Abstract
The ultimate aim of tissue engineering entails fabrication of functional replacements for damaged organs or tissues. Scaffolds facilitate the proliferation of cells, while also improving their various functions. Scaffolds are 3-D structures capable of imitating mechanical and bioactive behaviors of tissues extracellular matrix, which provides enabling environment for cellular bonding, proliferation, and distinction. Hence, scaffolds are often applied in tissue engineering with the aim of facilitating damaged tissue regeneration which is a very important aspect of bone repair. Polymers are broadly utilized in tissue engineering due to their inherent versatility. However, polymers cannot attain mechanical behavior comparable to the bone. Thus, polymer nanocomposites fabricated through inclusion of fibers/or uniformly distributed ceramic/metallic nanoparticles in the matrix are potential materials for bone scaffold fabrication because inclusion of fiber or nanoparticles enhances composites mechanical behavior, while also improving other properties. Hence, this article elucidates recent trailblazing studies in polymer fiber composites and nanocomposites applied in the medical field especially in tissue engineering and bone regeneration. Also insights into market prospects and forecasts are presented.
Introduction
Presently, the market estimation for regenerative implant surgeries, therapeutic cell culturing, and tissue mending is about US $23 billion, and it is expected to attain US $94.2 billion toward ending of 2025. 1 Scaffolds are used as matrices in tissue fabrication to fulfill certain essential parameters in bone tissue engineering, including osteoconductivity, high porosity, non-cytotoxicity, accurate pore dimension, efficient surface behavior which enables cell adherence, distinction, and proliferation, and other vital mechanical attributes pivotal to the maintenance of tissue architecture. 2
Bone tissue engineering extends to the use of appropriate biocompatible scaffold alone, or in conjunction with various cells or bioactive parameters as replacements for bone-grafts. 3 In a bid to replace traditional techniques used for treatment of bone damages, such as metallic prostheses and grafting, biodegradable and biocompatible polymers were developed as 3-D strategy for stimulating and sustaining bone regeneration in tissue engineering. 4 In recent years, advancement in development of composite materials has resulted in great improvements in architectural design of modern prosthetic and orthopedics devices. 5 Composites are engineering materials composed of two or more components, each providing a unique physical property, with synergistic composition. 6
Nowadays in orthopedics, fiber-filled polymer composites are multiphase materials commonly used. Recently, upper- and lower-limb prostheses have been constructed from polymer composites. 7 Polymer fiber composites are capable of effectively attaining both low elastic modulus and high strength, in addition to corrosion and fatigue resistance. The behavior of these materials can be configured through various techniques. Exhibition of superior strength and low elastic modulus are essential for bonding in fabrication of artificial limbs, and this is referred to as osseointegration. 8 Material biocompatibility reveals its performance capability when applied in a living system. 9 The most commonly implanted tissue apart from blood is the bone. 10 Hence, understanding bone activities with regard to its structural and physical behavior is imperative for the successful design of various types of implants. Bone is a naturally occurring and highly hierarchical composite material composed of collagen fibrils with hydroxyapatite (HA) nanocrystals interdistributed within the collagen fibers. However, properly tailored polymer-based composites can compete favorably with the bone properties and potentially used as a replacement material. 11
Easily available synthetic polymers for bone tissue engineering include highly porous polymeric matrices, capable of performing important functions to improve initiation of cells adherence, propagation, and novel tissue/fluids formation about the bone damages. 12 Biocompatible polymeric materials commonly utilized in fabricating polymer nanocomposites (PNC) for tissue engineering include polyhydroxyalkanoates (PHAs), polyetheretherketone, polycaprolactone (PCL), polyurethane, polyethylene (PE), and polysulfone. 13 These polymers enforce stability in the body, although PE is most commonly available. 14 Also, commonly applied synthetic polymers include poly(glycolic acid) (PGA), polyhydroxybutyrate (PHB), poly(lactic acid) (PLA), PCL, and their various copolymers. 15 These polymers possesses advantages, such as biocompatibility, which eliminates adverse reactions, and biodegradability, which enables its degradation at same duration as bone regeneration, until the scaffold is completely replaced by a fresh tissue. 16 Various methods applied in the fabrication of porous structures include thermally induced phase separation, solvent casting, emulsion-freeze-drying, supercritical gas foaming, electrospinning, leaching, and solid free-form fabrication. 17
On the other hand, these polymers also exhibit varying deficiencies such as low mechanical behavior when compared with bone tissue. To overcome these challenges, insights are taken from the natural bone structure, perceived as a form of nanocomposite, because of its composition of nanoscale part (HA crystals), in combination with a matrix composed of micrometric collagen. 18 Hence, organic and inorganic nanoscale particles are uniformly distributed in the polymeric matrix, with the intention of attaining superior properties in comparison with those exhibited by single components alone. Major advantages of nanoparticles include improvement of mechanical behavior of the scaffold, facilitating stimulation of cellular adherence, as a result of their large surface area, which is essential to bone tissue engineering. 19 Furthermore, some nanoparticles exhibit added antibacterial and antitumor functions, 20 which increases their added benefits. Recent studies have demonstrated that bioactive glasses (BGs) easily adhere to bone faster than other bioceramics. 21 HA, BG, and tri-calcium phosphate (TCP) have been widely used with biomaterials to enhance interaction with tissues and promote osteogenesis. 22
Therefore, the aim of this article is to elucidate recently emerging blue chip studies in the use of PNC filled separately with fibers, BG, HA, and other nanoparticles in the medical field, with emphasis on bone scaffolding and tissue regeneration procedures. Additionally, applications of polymer fiber composite materials are elucidated for hard- and soft-tissue procedures, in addition to use in tissue engineering and other prospective applications. Future view for next generation orthopedic medicine is summarily presented relative to current blue sky technological innovations in orthopedics advancements.
