Abstract
This paper deals with the effect of melamine cyanurate on the thermal and fire retardant behaviour of chopped glass fibre (40 wt.%) reinforced polyphenylene ether/polystyrene/nylon-6 (40/40/20 wt. %) which are processed in a twin screw co-rotating extruder. The composite specimens were fabricated by injection moulding. Addition of melamine cyanurate decreased the amount of toxic gases and organic volatile compounds, crystallinity, crystalline temperature and melt temperature but the same fire-retardant increased glass transition temperature and heat distortion temperature of the ternary alloy. Flame retardancy with V0 and V2 rating along with Limiting Oxygen Index 35% was observed in UL94 test for 5 wt. % and 10 wt. % addition of melamine cyanurate, respectively. Addition of melamine cyanurate increased tensile modulus and elongation at break but decreased the melt flow rate and the moisture content of the ternary alloy.
Keywords
Introduction
Polyphenylene ether (PPE) is an amorphous dimensionally stable polymer with limiting oxygen index (LOI) 28%, higher heat distortion temperature and lower specific gravity. The test and flame retardancy standards are evolved by Underwriters Laboratory. UL94 Standard provides a method for rating the ignition characteristics of plastic materials which are subjected to horizontal or vertical burning. To achieve V rating (V0, V1 or V2), the specimens, placed vertically with the test flame impinging on the bottom of the sample, must extinguish within specified times, not burning to the top clamp or dripping molten material which ignites a cotton indicator. It exhibited moderate flame retardancy with V2 rating 1 in UL 94 flame retardancy test.
PPE needs blending polymers such as polystyrene (PS) for processing in twin screw co-rotating extruder. PS exhibited LOI of 17% and did not pass UL 94 rating due to fire dripping. PPE/PS blends possess superior mechanical properties1-4 but are weakly resistant to solvents and halogens. Nylon-6 is a crystalline polymer with high melting temperature, good tensile strength, and excellent chemical resistance. It is hygroscopic due to the presence of amide groups.1-5 It showed LOI of 24% and UL 94 V2 rating in fire retardancy test.5, 6 PPE/nylon-6 blends possess high melt viscosity but are prone to colour shifting due to UV exposure. 7
For achieving superior mechanical, thermal and moisture resistance properties, ternary blends of PPE/PS/nylon-6 with 40/40/20 wt. % were fabricated and tested. The blends prepared without using coupling agent showed superior mechanical and thermal properties. 8 Injection moulded PPE/PS/nylon-6/glass (40 wt. % chopped fibres and without maleic anhydride) showed tensile strength of 112 MPa, flexural strength of 201 MPa and flexural modulus of 30.85 GPa. 9
Melamine cyanurate (MC) is a white, non-toxic, crystalline powder, inorganic non-halogen flame retardant additive with high thermal stability up to 320°C.4-6 MC undergoes endothermic decomposition above 320°C and it acts as a heat sink. It is widely used as a flame retardant with polymer blends in applications such as construction, automotive and electrical appliances. Several authors reported that MC strongly affected the hydrogen bonding network formation facilitating decomposition of nylon-6.10-19 MC is more effective when used in less than 10 wt. % with nylon-6 20 and enhances flame retardancy of polyamides. 21 MC primarily acts as a drip promoter in the combustion of polyamides in UL94 test. 12
Murashko et al., reported that addition of 15 wt. % phosphinate to PPE/nylon 66 blends enhanced flame retardancy but decreased mechanical proprieties of the blends due to weak interface between phosphinate and PPE/nylon 66. 5 Takeda K et al., reported that phosphinate is not an effective flame retardant with PPE/PS blends. 2 Polyamide with 13 wt. % MC showed fire retardancy of V0 rating as burning stopped within 10 s and LOI of 32.8%.12,20 Wu ZY et al., suggested minimum addition of 15 wt. % MC to nylon-6 to achieve V0 rating. Specimens of nylon-6/MC prepared via in situ polymerisation and observed that 6.8 wt. % MC specimens showed excellent mechanical properties and flammability with V0 rating in UL-94 test. 21 Glass fibre reinforced PPE/PS/nylon-6 ternary blend (TB) composites exhibited superior mechanical properties. 9 MC has high thermal stability up to 377°C. 4 Halogenated fire-retardants release dense smoke and toxic gases and hence they must be replaced with halogen-free alternatives. Extensive research is reported on enhancing fire safety of polymers by using halogen-free fire retardants such as melamine derivatives, phosphorus-based compounds and polyphosphazenes. Melanin cyanurate, which is a salt of melamine and cyanuric acid, is widely used as a non-halogenated flame retardant in polymer composites.22,23
Phosphinate as a non-halogenated fire retardant is widely used in polymer blends and composites, but with a compromise on their mechanical properties and thermal stability. MC decreases the melt temperature, releases low toxic volatiles during decomposition and increases Tg of PPE/PS/nylon-6 blends and composites. Effect of MC on flame retardant behaviour and thermal stability of binary or ternary blends of PPE/PS/nylon-6 and their composites is not yet reported. This research was aimed at enhancing thermal and fire retardant properties of PPE/PS/nylon-6/glass (chopped fibre) using MC as non-halogenated fire retardant filler. UL94 and LOI tests were conducted. Differential scanning calorimetry (DSC) for crystallinity and Tg, thermo gravimetric analysis (TGA) for thermal degradation temperature, Fourier-transform infrared spectroscopy (FTIR) for functional groups and scanning electron microscopy (SEM) for interfacial properties were used for the study. Heat distortion temperature, moisture resistance and mechanical properties of the composites were evaluated.
