Abstract
High swellable and biocompatible hydrogel composed of optimized composition of N-vinyl pyrrolidone (NVP), chitosan (CS), and itaconic acid (ITA) was prepared by the aid of gamma radiation. Certain optimized composition of the terpolymer hydrogel was used as a precursor for the preparation of its ZnO nanocomposite. The terpolymer hydrogel was characterized by various techniques FTIR, XRD, SEM, and TEM to evaluate the physical, chemical properties and morphology of the prepared hydrogels. FTIR proved the high interaction between the components of the terpolymer crosslinked hydrogel. Transmission Electron Microscope (TEM) confirmed the preparation of ZnO nanoparticles in the nanoscale with average particle size of 12.3 nm in the terpolymer matrix which is higher than the particle size of as prepared bare ZnO NPs. The gelability, crosslinking density, and high swellability of the prepared terpolymer hydrogel make it promising and suitable for application as wound dressing materials. In this respect, the NVP/ITA/CS terpolymer hydrogel first examined for controlled release of amoxicillin and second, the antibacterial test showed that neither Staphylococcus aureus nor Escherichia coli passed through the terpolymer nanocomposite hydrogel dressing; NVP/ITA/CS/ZnO nanocomposite hydrogel dressing could be considered a good blockade against bacteria. All results indicate that the nanocomposite hydrogel has great potential for biomedical applications, especially for wound dressings.
Keywords
Introduction
Preparation of gelable biocompatible and biodegradable materials with suitable swelling and mechanical properties is a very important characteristic for such materials to be applied as wound dressing materials. Many attempts have been made by many authors to prepare such materials using various types of polymers and polymer nanocomposites that are based on natural and synthetic polymers.
Chitosan (the N-deacetylation product of chitin) has been widely studied in pharmaceutical and biomedical fields because of its biodegradability, biocompatibility, nontoxic and interesting structural properties (presence of amino and hydroxyl groups).
1
This biopolymer can be easily obtained from chitin, an abundant and renewable material in the biosphere. The water sorption capacity of CS is limited, so that in order to obtain highly swellable CS, hydrogels mixtures with hydrophilic polymers such as polyvinyl pyrrolidone or poly(vinyl alcohol) (PVA) and polyelectrolyte complexes of CS with polyanions (carboxymethyl cellulose, alginate, pectin, poly(acrylic acid) have been prepared.2,3 Semi-interpenetrated or interpenetrated polymer networks with these polymers give rise to CS hydrogels with improved mechanical properties. Itaconic acid (ITA) is a dicarboxylic acid commercially produced by the fermentation of carbohydrate, primarily glucose, using
N-vinyl-2-pyrrolidone (NVP) is a water-soluble, nonionic monomer that can be used to prepare hydrogels. NVP-based materials have been of interest, for example, in biomedical applications. 6 NVP tends to homopolymerize under the effect of radiation and so mixing it with itaconic acid resulted in very loose with very poor mechanical properties hydrogels; these properties can be improved by mixing the NVP/ITA with chitosan which enhances the crosslinking under low doses of radiation. ITA easily copolymerizes with NVP monomer and provides polymer chains with carboxylic side groups, which are highly hydrophilic and able to form hydrogen bonds with corresponding groups in chitosan. ITA is expected to show high biocompatibility because of its natural source. 7 Coupling of temperature-responsive and pH-sensitive components allows a flexible control over the polymer phase behavior, resulting in multifunctional smart materials such as (NVP/ITA/CS copolymer hydrogels.
It is highly encouraged in the last few decades to apply gamma radiation technique as a clean tool for preparation of pure hydrogels containing only its main components without the need to add other chemical crosslinkers or initiators which can affect badly on the safety of the prepared hydrogels especially in case of biomedical application.
Additionally, the incorporation of metal oxide nanoparticles into the very loose polymer hydrogels increases the extent of crosslinking through the chelation of metal (Zn, Ca, Mg,…) with chitosan which resulted in hydrogels with improved physical and mechanical properties.8,9
Inorganic nanoparticles-containing hydrogel or nanocomposite hydrogel is one of several classes of unique materials that can replace antibiotics. These nanocomposite hydrogels are acting on the entire cellular membrane of bacterial cell, through electrostatic interaction and subsequent insertion of their metallic nanoparticle components into the bacterial membrane to generate pores which as a result lead to cell damage, destroying of cytoplasm, and then death of bacterial cell. 10 Recently, there has been a major advantage in generating antibacterial nanocomposite hydrogels by the incorporation of inorganic nanoparticles, like CuO, ZnO, MgO, and silver nanoparticles.11–13 The main advantage of using inorganic nanoparticles accompanied with the organic antimicrobial mediators is their strength, stability, and extended shelf life; besides, they can be used potentially for biomedical field. 14
It is evidenced that the antibacterial action of antibacterial nanocomposite materials passes through various mechanisms:15,16 Reactive Oxygen Species-Induced Cytotoxicity (ROS), Microbial Cell Envelope Interruption, Discretion of Nucleic Acid, Protein Production, and metal ion discharge. The detailed antimicrobial mechanism supposed of polymer nanocomposites is based on the steps clarifying how bacteria disappear in the physiological environment. The bacteria absorbed on the polymer surface and dissolved oxygen penetrates into the nanocomposites from the surrounding medium. Oxygen molecules reach the surface and the deterioration route with the embedded metal nanoparticles occurs. Metal ions are released and reach the polymer composite surface, thus initiating destruction to the bacteria membrane and are eventually spread into the bacteria17,18.
