Abstract
In this work, polyvinyl chloride (PVC) and 4-aminobenzoic acid (4ABA) modified PVC and titanium dioxide (TiO2) nanocomposites were prepared by solution blending method. TiO2 was added to PVC (0.5 g) at 0.01, 0.03, 0.05 and 0.1 g. PVC matrix was modified using 4-aminobenzoic acid (4ABA). Fourier transform infrared (FTIR) spectroscopy was used to confirm the modification of PVC with 4ABA. Scanning electron microscopy (SEM) images depicted homogeneous morphology of modified PVC/TiO2 nanocomposites. XRD results confirmed the formation of PVC/TiO2 nanocomposites. Moreover, the thermal decomposition temperature T10 (10 Wt. % weight loss) of non-modified PVC was 189°C however, modified PVC nanocomposites membranes had 196°C with similar TiO2 content. Furthermore, modified PVC-TiO2 with 0.05 g loading showed homogenous morphology with higher hydrophilicity as determined by porosity, solvent content, and shrinkage ratio. Modified PVC/TiO2 membrane with a lower contact angle (32°) could be used for water purification purposes. These studies suggest that optimum modifier concentration improved the interaction between PVC and TiO2.
Keywords
Introduction
Recently, polymer nanocomposites-based membranes have attracted enormous attention. A large number of polymers, including polyvinylidene fluoride (PVDF) and polyethersulfone (PSF) have been used in membranes for water purification because of their better chemical stability, high mechanical strength, and hydrophilicity.1-4
Polyvinyl chloride (PVC) is one of the most widely used polymers after polyethylene (PE) and polypropylene (PP). The properties of PVC can be altered by adding small content of plasticizer to provide elasticity and lower glass transition temperature.5,6 A plethora of reports have so far been communicated regarding the modification of PVC. Of particular interest is the case of dichlorination, which has been reported at 260°C by thermal degradation.7,8 Low thermal stability is overcome by adding selected additives such as plasticizers, stabilizers, and nanofillers. Moreover, the blending of PVC with other nanofillers has also been shown improved mechanical strength, processability, and thermal stability.9-12 However, the application of PVC in water purification is restricted without its modification and composites formation with nanofiller due to its hydrophobicity which leads to declining the permeability.13,14 Recently, Esmaeili et al. comprehensively investigated the effect of surfactants on the impact dynamics of water droplets and developed a fast and low-cost method for the fabrication of micro/nano-textured superhydrophobic surfaces.15,16 Modifying polymer matrix is an effective strategy to improve nanofiller dispersion, mitigate biofouling, and enhance hydrophilicity. 13 The problem with neat PVC arises from its high hydrophobicity, creating more shrinkage and affecting the membrane porosity, reducing the pure water flux.
The blending of PVC with inorganic nanoparticles has been reported, and several types of metal oxide nanoparticles such as SiO2, Al2O3, Fe3O4, and ZrO2 have been incorporated into membranes.17-20 Vatanpour et al. 21 improved water flux by introducing Graphene quantum dots (GQDs)/PVC nanocomposites membranes. Etemadi et al. 22 synthesized PVC/Al2O3 nanocomposites to reduce the irreversible fouling behaviour of membranes. Aji et al. 23 prepared PVC/Cellulose acetate nanocomposites membranes to overcome the hydrophobic nature of PVC in membrane fabrication. The membrane properties were assessed by pure water flux (PWF) and bovine serum albumin (BSA) rejection, respectively. The outcome showed that the membrane had a PWF of 85 L/m2 h, BSA rejection of more than 90% for the neat PVC membrane was recorded. In our previous works, we prepared modified PVC with graphene oxide (GO) to develop a membrane for water purification and realized good thermal stability, high water flux and water content, and low contact angle.24,25 However, expensive chemicals and multi-step preparation of these composite membranes limit its practical application. For the foreseeable future, membranes must demonstrate improved characteristics and provide a cost-effective, simple, and safe route for developing countries.
