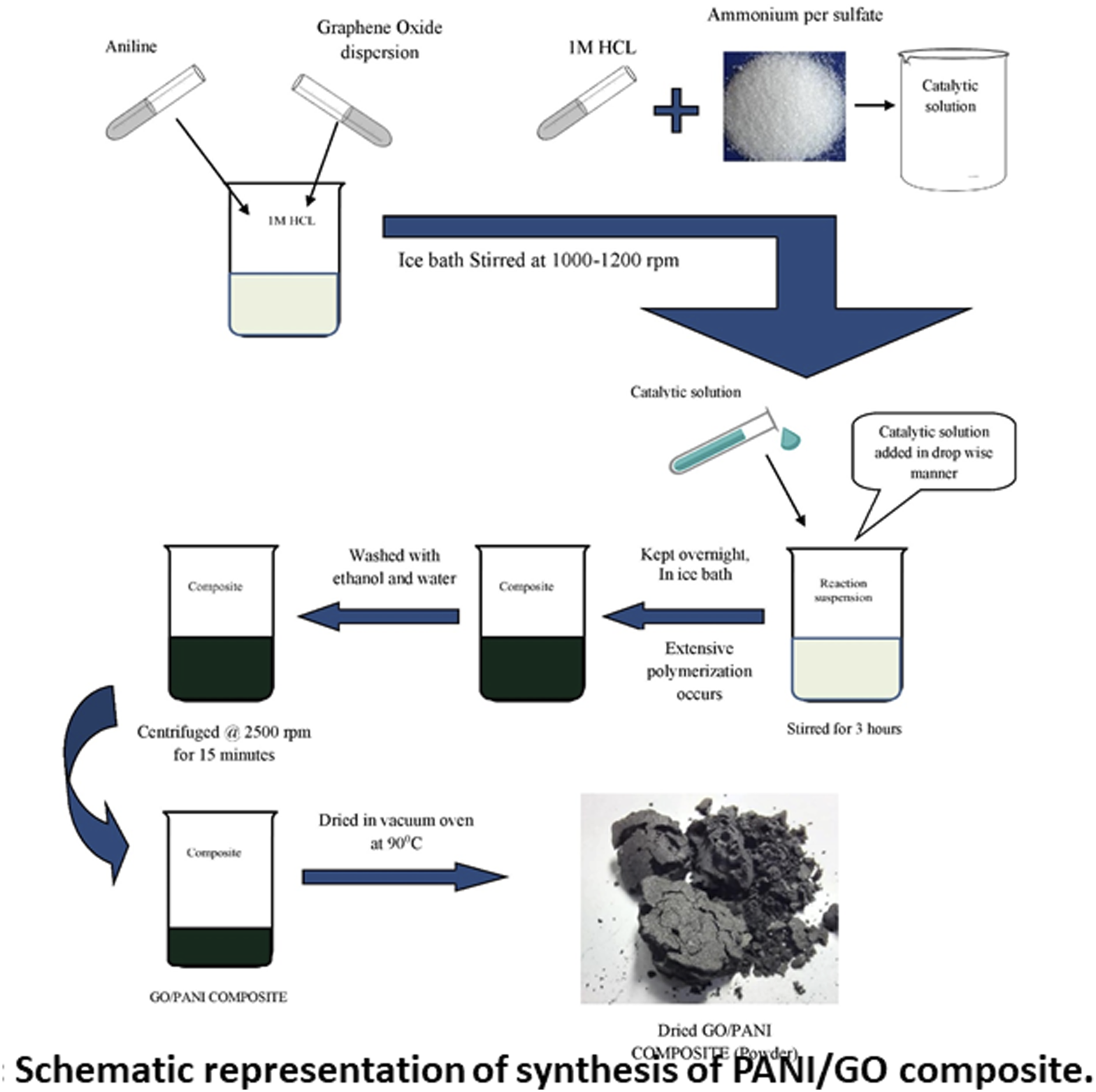
Synthesis of polyaniline/graphene oxide composite
Polyaniline/graphene oxide composite was prepared by in situ oxidative polymerization of aniline in presence of graphene oxide. A calculated amount of aniline and graphene oxide was suspended in 1M HCl and stirred at 1000∼1200 r/min for proper mixing of monomer and GO. The reaction vessel was kept in ice-bath. Ammonium persulfate (APS) was taken and dissolved in 1M HCl to prepare the catalyst solution. APS to aniline mole ratio was 1.20:1. The oxidant solution was added to the reaction suspension drop-wise with vigorous stirring. After 3 h of continuous stirring, the reaction vessel was kept in ice-bath for overnight with occasional stirring for extensive polymerization. Composite was prepared with 50% and 10% loading of graphene oxide. Three samples of composites each for GO-1 and GO-2 was prepared. The produced composite was washed with ethanol and water which was recovered from the water by centrifugation at 2500 r/min. Composite was dried in vacuum at 90°C overnight which gave solid composites (Figures 6, 7 and 8). Possible interactions among PANI and GO. Schematic representation of synthesis of PANI/GO composite. (a,b,c,d,e,f,g,h,i) Photographs of various composites prepared in this work.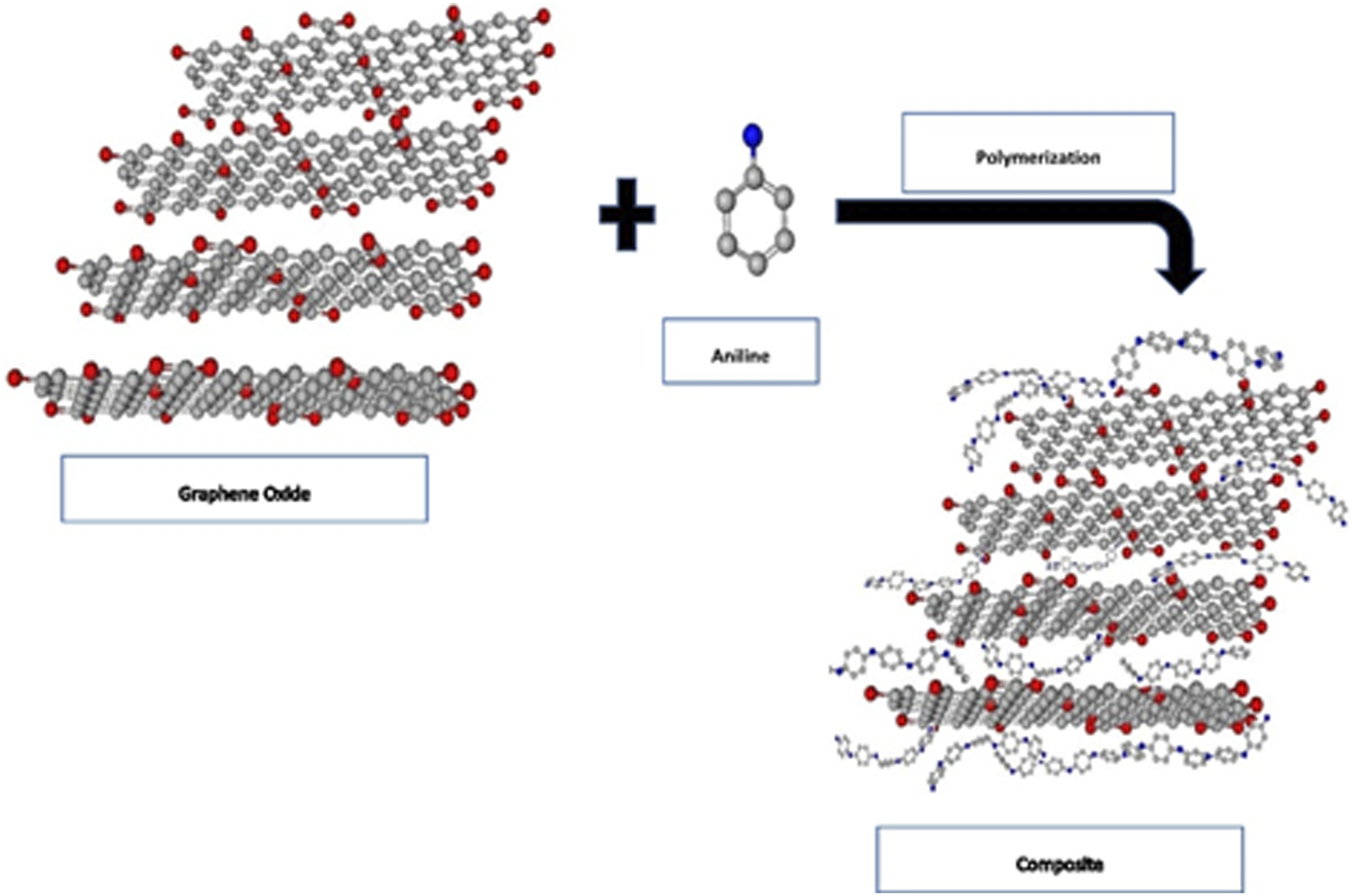

Casting of composite and adsorption experiment
The PANI/GO-1 and PANI/GO-2 were cast inside two disposable syringes using N-methyl-2-pyrrolidone (NMP) as a solvent. The cast composite was then dried in a vacuum oven set at 90°C for a whole night. The use of NMP as a dispersion agent was justified by the fact that it preserves the chemical stability and integrity of the composite. 22
Chlorpyrifos polluted water 250 mL each from the same batch of the solution was passed through the composite column and another 250 mL of unfiltered water was kept for analysis and comparison purpose. The set up for filtration mechanism and the mechanism of chlorpyrifos being attached and trapped into the composite matrix is represented in Figure 9. The pictorial representation of the composite column and pesticide polluted water being filtrated is represented in Figure 