This work deals with the preparation of NVP/ITA copolymer by the effect of gamma radiation then based on the gel fraction of the formed copolymers. The composition NVP/ITA with 10% itaconic acid was selected to prepare fast swellable NVP/ITA/CS with various contents of CS, and the resulted hydrogel with good mechanical and crosslinking density was used for incorporation of ZnO NPs for studying its use as wound dressing material.
Materials and experimental techniques
Materials
Chitosan (CS) (Mw: 100000-300000), N-vinylpyrrolidone (NVP) 100%, and itaconic acid (ITA) purchased from Sigma-Aldrich company. Zinc acetate, and sodium hydroxide purchased from Acmatic Co. Egypt. Doubly distilled water was used for all preparations.
Preparation of NVP/ITA hydrogel
N-vinylpyrrolidone (100%) (NVP) and itaconic acid 5% wt/v prepared by dissolving 5 g of ITA in 100 ml distilled water, were mixed with various compositions where the content of Itaconic acid (ITA) varied from 5%, 7%, 10%, 15%, upto 20% (v/v) and then exposed to 30 kGy irradiation dose. Based on the gel fraction of the formed NVP/ITA various hydrogels, the NVP/ITA composition contains 10% of ITA selected for the preparation of NVP/ITA/CS hydrogel.
Preparation of NVP/ITA/CS hydrogel
To the NVP/ITA (10%) composition mixture, different Chitosan (CS) (1% wt/v) contents 2%, 4%, 6%, 8%, and 10% v/v were added on a time and then exposed to 30 kGy of gamma radiation; then, the formed NVP/ITA/CS hydrogels were cut to nearly equal pieces, dried at room temperature, and kept in polyethylene bags for further use and application. Co-60 radiation source available in the national center for radiation research and technology (NCRRT), Cairo, Egypt, was applied for the preparation of all hydrogels at a dose rate 1.11 kGy/h.
Preparation of NVP/ITA/CS-ZnO nanocomposite
Incorporation of ZnO nanoparticles (ZnO NPs) within the NVP/ITA(10%)/CS(10%) polymer network was carried out in two steps: first, preparation of a stock solution of Zn acetate solution of 1% wt/v concentration by dissolving 1 g zinc acetate in 100 ml H2O, then soaking the NVP/ITA/CS hydrogel in Zn acetate solution for 24 h tell complete absorption; second, the resulted hydrogels loaded with Zn ions (colorless gels) were washed carefully and then 100 ml of NaOH (1M) was added to initiate formation of ZnO NPs where the colorless gels turn to cloudy white in color as indication to the formation of ZnO NPs as shown in Figure 1. Digital photographs of NVP/ITA/CS hydrogel before and after loading of ZnO NPs.
Characterizations
Gel percent of prepared hydrogels
First, (NVP/ITA) and (NVP/ITA/CS) hydrogel samples were kept in distilled water for 4 h under heating to remove soluble part of samples; then, the hydrogels were removed and again dried until constant weight is reached. The gel fraction percent (GF %) was calculated gravimetrically by using the following equation
Equilibrium swelling
The hydrogels samples with certain weights were soaked in distilled water and left to swell at room temperature for 24 h. Swollen gels were removed from water, then put in filter paper to remove outer surface water and weighed at several time intervals. The measurements were continued until a constant weight is reached for each sample (equilibrium stage). The swelling percent (Q) at equilibrium was calculated using the following equation
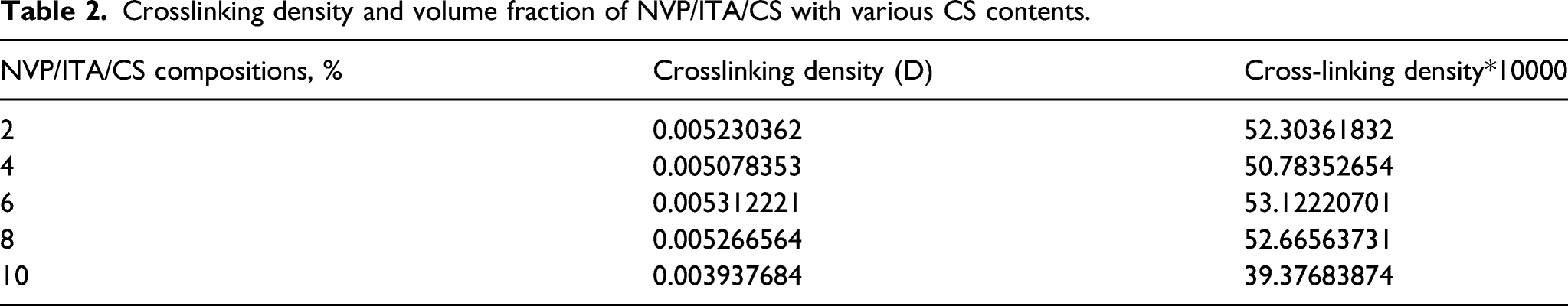
The crosslinking density of the prepared NVP/ITA and NVP/ITA/CS various hydrogel was calculated by applying the Flory–Rehner equation and its related derivatives (equation 3–6)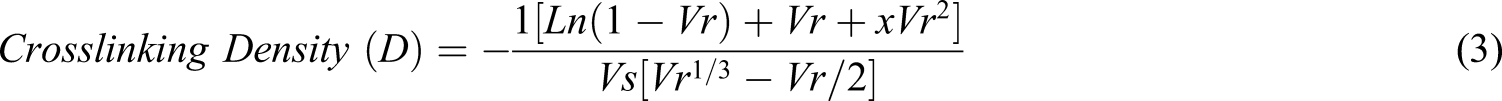

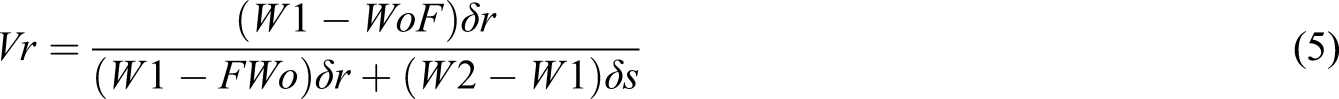

