Abstract
The aim of this study was to explore the preparation of porous nanocomposite scaffolds with simultaneous osteogenic – antibacterial properties by incorporating copper – doped bioactive glass nanoparticles into Poly (D,L-lactide-co-glycolide) lactide:glycolide. Bioactive glass nanoparticles were synthesized by using sol–gel technique from the SiO2 – P2O5 – CaO – Na2O – CuO system. Poly (D,L-lactide-co-glycolide) lactide:glycolide nanocomposite scaffolds with different nanoparticle contents were prepared by combined lyophilization/salt leaching. The in vitro bioactivity of the scaffolds was assessed in simulated body fluid, and cell viability and osteogenic differentiation assays were performed with stem cells. Antibacterial activity of the materials was assessed against
Keywords
Introduction
Efforts are continuously being made in the field of biomaterials and tissue engineering to find well-designed biomaterials for bone tissue regeneration. 1 Bioactive nanocomposite scaffolds prepared by incorporating bioactive ceramic nanoparticles into biodegradable polymeric matrices appear as interesting materials that combine the bioactivity of the inorganic nanoparticles with the supporting properties of the 3D polymeric matrix, which promote cellular attachment, and growth, and mineral deposition. 2 Recently, one of the most appealing approaches to design bone repair biomaterials is to combine in a single multifunctional biomaterial several abilities. 3 Bone healing is also substantially hindered when infection occurs, mainly after an open fracture. Also, in bone reconstruction surgery, osteomyelitis caused by bacterial infection is a severe and ever-present complication. 4 Another major cause of the infected bone defect is chronic bacterial infections, such as periodontitis. 5
Consequently, the development of bioactive materials with antibacterial properties would represent a valuable solution in preventing bone-related infections. Copper is an element that exhibits antimicrobial activity against several microorganisms, including bacteria, fungi, and viruses.
6
Besides, copper has antifouling property by decreasing the adherence of microorganisms and enhances proliferation of endothelial cells and angiogenesis in vivo.
7
The incorporation of copper ions into the bioactive glass structure is being explored as a strategy to generate bifunctional biomaterials with osteogenic-antimicrobial properties. Wu et al.
8
prepared microsized copper – containing mesoporous bioactive glass (MBG), which promoted the expression of osteogenic markers and inhibited the viability of
Experimental
Materials
Poly (D,L-lactide-co-glycolide) lactide:glycolide (PDLLA) (50:50), average molecular weight 406,000 g/mol, inherent viscosity 2.0 dL/g, and density 1.26 g/cm3 was purchased from Corbion-Purac Biochem (Gorinchem, Netherlands) and used without further processing. Tetraethylorthosilicate 98% (TEOS, Si(OC2H5)4, CAS78-10-4), calcium nitrate tetrahydrate (Ca(NO3)2·4H2O, CAS13477-34-4), ammonium phosphate monobasic (NH4H2PO4, CAS7722-76-1), sodium nitrate (NaNO3, CAS7631-99-4), 4-nitrophenyl phosphate disodium salt solution (pNPP, CAS 4264-83-9), and Bicinchoninic Acid Kit for Protein Determination (BCA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Copper nitrate trihydrate (Cu(NO3)2·3H2O, CAS10031-43-3), ammonia solution 28–30% (NH4OH, CAS1336-21-6), nitric acid (HNO3, CAS7697-37-2), sodium chloride (NaCl, CAS7647-14-5), sodium hydroxide (NaOH, CAS1310-73-2), magnesium chloride (MgCl2, CAS7786-30-3), and Tris (hydroxymethyl)aminomethane GR for analysis buffer (TRIS-buffer, CAS77-86-1) were purchased from Merck (Darmstadt, Germany). Dulbecco’s modified Eagle medium (Alpha-MEM) was purchased from Invitrogen Life Technologies (Carlsbad, CA, USA). 10% Gibco™ fetal bovine serum (FBS), 100 U penicillin, and 100 mg/mL streptomycin were purchased from Thermo Fisher Scientific (Asheville, USA). 3-(4,5- dimethyl-thiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H- tetrazolium] (MTS) and an electron coupling reagent (phenazine ethosulfate; PES) (CellTiter Aqueous One Solution cell proliferation assay kit) was purchased from Promega (Madison, WI, USA). Brain Heart Infusion (BHI) with 6.5% sodium chloride and BHI Agar were purchased from Becton Dickinson (Mountainview, CA, USA). Polysorbate 80 surfactant was purchased from Difco Laboratories (Tween® 80, Detroit, MI, USA).