In this work, 4-aminobenzoic acid (4ABA) as a source of the amino-functional group is used for PVC modification. One PVC/TiO2 and three PVC-4ABA/TiO2 series were prepared with nano-filler loading at 0.01, 0.03, 0.05, and 0.1 g per 0.5 g PVC. Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), X-ray diffraction (XRD), and thermogravimetric analysis (TGA) were used to analyze the functionality, surface morphology, crystallinity, and thermal stability of nanocomposites membranes, respectively. The membranes properties such as porosity, solvent contents, shrinkage ratio were measured by using appropriate formulas, while contact angles were measured by Drop Shape Analyzer (DSA100).
Experimental
Material
PVC (Mw - weight-average molecular weight ∼233,000), (Mn -number-average molecular weight ∼99,000), TiO2 (P25- Titanium dioxide nanoparticle average size ∼25 nm), 4-aminobenzoic acid (4ABA, 99.99%), pyridine (99%), tetrahydrofuran (THF, 99.5%) were purchased from Sigma Aldrich. Ethanol (C2H5OH, 99%), methanol (CH3OH, 99%), and ethyl acetate (C4H8O2, 99%) were obtained from Fluka.
Instrumentation techniques
Infrared (IR) spectra were recorded using Fourier transform infrared (FTIR) spectrometer, Model No. FTSW 300 MX, manufactured by BIO-RAD, California, USA (4 cm−1 resolution). Field emission scanning electron microscopy (FE-SEM) of freeze-fractured samples was performed using JSM5910, JEOL Japan. Thermal stability was verified by METTLER TOLEDO TGA/DTA thermogravimetric analyzer using 5 mg of the sample in Al2O3 crucible at a heating rate of 10 °C/min. X-Ray diffraction (XRD) study was carried out on Shimadzu XRD-6000 using crystal monochromatic Cu Kα radiation at a scanning rate of 8°/min. To analyze porosity, dry membrane weight was measured. Then, the membrane was soddened in distilled water for 24 h followed by mopping with blotting paper and weighing. Porosity was calculated using the following equation (1).





Surface wettability (contact angle) was measured by drop shape analyzer (DSA 100). A small water drop (one μL) was placed on the surface of the membranes, and several images were taken. Solid-liquid extraction tests were carried out using neat polyimide and nanocomposites membranes using chloride salts of Pb2+, Hg2+, Cd2+, Co2+, Fe3+, Ni2+, and Cu2+. The concentration of each cation in the liquid phase was determined after filtration by inductively coupled