FTIR Spectroscopic Measurements
The FTIR spectra of (NVP/ITA) and (NVP/ITA/CS) hydrogel over the range 400–4000 cm−1 were recorded using Fourier-Transform Infrared Spectroscopy FTIR-Vertex 70 spectrophotometer (Bruker, Germany).
Scanning Electron Microscopy
Scanning electron microscope (SEM.JSM-5400, Japan) was used to investigate the morphology of the surfaces of the (NVP/ITA) and (NVP/ITA/CS) hydrogel and (NVP/ITA/CS)-ZnO nanocomposite. A sputter coater was used to pre-coat conductive gold onto the fracture surfaces before observing the micrographs at 30 kV.
X-ray Diffraction
X-ray diffraction measurement was conducted for (NVP/ITA/CS)-ZnO nanocomposite, using X-ray diffraction operated at Cu K at wavelength of 1.54 A, 30 nmA and 40 keV. The X-ray data were recorded in the range from 4 to 80 (degree) 2θ with continuous scanning mode and scanning speed 8 (deg/min).
TEM Measurements
Nanocomposite (NVP/ITA/CS)-ZnO nanocomposite was powdered and suspended in water, and a drop of the resultant mixture was deposited on an ultrathin carbon supported Cu grid, and air-dried. Energy filtered electron powder diffraction used TEM JEOL: JEM-100cx (available at NCRRT).
Drug loading and release of NVP/ITA/CS-ZnO nanocomposite
Drug loading
Amoxicillin drug was dissolved to prepare drug solution at a concentration of 100 mg/ml. 0.1 g of NVP/ITA/CS/ZnO nanocomposite was immersed in 50 ml of prepared drug solution. The drug loading was studied at different physiological conditions at 24°C. The concentration of the drug was determined using UV–vis spectrophotometer. The drug loading % and loading efficiency % of hydrogel was calculated using following relations as
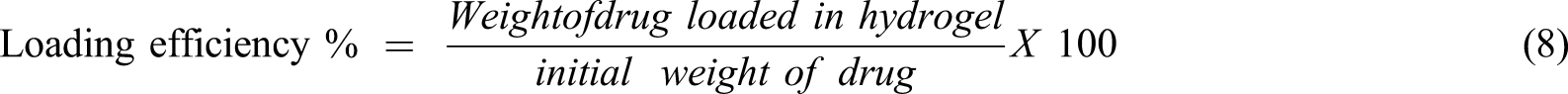
Drug release measurements
In vitro drug release rates were conducted at 37°C. Release experiments were done by placing the NVP/ITA/CS/ZnO nanocomposite samples loaded with drug (conc. 100 mg/ml) into 30 ml buffer solutions of pH 2.2 and 7.4. The amount of drug released from nanocomposite hydrogels was calculated using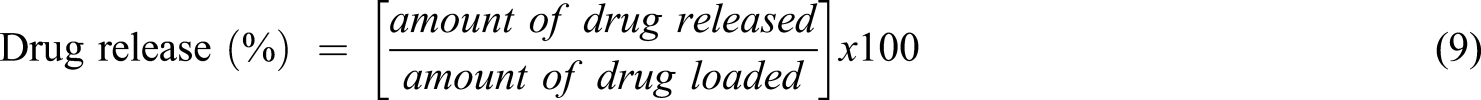
Kinetics of drug release
The mechanism of drug release was evaluated by using the well-known Fick’s equation
Porosity studies
Porosity studies were carried out by solvent replacement method to evaluate and analyze the porosity of hydrogel discs. Weighed dried (W1) samples of (NVP/ITA/CS) and (NVP/ITA/CS)-ZnO nanocomposite hydrogels were immersed in absolute ethanol of purity >99.9% for 48 h. After 48 h, discs of hydrogels were removed, blotted with tissue paper to remove excess solvent, and accurately weighed (W2). Thickness and diameter of discs were also measured. Hydrogel porosity was calculated by using the following formula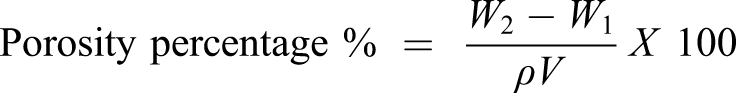
ρ refers to the density of absolute ethanol, and V is the volume of hydrogel after swelling
In vitro biodegradation of hydrogels
The biological stability of the hydrogels was measured by biodegradation tests in PBS solution. Specifically, an initial weight of the (NVP/ITA/CS) hydrogel and (NVP/ITA/CS)-ZnO dressings, approximately 0.1 g, was immersed in 20 mL of PBS solution at 37°C. Weight checking was achieved on days 1, 3, 5, and 14. At each recorded time interval, degradation was interrupted by removing the (NVP/ITA/CS) hydrogel and (NVP/ITA/CS)-ZnO dressings from the PBS solution and submerging it for 15 min four times in distilled water to remove the PBS solution adhered on the hydrogel. Then, it was sapped on filter paper to remove any excess water from the surface. Then, the (NVP/ITA/CS) hydrogel and (NVP/ITA/CS)-ZnO sponges were dried and weighed. The in vitro degradation % was calculated as a percentage of weight loss. The data were substituted into the following equation (11)