Nanotechnology in tissue engineering
Nanotechnology enhances the bioactivity of scaffolds because of the higher surface area possessed by nanoparticles. 23 Moreover, there exist the feasibility of functionalizing them using proteins and initiators capable of stimulating adhesion, proliferation, and differentiation of bone cells. 24 Nanotechnology is a reoccurring decimal in the great progress observed in biomedical field in previous years. Hence, nanomaterials are presently undergoing massive industrial production, while some products are already being commercialized. However, present challenges to do with toxicity of these nanomaterials still persists.
Nanoparticles, specifically those having dimensions below 100 nm, demonstrate potentials of clusters formation as a result of van der Waals forces, governing the nanoscale. 24 It is imperative to apply a method guaranteeing uniform distribution of nanoparticles in the matrix to ensure effective results during fabrication of scaffold, otherwise the nanoparticles will form micrometric scale agglomerates and probably nullify their nanoscale influence. Thus, the higher the dispersion, the more improved the properties exhibited by the nanocomposites. 25 Nanoparticles applications in treatment of infection can be divided into two strategies, namely: use of materials possessing inherent bactericidal attributes at the nanoscale 26 and encapsulation of therapeutics in nano-vesicles to improve efficiency. 27 The European Commission in 2011 expressed nanomaterials as naturally occurring, accidental, or manufactured materials containing particles, by themselves or in agglomerates containing at least one dimension between 1 nm and 100 nm. 28
Nowadays, nanocomposites have attained high recognition in bone tissue engineering, as living bone tissue is perceived as nanocomposite, exhibiting an intricate hierarchical architecture, composed of an organic medium of fibrous collagen, and nanocrystals and HA mineral medium. 29 Thus, bone cells naturally react with nanostructured materials, in as much as they possess a rough surface and composed of pores of about 2100 nm. 29 Polymers with inclusion of nanoparticles have capability of mimicking this roughness.
Nanomaterials used in biomedicals
Nanomaterials utilization in biomedicine have rapidly developed in response to increasing threat of bacteria having affinity for developing immunity to antibiotics. 30 Due to nanoparticles physicochemical behavior, large surface area, and nanoscale dimension, they exhibit ability of interacting with the cell membranes of microorganisms, proteins, and enzymes capable of hindering the development of pathogens. 31 Also, as a result of their ability to mimic minerals already existing in the bone, calcium and phosphate ceramics such as HA (Ca10 (PO4)6(OH)2), biphasic calcium phosphate, and b-TCP (Ca3(PO4)2) are broadly used for bone tissue engineering. These ceramics possesses absorbability, and excellent bio-conductivity, while exhibiting mechanical behavior similar to those of living bone. 32
Metallic nanoparticles such as zirconia, titanium, silver, and zinc demonstrate antibacterial and antifungal behavior and, thus, are vastly used in tissue engineering. 33 Recent investigations have demonstrated that these nanoparticles enhance mechanical behavior while stimulating adherence of cells. 34 Zirconium oxide (ZrO2) is largely available on earth, and in the human body, it is available in bone tissue to about 2–20 mg/kg. 35 ZrO2 has been used as a biomedical material because of its versatility.
Titanium oxide (TiO2) is an organic ceramic nanomaterial exhibiting tri-crystalline architecture, namely: brookite (orthorhombic), anatase (tetragonal), and rutilo (tetragonal). 35 However, the brookite architecture is not stable, and not easily synthesized, while rutilo and anatase are widely investigated, can undergo laboratory synthesis while exhibiting semi-conductivity and catalytic activity. Titanium oxides are widely applied in tissue engineering as a result of their nil-toxicity, low cost, photocatalytic mode, and mostly as a result of their bactericidal attributes. 35
Zinc is a fundamental element also found in the human body, especially in the bone. 36 Zinc also facilitates monitoring, architectural and alkaline phosphatase (ALP) catalization, thereby playing a vital role in osteogenesis and mineralization. Zinc also enhances the ability to restrain osteoclastic reabsorption procedure. 37 This nanomaterial exhibits effective compatibility, chemical stability, and antibacterial and antifungal activity, while remaining nil-toxic in vitro and in vivo. Because of its exhibition of fluorescence, low cost, and ease of availability, zinc has been widely applied in preclinical studies.
Additionally, silver and gold nanoparticles are broadly applied in biomedicine for treatment of cancer, due to their antitumoral effects. 38 Hence, the potential of utilizing these nanomaterials in bone tissue regeneration is large because of the prospects of facilitating simultaneous bone cancer treatment responsible for bone deterioration and subsequent fracturing. 37 The inclusion of gold nanoparticles into fibrous PCL fibers demonstrate improved elucidation of a-sarcomeric-actinin, a marker linked with the contraction of the cardiac muscle, which interact to notably reduce electrical fields in comparison with controlling post culturing for a week. 38
Nanoclays are widely used as nano-charges in the fabrication of PNC. 39 Montmorillonite (MMT) is a renowned nanoclay. MMT is a lamellar silicate composed of an octahedral layering of alumina within two tetrahedral silica layering. 40 Nanoclays are highly efficient in biomedical applications, due to their superior mechanical behavior, biocompatibility, biodegradation, and osteoconductivity. 41
Carbon nanotubes PNC in tissue engineering
Since its discovery, carbon nanotubes (CNTs) have undergone a great deal of investigation to enable exploitation of its inherent potential applications in biomedical and biological sectors. CNT is recognized for its chemical and thermal stability, high aspect ratio, mechanical strength, lightweight, and superior electrical properties. 42 -44 In a bid to enhance mechanical behavior of polymer matrices, CNT exhibits one of the highest values, due to inherently high mechanical behavior and fibrous architecture. 44 CNTs are cylindrically structured carbon derivatives composed of a length/diameter ratio of 28,000,000/1. They are categorized into single wall nanotubes (SWNT) and multiwall nanotubes (MWNT). CNTs have demonstrated efficiency as fillers or reinforcements in nanocomposites development. With regard to cardiac tissue engineering, SWNTs and MWNTs are used in fabricating scaffolds exhibiting appropriate elastic modulus. 44