Synthesis of nanoparticles
nBG nanoparticles with a molar composition of 60SiO2: 25CaO:11Na2O: 4P2O5 (mol %) were prepared by the sol-gel method as out as follows: a calcium-based solution was prepared by dissolving Ca(NO3)2 × 4H2O and NaNO3 in distilled water at room temperature. A second solution was prepared by diluting TEOS in ethanol, and it was added to the calcium nitrate solution, and the pH of the resulting solution was adjusted to 1–2 with nitric acid. This transparent solution was slowly dropped under vigorous stirring into a solution of NH4H2PO4 in 1500 mL of distilled water. During the dripping process the pH was kept at around 10 with aqueous ammonia. The mixture was stirred for 48 h and aged for 48 h at room temperature. The precipitate was separated by centrifugation (12,000 rpm) and washed by three centrifugation-redispersion cycles with distilled water. This suspension was freeze dried and then calcined at 700°C for 3 h to obtain a fine white nBG powder. To prepare Cu-nBG nanoparticles 1 or 2 mol of calcium was replace in first solution by Cu(NO3)2.
Preparation of nanocomposite scaffolds
The PDLLA nanocomposite scaffolds were fabricated using solvent casting-particulate leaching and freeze dryer processes from mixtures of PDLLA and nanometric bioactive glass. Briefly, PDLLA was dissolved in DMC solvent under magnetic stirring at room temperature for 2 h, using a polymer solvent at a ratio of 8% (w/v). Then, the nanoparticle powders (5 or 10 wt.%) were added to the polymer solution, and the resulting mixture was stirred for 30 min and sonicated for 15 min to ensure a homogeneous distribution of the glass particles in the polymer solution.
Then, NaCl particles sieved between 100 and 400 μm was added to the suspension in a ratio of 9:1 and stirred for 30 min. This mixture was then transferred to a Teflon dish and maintained at −80°C for 3 h before freeze dryer for 24 h. After that, the scaffolds left immersed in sterilized double-distilled water for 48 h, replacing it every 4 h to dissolve the salt completely and to leave a porous structure. Finally, porous discs of 150 mm in diameter and 4.5 mm in thickness were obtained, and these were dried at 30°C with vacuum for 48 h. For characterization, the scaffolds were cut using double‐edged foil razor blades.
Material characterization
The materials and apatite formation in SBF were analysed by scanning electron microscopy (SEM) in an JEOL JSM-IT300LV microscope equipped with X-ray dispersive energy elemental microanalysis (EDX). Average particle size of nanoparticles was calculated from the arithmetic mean of 20 measurements using the software of the JEOL JSM-IT300LV microscope. Nanoparticle X-ray diffraction (XRD) analysis was carried in a Siemens D5000 X-ray powder diffractometer using CuKα radiation. The chemical structure of the materials was characterized by total attenuated reflectance with Fourier transform infrared spectroscopy (ATR-FTIR) using an Agilent Cary 630 ATR-FTIR spectrometer. Mechanical compression tests of 4 × 4 × 5 mm scaffolds were performed with DEBEN microtest machine (Suffolk, UK) using a 2 N load cell in compression mode. Elastic modulus was obtained as the mean value of measuring four specimens.
The porosity of the scaffolds was estimated using the method of displaced volume. The densities of PDLLA non-porous films (ρmat) and of absolute ethanol (ρe) were measured with a pycnometer. A small piece of scaffold was weighed (Ws) and its volume calculated using Vs = Ws/ρmat. Then, scaffold was immersed for 20 min in absolute ethanol. The excess liquid was removed from the scaffold and the wet weight (Wf) was obtained. The weight (We) and volume (Ve) occupied by the ethanol inside the scaffold’s porous network were calculated from the formula: We = Wf - Ws and Ve = We/ρe, respectively. The porosity percentage of each scaffold was calculated as follows: Porosity (%) = (Ve/(Ve+Vs)) × 100.
In vitro bioactivity assays
The ability of the nanocomposites to induce the formation of apatite was assessed in acellular SBF, which has inorganic ion concentrations like those of human extracellular fluid. The SBF solution was prepared as described by Kokubo et al. 17 using the standard ion composition and pH 7.4. The cylindrical nanocomposite samples (1.0 cm × 0.5 cm) were individually soaked in 50 mL of SBF in polyethylene containers at 36.5°C using a thermostatic bath. After incubation for 7 days, the scaffolds were removed from SBF, rinsed with distilled water, and dried at 60°C.