Antibacterial test
The antimicrobial activity of neat NVP/ITA/CS hydrogel, (NVP/ITA/CS)-ZnO nanocomposite, and the (NVP/ITA/CS)-ZnO/drug, against Gram-positive
Results and discussions
Gel fraction, equilibrium swelling%, and crosslinking density
Gel Fraction % and equilibrium swelling % of NVP/ITA/CS various hydrogels.

Crosslinking density and volume fraction of NVP/ITA/CS with various CS contents.
IR spectroscopy
The FTIR spectra of NVP/ITA/CS hydrogel and its components formulations NVP/CS and NVP/ITA are represented in Figure 2. For chitosan 100% the characteristic bands at 3462 cm−1, 2929 cm−1, 1662 cm−1, 1440 cm−1, 1270 cm−1, and 1185–994 cm−1 which corresponding to hydroxyl group, CH2 stretching vibration of pyranose ring, C=O in amide group,NH2 group, CH3 in amide group and C-O-C in glycosidic linkage are represented, respectively. Itaconic acid (100%) showed the characteristic bands for OH, C=O at wave numbers 2973 cm−1 and 1695 cm−1 respectively. The IR spectra of the NVP/CS, NVP/ITA, and NVP/CS/ITA hydrogel show the combination of the characteristic bands for its components with overlapping of the bands at the same wave number; also, it is noticed the dimensioning in the intensity of all bands was due to the strong interaction between the polymer components with each other. IR spectra of itaconic acid, Chitosan, NVP/CS, and NVP/ITA/CS hydrogel.
Morphological study of NVP/ITA/CS hydrogel and its NVP/ITA/CS-ZnO nanocomposite
Scanning electron microscopy
Scanning electron microscopy (SEM) is a traditional tool used to describe the morphological change that took place after mixing, copolymerization, or grafting of polymeric materials. In this regard, SEM photos of NVP/ITA/CS copolymer hydrogel and its ZnO nanocomposite are shown in Figure 3. Mixing of NVP, ITA, and CS and then exposing the mixture to gamma radiation resulted in a copolymer with rough surface and full of polymer aggregates with some cavities and hills due to the inhomogeneous effect of radiation during crosslinking processes (Figure 3a). In Figure 3b, incorporation of ZnO NPs within the NVP/ITA/CS copolymer hydrogel changes the morphology completely and gives rise to a very rough surface with some parts look like reefs with wires like appearance and full of small particles and agglomerated ones due to the imbedded ZnO NPs.
19
SEM photos of a NVP/ITA/CS hydrogel and b NVP/ITA/CS-ZnO nanocomposite.
Transmission electron microscopy
TEM images of pure ZnO nanoparticles and ZnO NPs imbedded in the NVP/ITA/CS polymer network are illustrated in Figure 4. By studying the TEM image of pure ZnO NPs, it appears highly dispersed and the size of the particles is nearly equal with similar shape. The estimated particle size of pure ZnO NPs from TEM image is 11.4 nm. On the other hand, the examining of the TEM image of ZnO NPs embedded in the polymer network of NVP/ITA/CS hydrogel shows some agglomeration of ZnO NPs with spherical NPs varied in size and shape due to coating with Polymer chains and the estimated average particle size was 12.3 nm. TEM images of pure ZnO nanoparticles and ZnO NPs imbedded in the NVP/ITA/CS hydrogel.
X-ray diffraction
XRD patterns of the prepared pure ZnO nanoparticles and ZnO incorporated in the crosslinked matrix of NVP/ITA/CS hydrogel are shown in Figure 5. Figure 5 shows the characteristic peaks of 32.06o (100), 34.98o (002), 36.66o (101), and 48.32o (102) corresponding to pure hexagonal wurtzit structure of ZnO NPs which corresponding to the ZnO diffraction peaks that confirmed by the (JCPDS NO. 36-1451).