plasma mass spectrometry (ICP-MS, Agilent 7500). Repeated dilutions of sample aliquots with pure water/nitric acid (5 %v/v) were made to reach concentrations in the range of calibration curve (0–40 ppb), and extraction percentages of metal ions were collected directly. The extraction of environmentally toxic heavy metal ions was investigated in terms of distribution coefficient (Kd′) and solid-liquid extraction selectivity (αS, L). Accordingly, Kd′ is the measure of the capacity of a material to extract cations under competitive conditions.

Modification of PVC
For PVC modification, 0.5 g PVC and 10 mL THF were mixed in a round bottom flask. Three different formulations were prepared by adding 4ABA at 0.05, 0.1, and 0.3 g with five drops of pyridine to the mixture and stirred for 2 h at room temperature. Then, reflux was made at 80°C for 6 h. Finally, the films were cast in Petri dishes and dried at room temperature. Figure 1 shows the modification process of PVC with 4ABA. PVC modification with 4-aminobenzoic acid (4ABA).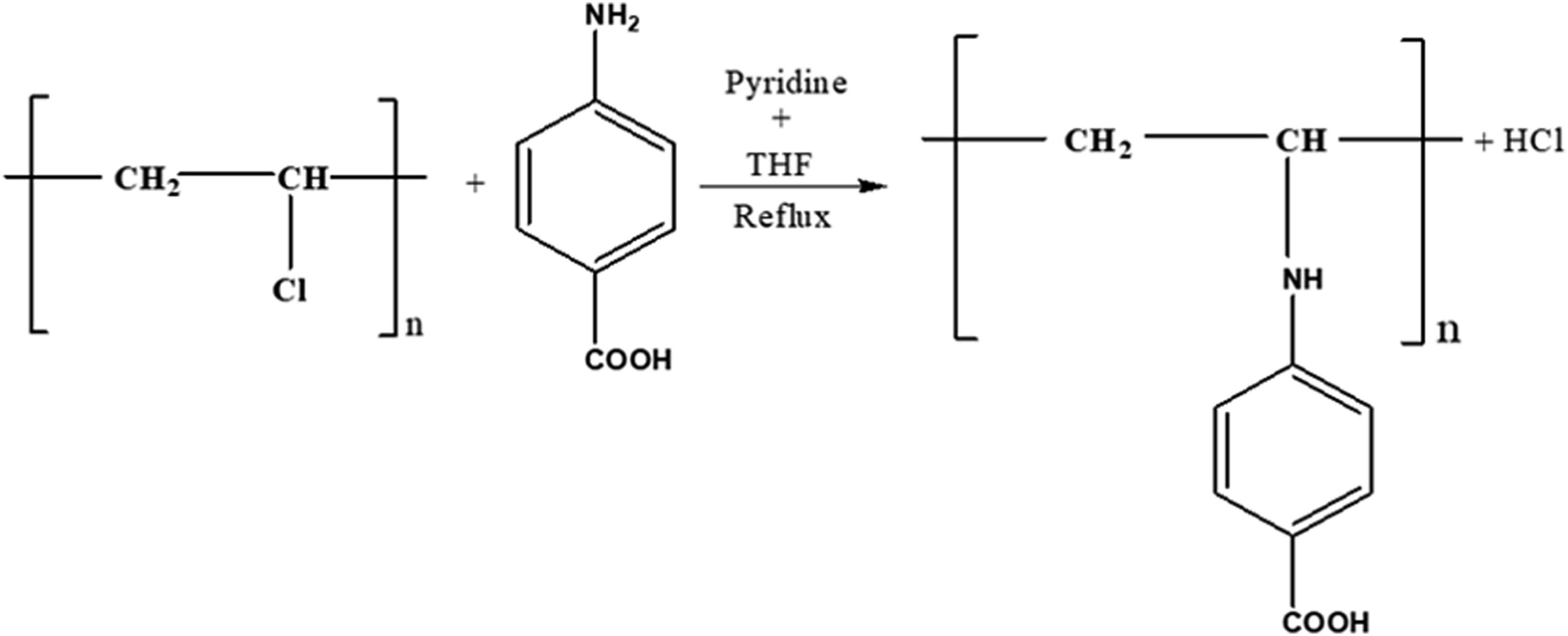
Preparation of PVC/TiO2 nanocomposites membranes
For PVC/TiO2 composites preparation, 0.5 g PVC was dissolved in 10 mL THF at room temperature for 2 h. The desired TiO2 contents such as 0.01, 0.03, 0.05, and 0.1 g were added to the solution. The mixture was refluxed at 80°C for 6 h with continuous stirring. The evenly dispersed solution was poured into Petri dish. Finally, the membranes were obtained after 12 h drying at room temperature. Figure 2 illustrates the chemical route for PVC/TiO2 nanocomposites membranes. Preparation of PVC/TiO2 nanocomposite membranes.
Preparation of PVC-4ABA/TiO2 nanocomposites membranes
For PVC modified nanocomposites, 0.5 g polymer was dissolved in 10 mL THF with constant stirring at room temperature for 2 h. Then, PVC was modified using three different 4ABA contents such as 0.05, 0.1, and 0.3 g according to the procedure mentioned in the PVC modification section. The desired TiO2 content (0.01, 0.03, 0.05, and 0.1 g) was added to the mixture, and then it was refluxed at 80°C for 6 h with constant stirring. The membranes were cast by pouring the dispersed solution into Petri dishes and drying for 12 h at room temperature. Figure 3 illustrates the chemical route for the preparation of PVC-4ABA/TiO2 nanocomposite membranes. Preparation of PVC-4ABA/TiO2 nanocomposite membranes.