Cell culture
Stem cells isolated from human dental pulp (DPSCs) were used to evaluate cell proliferation and differentiation at 3, 7, and 14 days of incubation with the scaffold materials. DPSCs were cultured with the scaffolds in Alpha-MEM containing 10% FBS, 100U/mL penicillin, and 100 μg/mL streptomycin in absence of osteogenic supplements. Cell viability was measured using the MTS colorimetric assay (CellTiter 96® Aqueous MTS Reagent, Promega) at 490 nm. The enzymatic activity of alkaline phosphatase (ALP) was measured using the colorimetric ALP reaction buffer (Tris (0.1
Antibacterial activity
The nanocomposite scaffolds were incubated in a 0.5 McFarland
Statistical analysis
Cell results were expressed as mean ± SD, with
Copper release measurements
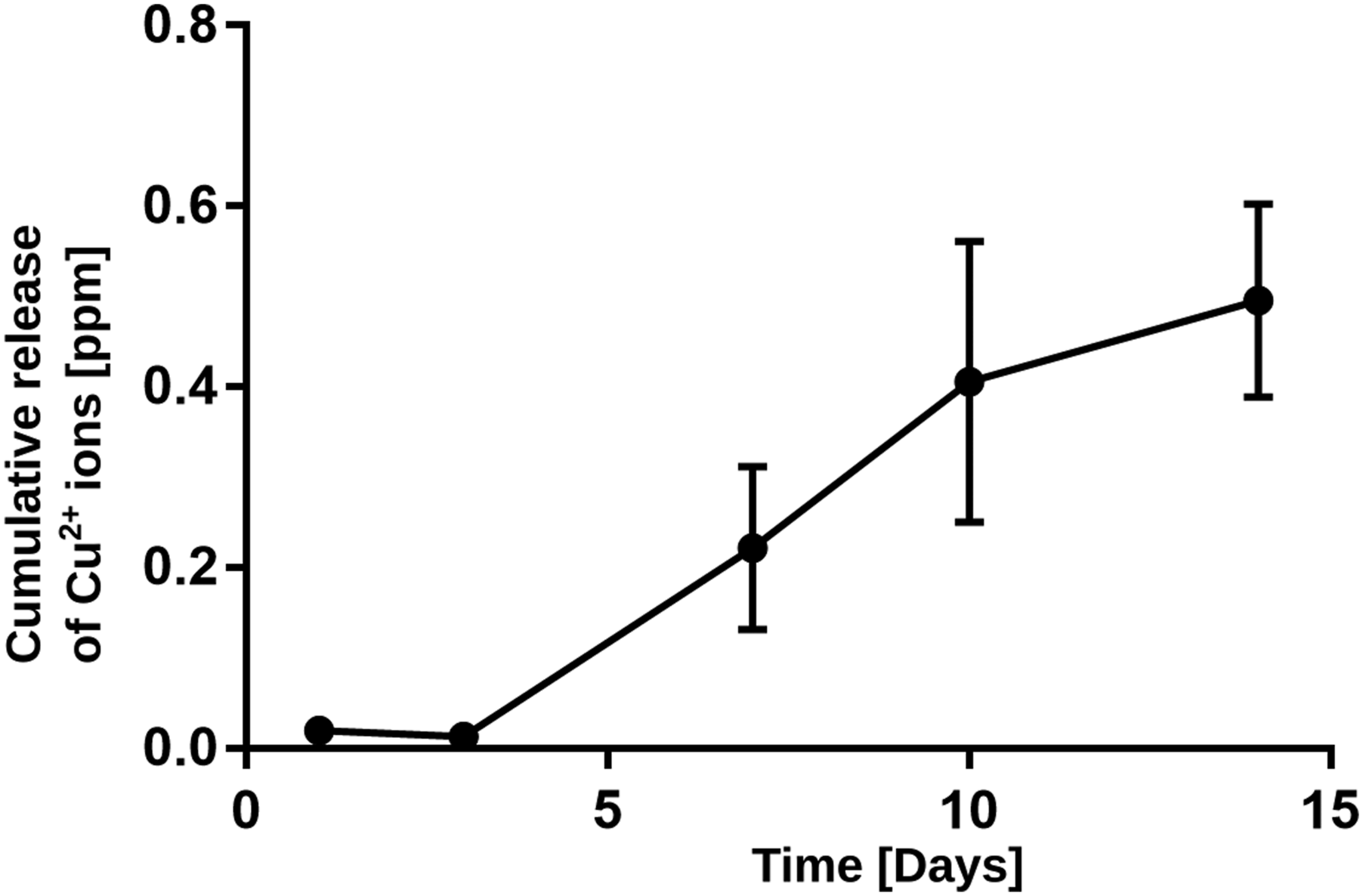
The release of Cu ions from a representative nanocomposite scaffold was measured in phosphate buffered saline (PBS) (pH 7.4) at 37°C. 2 × 2 × 2 cm scaffold piece was immersed in 20 mL of PBS up to 14 days of incubation. The Cu ion concentration in the aqueous phase was analyzed at different time intervals with a Cupric Sulfide/Silver Sulfide Solid – State Combination Ion Selective Electrode (Hanna HI4108).