8
Examining the diffractograms of NVP/ITA/CS-ZnO nanocomposites shows the presence of a broad peak at nearly 2-Theta: 20.24o which corresponds to the amorphous crosslinked polymer matrix of NVP/ITA/CS in addition to the characteristic peaks of ZnO NPs at 2-Theta: 30.82o, 34.56o, 36.42o, and 38.32o.20,21 XRD patterns of pure ZnO NPs and NVP/CS/ITA–ZnO nanocomposite hydrogel.
Biodegradation
The prepared hydrogels and NVP/CS/ITA and its nanocomposite NVP/CS/ITA –ZnO were examined for their biodegradation in PBS solution at pH 7.4 for comparison. Figure 6 depicts that the degradation of NVP/CS/ITA hydrogels and NVP/CS/ITA–ZnO nanocomposite in PBS solution showed progressive weight loss. After a period of 2 weeks of incubation, significant differences between the different samples were observed. The highest biodegradation values were found in NVP/CS/ITA–ZnO nanocomposite hydrogels with an average weight loss of nearly 73%. This may be due to the highly porous structure obtained by incorporation of ZnO NPs into the NVP/CS/ITA polymeric matrix. The biodegradation of NVP/CS/ITA and its nanocomposite NVP/CS/ITA–ZnO in KBS solution at pH 7.4.
Porosity studies
Porosity of a polymeric hydrogels depends on the number of pores present in its network structure and consequently on the crosslinking density of the prepared hydrogels. The percentage of porosity for NVP/CS/ITA hydrogel and NVP/CS/ITA/ZnO nanocomposite hydrogel is shown in Figure 7. It is demonstrated that from Figure 7, the incorporation of the ZnO nanoparticles with its hexagonal wurtzit structure has a great effect on the porosity of the resulted nanocomposite hydrogel where it increased by 20% than that for the NVP/CS/ITA hydrogel. Viscosity of polymers affects the porosity of the developed hydrogels. As the viscosity of the system is increased, the removal of bubbles from the polymeric hydrogels is decreased up to certain extent, which generate pores in the fabricated hydrogels and as a result porosity is increased.
11
The percentage of porosity of NVP/CS/ITA and its nanocomposite NVP/CS/ITA–ZnO.
Amoxicillin loading and release by NVP/CS/ITA–ZnO nanocomposite hydrogel
Two methods can be followed to introduce healing ability to hydrogels, one in which the molecules, for example, drugs are combined with the polymer solution by simple mixing or the second in which the drug molecules are covalently bound to the hydrogels, providing those new healing properties and drug release capabilities. This section focuses mainly on the first methodology. Since hydrogels are directly applied to the wound bed, they can be excellent drug carriers. 22 Amorphous hydrogels and in situ gelable hydrogels are especially appropriate, as both drug delivery and wound covering applications can be flawlessly combined. However, the development of gels that can gelate in situ under favorable conditions for the purpose of drug delivery has been difficult to achieve. The properties of the hydrogel can influence the behavior and efficiency of loaded drugs and must, therefore, be considered23,24. In this respect, the properly optimized NVP/ITA/CS/ZnO nanocomposite hydrogel which has high swelling ability with sticky behavior suitable for wound dressing application examined for the controlled release of amoxicillin as a model. Amoxicillin is used to treat different kinds of infections caused by bacteria, such as tonsillitis, bronchitis, and infections of the ear, nose, throat, skin, or urinary tract.
Loading efficiency of amoxicillin into the NVP/ITA/CS/ZnO nanocomposite hydrogel was studied in distilled water at room temperature. Maximum drug loading by NVP/ITA/CS/ZnO nanocomposite hydrogel reached 77% after 24 h. Therefore, amoxicillin along with an antibacterial agent ZnO NPs that was incorporated in NVP/ITA/CS hydrogel could be very helpful in hastening of wound healing process.
Drug delivery of the prepared NVP/ITA/CS/ZnO nanocomposite hydrogels was studied using cumulative release experiments and is shown in Figure 8. The amoxicillin drug release was carried out in two different pH values, 2.1 and 7.4, similar to the pH of stomach and intestine, respectively.
4
Figure 8 shows the change in drug release profile of NVP/ITA/CS/ZnO nanocomposite hydrogel with time. It is noticed from Figure 8 that at pH 2.1, the release profile of amoxicillin was very slow and the total amount released after 240 min does not exceed 33.5% due to the shrinking of the polymer matrix at low pH values, while in case of pH 7.4, it is noticed that the drug release was fast where nearly 67% of drug loaded released in the first 60 min; only then, the release process started to be slower and the other 30% of loaded drug released in a time period of 240 min reaching a total amount of drug released 93%. This behavior of the prepared nanocomposite hydrogel in the releasing of drug is suitable for fast healing of wounds especial at earlier stages. Drug release profile of amoxicillin at different pH by NVP/ITA/CS/ZnO nanocomposite.