Graphene PNC in tissue engineering
Graphene is a synthetic sp2 bonded carbon atoms composed of graphite layering arranged in a honeycomb lattice architecture. 45,46 Graphene is a single sheet of aromatic carbon atoms arranged in a 2-D lattice architecture. Nowadays, due to their osteogenic and regenerative attributes, graphene and its derivatives are prospectively applied in construction of bone tissue engineered scaffolds. Graphene can undergo combination with natural and synthetic biomaterials to enhance the osteogenicity and mechanical strength of tissue engineered scaffolds. 46
Studies of graphene and its derivatives have garnered immense interest industrially and in the academia as a result of its wonderful biomechanical attributes. Globally, great interest in graphene has continued to escalate especially in bone tissue engineering and biomedical applications. Moreover, graphene-oriented materials have also garnered versatile applications in the field of biomedicine and drug conveyance. As a result of its excellent biocompatibility under approved inclusions, graphene and its derivatives have demonstrated potential results in the regulation of cell tendencies, drug genes loading, and releasing. Furthermore, it is also perceived to assist in cell differentiation through surface modification of scaffold materials. Graphene enhances the adherence, development, proliferation, and differentiation of osteoblast. 47 They also exhibit special physiochemical and mechanical behavior, which enables their utilization in varying devices and as scaffold materials in enhancing stem cells proliferation for effective bone regeneration. 48 Graphene oxide (GO) assist in bone regeneration through improvement of the osteogenic differentiation of progenitor cells. This is attained via electrostatic and hydrophobic interactions with microenvironment proteins. 49
Reduced GO similar to GO consists of surface functional groups with peculiar electrical behavior in comparison with GO. Importantly, graphene-based materials have demonstrated capability of cell growth improvement, cell differentiation, and cell proliferation. A research has investigated the mechanical behavior and in vitro cytotoxicity of 2-D GO nanoribbons and nanoplatelets filled with porous PNC using porous poly(propylene fumarate) (PPF) nanocomposites. 50 Results revealed that 2-D graphene-filled porous PPF nanocomposites demonstrated superior mechanical behavior, cytocompatibility, and improved protein adsorption. The beneficial cytocompatibility behavior reveals novel routes for in vivo safety and healing effects for bone tissue engineering applications. 51
Fibrous scaffold structures have garnered interest for application in soft tissue engineering and in advanced bone tissue engineering. However, known approaches for treatment of damaged bones include autograft, allograft, vascularized grafts, and bone marrow substitution. 52 In vivo research has revealed that reduced GO demonstrated higher bioactivity when compared with GO upon coating on scaffolds made of collagen. 52 3-D printed GO and chitosan (CS) scaffolds enhanced bone regeneration in mouse calvarial damages. Investigation reveals improvement in osteogenic procedure and mending of the critical size damage. 49,50 The inclusion of GO with CS revealed improvement in ALP procedure, elucidation of bone morphogenic protein, and RUNX-2 factor. 50 Collagen scaffolds have been coated with varying GO concentration and studied for bioactivity, cell proliferation, and differentiation both in vivo and in vitro. 52 The results of subcutaneous implant examination in rats reveal improved cell growth at low GO inclusion. Severe biological effects where observed at increasing inclusions of GO. Thus, scaffolds which are treated with appropriate concentration of GO are effective bioactive material for tissue engineering.
Natural and synthetic biomaterials in tissue engineering
Naturally occurring and synthetic 3-D printed biomaterials have been utilized in tissue engineering. Varying biomaterials have been studied as scaffold materials for bone tissue engineering. Main parameters to consider during selection of appropriate materials for scaffold include biodegradability, biocompatibility, and appropriate mechanical strength to sustain the forces acting on the material. 51
Natural biomaterials applied in bone tissue regeneration include proteins such as silk, collagen, gelatin, fibrin, fibrinogen, elastin, keratin, actin, and myosin. 50 -53 The most reoccurring polysaccharides investigated include CS, cellulose, and alginate, while the protein-oriented polymers commonly researched include silk, collagen, and gelatin. 53 -55 However, polysaccharides, including dextran, hyaluronic acid, agarose, glycosaminoglycans, CS, amylose, alginate, cellulose, and chitin, and polynucleotides, such as DNA, RNA, CS, hyaluronic acid, alginate, and agarose, are also utilized. 56 The use of biomaterials has aroused interest in tissue engineering and regenerative medicine. Nowadays, biomaterials are applied in bones, artificial skin, cardiovascular and ureteral stents, cornea, nerve tissues, and so on. Bone tissue engineering is based on biomaterial usage with regard to prostheses, scaffolds, hydrogels, cells, and developmental factors used for treating bone losses caused by osteoarthritis, neoplasms, fractures, and osteoporosis. 57
Synthetic biodegradable polymers offer a variety of advantages for scaffolds development for tissue replacement, including tailorable mechanical properties and degradation kinetics. The most commonly used synthetic biopolymers for tissue engineering are members of the polyester family such as poly(
Nowadays, PHAs, natural polyesters synthesized via bacterial fermentation of sugar or lipids, and polyesters oriented on fumaric acid, a constituent of the Krebs cycle, have developed various interest for biomedical usage as a result of their superior biocompatibility and biodegradability.