10. The filtrated water was analysed by GC-MS (Ultima 1310 & Thermo TQS 8000 with autosampler Triplus RSH) and amount of chlorpyrifos was compared to the unfiltered polluted water. Adsorption of Chlorpyrifos mechanism. Photographs of Adsorption experiment set-up.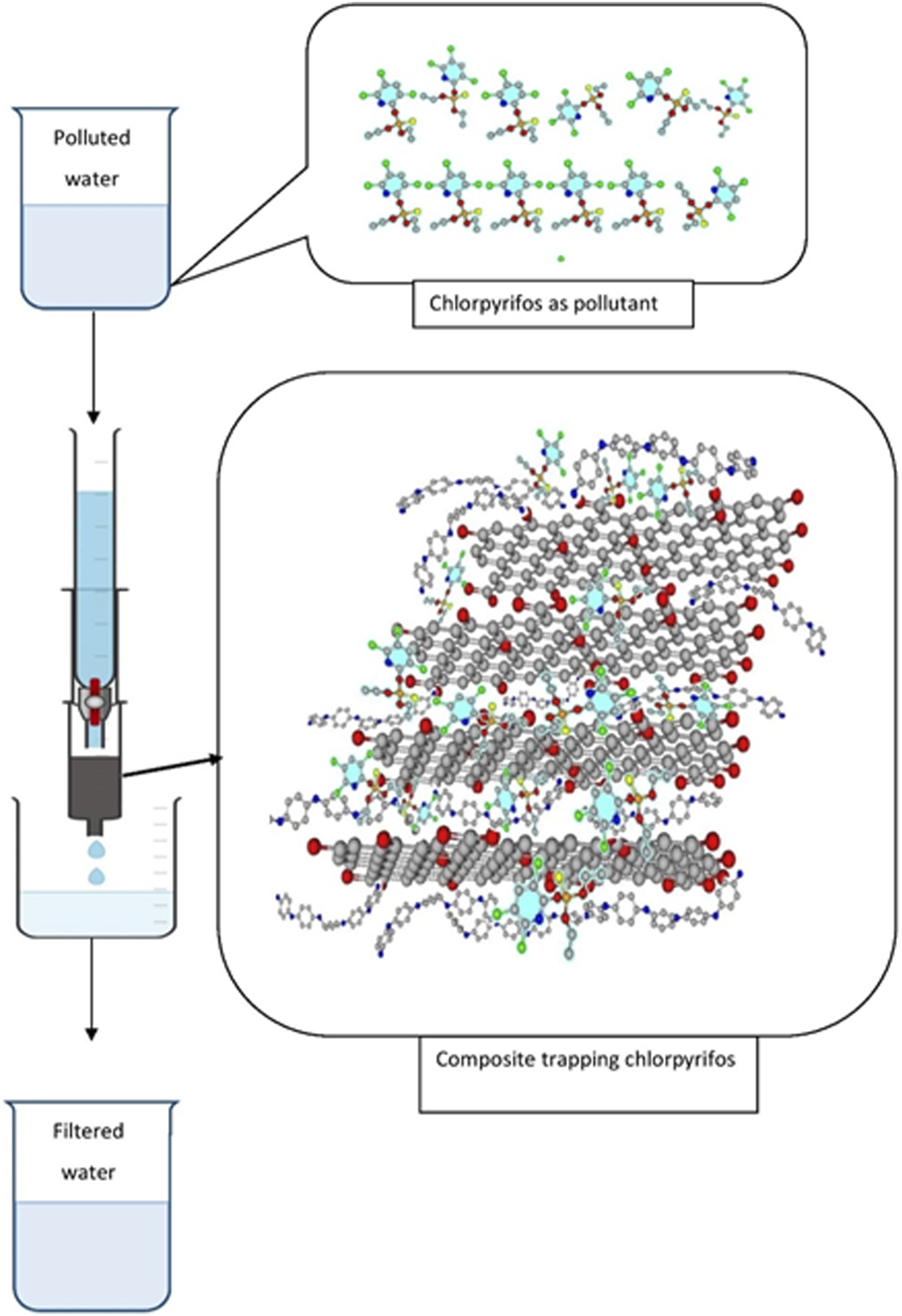

Material characterization
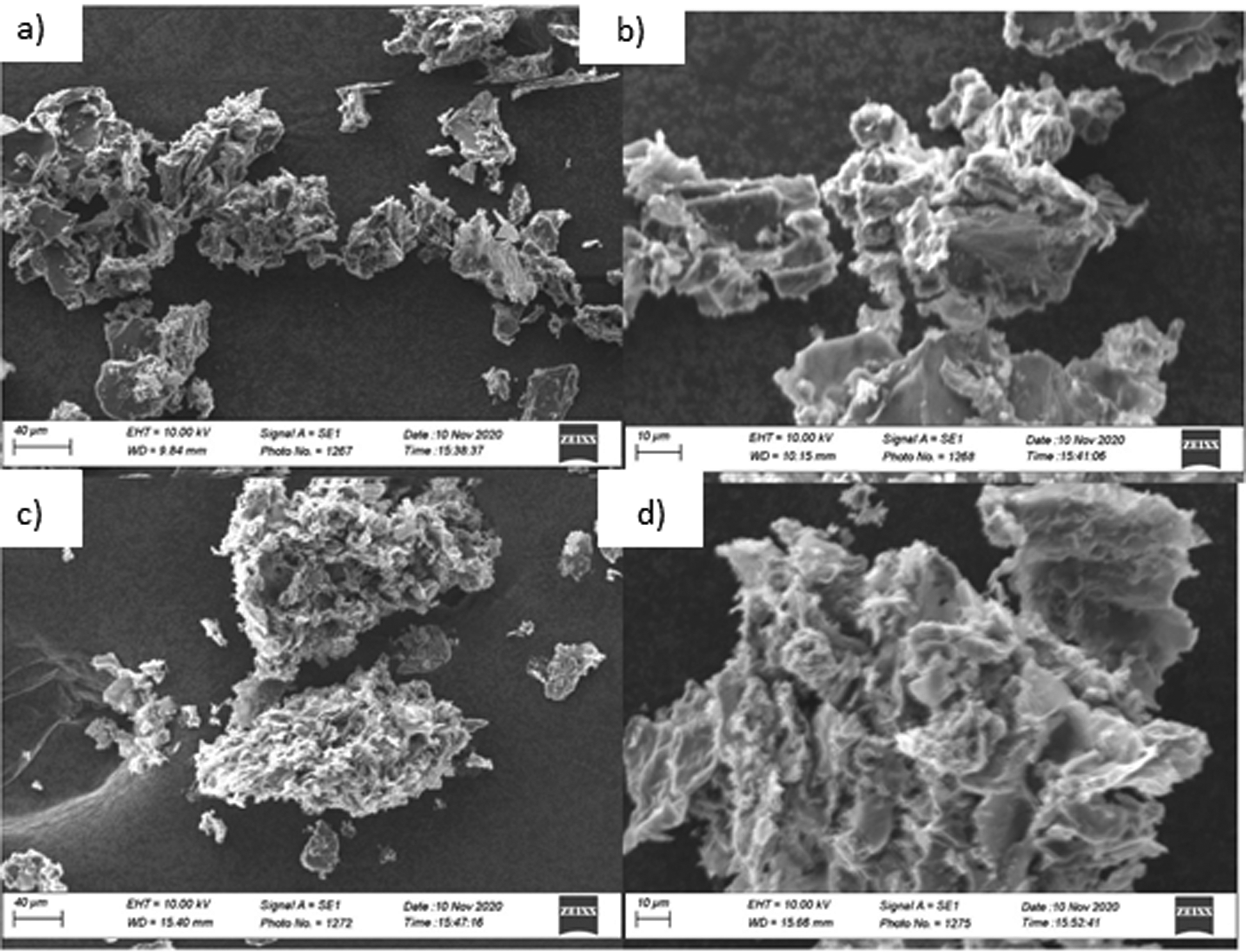
Thermo Gravimetric Analysis (TGA) was done for composite to know about thermal behaviour of composite in comparison to polyaniline. Figure 11 Fourier transform infrared (FT-IR) spectra of the powdered samples were recorded. Figure 12 XRD patterns of graphene oxide and PANI/GO composites were also recorded. Surface morphology was studied by Figure 13 scanning electron microscopy (SEM). FTIR spectra of composites and PANI. XRD pattern for PANI & composites. SEM images of composites with 1:1 polymer to GO ratio.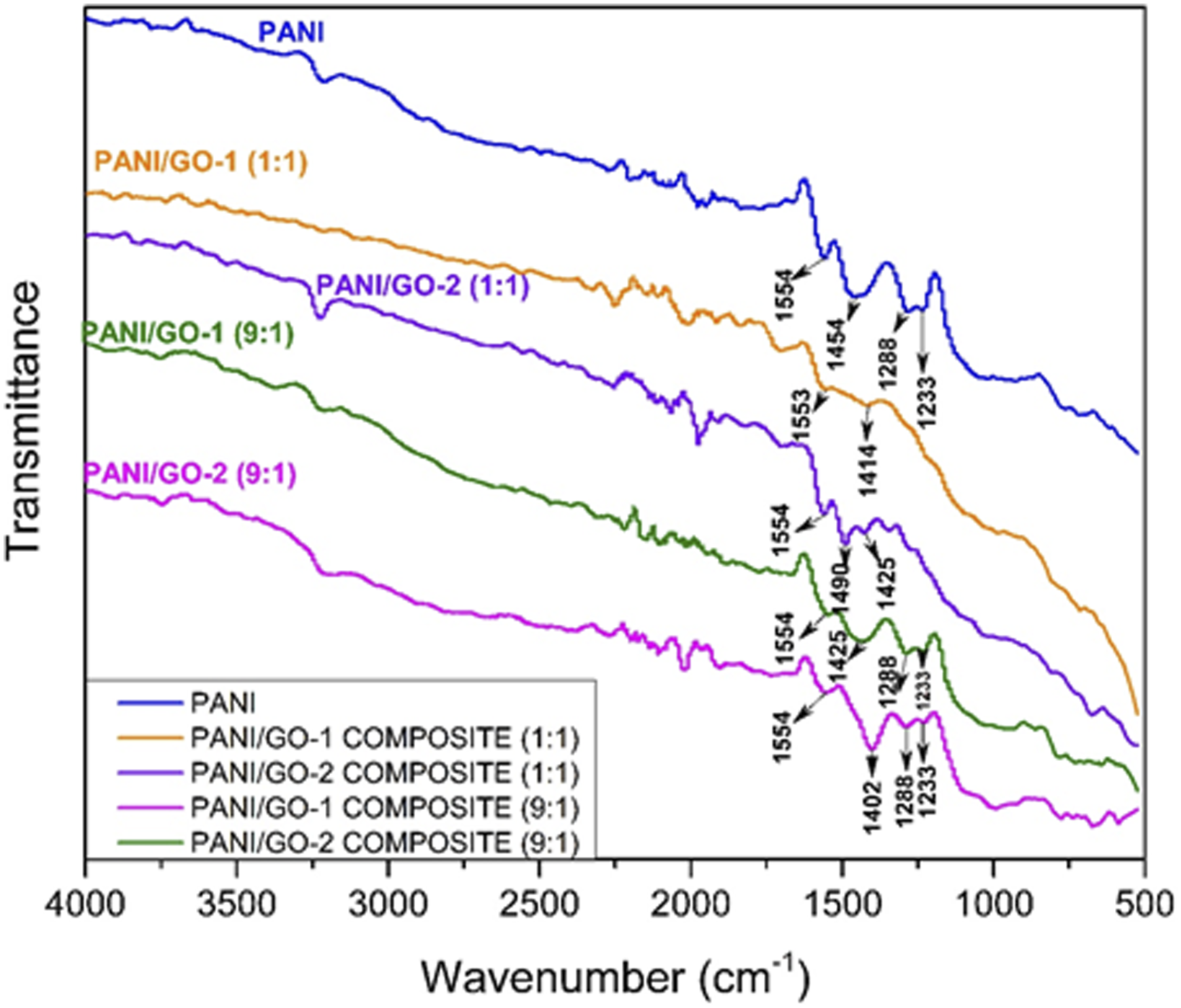
Results and discussion
Material characterization and properties
Thermal sability-TGA