Results and discussion
FTIR analysis
The structure and bonding of as-prepared nanocomposites were investigated using FTIR spectroscopy. The FTIR spectra of PVC/TiO2-0.01, and PVC/TiO2-0.05 nanocomposites in Figure 4 represent the presence of band at 2914 cm−1, which indicates the aliphatic stretching vibration of C-H bonds. The band at 1492 cm−1 represents C-H scissoring vibration. The deformation vibration of H-C-Cl or C-H bond is observed at 1308 cm−1. The peak detected at 794 cm−1 and 603 cm−1 is assigned to the C-Cl and Ti-O-Ti stretching vibrations, respectively. FTIR spectra of PVC/TiO2 0.01, and PVC/TiO2 0.05 nanocomposites.
The FTIR spectra of PVC-4ABA-0.05/TiO2-0.01, PVC-4ABA-0.05/TiO2 0.05 nanocomposites are given in Figure 5. The bands observed at 3001 cm−1 and 2909 cm−1 indicate the aromatic and aliphatic stretching vibration of C-H, respectively. Moreover, a C=C stretching peak appeared at 1677 cm−1. The peak at 1429 cm−1 is due to the C-N stretching, which confirms the modification of PVC. The band at 1251 cm−1 and 1162 cm−1, related to C-O, C-C aliphatic stretching vibration of the main chin of PVC, and a peak at 609 cm−1 is assigned to Ti-O-Ti stretching vibration. FTIR spectra of PVC-4ABA-0.05/TiO2 0.01, and PVC-4ABA-0.05/TiO2 0.05 nanocomposite.
Morphological studies
SEM images of PVC/TiO2-0.01 and PVC/TiO2-0.05 nanocomposites membranes are shown in Figure 6. The agglomeration of TiO2 nanoparticles in unmodified PVC can be observed with feeble adhesion between TiO2 and PVC matrix due to weak interaction and less interface incompatibility between the nanoparticles and polymer. This non-homogeneous morphology can be attributed to the lack of interaction between the nanofiller and polymer matrix. SEM images of PVC/TiO2 0.01 (A, B) and PVC/TiO2 0.05 (C, D) nanocomposite membranes.
The SEM images of PVC-4ABA-0.05/TiO2-0.01 in Figures 7(a) and (b) show an excellent dispersibility of TiO2 nanoparticles without any huge phase separation in the polymer matrix and indicate better homogeneous morphology. The significant accretion on the surface was observed at small nanoparticles content in the polymer matrix due to the active size for interaction with the polymer, which hinders the viscosity increment. Figures 7(c) and (d) illustrates the SEM images of PVC-4ABA-0.05/TiO2-0.05. It indicates that nanofiller is completely embedded in the polymer matrix as a result, a uniform and homogenous morphology was achieved. The better morphology of TiO2 nanoparticles was due to small filler loading, which can possess a sizeable surface-active area for interaction with the modifier and polymer moieties. These results indicate that TiO2 nanoparticles have better interaction with modified PVC than unmodified polymer networks. SEM images of PVC-4ABA 0.05/TiO2 0.01 (A, B), PVC-4ABA 0.05/TiO2 0.05 (C, D) nanocomposite membrane.
Thermal analysis
Thermal stability of nanocomposites was studied by thermogravimetric analysis (TGA), and the obtained thermograms are given in Figure 8. It can be seen that initial decomposition temperature (To) for all three samples (PVC/TiO2-0.05, PVC-4ABA-0.05/TiO2-0.05, and PVC-4ABA-0.05/TiO2-0.1) was observed at 99°C, which is due to loss of moisture and volatile impurities. For PVC/TiO2-0.05, 10% weight loss temperature (T10) was 189°C. The obvious enhancement was observed at 196°C and 205°C for PVC-4ABA-0.05/TiO2-0.05 and PVC-4ABA-0.05/TiO2-0.1, respectively. The neat PVC used in this study has T10 equal to 178°C, i.e. lower than its composites, given that the increased filler loading and modified PVC composites were highly stable against heat. TGA curves of neat PVC, PVC/TiO2 0.05, PVC-4ABA-0.05/TiO2 0.05, and PVC- 4ABA-0.05/TiO2-0.1 nanocomposites.