60,61
Another widely studied material is PPF, a linear polyester including two ester-bonds and one unsaturated carbon–carbon double bond, which enables cross-linking either by free radical polymerization using monomers of methyl methacrylate or
Synthetic biopolymers in tissue engineering
Synthetic polymers are materials usually utilized for tissue regeneration because of their inherent porosity, rapid time of degradation, and high mechanical attribute. They exhibit some benefits over naturally occurring polymers, such as prolonged shelf-life, low cost, flexibility and ease of manipulation to attain a desired shape via milling and printing, rapid mass production under monitored conditions, exhibition of effective cell differentiation behavior, pore attributes, and mechanical behavior. However, deficiencies of synthetic biomaterials include lack of cell bonding sites and need for chemical treatment to enhance cell bonding. The commonly applied synthetic materials include polylactide-co-glycolic acid (PLGA), PGA, PLA, PCL, HA, TCP, HA nanoparticles, BG, and glass-ceramics. These polymeric materials exhibit varying levels of biodegradability, biocompatibility, and mechanical strength, but no single polymer optimally exhibit all these three essential attributes. 63
Bioglass in tissue engineering
Originally, 45S5 glass was invented by Larry Hench about 50 years ago. 64 It demonstrated the ability to bond with natural bone and ability to stimulate osteogenesis by releasing biologically active ions. 64 BGs initiated a revolution in medicare and opened the window for enhanced biomaterial-initiated regenerative medicine. 65 45S5 glass products have successfully been implanted in millions of patients globally, in a bid to mend bone and dental damages. In previous years, numerous other BGs have undergone proposition for advanced biomedical uses in soft tissue mending and drug conveyance. 65
In the United States, the foremost 45S5 Bioglass® device approved for clinical use had the objective of replacing small bones of the middle ear so as to repair a conductive hearing impairment. 66 This implant attained US food and drug administration (FDA) recognition in 1985 and subsequently went commercial under the trademark of “Middle Ear Prosthesis” MEP® or Bioglass Ossicular Reconstruction Prosthesis.” 67 This device closely adhered to living tissues due to the ability of 45S5 Bioglass to adhere to hard calcified tissues such as bone and to collagenous soft tissues such as eardrum. 68 A remodeled version of the foremost MEP device, Douek-MEDTM; 45S5 Bioglass cones of three varying dimensions is presently commercially offered in some European countries. Other notable Bioglass trademarks include Ceravital® glass-ceramics synthetic devices, which can be used in replacing the small bones of the middle ear, 69 Bioglass-EPI (extracochlear percutaneous implant), 70 PerioGlas® (NovaBone Products LLC, Alachua, Florida, USA), 71 NovaBone® (NovaBone Products LLC), 72 Biogran®, 73 StronBone® (44.5 SiO2–4 Na2O–4 K2O–7.5 MgO–17.8 CaO–4.5 P2O5–17.8 SrO mol.%, RepRegen, London, UK), 74 TheraGlass® (MedCell, Burgess Hill, UK), 75 and 45S5 Bioglass. 76 However, the sinterability technique of 45S5 Bioglass is limited and cannot be successfully sintered without devitrification procedure. 77 Notable BG trademarks also include NovaMin® (NovaMin Technology, Florida, USA; presently owned by GlaxoSmithKline, Brentford, UK), which was produced and included into toothpaste with the objective of mending dental hypersensitivity. 77 BGs have also been utilized in the production of glass polyalkenoate cements for dental and bonding of joint prostheses to bone. 78
BGs have garnered great attention from researchers, as their ionic dissolving products were known to stimulate angiogenesis, which plays a critical function in wound healing and in some ophthalmic infections. Two BG-oriented commercial products have been noted to perform angiogenic functions. Biodegradable cotton-candy-borate-BG (Mo-Sci Corp., Rolla, Missouri, USA), imitating fibrin clot microstructure has been reported to enhance wound repairs in both animals and human patients. These nanofibrous BG of basic 13-93B3 composition: 53 B2O3–6 Na2O–12 K2O–5 MgO–20 CaO–4 P2O5 wt%, with the trade name of DermaFuse™-Mirragen™, assist immensely in prolonged mending of venous stasis ulcers in diabetic patients, not responding to conventional treatment. 79
Other BG products include porous PE (Medpor® implant) marketed as Medpor-PlusTM (Porex Surgical, Newnan, Georgia, USA), 80 resorbable Ag-included-phosphate-glasses in combination with a polymeric adhesive for wound dressing referred to as Antimicrobial-Arglaes® film, and antimicrobial-Arglaes (Medline Industries, Northfield, Illinois, USA) or with alginate for topical flour (Arglaes powder, Medline Industries) enabling long-term control of diseases via sustained silver release, a strong antibacterial agent have been recently put on the market. 81
A resorbable tube composed of Na2O–CaO–P2O5 glass (Corglaes®, Giltech Ltd., Ayr, UK) has undergone analysis for the mending of a pieced sheep facial nerve as replacement for end closed suturing. 82 Presently, radioactive glass-microspheres with the trade name of TheraSphere® have been clinically tested for treatment of hepatocellular carcinoma and metastatic liver cancer in numerous specialized clinics in North America and Europe after attaining FDA recognition in 1999. 83
HA PNC in tissue engineering
HA possesses a compositional and structural architecture similar to natural bone mineral and has been perceived as a material appropriate for fabrication of bone tissue engineering scaffold as a result of its osteoconductivity and osteoinductivity. However, its brittleness and inferior mechanical behavior hinder its application in the regeneration of non-load-carrying bone damages. 84,85 A study involving PCL/nano-HA scaffolds has demonstrated that high hydrophilicity of HA nanoparticles improved water infiltration in scaffolds. 86
Synthetic biopolymer/graphene derivatives scaffolds in bone tissue engineering
PCL is a hydrophobic polymer exhibiting melting temperature of about 60°C. PCL has been widely utilized in biomedical application because of its blend compatibility and superior solubility. Although PCL exhibits numerous benefits such as hydrophobicity, gradual degradation, and resorption, it is still very appropriate for use because of inherent osteogenic prospects and biocompatibility. 87 Studies confirming synthetic biopolymer/graphene derivatives as efficient scaffold materials have been conducted. 87 -89 BG has been widely utilized in bone tissue engineering due to its affinity in forming bioactive HA layering, thereby creating a cohesive bond between implant and the surrounding bone. 64 -73 BG exhibits poor mechanical behavior in bone tissues replacement and requires inclusion of either organic or inorganic nanomaterials to improve their mechanical strength. 70 Graphene has demonstrated excellent mechanical strength and good biocompatibility and does not induce any inflammatory variation to the encumbering microenvironment. 90 -92
PCL/HA nanocomposites in tissue engineering
PCL and PCL/PLA scaffolds were fabricated using a new inclusive bio-fabrication strategy referred to as Bio-Cell printing. 92 -94 PCL/PLA blending occurred via melt blending and solvent casting techniques. The fabrication techniques are shown in Figure 1, while Figure 2 reveals the surface characteristics of PCL/PLA scaffolds.