Results and discussion
Figures 1 and 2 show the structural characterization of the nBG particles and those synthesized in the presence of copper ions. Average particle size of 1Cu-nBG and 2Cu-nBG was of ∼70 nm as estimated from SEM measurements, and their EDX copper contents were of 1.2 and 2.2 wt. %, respectively (Figure 1). XRD patterns of the nanoparticles exhibit reflections associated with structural ordering in the glass. Although nBG with an amorphous structure is typically obtained from the sol-gel synthesis composition SiO2–CaO–P2O5,
15
bioactive glasses with crystalline structures have been observed when the quaternary system 60SiO2: 25CaO:11Na2O: 4P2O5 is used.19,20 Identification of the XRD reflections revealed the presence apatite – related crystalline phases in the nanoparticles (Figure 2), which are also responsible of the FTIR vibrations at 590, 601 and 1100 cm−1 corresponding to the P – O deformations modes of phosphate in apatite.
21
The results also indicate that no changes in the crystal structure of nBG occur after copper incorporation. Scanning electron microscopy images and X-ray dispersive energy elemental mapping of nBG, 1Cu-nBG, and 2Cu-nBG nanoparticles. X-ray diffraction and total attenuated reflectance with Fourier transform infrared spectroscopy analyses of nBG, 1Cu-nBG, and 2Cu-nBG nanoparticles.
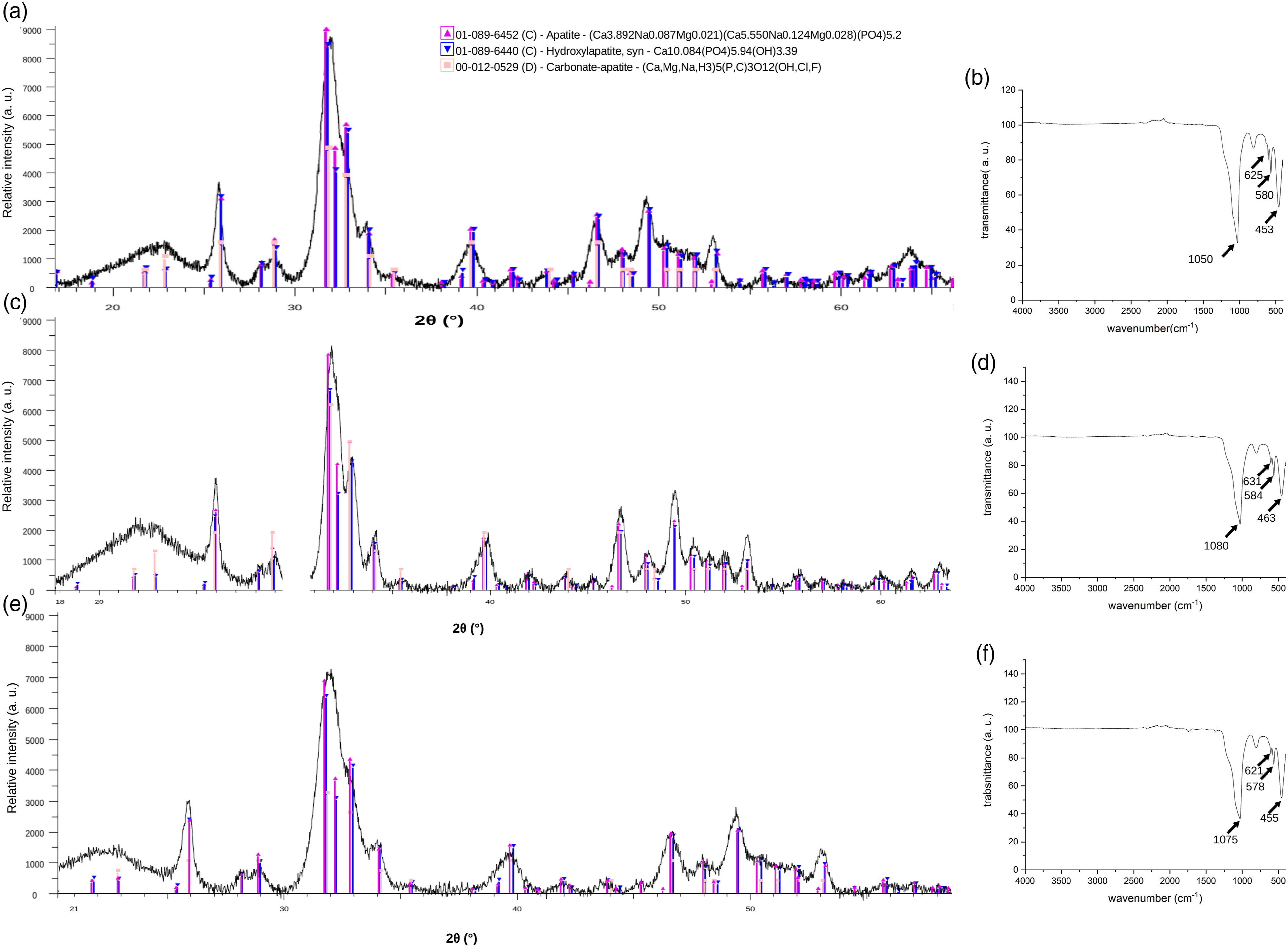
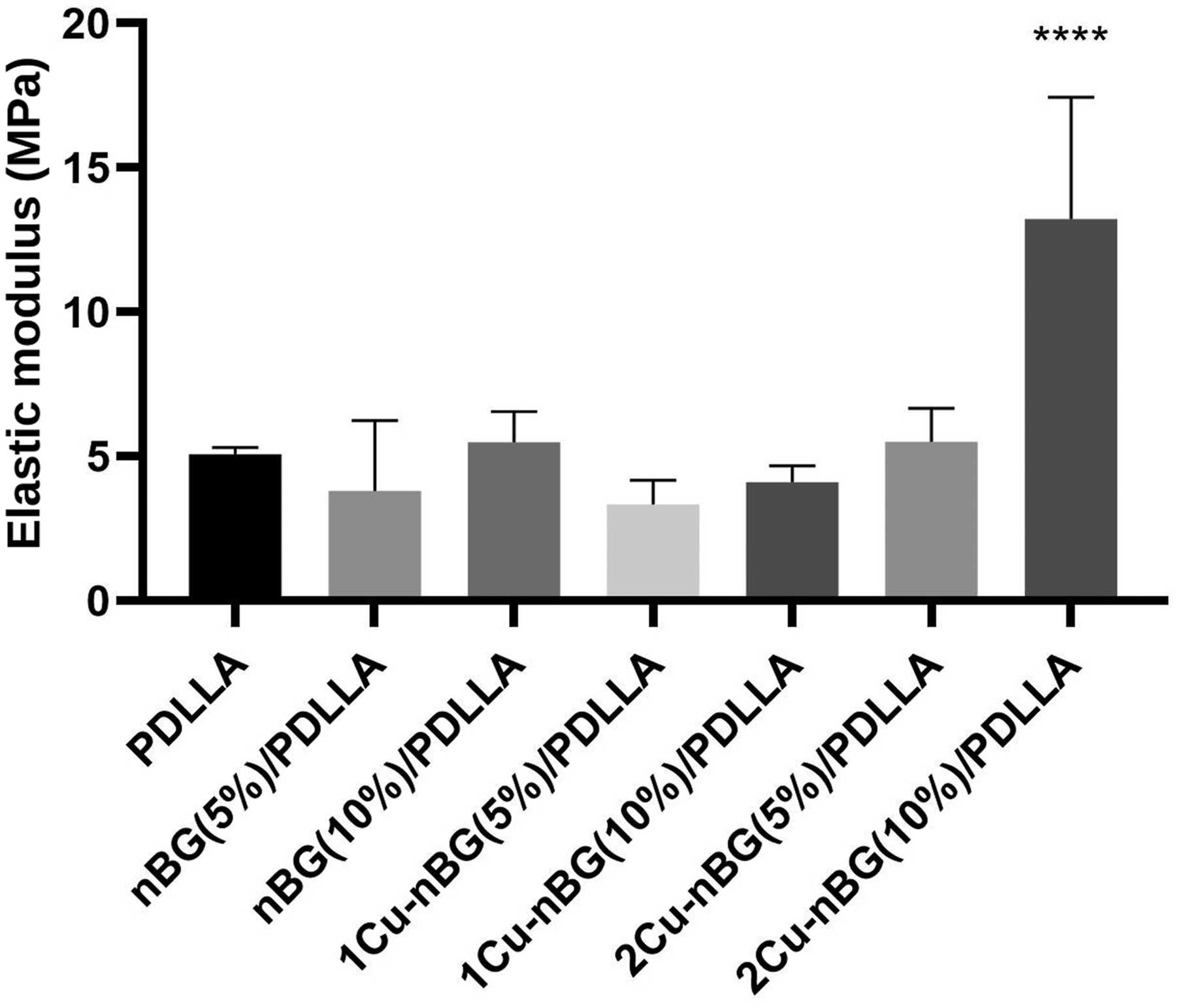
Figure 3 shows the SEM-EDX and ATR-FTIR analysis of the neat PDLLA scaffold and nanocomposites prepared with the 5 wt. % nanoparticle content. From SEM images, it can be observed that the scaffolds are composed by elongated pores with relatively uniform diameters in the 50–120 μm range. Although microsized agglomerations of the nanoparticles can be detected on the polymer walls, EDX mapping of Si, P and Cu suggests a relatively uniform