When temperature was further increased, 50% weight loss was observed in the range of 256°C in PVC/TiO2-0.05 while it increased to 264°C and 305°C for PVC-4ABA-0.05/TiO2-0.05 and PVC-4ABA-0.05/TiO2-0.1, respectively. Moreover, the novel modified PVC nanocomposites showed better thermal stability than neat PVC and unmodified PVC composites.
XRD analysis
Figure 9 shows the XRD patterns of neat PVC, TiO2 nanoparticles, and its composites series with different contents of TiO2 nanofiller. In the given series, the amorphous nature of PVC was observed at 2θ=25. Figure 9(b) shows the crystalline nature of TiO2 at 2θ=25.5 (101), 2θ=37.8 (004), 2θ=47.9 (200), 2θ=53.5 (105) and 2θ=62.6 (204). All the peaks in the XRD patterns can be indexed as anatase phases of TiO2, and the diffraction data were in good agreement with JCPDS No#21–1272 (anatase TiO2). The crystallite size of TiO2 was obtained (26 nm) by the following Scherrer formula, D=Kλ/(βcosθ). The loading of TiO2 nanoparticles changed the amorphous behavior of the PVC membrane. The diffraction peaks of PVC/TiO2-0.05 and PVC/TiO2-0.1 composite membrane were appeared at around 2θ = 25.8°, 38.5°, 48.2°, 54.5°, and 63.3°. The broad peak intensity was increased in nanocomposites with increasing TiO2 content from 0.05 g to 0.1 g. The peak slightly shifted and became broadened, revealing the existence of strong interactions between polymers and TiO2 nanoparticles. The small and broad peaks of PVC-4ABA-0.05/TiO2-0.05 and PVC-4ABA-0.05/TiO2-0.1 