Figure 14 shows the TGA curve of PANI and PANI/GO. Weight loss below 100°C is due to the removal of water. Weight loss between 130°C and 300°C is attributed to the removal of oxygen-containing groups in the PANI/GO composite. Drastic weight loss after 300°C in the PANI is attributed to the decomposition of PANI backbone. The weight loss rate is lower in the case of composite indicating the increase in thermal stability may be due to the formation of bonds between GO and PANI. Work done by Qiahui Fan et al also showed a similar type of TGA curve however with different rate of degradation.
23
This different may be due to different synthesis conditions etc. TGA curve for PANI & PANI/GO composite.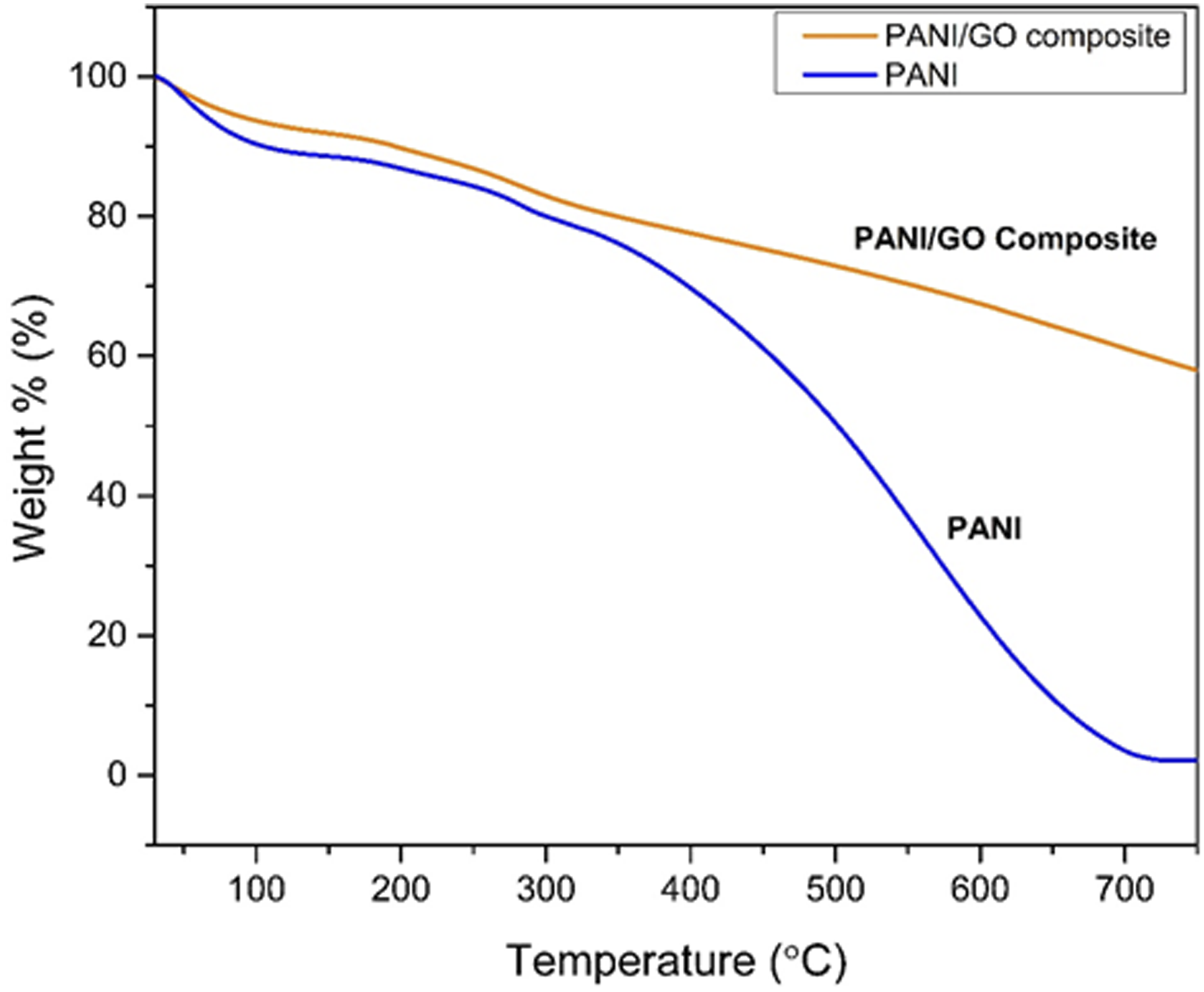
Structural analysis of GO and composites-FT-IR analysis
FTIR spectrum of GO-1 and GO-2 (Figure 15) shows peaks corresponding to some characteristic bonds. Broad peaks at 3238 cm−1 correspond to O‒H stretching in the carboxylic groups which are present in both samples of graphene oxide. Peaks at 1708 cm−1 correspond to C = O stretching in carboxylic groups. Adsorption peak at 1620 cm−1 for GO-2 and 1575 cm−1 in GO-1 attributes to C = C aromatic