distribution of the nanoparticles into the polymer matrix. The nanocomposite scaffolds also exhibited porosities values around 90% without significant differences with respect to that of the neat scaffold (Table 1). These high porosity values have been found to be suitable to facilitate the process of vascularization and bone tissue reconstruction when polymeric scaffolds are in vivo implanted.22,23 On the other hand, elastic modulus of PDLLA scaffold was significantly increased only when 10 wt. % of 2Cu-nBG nanoparticle was incorporated into the polymer matrix (Figure 4). Mechanical reinforcement effect in the polymer nanocomposites depend on the chemical nature, size, content, and distribution of the particles, which determine interfacial interactions and restrictions in the mobility of the polymer chains.
24
It has been found that the rigidity of some inorganic fillers may reduce the interfacial energy and consequently enhance the polymer mechanical properties as compared to softer fillers.25,26 This effect could explain why 2Cu-nBG nanoparticle having higher metal content produces a more significant mechanical reinforcement in the PDLLA polymer matrix. Scanning electron microscopy images and X-ray dispersive energy elemental mapping of PDLLA, nBG (5%)/PDLLA, 1Cu-nBG (5%)/PDLLA, and 2Cu-nBG (5%)/PDLLA nanocomposite scaffolds. PDLLA: Poly (D,L-lactide-co-glycolide). Porosity measurements of neat poly (D,L-lactide-co-glycolide) and nanocomposite scaffolds. PDLLA: Poly (D,L-lactide-co-glycolide). Compressive elastic modulus of neat poly (D,L-lactide-co-glycolide) and nanocomposite scaffolds.

The ability of the nanocomposite scaffolds to induce the formation of bone-like apatite on its surface was assessed in SBF at 7 days of incubation. The evolution of apatite formation on the scaffold surfaces was analysed by XRD, ATR-FTIR, and SEM (Figure 5). Most of the XRD diffractograms of scaffolds immersed in SBF exhibited a peak around 29° associated to the formation of calcite (CaCO3),