Kinetics of drug release
When a swellable polymeric hydrogels such as chitosan hydrogels is brought into contact with water, the water will diffuse into the polymeric hydrogel and the hydrogel expands leading to its swelling. This diffusion involves the migration of water into pre-existing or dynamically formed species between the polymeric chains of the hydrogel. The swelling of these hydrogels involves larger scale segmental motion, leading to an ultimate increase in separation of the polymeric chains of the hydrogels. The mechanism of drug release was evaluated by using the well-known Fick’s equation (4). A Fickian diffusion has an n value of 0.5 (case I transport) and 0.5<n<1.0 for an anomalous or non-Fickian diffusion. N<0.5 for a less Fickian diffusion (also classified as Fickian diffusion) and n = 1 for a relaxation controlled (case II transport) release. A super case II transport which occurs occasionally is attributed to cases when n>1.0 where the polymer chain relaxation in the hydrogel controls the release process. Plots of log (Mt/Meq) against logt gave a straight line as presented in Figure 9 for the amoxicillin drug by NVP/CS/ITA/ZnO nanocomposite at the two physiological pH 2.1 and 7.4 and the values of n for the NVP/CS/ITA/ZnO nanocomposite hydrogels were found from the slopes of the plots and are presented on the figure. From the values of correlation coefficients Kinetics study of the amoxicillin release by NVP/CS/ITA/ZnO nanocomposite hydrogel.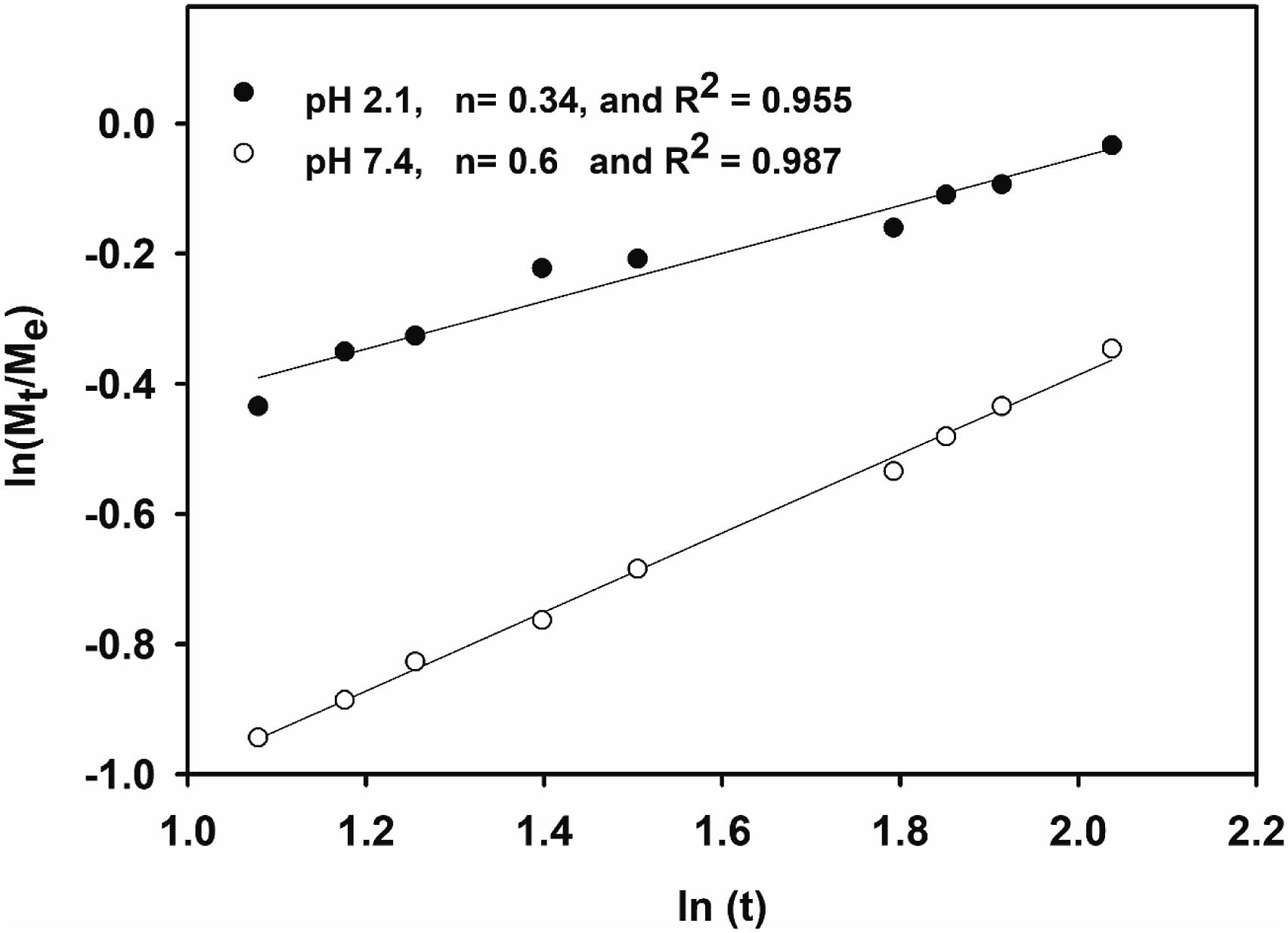
Antibacterial test and mechanism
The three samples of the prepared hydrogels NVP/CS/ITA, NVP/CS/ITA/ZnO, and NVP/CS/ITA/ZnO-Drug were examined for their antibacterial ability toward the Gram-negative bacteria (E. Coli) and Gram-positive (S. aureus). The disc diffusion method against E. coli (Gram-negative) and S. aureus (Gram-positive) bacteria for NVP/CS/ITA, NVP/CS/ITA/ZnO, and NVP/CS/ITA/ZnO-Drug are illustrated in Figure 10 and the inhibition zone calculations are summarized in Table 3. As can be seen from Figure 9 and Table 3, the NVP/CS/ITA/ZnO and NVP/CS/ITA/ZnO-Drug have a good antibacterial ability