bond, present in cyclic rings of graphene sheets. Peaks at 1126 cm−1 are attributed to C‒O stretching in alkoxy group. Peaks at 1027 cm−1 correspond to C‒O‒C bond.The plot is similar to that of work done by Shouwei Zhang et al and Syed Shahabuddin et al, however, peaks are observed at slightly different wave number which can be attributed to various factors.11,17 FTIR spectrum of GO-1 and GO-2.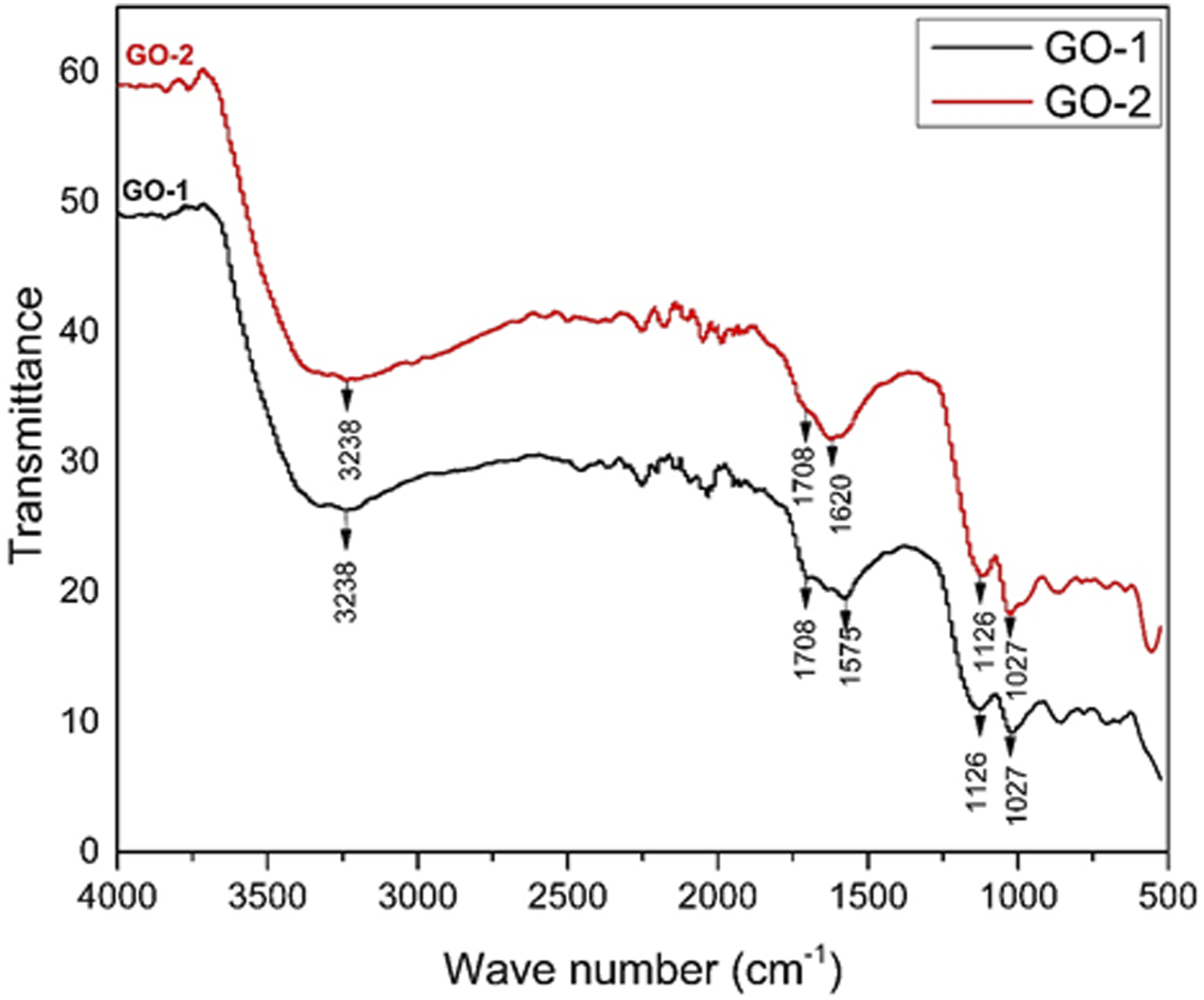
FTIR spectrum for composites of various compositions in Figure 11 shows the disappearance of the broad peak at 3238 cm−1 which was present in GO. This may be attributed to the reaction between ‒OH group in graphene sheets with aniline during polymerization. Similarly, peaks at 1708 cm−1 also disappeared, showing possible bonding between the carboxylic groups in graphene oxide and ‒NH2 group of aniline during polymerization. Peaks at 1288 cm−1 and 1233 cm−1are attributed to C‒N stretching which are characteristic peaks for PANI. Peaks at 1554 cm−1 and 1402∼1490 can be attributed to C = C/C‒C stretching of quinoid-rings and benzenoid-rings respectively.