Experimental Details
Materials and processes
PPE of grade PX-100L (Mitsubishi Engineering, Singapore), PS of grade SC201LV (Supreme Petrochem Ltd, Mumbai, India), nylon-6 of grade PA6 (LUHS1, Loxim, Ahmadabad, India), e-glass chopped fibre of grade 300, diameter 14–17 μm (Jushi, China) and MC white powder grade MC25 particle size 5–7 μm (Unibrom, China) were used for the experimental study.
Ternary blends and glass fibres with MC were compounded in a twin screw co-rotating extruder (TSE), Figure 1(a) (OMEGA 40, Steer, Bangalore, India), length to diameter ratio of screw 1:44, screw speed 300 r/min and barrel temperature 160°C–280°C. Vacuum was applied at barrel-8 before the metring zone. The three polymers were fed directly to the intake barrel (barrel 1). MC and chopped glass fibres were fed through side feeders (barrel four and six, respectively). Melting of the materials occur at barrel 3. Dispersion and distribution occur at barrel five and seven, respectively, due to the conveying and the kneading elements (Figure 1 (b)). Dynamic stirring elements and fractional mixing elements were employed in the TSE.
8
These elements perform mixing, fibre wetting and fibre distribution which favour retaining fibre length and improving mechanical properties. Lower process energy and shear rate due to the elements result in lower temperature of the melt avoiding possible discolouration of the melt. The hot melt was passed through a die and water bath for quenching and pelletising. The composite pellets were passed through injection moulding machine (L&T, 100 tonnages, S- series) at mould temp 260°C and back pressure 1.0 MPa. The injection moulded specimens were aged at 23°C for 16 h as per ISO 527. (a): Preparation PPE/PS/nylon-6 ternary blends with chopped glass fibre and MC using TSE, and (b): Zones/barrels of extruder used for compounding.
Testing of specimens
Thermal Characterisation of PPE/PS/Nylon-6/glass with MC
Glass transition temperature (Tg) and melt temperature (Tm) were determined using DSC as per ISO 11,357 (Model TGA 4000 Perkin Elmer, Bangalore, India). Specimens measuring 10 mg each was subjected to heating and cooling from 30°C to 300°C at 10 °C/min using N2 gas @ 50 mL/min.
Crystallinity of the specimens was computed using equation (1)
TGA as per ISO 11,358 was performed on 10 mg specimens by heating from 30°C to 900°C at 10 °C/min using N2 gas @ 50 mL/min (Model Q 200, TA Instruments Bangalore, India).
FTIR spectroscopy was performed as per ASTM E168, in transmission-mode with Perkin Elmer Spectrum 100 FTIR spectrometer. The device is fitted with a single-bounce diamond/ZnSe ATR accessory and room temperature detector. Four background and four sample scans were performed. The spectral resolution was four wave numbers. The default scan range is 750–4000 cm−1 at a scan speed of 0.2.
