Abstract
Polyimide is a class of resins with high heat resistance, and their composites have a wide range of applications in military and civilian. However, the application of polyimides as an ablation and thermal insulation material has always been a challenge due to their low char yield during ablation in the air. Herein, we described the first application of polyimide composites—albite/glass powder doped polyimide hybrid materials - as an ablation and thermal insulation material. The key to the success of this case is that the process of the ceramization of albite/glass powder can greatly reduce the carbon loss of polyimide during the ablation process in the air. Moreover, the mechanical properties, thermal stability, ablation and thermal insulation properties of these polyimide hybrid materials have been studied systematically. This work not only opens up a new application for polyimide, but also provides a new idea for the design of novel ablation and thermal insulation material.
Introduction
Ablation and thermal insulation materials are the foundation of thermal protection systems in the aviation and aerospace industries.1-3 Therefore, with the rapid development of aviation and aerospace industries, people’s demand for new ablation and thermal insulation materials is unprecedentedly huge. Up to now, the ablation and thermal insulation materials that have been used mainly include metal-based, ceramic-based, carbon-based, and resin-based ablation and thermal insulation materials. Among of these, the resin-based ablation and thermal insulation materials are attracted considerable attention due to their light weight, high strength, and high-temperature resistance.4-8 Their thermal protection method is active heat protection, which mainly achieves the heat protection effect by carbonizing of the resin at high temperature to form a stable carbon layer.9-12 Therefore, for the resin-based ablation and thermal insulation materials, the char yield of the resin during the ablation process in the air is an important indicator to measure their thermal insulation effect.
As a class of high-performance resins, polyimides possess the advantages of high-temperature resistance and benign mechanical properties. Theoretically, they can become an ideal matrix for resin-based ablation and thermal insulation materials. However, so far, their application in this field is still a blank.13,14 The reason can be attributed to the following two factors: (1) the structure of polyimides contains a large number of heteroatoms such as oxygen and nitrogen atoms; (2) the carbon loss of polyimides during the ablation process in the air is high. These two factors can make the char yield of polyimides at a very low level, making them difficult to meet the requirements for the ablation and thermal insulation materials.15,16 Thus, designing a resin structure with fewer heteroatoms and reducing the carbon loss of the resin during the ablation process in the air are the keys to realizing the application of polyimides in ablation and thermal insulation materials.
In the present reports, the ceramization-carbon fixation strategy is an effective method for reducing the carbon loss of resin-based ablation and thermal insulation materials during the ablation process in the air. This strategy is to add a ceramic precursor to the resin to reduce the carbon loss of the resin in two ways: (1) the ceramization reaction of ceramic precursor and the activated carbon produced by the decomposition of the resin at high temperature can fix carbon atoms; (2) the ceramic structure formed at high temperature can prevent the activated carbon generated by the decomposition of the resin from contacting air, thereby reducing the carbon loss. In 2012, Huang and co-workers17,18 used aluminosilicate ceramic precursors as fillers and achieved the preparation of carbon-based ceramic composites with a high char yields for the first time through the ceramization-carbon fixation strategy. Subsequently, Chen group19,20 and Shi group, 21 respectively, used this strategy to prepare carbon/phenolic resin-based ZrB2 composites and boron-modified phenolic resin-based MgO-Al2O3-SiO2 composites with high char yields. Inspired by the above reports, we speculated that the ceramization-carbon fixation strategy might reduce the carbon loss of the polyimide resins during the ablation process in the air. Herein, we designed a new type of polyimide-based albite/glass powder composites, which can give a ceramized material through a ceramization reaction in the air. The ceramized material exhibits high char yield and benign heat insulation effect, and fully meets the requirements of ablation and thermal insulation materials. As the best of our knowledge, this is the first example of the application of polyimide in the ablation and thermal insulation materials.
Experimental
Materials
4,4′-oxydiphthalic anhydride (4,4′-ODA, 99%) and 4,4′-oxydiphthalic anhydride (4,4′-ODPA, 95%) were purchased from Changzhou Sunlight Pharmaceutical Co., Ltd. cis-5-norbornene-endo-2,3-dicarboxylic (NA, 97%) were purchased from Hebei Budweiser super fine materials Co., Ltd. ethanol (analytical reagent) were purchased from Tianjin Fuyu Fine Chemical Co., Ltd. Glass powder (B, melting point of 600°C) and albite (E, melting point of 1100°C), the average particle size of both are 20 μm. They are all purchased from Shanghai Silicate Research Institute. Short cut high silica fibers (GF, softening point is 1700°C) are purchased from Shenzhen Xinxian Technology Co., Ltd., the length is 12 mm, and SiO2 content is higher than 96%. All chemicals were used as received.