Structural analysis-XRD
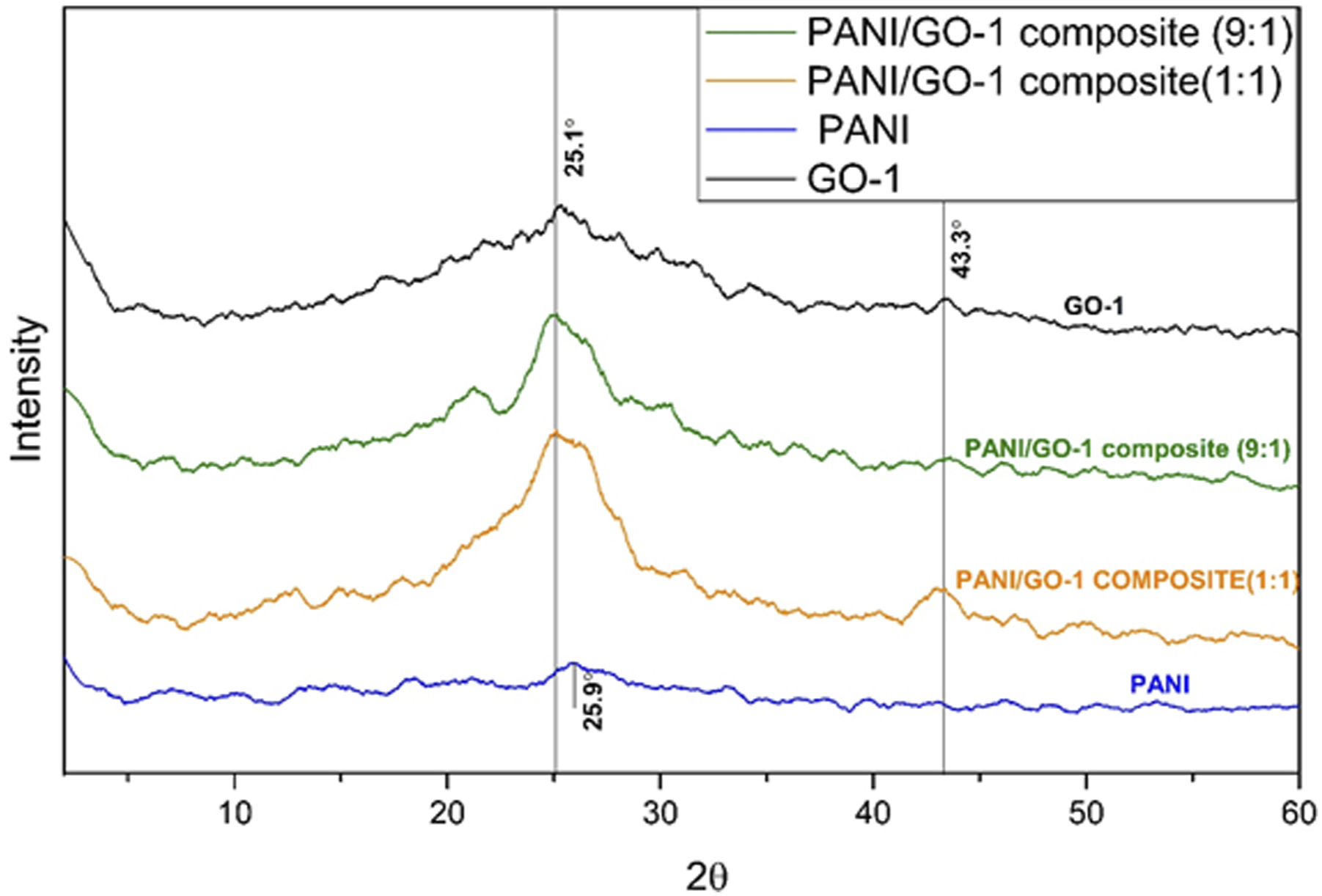
In Figure 12 XRD pattern of GO-1, Polyaniline and graphene oxide in comparison to composite shows good interaction between GO and PANI due to appearance of broad peaks centred around 43.3° indicating the presence of graphite sheets. 24 Peaks centred around 25.5° indicates the presence of PANI which is also shown at peak 25.9° in the PANI peak. 14 The peaks at 25.9° shifts to a lower angle of 25.1° which may be due to the interaction between the PANI crystals and graphite sheets. Using Bragg’s equation, the space between crystal planes was found to be 0.35 nm for composites.
Morphology analysis of GO and composite-SEM
Morphology of composites was studied by Scanning Electron Microscopy. Morphology of two composites with 50% and 10% GO loading is shown in Figures 15(a)–(d) and 16(a)–(d) respectively. Graphene Oxide sheets are visible in all these composites, However, the sheets of GO in the case of 50% loading are visible prominently. In the case of 10% loading, PANI content is higher, which is evident from the smaller sheets of GO and higher porosity. Composites with 50% loading of GO has lower porosity level. GO sheets are joined together by the mesh-like structure of PANI which are visible in Figure 15(2)a, 15(2)b, 15(2)c, 15(2)d containing composite with 90% of PANI content. SEM images of composites with 9:1 polymer to GO ratio.