Design factors. 95

Scanning electron microscopic images of the scaffolds: (a and b) PCL scaffold; (c and d) PCL/PLA scaffold (prepared through melt mixing); (e) PCL/PLA scaffold (blends prepared by solvent casting); (f) PCL/PLA surface characterization scaffolds; (g) optical images of both the silicon cantilever and the PCL/PLA scaffolding fabricated through melt extrusion; (h) AFM images of PCL/PLA scaffold prepared via melt mixing; (i) optical images of both the silicon cantilever and the PCL/PLA scaffold prepared through solvent casting; and (j) AFM micrographic images of PCL/PLA scaffolds prepared through solvent casting. 95
Furthermore, studies have shown that collagen as human tissue demonstrated biocompatibility in the body. Results reveal that surface modification technique used in this research facilitated collagen immobilization on the surface of PCL-scaffolds, which implied that PCL-Col scaffolds could be used for regenerative medicine and tissue engineering. Figure 3 reveals the SEM images of PCL scaffold and PCL-Col scaffold. 96
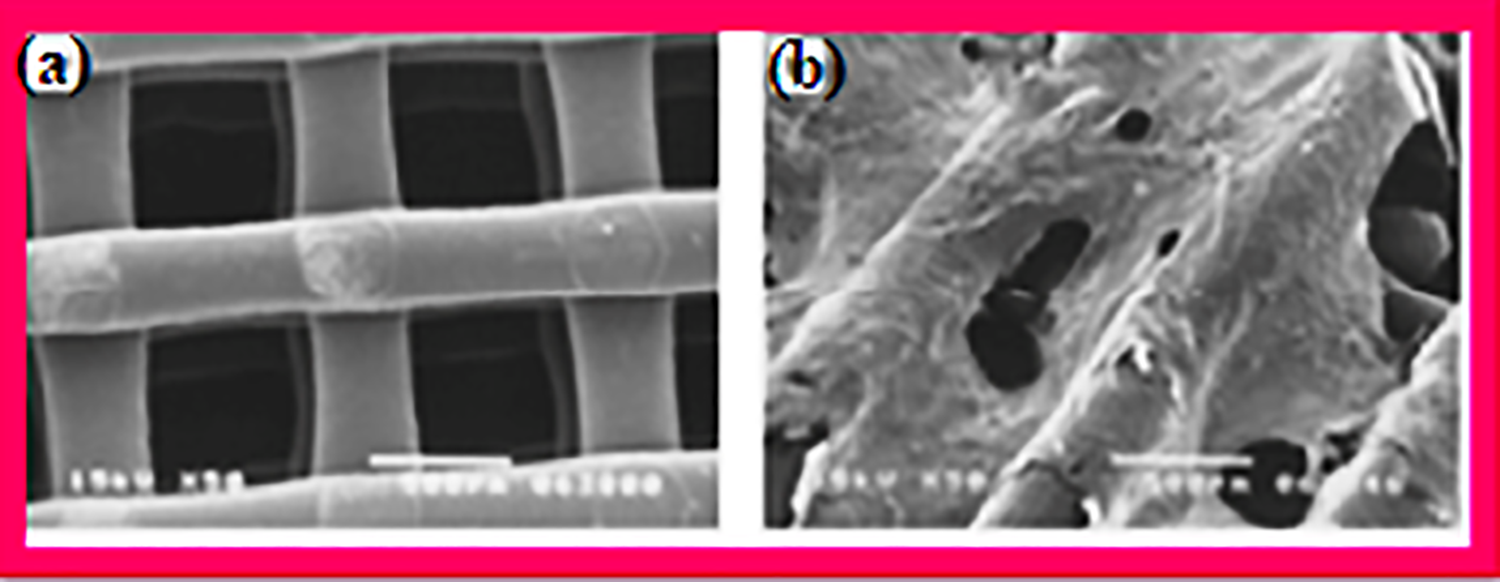
SEM images of (a) PCL scaffold and (b) PCL-Col scaffold. 96
In another study, the fabrication of new biodegradable porous scaffold composed of PCL and magnesium (Mg) microparticles has been conducted. 97 Incorporation of 29% Mg particles enhanced PCL porous scaffold compressive modulus by 150% and further increased by 236% on addition of silane-Mg particles. Figures 4 and 5 elucidate morphological behaviors of the fabricated composites. 97

(a) (a′) SEM images of surface architecture of virgin PCL and hybrid Mg/PCL porous scaffolds and (b′) micro-CT-3-D reconstructed model of the fabricated scaffold. (b) Microscopic view of (a′ and d′) virgin PCL (b′ and e′) TMSPM-coupled 45-µm particle Mg/PCL scaffold, and (c′ and f′) TMSPM-coupled 150-µm particle Mg/PCL scaffold after culturing using GFP mouse osteoblasts for 3 days; (a′ to c′) show the scaffolds with reduced magnification, while (d′ to f′) show the scaffolds with higher magnification. (c) SEM of (a′) virgin PCL and (b′) TMSPM in combination with 45-µm particle Mg/PCL scaffold, and (c′) TMSPM reinforced with 150-µm particle Mg/PCL scaffold. PCL chemical composition, silane/Mg/PCL porous scaffolds analyzed via energy-dispersive X-ray spectroscopy. (c1 and c2) post immersion effect on pristine PCL scaffold. 97

Histological images of bony tissue around pristine PCL, TMSPM-reinforced 45-µm particle Mg/PCL porous scaffold, and TMSPM-filled 150-µm particle Mg/PCL porous scaffold after 3 months implantation, (a to c) scaffolds position in red line (40 magnification) and (d to f) novel bone fabricated amidst osteoblast-alike cells (green arrows) within scaffolds (200 magnification). 97
The fabrication of composite electrospun membranes comprising of PCL and a BG (F18) was carried out. 98 The PCL-F18 composite membranes demonstrated zero cytotoxicity, while improving osteogenic prospects in comparison with virgin PCL membranes. Figure 6 reveals the morphological micrographs. 98

(a) SEM images of pristine PCL membranes. The image demonstrates even morphology, while the fibers are arranged unevenly, demonstrating a smooth and continuous architecture (magnification 3000). (b) SEM images of PCL-F18 membrane. The observed architecture is slightly varied in Figure 6 (a). The fibers are irregularly arranged, while beads and fiber discontinuities are more prominent (magnification of 3500); optical micrographs of stained membranes for ALP behavior. From the top to the bottom, varying degrees of staining and color differentiation between PCL-F18 (c), PCL in osteogenic phase (d), and virgin PCL membrane (e). Post 2 weeks, PCL-F18 (c) specimen showed a similar effect on MG-63 cells as the control osteogenic phase (d). 98
A 3-D fibrillated porous and interlinked PCL scaffolds possessing proper pore-dimension and appropriate porosities have been fabricated via PCL blending with water-soluble poly(ethylene oxide) (PEO) as sacrificial material, followed by micro-cellular injection molding and polymer leaching methods as elucidated in Figure 7. PCL/PEO scaffolds characteristics with regard to immiscibility, structural, mechanical property, hydrophilicity, and biocompatibility demonstrated that the materials were immiscible and useful for tissue engineering. 99