for both bacterium and NVP/CS/ITA/ZnO-Drug exhibited higher inhibition zone than in case of NVP/CS/ITA/ZnO due to loaded drug. Antibacterial test of 1) control. NVP/CS/ITA, 2) NVP/CS/ITA/ZnO and 3) NVP/CS/ITA, NVP/CS/ITA/ZnO-drug samples against Escherichia coli and Staphylococcus aureus. Inhibition zone of NVP/CS/ITA, NVP/CS/ITA/ZnO and NVP/CS/ITA, NVP/CS/ITA/ZnO-drug against various tested bacteria.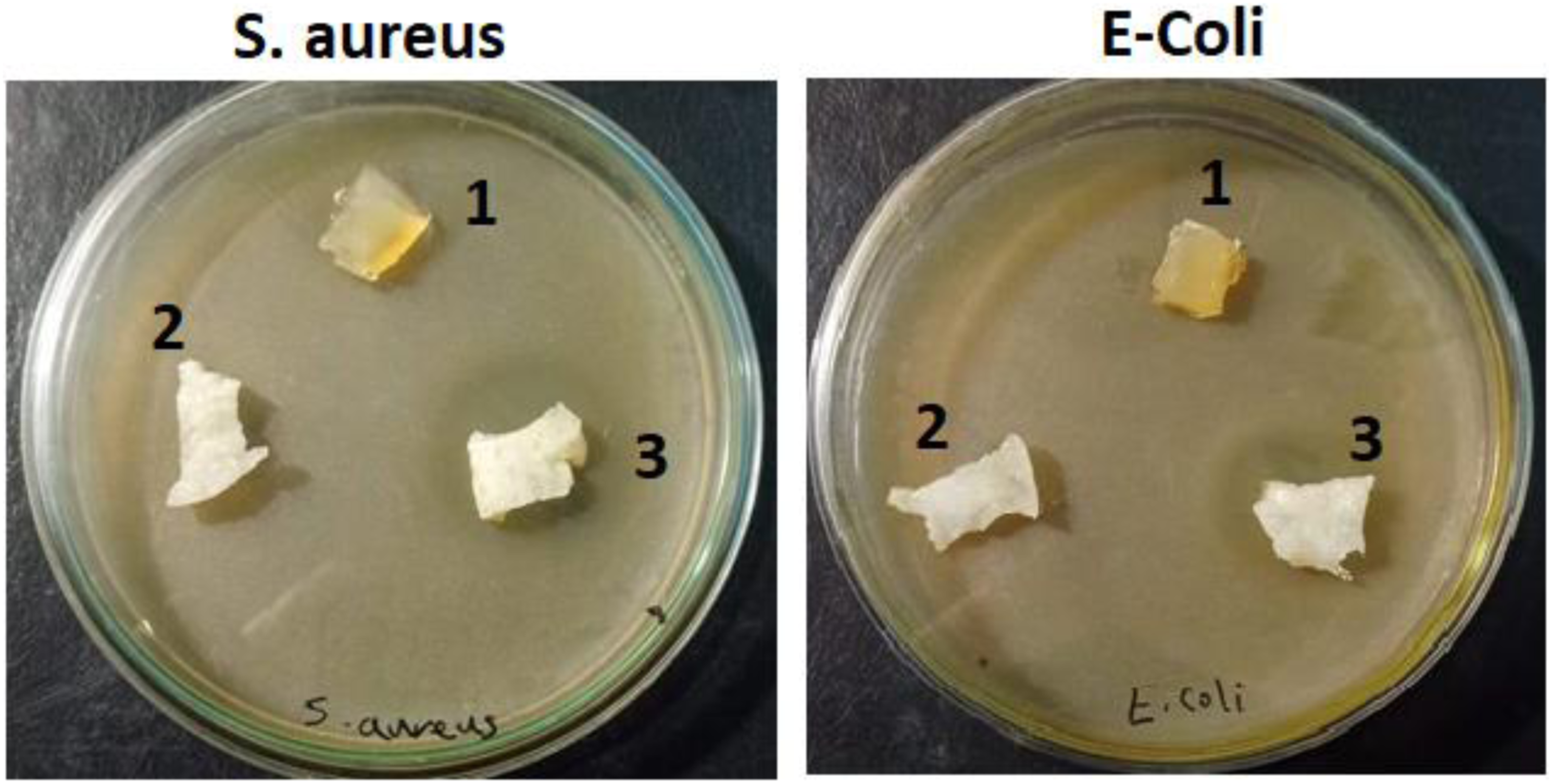
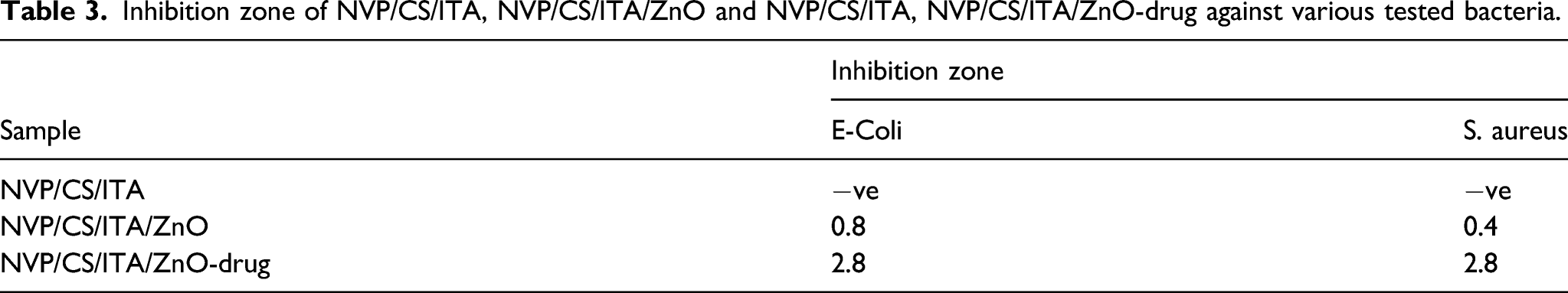
The incorporation of amoxicillin drug with NVP/CS/ITA/ZnO has extremely obvious antibacterial activities against both tested types of bacteria (E− Coli and Staphylococcus aureus (S. aureus). Previous studies have demonstrated that the peptide linkages in Escherichia coli cell membrane were destroyed by the generation of superoxide ions on the surface of bacterium cell resulting in the leakage of their intracellular content and eventually death. 25
According to Figure 10 and Table 3, NVP/CS/ITA/ZnO/Drug nanocomposite hydrogel showed a good and higher antibacterial activity than NVP/CS/ITA/ZnO nanocomposite hydrogel, toward both S. aureus (Gram-positive) and E. coli (Gram-negative) bacteria due to the expected effect of drug and due to the combined effect of ZnO nanoparticles and amoxicillin drug can be explained by various mechanisms such as changing the chemistry of the surface area of the particles, consequently, changing their biological characteristics.
Additionally, the sample of NVP/CS/ITA/ZnO exhibited slightly lower activity against
Mechanism of the antibacterial action of nanocomposite hydrogel
It is reported that the mechanism of antibacterial action of metal oxides in this case ZnO NPs is generally passed through many path ways: reactive oxygen species (ROS), metal ion release, particle internalization into bacteria, and direct mechanical destruction of bacterial cell wall.16,27,28 These path ways of antibacterial effects of ZnO NPs are generalized according to literatures as follows: 1) disruption of the bacterial cell membrane; 2) generation of ROS; 3) penetration of the bacterial cell membrane; and 4) induction