27
and some of them exhibit the more characteristic apatite peak at 31.7°, corresponding to the 211 reflections of the apatite crystal (JCPD 205166). ATR-FTIR analysis only detected the formation of apatite on the nanocomposite scaffolds loaded with 10 wt. % of nanoparticles, as judged by the bands at 560 cm−1 and 610 cm−1, assigned to the P-O bending vibration in crystalline apatite.
21
SEM examination of the 10 %wt. nanocomposites confirmed the presence of mineral deposits associated to the formation of apatite on their surface (Figure 5). This effect is consequence of the already demonstrated capacity of nBG to induce rapid crystallization of apatite when incorporated into different biodegradable polymer matrices.15,16 Interestingly, copper incorporated into the nBG structure does not alter its in vitro bioactivity, which can be explained by the comparable crystallinity of nBG with those doped with Cu, which would produce a similar dissolution rate of the nanoparticles during the apatite formation process. Cytocompatibility of the scaffolds was assessed using DPSCs until 14 days of incubation. In general, MTS absorbance values of mitochondrial activity of cells cultured with the nanocomposites (Figure 6) tended to be lower than of those incubated with the neat PDLLA scaffold. At 14 days, no significant differences were detected between viability of cells cultured with nBG/PDLLA and most of the Cu-nBG/PDLLA samples. This result suggests that cell viability decrease is related with the incorporation of nanoparticles into the polymer matrix rather to the presence of Cu in the nBG structure. The preparation and bioactive properties of PDLLA scaffolds loaded with nanosized BG has been scantly studied. Hong et al.