Preparation of polyimide and composites
Monomer ratio of polyimide.


Synthesis of polyimide.
Processing of polyimide and composites
Polyimide and composites were compression-molded in stainless steel mold (100 mm×100 mm) with a hot press. Molded parts of polyimide (PI-0) by polyimide powder (90 g) were heated at 310°C for 20 min on a hot plate. Then, 1 MPa pressure was applied and the temperature was raised to 350°C in 2 h. The cured resins were cooled to about 150°C under pressure, and the pressure was subsequently released. The polyimide sheets were removed from the mold and stored under ambient condition before characterization. The composites of polyimide (90 g of PI-GF, PI-BE40 and PI-BE40-GF) were heated at 320°C for 10 min on a hot plate. Then 10 MPa pressure was applied and the temperature was raised to 350°C in 2 h. The cured resins were cooled to about 150°C under pressure, and the pressure was subsequently released. The polyimide sheets were removed from the mold and stored under ambient condition before characterization.
Characterization
The structure of polyimide was characterized by FTIR (Bio Rad Digilab Division FTS-80). Potassium bromide was used to compress the polyimide. The wave number range was 400–4000 cm−1. The cross-sectional morphology of the samples was identified by scanning electron microscopy (FEI XL-30). Before observation, the samples were sputtered with gold. The thermal stability of the samples was tested by thermogravimetric analyzer (TA Q600), and the heating rate was kept at 10°C min−1 in air atmosphere. The samples were analyzed at the temperature between room temperature and 800°C. Tensile, flexural and compression were evaluated on an Instron material testing system (Model 5982, Instron, USA) with a load cell of 50 kN and referencing to ASTM 638D standards. The tensile tests were carried out according to the GB/T1447-2005 and flexural tests were carried out according to the GB/T1449-2005. At least five samples were subjected to tensile and flexural testing at strain rate of 2 mm min−1. The compression tests were carried out according to the GB/T 1451-2005 standard under a constant displacement rate of 2.0 mm min−1. At least five independent and replicated experiments were conducted to analyze the statistical