of intracellular antibacterial effects, in addition to interactions with DNA and proteins of the bacteria cell causing its death. Figure 11 shows graphical illustration for the effect and mechanism of antibacterial activity of ZnO NPs. Diagrammatic illustration of the preparation and mechanism of antibacterial action of NVP/ITA/CS/ZnO-drug nanocomposite.
This work focuses on the effect of the surrounding NVP/CS/ITA hydrogel network, ZnO NPs and loaded drug on antibacterial activity toward Gram-positive and Gram-negative bacteria. NVP/CS/ITA network matrix, due to the presence of carboxylic and hydroxyl groups, can help and enhance the generation of the reactive oxygen species which may facilitate the release of ZnO NPs and drug molecules and consequently cause bacterial cell death. 29
In general, the antibacterial activity of ZnO is a consequence of visible light irradiation and its direct contact with the microbial cell wall resulting in destruction of bacterial cell integrity, release of antimicrobial Zn2+ ions, and generation of ROS.19,30
Conclusion
In conclusion, a new wound dressing nanocomposite hydrogel composed of NVP/CS/ITA hydrogel that combines with in situ prepared ZnO NPs was examined as a fast drug delivery system for amoxicillin as a model drug and to produce an improved antimicrobial activity against both Gram-negative and Gram-positive bacteria. The prepared nanocomposite hydrogel was studied by various techniques for structural and morphological evaluations. TEM and XRD confirmed the formation of crystalline pure ZnO NPs in the nanoscale. According to obtained results of drug loading and release and antibacterial activity, the investigated NVP/CS/ITA/ZnO and NVP/CS/ITA/ZnO-Drug hydrogel nanocomposites manifest a real potential as an antimicrobial material in a wide variety of bio applications such as wound dressing.
Footnotes
Acknowledgments
The authors express deep gratitude to Dr. Rasha Fathy for her great assistance to carry out the antibacterial test for the prepared NVP/CS/ITA/ZnO and NVP/CS/ITA/ZnO-Drug hydrogels.
Author’s contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