28
prepared nBG/PDLLA scaffolds with good in vitro bioactivity in SBF, however no cell assays were performed. In the current study, the viability decrease observed in the cells cultured with the nBG nanocomposite scaffolds could be due to the use of quaternary nBG, which has shown lead to higher pH value of the culture medium
29
than the ternary nBG.
27
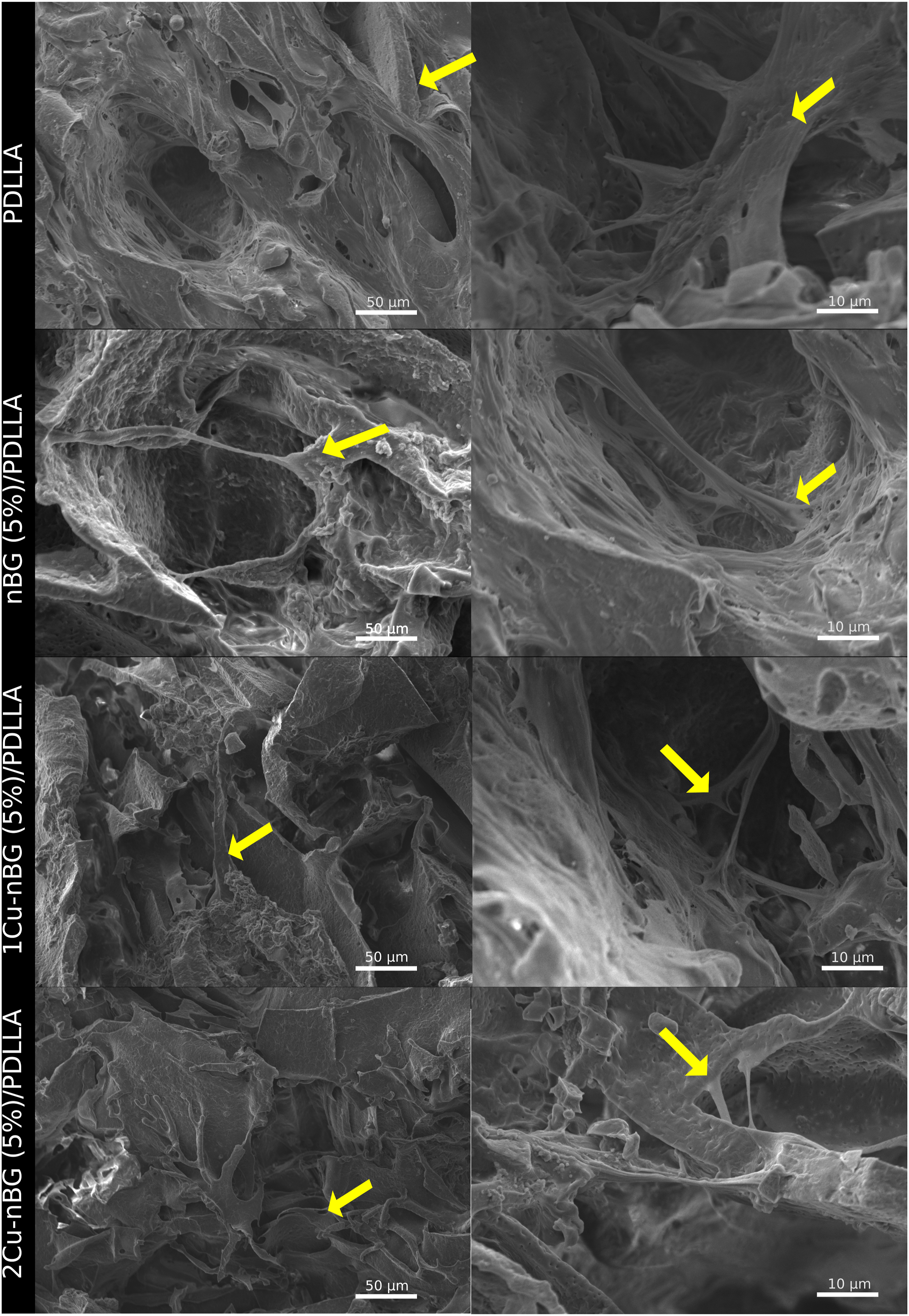
Despite the MTS viability decrease detected in cells cultured with the nanocomposites, SEM analysis (Figure 7) revealed the presence of cells intimately adhered on the surface of nanocomposite scaffolds, which is also considered an indicator of the material bioacompatibility. It is for that reason that the nanocomposites were able to stimulate the expression of ALP enzyme (Figure 8), producing some ALP activity values higher than those achieved with the neat PDLLA scaffold. The results also show that the presence of Cu in the scaffold materials does not impair the ALP activity in the DPSCs. ALP is a typical marker of the osteogenic differentiation process of stem cells, which is produced when specialized bone – forming cell lay down the bone extracellular matrix.