Schematic illustration of the PCL tissue scaffold fabrication method. 99
PCL/HA nanocomposites in tissue engineering
Synthetic PCL demonstrating biodegradability and biocompatibility exhibits a slow rate of deterioration and is a proper material for fabricating fibrous scaffolds. 92,100,101 Inclusion of HA into PCL fibers could exhibit superior behavior in comparison with PCL fibers separately. Hence, the fabrication of nano-HA/PCL composite microfibers via electrospinning technique has been conducted. 102 Figure 8 reveals elemental mapping and energy dispersive x-ray analysis (EDX) of the composition, while Figure 9 reveals morphological structure post 1-week culturing. The elemental mapping additionally confirmed distribution of nHA in PCL nanofiber matrices (Figure 8). EDX analyses at varying points of electrospun composite fibers also reveals availability of nHA particles when effectively combined with polymer fiber surfaces, whereas after 1-week culturing, the architecture of L929 cells adherence to PCL and HA/PCL electrospun fibers is shown in Figure 9. At day 7, L929 cells had evolved into a component forming a monolayer. 102

Elemental mapping of (a) calcium and (b) phosphorus, and (c) EDX spectrum of 10% HA in 7.5 w/v% HA/PCL composite fibers at 1 mL/h, 22 kV. 102

After 7 days of culture with L929 cells seeded on (a and b) PCL and (c and d) HA/PCL microfibers. 102
A porous PCL/HA composite scaffolds’ for bone regeneration, with ability of tailoring the macroporous/microporous structure and possessing high mechanical properties and excellent in vitro bioactivity using nil-solvent-induced phase separation focused on 3-D plotting, has been studied. 103 This new 3-D plotting technique can form strong microporous PCL/HA composite filaments via inducement of the unique phase-separation in PCL/HA solutions via non-solvent–solvent exchange mechanism. The fabricated PCL/HA composite scaffolds demonstrated high microporous architecture as shown in Figures 10 and 11. Mechanical behavior, including ultimate tensile strength and compressive yield strength, was improved with increasing incorporation of HA in PCL. Also, incorporation of bioactive HA particles into PCL resulted in notable enhancement in vitro apatite-forming potential. 103

FESEM images of macroporous/microporous PCL/HA composite scaffolds prepared by including varying amounts of HA: (a) 0 wt%, (b) 10 wt%, (c) 15wt %, and (d) 20 wt%, revealing structure of straight PCL/HA struts and macro-channels. Insets in Figure 10 (a to d) show optical images of porous scaffolds. FESEM images of macroporous/microporous PCL/HA composite scaffolds fabricated through inclusion of HA (e) 0 wt%, (f) 10 wt%, (g) 15 wt%, and (h) 20 wt%, revealing the cross-sectional morphology. 103
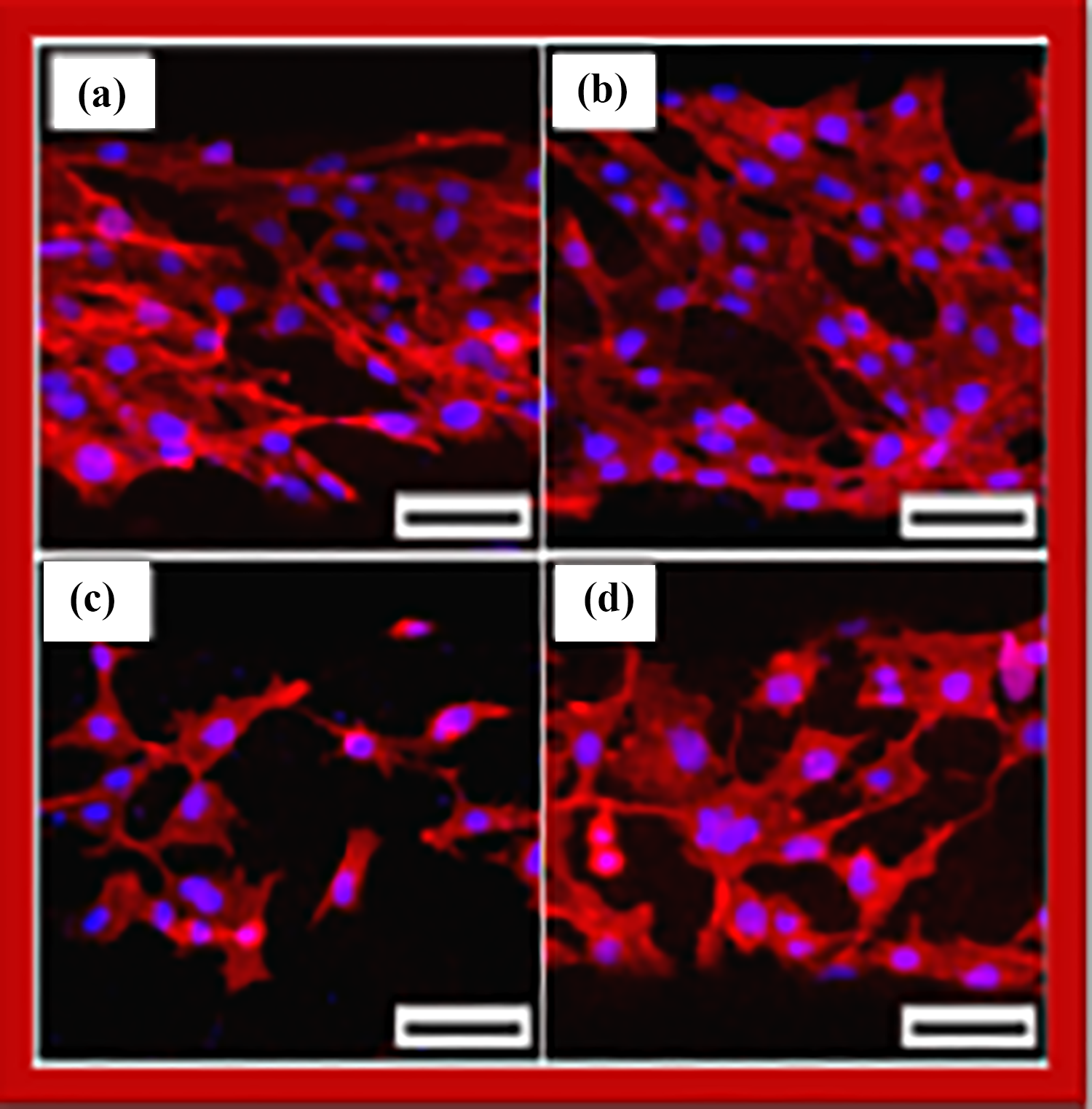
Confocal laser scanning microscopic images of MC3T3-E1 cells on macroporous/microporous PCL/HA composite scaffolds fabricated using various HA compositions (a) 0 wt%, (b) 10 wt%, (c) 15 wt%, and (d) 20 wt% after 24 h of cell culturing (scale = 100 m). 103
PHAs composites
PHAs are biodegradable polymers derived from microorganisms. Varying forms of PHA can add to biodegradability, biocompatibility, and mechanical and chemical behavior of composites. 59 This has resulted in escalating interest in PHAs usage for varying biomedical applications over the years. PHAs are a category of polyesters capable of natural release by bacteria and exhibit superior biodegradability and biocompatibility capability while garnering high interest for usage in the biomedical field. 61 PHAs can broadly be categorized into two groups; short-chain length PHAs possessing 3–5 carbon monomers or medium-chain length PHAs possessing 6–14 carbon monomers. 104 PHAs have been widely studied for usage in tissue engineering as cardiovascular patches, sutures, wound-dressings, and recently, for bone-scaffolds. 62
PLA/BG nanocomposites for bone replacement