Flammability of PPE/PS/Nylon-6/glass with MC
PPE/PS/nylon-6/glass with MC specimens were injection moulded (L&T Demag 100 Ton, 2004 model). Specimens (ASTM D 3801) for fire retardancy test were prepared with dimensions 125 ± 5 mm x 13.0 ± 0.5 mm x 1.6 ± 0.1 mm. A total of 10 (2 sets) of specimens for each condition were tested at 23 ± 2°C, RH 50 ± 5% for 48 h, and another 10 sets of samples were prepared for ageing in air oven at 70 ± 2°C for 168 h ± 2h as per the UL 746B. The aged samples were kept in desiccators for 4 h/23±2°C and Relative humidity (RH) below 20%. Each specimen was mounted with longer axis vertical and supported its lower end at 10 mm above Bunsen burner tube. A 20 mm high blue flame was applied to the centre of the lower edge of the specimen for 10 s and withdrawn. As per the UL 94 test procedure, if burning ceases within 30 s, the flame is reapplied for an additional 10 s. If the specimen drips, particles are allowed to fall onto a layer of dry absorbent surgical cotton placed 300 mm below the specimen. Limiting oxygen index specimens with dimensions 100 ± 5 mm x 10.0 ± 0.5 mm x 4.0 ± 0.25 mm was carried out as per ASTM D 2803 using Stanton red croft.
Mechanical testing and Heat distortion temperature of PPE/PS/nylon-6/glass with MC
Tensile modulus of the specimens (150 × 10 × 4 mm3) was determined using Instron Universal Testing machine, model 5569 A, (TA Instruments division, Bangalore, India), as per ISO 527 at cross head speed 5 mm/min. Tensile fractured surfaces were examined using SEM (1 × 105 X) after gold sputtering to impart conductance (JSM-IT, model 300LV, Mysore, India). Un-notched Izod impact tests were conducted on the specimens (80 × 10 × 4 mm3) using model 7611 (CEAST, Bangalore, India) as per ISO 180, hammering with 5.5 J. Density of the specimens was determined on 10 mg specimens at ambient temperature as per ASTM D792 using (Mettler Toledo, Chennai, India).
Heat Distortion Temperature (HDT) of the specimens (80 × 10 × 4 mm3) was measured at two loads, 0.45 MPa as per ISO75, using (model 6921, CEAST, Bangalore, India) by increasing temperature at 2°C/min till the specimen deflected by 0.25 mm.
Moisture content was determined as per ASTM D 6930 using (model MA160-1, Sartorius, Bangalore, India). Melt flow index (MFI) was determined as per ISO 1133 (model 7028, CEAST, Bangalore, India). MFI measures the capability of the melted polymer to flow when pressure is applied. MFI is inversely related to viscosity of melted polymer at the conditions of the test, that is, 280°C at 5 kg load.
Results and Discussion
Thermal Stability of PPE/PS/nylon-6/glass with MC
TGA was performed to study the thermal stability and volatile organic compound of PPE/PS/Nylon-6/glass with MC (Figure 2). Results of decomposition temperature and residual are presented in Table 1. TGA of PPE/PS/nylon-6/glass with MC TGA of PPE/PS/Nylon-6/glass with MC TB: PPE/PS/nylon-6 ternary blend, VOC: Volatile organic compound.

PPE, PS and nylon-6 molecules have phenyl and acylamino groups, whereas MC molecules possess carbonyl, amino, and hydroxyl groups. PPE has greater flame resistance than that of PS or nylon-6. 1 PPE/PS/nylon-6 (40/40/20 wt.%) blend showed decomposition onset temperature of 425.18°C and addition of 40 wt.% glass fibre to the blend resulted in slight increase in decomposition onset temperature. PPE/PS/nylon-6/glass with MC showed two decomposition onset temperatures, that is, 336°C corresponding to MC and 416.24°C corresponding to the composite due to replacement of carbonyl groups with amine groups in cyanuric acid. MC completely disappeared above 450°C without any residual char.12,20 PPE/PS/Nylon-6/glass with MC 10 wt. % showed less volatile organic compound of 0.14. More number of hydrogen bonds was present in the PPE chain, which increased the thermal stability of PPE/PS/nylon-6/glass/MC (Figure 2).
Differential Scanning Calorimetry and Crystallinity
DSC results of PPE, PS, nylon-6, blends and composites with MC

TB: PPE/PS/nylon-6 ternary blend, Tg: Glass transition temperature, Tm: Melt temperature, Tc: Crystalline temperature.