composite membrane were appeared at around 2θ = 25.5°, 38.8°, 48.4°, 54.7°, and 63.5° due to the low content of TiO2 (0.05) in the polymer matrix. In contrast, the intensity of diffraction peak increased for 0.1 g TiO2 which is due to better crystallinity. The increase in peak intensity from 0.05 to 0.1 g is not prominent as PVC/TiO2, which showed that PVC-4ABA/TiO2 had a less crystalline nature than the PVC/TiO2 owing to the strong interaction of TiO2 with modified PVC matrix, which diminishes the crystalline nature. Hence, it can be revealed that TiO2 is strongly embedded or dispersed in modified PVC than unmodified polymer. XRD pattern of PVC, TiO2, PVC/TiO2-0.05, PVC/TiO2-0.1, PVC-4ABA-0.05/TiO2 0.05, and PVC-4ABA-0.05/TiO2-0.01 nanocomposites.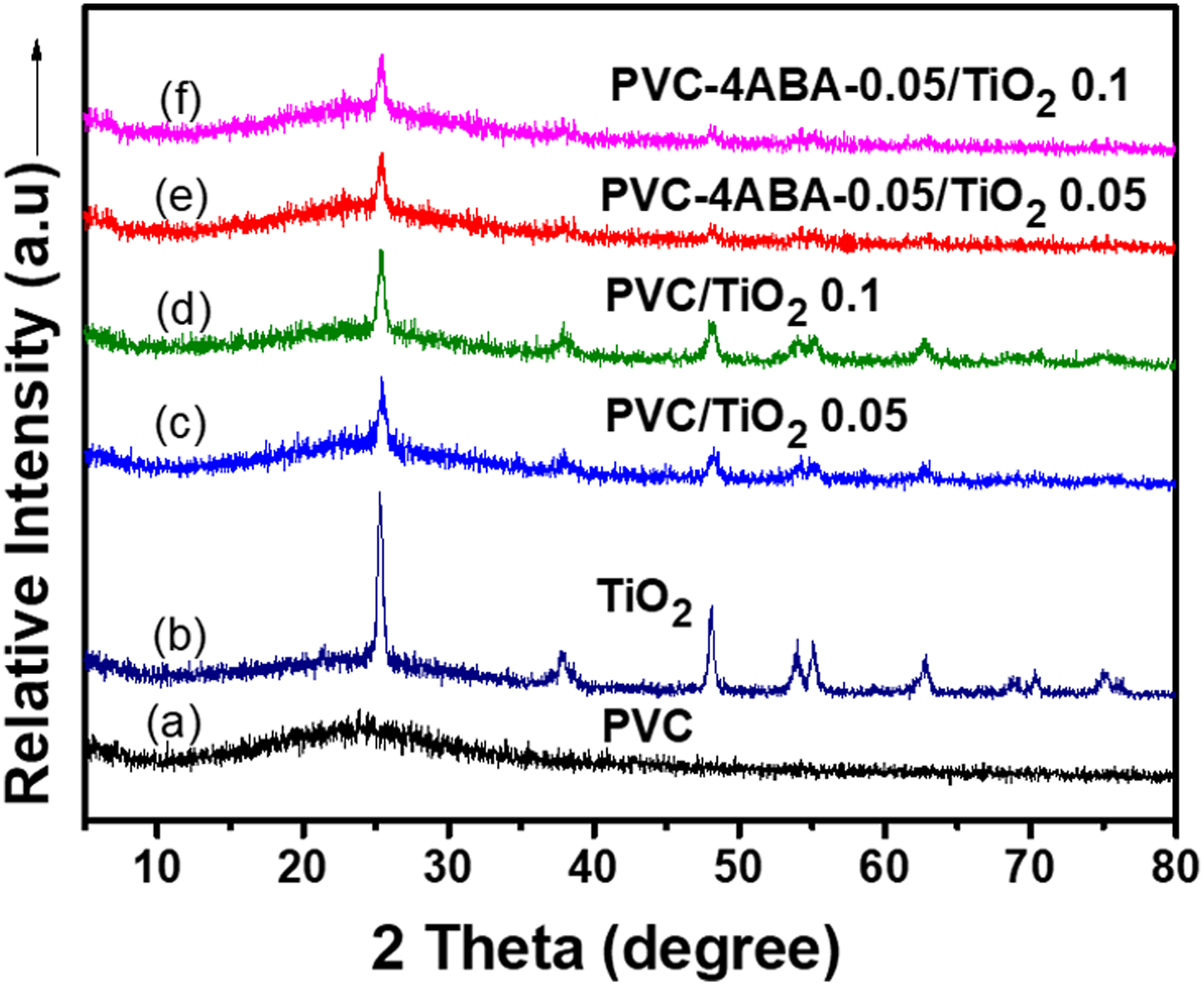
Membrane properties
The properties of membranes were studied to investigate if they are suitable as a novel membrane for water purification. Porosity was measured by respective formulas, as reported in the literature.24-26 Membranes have physical spaces in their structure called “pores”. The dimension of these pores can be in the range of micron to a nanometer scale. Transport of fluid through membranes strongly depends on membranes properties and fluid hydrodynamics.
Sample code and composition of nanocomposites.

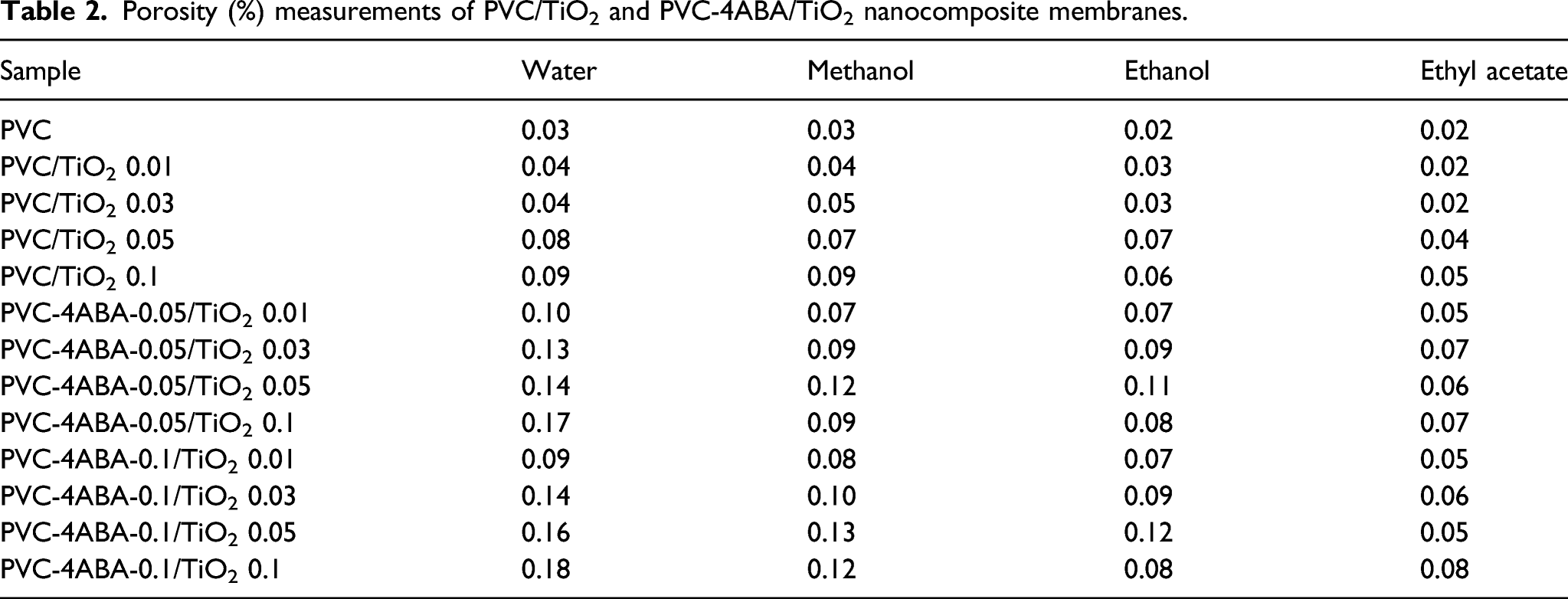
Porosity (%) measurements of PVC/TiO2 and PVC-4ABA/TiO2 nanocomposite membranes.