variability. The data are presented as averages. The ablation and thermal insulation properties of polyimide composites were tested by coal gas/oxygen ablation test system. The sample size was 110 mm×110 mm×4 mm. The vertical distance of the nozzle to the sample was 10 mm and the inner diameter of the nozzle was 2.0 mm. The surface temperature of the samples was approximately 1800°C and was double platinum rhodium B thermocouple (Shenyang Dongda Sensing Technology Co., Ltd.) was used for flame temperature correction, and K-type high-temperature thermocouple (produced by Shenyang Dongda Sensing Technology Co., Ltd.) is used for back temperature measurement. The range of test temperature of K-type high-temperature thermocouple is 10–900°C and the accuracy is 1°C. The testing time (measured by Ta260 multi-functional electronic stopwatch, Shenzhen jintuojia Electronic Technology Co., Ltd.) was fixed at 10 min and the temperature point was collected every 15 s. Figure 1 showed a schematic diagram of the ablation test and specimen under the test. The linear and mass ablation rates were calculated according to the following formulae Coal gas/oxygen ablation test system.

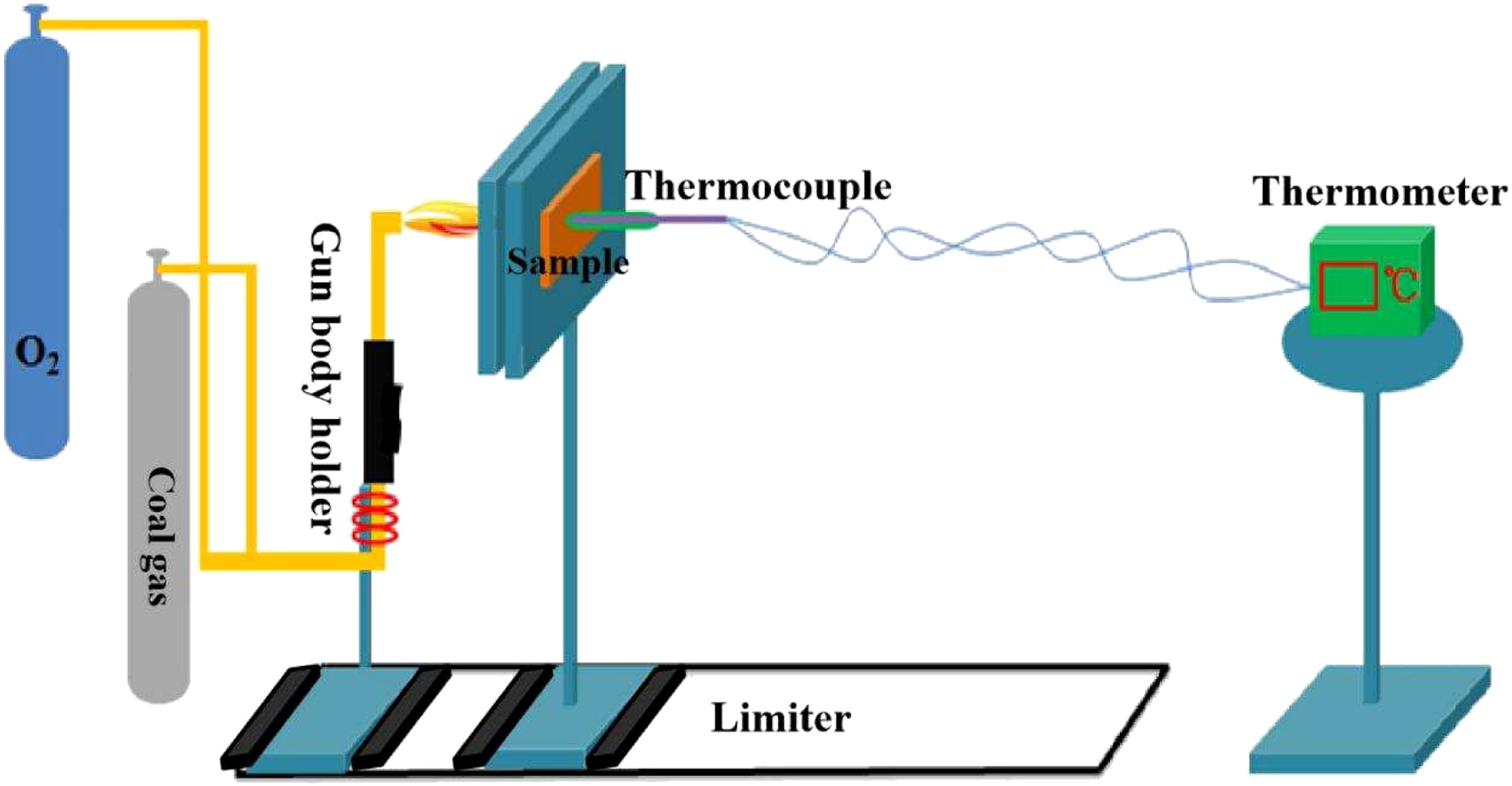
Results and discussions
Compositional analysis of PI by FTIR spectroscopy
The infrared spectrum of polyimide is shown in Figure 2. The absorption peak at 1780 cm−1 is attributed to the asymmetric stretching vibration of carbonyl group in aromatic imide, the absorption peak at 1720 cm−1 is attributed to the symmetrical stretching vibration of carbonyl group, the absorption peak at 1380 cm−1 is attributed to the stretching vibration of carbon nitrogen bond, and absorption peak at 725 cm−1 is attributed to the bending vibration of carbonyl group. These characteristic absorption peaks indicate that the polymer is polyimide.22,23 FTIR spectrum of polyimide.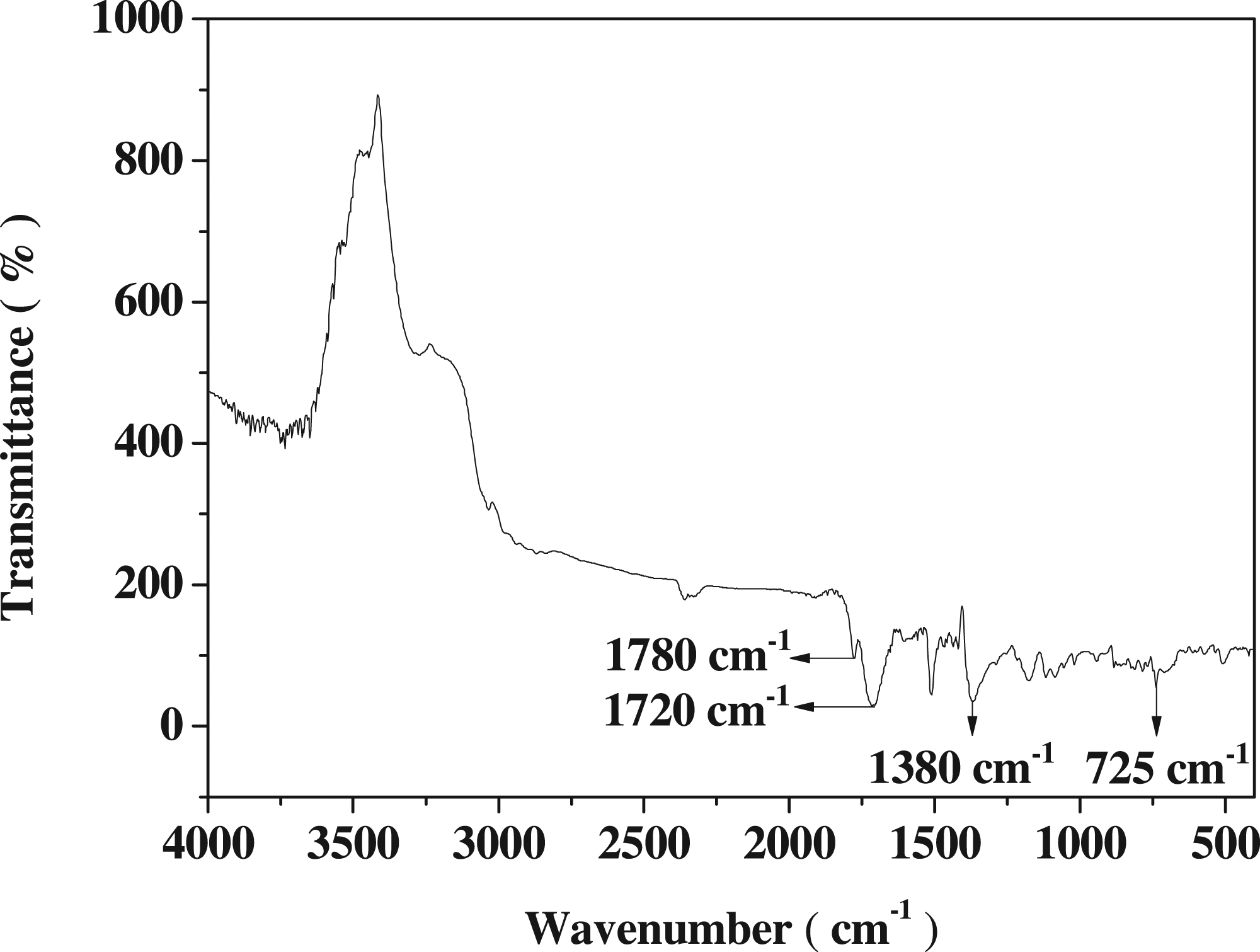
Microstructures of the samples