DSC: (a) PPE/PS/nylon-6 and (b) PPE/PS/nylon-6/glass with MC
PS and PPE are amorphous in broad range of Tg and nylon-6 is crystalline and hence showed sharp melting point. PPE/PS/Nylon-6 blend with 40/40/20 wt. % exhibited lower Tg (127°C) and homogeneous phase (Table 2). PPE/PS have phenolic OH group which easily dissolve to form lower Tg. Glass fibre (40 wt. %) addition to PPE/PS/nylon-6 blend increased their Tg to 141.19°C (Figure 3(b)). Glass fibre decreases the melt flow rate and increases the back pressure which restricted the chain flexibility (Random chain) to form amorphous regions in PPE/PS/nylon-6/glass. Addition of MC to PPE/PS/Nylon-6/glass gradually increased glass transition temperature but decreased crystallinity, crystalline temperature and melt temperature due to higher amorphous content (PPE/PS). Lower crystallinity of PPE/PS/nylon-6/glass/MC is due to the irregular arrangement of molecular chain.
Fourier Transform Infrared spectroscopy
FTIR results of PPE/PS/nylon-6/glass with MC

WN: Wave number, FG: Functional group.
FTIR spectrum of PPE/PS/Nylon-6 shows functional group and wave number (Figure 4 (a)). PPE has CH3 group of PPE at 3699 cm−1, CH2 group of PS at 2921 cm−1 and NH3 group
24
of nylon-6 at 2360 cm−1 evidence homogeneous blending of PPE/PS/nylon-6.
25
Shift of OH stretching group from 3713 cm−1 to 3699 cm−1 is due to the Hydrogen bonding between the phenolic groups. PPE aromatic ring stretching at 1643 cm−1 overlaps with the carbonyl functional group of nylon-6 segment at 1548 cm−1. The ring stretching of PS segment at 1514 cm−1 is completely miscible with PPE segment at 1643 cm−1 resulted in increasing the wave number to form homogeneous blend as shown in Figure 4 (a).
26
Chopped glass fibre showed composition of SiO2 as 54 wt. % and CaO MgO as 22 wt. % as shown in Figure 4 (b). Glass fibre has functional group of Si-O-Si at 860-1085 cm−1. Bending and stretching of Si-OH group at 823 cm−1 increased the mechanical properties. Addition of melamine cyanurate to PPE/PS/Nylon-6/glass showed N-H groups at 3302 cm−1, OH group at 3712 cm−1, C=O at 1715 cm−1, C=N at 1665 cm−1 and C-N at 1448 cm−1 evidence homogeneous blending. Higher peak intensity was observed at C-N and Si-O-Si function group at 1448 cm−1 and 1185 cm−1, respectively, due to higher portion of MC (10 wt.%) (Figure 4 (b)). Changes in intensity were observed amongst PPE/PS/Nylon-6, PPE/PS/Nylon-6/glass and PPE/PS/Nylon-6/glass/MC at 3387, 3298, 3200, 3100, 1720, 1638, 1534 and 1448 cm−1. FTIR spectrum of: (a) PPE/PS/nylon-6, and (b) PPE/PS/nylon-6/glass with MC
UL94 Vertical Burning test and LOI
Vertical burning and LOI results of PPE/PS/nylon-6/glass with MC

TB: PPE/PS/nylon-6 ternary blend, T1: the self-extinguishing time after the first ignition, T2: the self-extinguishing time after the second ignition.

UL94 vertical burning test – Burnt specimens L1, L2, L3 and L4.
PPE/PS/nylon-6 specimen was fixed vertically and the flame was applied for 10 s at the lower edge of the specimen. Burning ceased in 20.8 s. The flame was reapplied for additional 10 s. Specimen L1 (Figure 4) did not show UL94 rating. The same procedure was used for testing PPE/PS/nylon-6/glass (L2). Glass fibre reinforced specimens reduced melt dripping with no UL94 rating. PPE/PS/nylon-6/glass/MC (5wt %) L3 specimen showed V1 UL 94 rating (no melt dripping no cotton ignition), Figure 5. PPE/PS/nylon-6/glass/MC (10 wt. %) L4 specimen showed V0 UL 94 rating, Figure 5. MC has NH2 and NH groups which help in the formation of hydrogen bonding between the phenolic, amine, Si-O-Si groups to form carbon layer on PPE/glass. The hydrogen bonding so formed acts as thermal barrier or char, preventing heat transfer to PPE/PS/nylon-6/glass.
LOI tests were performed to determine minimum oxygen required to start combustion of the specimen and keep it burning. Higher LOI indicates superior flame retardency. 19 PPE/PS/nylon-6 showed lower LOI of 23% and addition of glass fibre increased LOI of 28% (Table 4). PE/PS/Nylon-6/glass/MC 10 wt. % showed highest LOI of 35%.