The fabrication of better scaffold material is a major challenge in bone tissue engineering. PLA is a good biopolymer material for bone tissue engineering application due to its biocompatibility and biodegradability. 104 However, PLA shows some deficiencies such as slow degradation rate and cell adherence as a result of its hydrophobity and inflammatory reactions in vivo because of lactic acid, its degradation product. The inclusion of bioactive reinforcement is applied as a strong technique in improving PLA properties. 105
PLA/HA nanocomposites for bone replacement
A bioactive material is susceptible to undergoing unique surface varying interactions, when implanted in the human system, resulting in formation of a material similar to HA layering while causing the formation of a firm bond with hard and soft tissues. 100 -105 Ceramic nanomaterials such as HA, calcium phosphates and BG, especially BG® are commonly available bioactive fillers applied in biocomposites. 106 These nanoparticles are bioactive as a result of their capability to adhere to bone and improve bone tissue formation. 15
CS composite materials for bone tissue engineering
Composite materials are currently exercising vital functions as scaffolds in bone tissue engineering. 92,93 CS has varying beneficial attributes for orthopedic applications, making them effective as bone graft materials. 99 Recent developments in the field of composite materials with CS mimic all bone properties. 92,99
Varying techniques have been utilized in preparing CS/HA nanocomposites, improving their functional biomedical properties and biomaterial usages. 48,49,53 CS/HA nanocomposites possessing uniform micro-architecture have undergone preparation via in situ synthesis, precipitation, in situ co-precipitation via electrospinning procedure, solvent casting and evaporation method, simple in situ hybridization, in situ chemical, freezing and lyophilization, and so on. 48,49,53,92,93,99
CS/CNT tissue engineering
It has been noted that CNT combination with CS results in enormous improvement in composite mechanical strength. 107 CNTs have garnered great attention with regard to biomaterials, specifically those placed in contact with natural bone such as prostheses used for arthroplasty, plates/screws used in fracture fixation, drug conveying systems, and scaffold for bone regeneration. 108 The most vital aspect of CNTs usage in biomaterials includes tissue safety; however, investigation into CNTs toxicity has not often been carried out. 109
Specifically, bone tissue compatibility is highly vital when using CNTs in biomaterials. Investigations into CNTs have revealed that these tubes exhibit effective bone tissue compatibility and have ability to allow bone mending when in contact with bone tissue, while accelerating bone formation and stimulating bone morphogenetic protein. 110
However, a notable challenge in bone tissue engineering is ascribed to scaffold mechanical strength enhancement. A major challenge when preparing CS-based composites is enhancement of mechanical behavior. Many studies have been conducted in fabrication of CNT/PNC for scaffolds used in tissue engineering. 107 -110
Nondegradable PNC for bone replacement
Ultrahigh molecular weight polyethylene
Ultrahigh molecular weight polyethylene (UHMWPE) is an effective material used in arthroplasties. 88 This is because it combines excellent wear resistance and very high fracture toughness and biocompatibility when compared with other polymeric materials. UHMWPE is a unique polymer exhibiting high physical, chemical, and mechanical attributes. 111 Other notable properties include impact inhibition, lubricity, chemical inertness, and abrasion hindrance. UHMWPE is a semicrystalline polymer and commonly applied as carrier in human joint replacing procedures and as synthetic bones. These special characteristics have led to its versatile application in medicals especially in orthopedic devices. Orthopedics has found effective use for UHMWPE especially as load carrier in synthetic joints. UHMWPE demonstrates high wearing resistance, minimal frictional surface, biological neutrality, excellent strength, creep resistance, and low frictional co-effcient. 112,113 Various inorganic nanomaterials have been used as fillers in UHMWPE nanocomposites. These materials include MMT, kaolin, aluminum oxide, quartz, carbon, and HA.