Solvent content measurements of PVC/TiO2 and PVC-4ABA/TiO2 nanocomposite membranes.

Shrinkage ratio measurement of PVC/TiO2 and PVC-4ABA/TiO2 nanocomposite membranes.
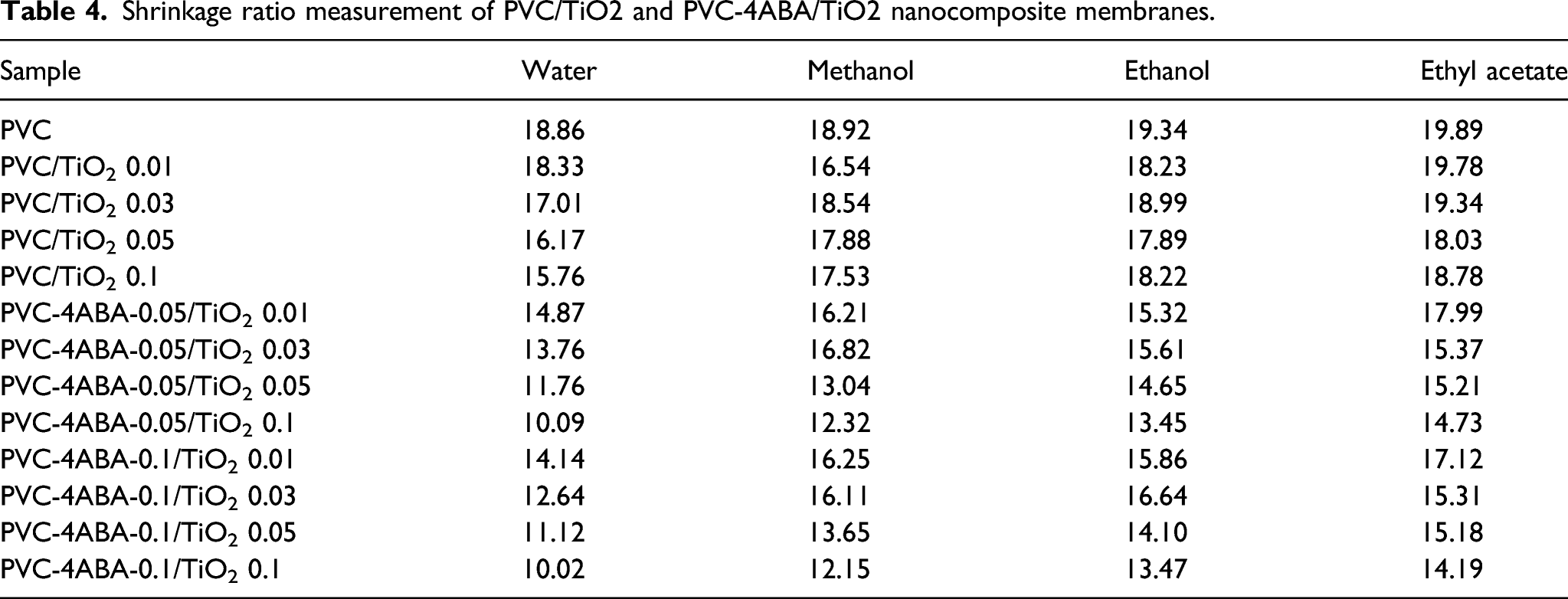
For PVC-4ABA-0.05/TiO2-0.01, the shrinkage ratio measured using water as solvent was 14.87, which decreased with TiO2 loading to 10.09 in PVC-4ABA-0.05/TiO2 0.1. It is also observed that the shrinkage ratio was increased when shifted from water to ethyl acetate. The shrinkage ratio of PVC/TiO2-0.01 were 18.33 (water),16.54 (methanol), 18.23 (ethanol) and 19.87 (ethyl acetate). Also, the shrinkage ratio of PVC-4ABA-0.05/TiO2-0.01 were 14.87 (water), 16.21 (methanol), 15.32 (ethanol) and 17.99 (ethyl acetate). Consequently, membranes with a low shrinkage ratio, higher solvent content, and porosity are the best choice for water purification. Contact angle is the measure of the hydrophilicity of membranes. The higher the contact angle, the lesser will be the hydrophilicity.
Contact angle measurements of neat PVC, PVC/TiO2, and PVC-4ABA/TiO2 nanocomposite membranes.


Optical micrograph of water contact angle on the surface of PVC (A), PVC/TiO2 0.01 (B), PVC/TiO2 0.05 (C), PVC/TiO2 0.1 g (D) and PVC-4ABA-0.05/TiO2 0.05 (E), PVC- 4ABA-0.1/TiO2 0.05 (F), PVC-4ABA-0.3/TiO2 0.1 g (G) nanocomposite membranes.