Burning mechanism of PPE/PS/nylon-6/glass/MC
PPE/PS/nylon-6/glass/MC was processed using twin screw co-rotating extruder by melt blending. PPE and PS are thermodynamically compatible due to phenolic group. PPE is made of phenylene rings linked together by ether linkages in 1, 4 or para-positions, with a methyl group attached to carbon atoms in 2 and 6 positions and PS has styrene (CH2) functional group to form PPE/PS blend (Figure 6). Char mechanism PPE/PS/nylon-6/glass/MC.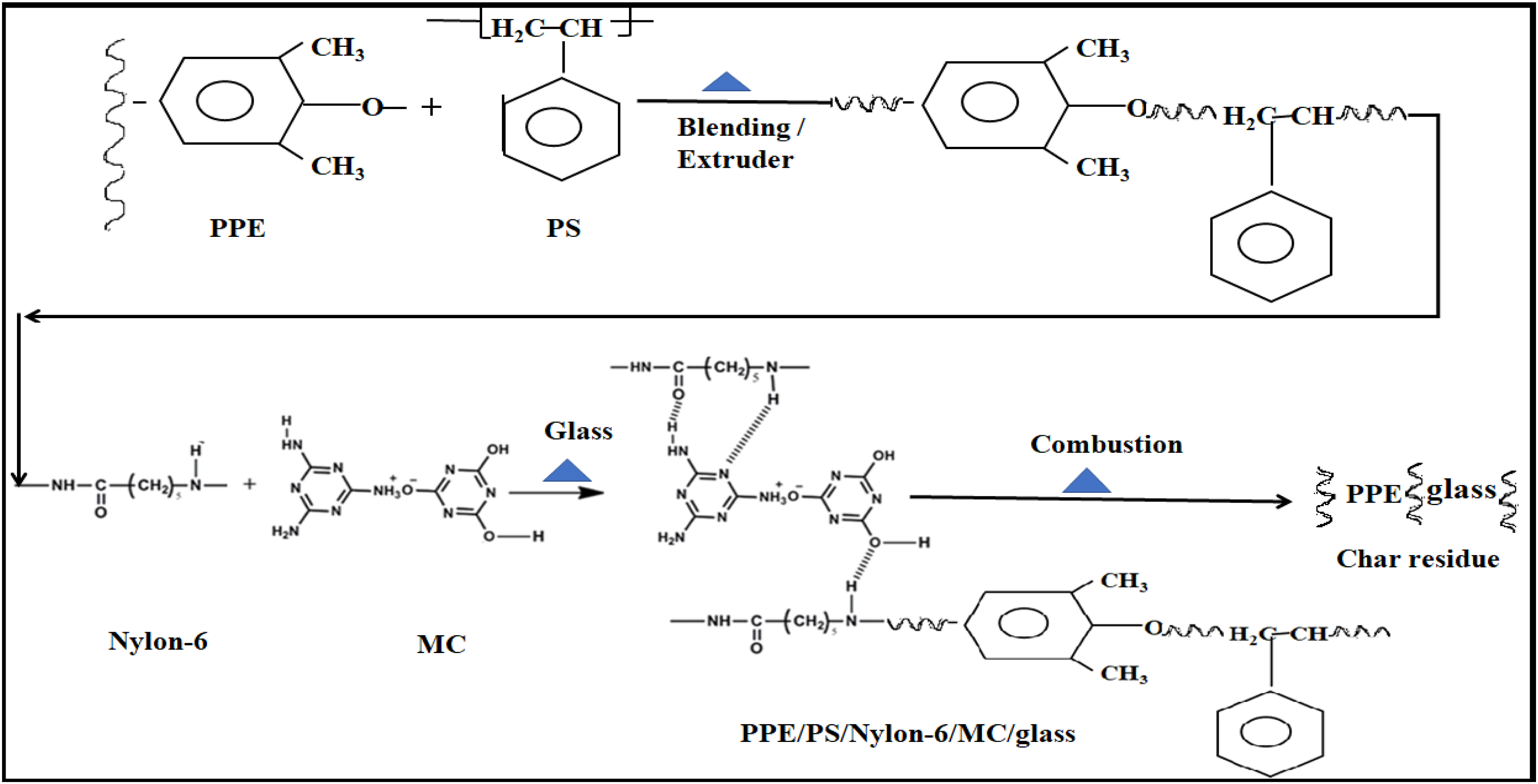
The main chain of PPE/PS blend is attached to nylon-6 in which the methylene bridge bonds in place of the ether bond. Nylon-6 has amino (NH2) functional group and hence it blends with PPE/PS to form PPE/PS/nylon-6 blend. TGA of PPE/PS/Nylon-6/glass/MC showed decomposition temperature of MC of 336°C. In the process of degradation, MC released non-inflammable gases and less volatile components. PS/nylon-6 started decomposing at 450°C leading to the formation of styrene, caprolactam, alkyl cyanides and NH3 groups. 18 However, the residue or char formation on the surface of PPE/glass acted as barrier of heat transfer to MC. 2
Fire-retardant mechanism on PPE/PS/nylon-6/glass/MC
To study the fire retardant mechanism of PPE/PS/nylon-6/glass/MC, the specimen was initially weighed and kept in the furnace at 300°C for 30 min. The specimen was withdrawn from the furnace, cooled in a desiccator and weighed again. The procedure was repeated (Figure 7) with increase in furnace temperature in steps of 50°C up to 450°C, that is, TGA of MC. The specimen colour changed from white to brown at 300°C (Figure 7). At 350°C, specimen colour changed from brown to ‘charcoal gray’ and released gases. At 450°C, the specimen colour changed from light dark to dark. The MC layer formed thermal barrier and prevented PPE/PS/nylon-6/glass from burning. Fire retardant mechanism of PPE/PS/nylon-6/glass with MC
Mechanical and physical properties and HDT
Mechanical, physical and HDT properties of the PPE/PS/nylon-6/glass/MC.

TB: PPE/PS/nylon-6 ternary blend, TM: Tensile Modulus, EB: Elongation at break, IS: Impact strength, HDT: Heat Distortion Temperature, MFR: Melt flow rate.