Adsorption of chlorpyrifos-GC-MS analysis
Water contaminated with chlorpyrifos had a characteristic milky-white color that changed to a transparent liquid after going through the composite, suggesting that the pollutant may have been remedied in the composite structure depicted in Figure 17. The purification method is shown in Figure 17, and the potential interactions between the chemical groups in the composite are what primarily drive the purification capabilities. The chlorpyrifos molecule can form hydrogen bonds with the amine group and hydrogen atoms in the chloropyrifos in addition to the π-π staking, electrostatic contact, and hydrogen bonding found in PANI/GO composite. Furthermore, the nitrogen group in the aniline and the hydroxyl, carboxylic group in GO interact with the sulfur, oxygen, chlorine, and hydrogen groups found in chloropyrifos. Chloropyrifos was eventually adsorbed from water in 99.76% of cases thanks to these robust interactions. Mechanism of adsorption efficiency of PANI/GO composite.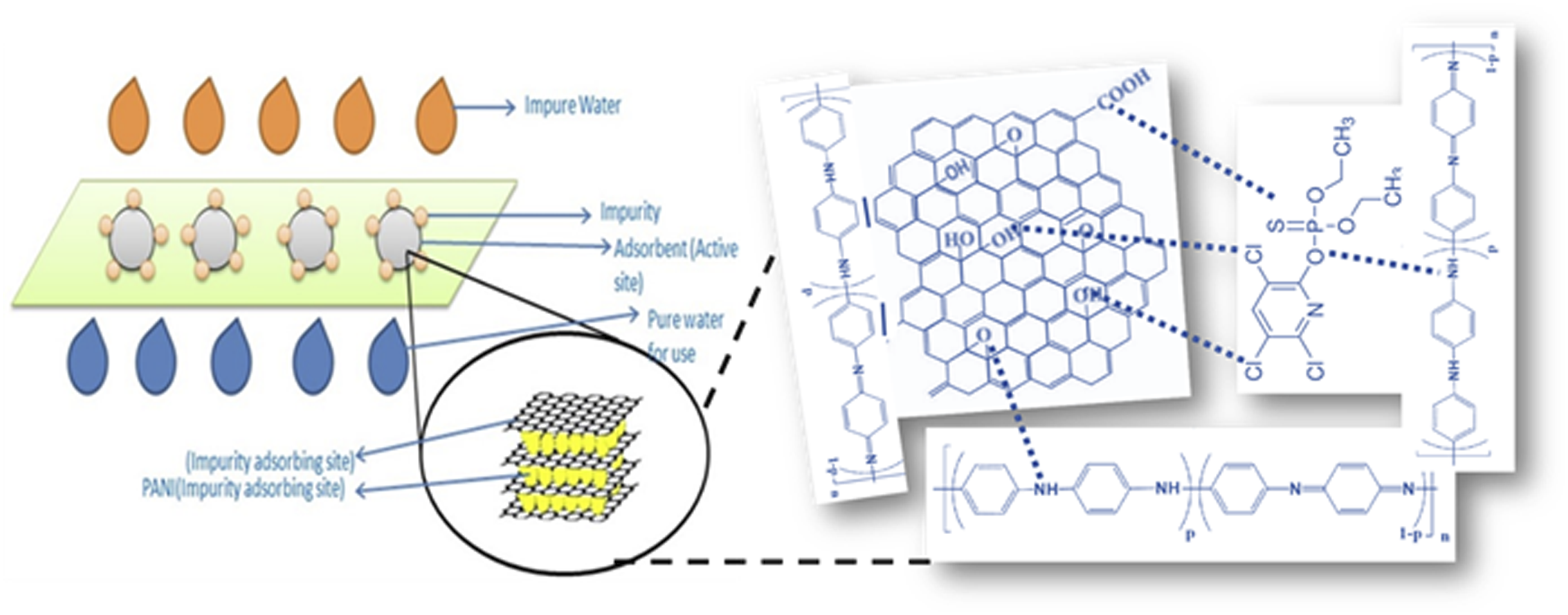
PANI/GO composites have demonstrated a significant improvement in adsorption capacity when compared to their constituent parts. Figure 18 Graphene oxide has a large surface area and a large number of functional groups. Polyaniline has high adsorption capabilities due to its π-conjugated structure.25–27 Picture of polluted water and filtered water.
GC-MS analysis of unfiltered water filtered water from 1:1 composite and 9:1 composite respectively in Figure 19(a)–(c) respectively. The data collected, are compiled in Table 1. (a) GC-MS analysis of polluted water. (b) GC-MS analysis of filtered water from PANI/GO composite (1:1 composition). (c) GC-MS analysis of filtered water from PANI/GO composite (9:1 composition). Water samples and Chlorpyrifos amount detected by GC-MS.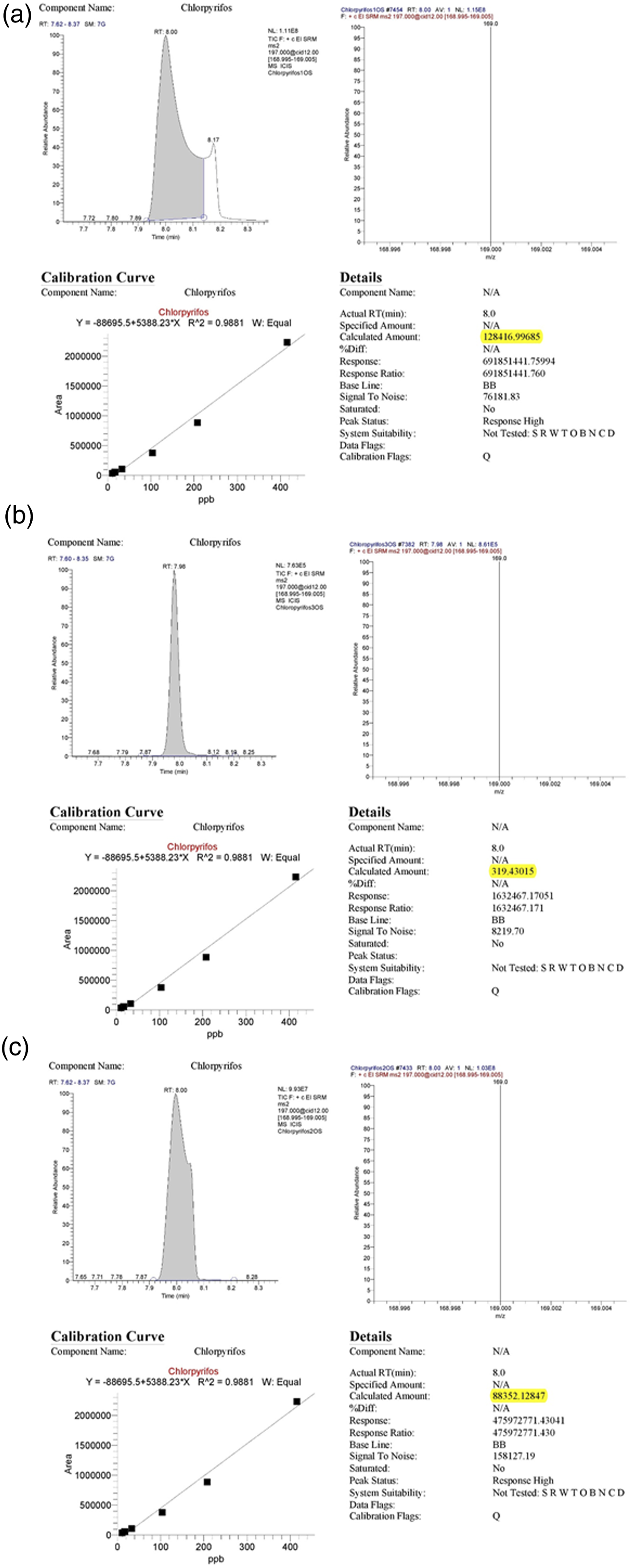

From this analysis, it is evident that composite with 50% loading of GO was able to remove 128,097.5 ppb of chlorpyrifos after filtration. Which is 99.75% of the chlorpyrifos found in the unfiltered water sample. Therefore, we can say composite with 50% GO loading was 99.75% efficient for removing chlorpyrifos.