30
Although some works have reported an increase of ALP activity attributed to copper ions released from the material toward the cell culture medium,10,31 most of the studies indicate that Cu does not improve the osteogenic differentiation process.11,32–34 So, the capacity of the nCu/PDLLA nanocomposite scaffolds to stimulate the osteogenic cell differentiation should be consequence of the ionic dissolution products of nBG. Specific concentrations of silicon and calcium ions produced by BG recreate an extracellular environment capable of driving the stem cell differentiation toward an osteoblast phenotype.35–37 Antibacterial properties of the scaffolds were assessed by measuring the growth of X-ray diffraction patterns, total attenuated reflectance with Fourier transform infrared spectroscopy spectra, and SEM images of PDLLA, nBG (5%)/PDLLA, nBG (10%)/PDLLA, 1Cu-nBG (5%)/PDLLA, 1Cu-nBG (10%)/PDLLA, 2Cu-nBG (5%)/PDLLA, and 2Cu-nBG (10%)/PDLLA nanocomposite scaffolds after conditioning in simulated body fluid for 7 days. PDLLA: Poly (D,L-lactide-co-glycolide). MTS viability of dental pulp stem cells cultured with PDLLA, nBG (5%)/PDLLA, nBG (10%)/PDLLA, 1Cu-nBG (5%)/PDLLA, 1Cu-nBG (10%)/PDLLA, 2Cu-nBG (5%)/PDLLA, and 2Cu-nBG (10%)/PDLLA nanocomposite scaffolds. PDLLA: Poly (D,L-lactide-co-glycolide). Scanning electron microscopy images of dental pulp stem cells adhered on the surface of neat PDLLA and on that of nanocomposite scaffolds loaded with 5% of nanoparticles after 7 days of incubation. Alkaline phosphatase activity of dental pulp stem cells cultured with PDLLA, nBG (5%)/PDLLA, nBG (10%)/PDLLA, 1Cu-nBG (5%)/PDLLA, 1Cu-nBG (10%)/PDLLA, 2Cu-nBG (5%)/PDLLA, and 2Cu-nBG (10%)/PDLLA nanocomposite scaffolds in the absence of osteogenic supplements. PDLLA: Poly (D,L-lactide-co-glycolide). Antibacterial activity of the scaffolds expressed as the capacity of the material to inhibit the growth of viable colonies of Cumulative Cu2+ release from 1Cu-nBG (10%)/poly (D,L-lactide-co-glycolide) nanocomposite scaffold immersed in phosphate buffered saline.



Conclusions
Synthesis of Cu-containing BG nanoparticles using the 60SiO2: 25CaO:11Na2O: 4P2O5 sol-gel system produces particles with relatively crystalline structure. PDLLA nanocomposite scaffolds prepared with Cu-nBG nanoparticles are cytocompatible, promote the formation of bone-like apatite in SBF and stimulate the osteogenic differentiation process of stem cells in vitro. Development of greater antibacterial activity in the nanocomposite scaffolds would requires further research primarily related to the synthesis of Cu-nBG nanoparticles with more amorphous and soluble structure.
Footnotes
Acknowledgements
The authors acknowledge the financial support of U-Redes Consolidación Project URC 026/16 and PRI-ODO 18/004 from University of Chile, and FONDEQUIP EQM130076.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by University of Chile (Project URC 026/16 and PRI-ODO 18/004) and FONDEQUIP (EQM130076). CONICYT for funding of Basal Centre, CeBiB, FB0001 and P09-022-F from ICM-ECONOMIA, Chile.