PPE/PS/nylon-6 (40/40/20) wt. % showed tensile modulus of 3.63 GPa, elongation at break 2.49 and impact strength 20.82 KJ/m2. 8 The corresponding properties of PPE/PS/nylon-6/chopped fibre (40 wt. %) were 11.11 GPa, 1.28 and 13.93 KJ/m2. 9 Addition of MC to PPE/PS/nylon-6/glass increased these properties along with HDT (Table 5). Additionally, MC decreased moisture content, increased density and decreased MFR (due to decreased viscosity) of the specimens.
Dispersion of MC in PPE/PS/nylon-6/glass
Scanning electron micrographs of tensile fractured PPE/PS/nylon-6, PPE/PS/nylon-6/glass and PPE/PS/nylon-6/glass with 5 wt. % and 10 wt. % of MC are shown in Figure 8 (a) to (d). At higher magnification PPE/PS/nylon-6 (40/40/20) showed homogeneous phase (Figure 8 (a)), which is evidenced by single Tg in DSC results and superior mechanical properties. PPE/PS/nylon-6 with glass 40 wt. % showed smooth surface, good fibre wetting and distribution, absence of voids (Figure 8 (b)), evidenced by superior tensile and flexural strength and flexural modulus.
9
Scanning Electron Micrographs of Figures 8 (c) and (d) revealed even distribution of MC with no voids which is evidenced by superior fire properties. SEM micrographs of tensile fracture surfaces of flame retardant/glass fibre reinforced PPE/PS/nylon-6 (40/40/20) composites, a) to d) of L1 to L4 specimens.
Conclusion
Effect of halogen-free melamine cyanurate on the thermal and fire retardant behaviour of PPE/PS/nylon-6/glass was studied by preparing the composite specimens with 5 wt. % and 10 wt. % melamine cyanurate and 40 wt. % chopped glass fibre. The specimens were fabricated by twin screw co-rotating extrusion and injection moulding. The specimens were studied for thermal decomposition, crystallinity, flame retardant behaviour, mechanical properties and morphology.
PPE/PS/nylon-6/glass with 10 wt. % melamine cyanurate showed least volatile organic compound of 0.14 wt. % and superior thermal stability indicated by high degradation temperature. These specimens showed highest glass transition temperature of 146.50°C due to melamine cyanurate and the phenolic group of PPE/PS. The specimens showed UL 94 V0 rating, Limiting Oxygen Index of 35%, superior tensile modulus, elongation at break, impact strength and heat distortion temperature. SEM micrographs showed even distribution of MC without voids.
Footnotes
Acknowledgements
The special thanks Dr Babu Padmanabhan STEER engineering private limited, Bangalore
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