Solid-liquid extraction selectivity (αS,L) and distribution coefficient (Kd′) of membranes were tested27, 28 and % E for PVC-4ABA 0.05/TiO2-0.1 was found up to 75% for extraction of Cd2+, Co2+, Ni2+, Fe3+. The high extraction level (100%) of Pb2+ was due to the mutual effect of PVC modified with amino-functionalized and filler. Overall, the attained results revealed the effectiveness of such membrane for removal of selective cation decontamination of water on an industrial scale.
Conclusions
PVC/TiO2 and PVC-4ABA/TiO2 nanocomposites were successfully prepared by a facile solution blending technique using TiO2 as the nanofiller at the loading of 0.01, 0.03, 0.05, and 0.1 g per 0.5 g PVC. The 4-ABA modifier was used to modify the PVC. FTIR analysis confirmed the modification of PVC. Moreover, the SEM images showed the homogenous morphology of modified PVC/TiO2 compared to un-modified PVC/TiO2 nanocomposites. XRD analysis explained that the modified PVC with 4ABA has a better interaction than unmodified PVC/TiO2 nanocomposites. TGA confirmed the excellent thermal stability of modified PVC. It was found that the properties of the membrane, such as porosity and solvent content values increased with increasing TiO2 loading from 0.01 to 0.1 g and modifier 4ABA. In contrast, shrinkage ratio and contact angle values decreased with TiO2 nanofillers. These findings showed that the amino-functionalized PVC/TiO2 nanocomposites could work as nanofiltration membranes for water purification.
Footnotes
Acknowledgements
The authors acknowledge the support provided by Nanoscience and Technology Department, National Centre for Physics (NCP).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