Comparative analysis of different polymer composites reported for the removal of chlorpyrifos.
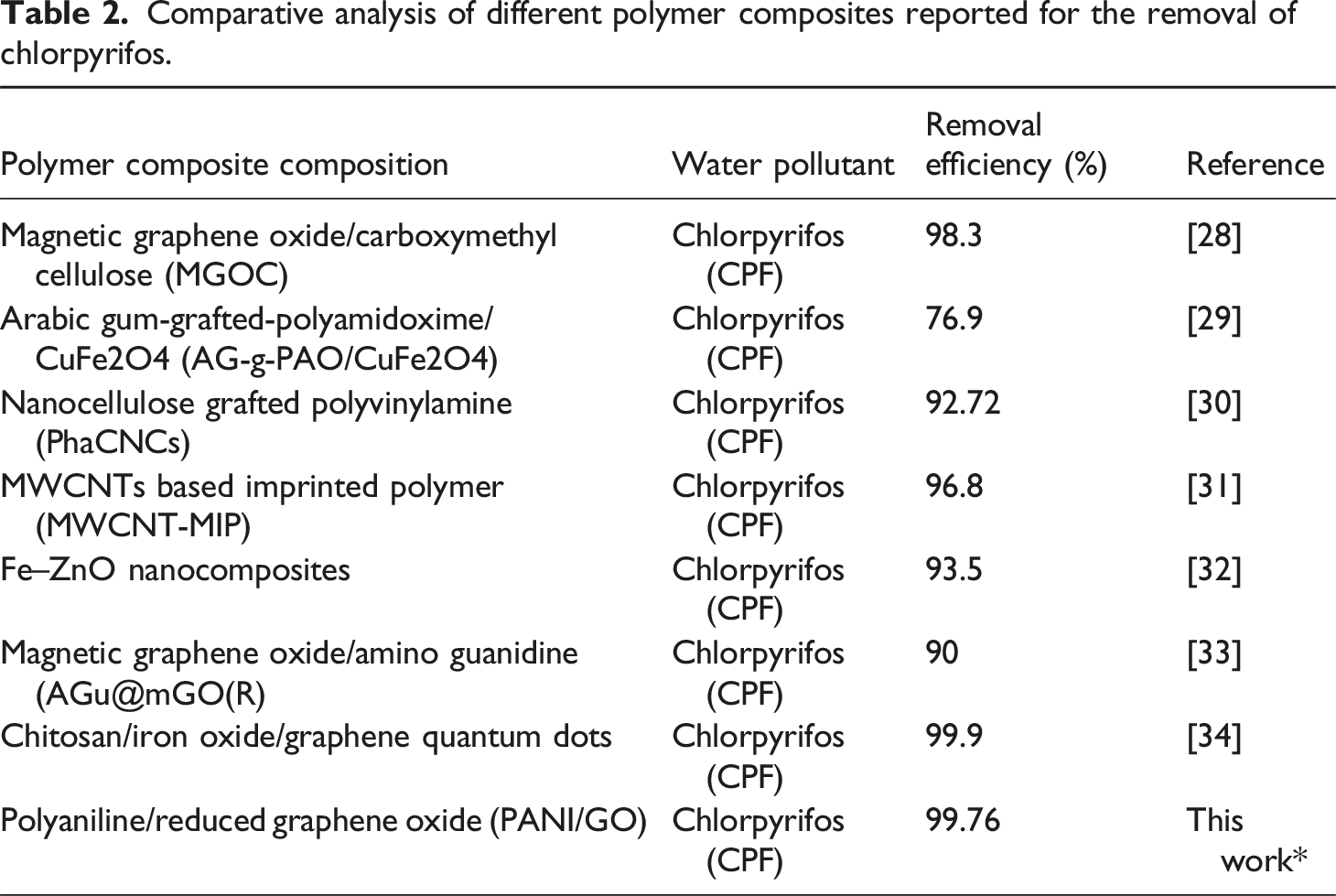
The lower efficiency of the composite with less amount of GO may be due to the lower number of graphene sheets and higher porosity of the composite matrix.
This adsorption property of composite can be attributed to the bulky size of chlorpyrifos which may get trapped inside the matrix of the composite. The presence of π bonds and lone pair containing atoms may get attached to the composite substrate due to interaction between the functional groups present in the composite substrate. π-π interaction may be one of the causes for this behaviour of composite.35,36
Conclusion
In addition to creating composites of polyaniline and graphene oxide (PANI/GO), the current study showed promising potential for treating water contaminated by the organophosphate pesticide chlorpyrifos. Graphene oxide was successfully synthesized by the modified Hummers method with different levels of oxidation. In situ polymerization of aniline in presence of graphene oxide was carried out to get PANI-GO composite. Adsorption experiment for water polluted with chlorpyrifos pesticide was carried out and water samples before and after filtration were analyzed by GC-MS. Polluted water had a milky-white colour, which changed to transparent liquid after filtration indicating adsorption of chlorpyrifos. GC-MS analysis study showed chlorpyrifos amount decreased after filtration for both the cases of composites. PANI/GO-1 composite with a 1:1 ratio was able to remove 99.75% of chlorpyrifos from the polluted water and a composite with 9:1 ratio was able to remove 31.19% of chlorpyrifos. PANI/GO composites show selective adsorption in the direction of chlorpyrifos because of particular interactions between the functional groups on the surface of the composite and the pesticide molecules. These adsorption properties of composites indicate that it can be an excellent filtration material that can be applied for the removal of pesticides or chemicals having similar structures from wastewater. Recycled and repurposed PANI/GO composites may not appreciably lose their adsorption capacity, however the studies are futuristic. The synthesis of the PANI/GO and the water purification is more economical and sustainable, making it suitable for long-term water remediation applications. In addition to chlorpyrifos, PANI/GO composites may be helpful in the adsorption of other organic pollutants and poisons that are present in water, such as heavy metals, dyes, and pharmaceuticals.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 1819-Innovation, technology Development And Deployment; DST/TMD(EWO)/OWUIS-2018/RS13(C).
Data Availability Statement
The data will be available as per request.