Polyethlene/HA composites for tissue engineering
Fundamentally, inclusion of nanoparticles into polymer matrices has improved the properties of PNC such as flame retardancy, electrical, thermal conductivity, thermal expansion resistance, and so on. 114 -131 These attributes have resulted in effective use of these materials in automobiles, built-tech, aerospace, packaging, biomedicals, pharmaceuticals, and so on. Two forms of polymer materials used in fabricating polymer/HA composites are nonbiodegradable and biodegradable polymer composites. As already elucidated in previous sections, biodegradable polymer matrices include PLGA, PCL, PHB, among others. Another class of nonbiodegradable polymeric matrices includes high density polyethylene (HDPE), polymethylmethacrylate (PMMA), polyamide (PA), ultra high molecular weight polyethylene (UHMWPE), and others. In a recent study, porous PE scaffolds have been studied as bone replacement in vitro and in vivo in nano-dimensions for calvarial bone damage in transgenic rats. Microscopic analysis shows that the pores looked similar to a network connection linked with the material, placing them effective cell-development materials. 14 Bone regeneration activity at the border was revealed, showing that PE functioned as anosteo-conductive bone-graft. In addition, bone formation in the pore interior of coated PE significantly enhanced osteo-integration processes. Figure 12 shows varying micrographs garnered from the study. 14

SEM images of porous PE. Surface scale bar = 100 m. (b) Microradiograph for CS-HA and SEM powder showing CS in rectangular and rod-like shapes (arrows) and smaller particles of HA. Scale bar = 100 m. CS microradiographic images for CS-HA SEM, which attained compact structures of well-interconnected crystals. The HA crystals are shorter than the CS crystals. Scale bar = 2 m. Microradiograph for SEM of porous PE. (c and d) Cut section of non-coated porous PE; the variation in pore size is noted to vary within the range of 50–400 m (c and d) scale bars = 500 and 100 m; (e and f) coated porous PE. 14
Conclusion and future prospects
Presently PNC are widely utilized in tissue engineering and all fields of advanced orthopedic medicare, especially in dentistry and fabrication of lower-limb sports prostheses. The major benefit of utilizing polymer-fiber composites for orthopedic functions is linked with their excellent specific strength and biocompatibility behavior. Scaffolds essential for bone tissue engineering are intricate. Numerous factors including interfacial chemistry, micropore architectures, mechanical strength, rate of degradation, porosity, and topographical parameters are to be placed in careful perspective to enable effective design and fabrication of scaffolds to meet bone tissue engineering applications. Nanocomposites have widely been researched as scaffolding materials because the polymers used as matrices are easily processable in comparison with ceramics and metals, aside being biodegradable, biocompatible, and bioresorbable. Additionally, with regard to bone–cells interaction with nanostructured materials, inclusion of certain nanoparticles in polymers enhances biological behavior of the scaffolds. Due to their nanoscale dimensions, nanoparticles larger surface area result in superior mechanical behavior, which, when combined with polymer, are capable of mimicking the properties of natural bone. Thus, PNC are widely utilized in bone tissue regeneration. Numerous challenges exist, such as achievement of uniform nanoparticles dispersion in the polymeric matrix and compatibility stability between the phases. Moreover, use of conventional techniques including electrospinning, solvent-casting and particulate-leaching, phase-separation, and so on for scaffolding has limited prospects for controlling the interior and exterior microstructure of scaffolds including pore size, pore morphology, pore interconnectivity, and porosity. Thus, the quest for bone tissue engineering has emanated so as to replenish deficiencies of present treatment techniques. Moreover, majority of scaffolds fabricated via conventional techniques exhibit inferior mechanical behavior. However, 3-D-scaffolds progress also depends on a combination of essential materials with proper design and production techniques. The evolvement of UHMPE/HA composites for full- or partial-joints replacement is a novel and enchanting field. The incorporation of nano-HA has been revealed as an effective route for improvement of the wearing resistance of UHMWPE/polymer matrix without essentially compromising other mechanical properties. Polymeric biodegradable/bioresorbable materials such as PHAs, PCL, PGA, and PLA are increasingly being applied for craniomaxillofacial fixation, sutures, interference screws, fixation plates and pins, and meniscal mending. Nowadays, CS-based composite materials have domineered the field of bone tissue engineering. The mixing of biocompatible polymers with bioresorbable ceramic materials can mimic the natural function of bone. CS-HA composites are confirmed prospective bone implant materials with effective osteoinductive, osteoconductive, and osteogenic properties. The architectural, mechanical, chemical behavior and in vitro investigation of CS, HA, and CNT have been conducted for bone tissue engineering, though some challenges, especially mechanical parameters require further research to attain viable solution. Hence, studies are still ongoing to address the mechanical parameters challenges. Further research is still essential in addressing CNTs cytotoxicity. Although some challenges still abound, CNT inclusion to enhance mechanical behavior of CS-ceramic composites would enable facilitation and stimulation of the natural bone functions. Technical improvements in the efficiency of CS composites will open the window for greater opportunities in the future for bone tissue engineering.
Footnotes
Acknowledgements
The author wish to acknowledge Prof. Charles Esimone, FAS, Vice Chancellor, Nnamdi Azikiwe University, Awka, Nigeria, and Prof. Azman Hassan of Universiti Teknologi Malaysia for provision of enabling environment for the success of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