SEM results showed the microstructure of B, E, PI, PI-BE40, PI-GF, PI-BE40-GF samples. Glass powder and albite powder have irregular block structure, which are unifromly dispersed in polymer matrix and well bonded with matrix, as shown in Figure 3(d). The tensile strength of polyimide containing BE is 9% higher than that of pure polyimide (55 MPa). This is mainly due to the physical entanglement between BE and polyimide chain limits the fluidity of the chain, resulting in an increase in tensile strength but a decrease in flexibility (4.6 vs 0.9%). When short cut high silica fiber (GF) is introduced into the composite, BE still has a good dispersion and bonding with resin. For example, the Figures 3(e) and (f) show that the combination between polyimide and chopped high silica fiber presents macroscopic network interpenetrating structure due to the addition of BE. The mechanical property test results also show that due to the addition of BE, the bonding between polyimide and chopped high silica fiber presents a macroscopic network interpenetrating structure, which leads to significant increase of tensile strength and flexural strength of the composites. SEM characterization of series samples. (a) B (glass powder) particle size distribution. (b) Size distribution of E (albite). (c) PI pure resin sample block. (d) PI-BE40. (e) PI-GF. (f) PI-BE40-GF.
Mechanical properties of samples
Mechanical property test of samples.

Thermogravimetric analysis of samples
All data (Figures 4 and 5 and Table 3) determined by TGA at a heating 10°C/min in air atmosphere; T5% was generally taken as the temperature of 5% weight loss; T10% was generally taken as the temperature of 10% weight loss; Tmax was generally taken as the temperature of maximum rate of degradation; R800 was generally taken as the char yield of 800°C. TG curves of samples. DTG curves of samples. Comparison of thermal decomposition properties of samples.


Under 400°C, the mass loss of all samples is very small. The decomposition temperature T5% and T10% are defined as the temperature of 5% and 10% weight loss, respectively, and the temperature of 5% weight loss is defined as the initial decomposition temperature. The T5% of pure polyimide in air is 446°C and T10% is 479°C, T5% of PI-BE40 and PI-BE40-GF composites in air is 475°C and 480°C, T10% is 495°C and 513°C, respectively. The results showed that the addition of albite and glass powder can significantly improve the thermal stability of polyimide. This is due to the high-temperature porcelain forming reaction between albite and glass powder provides the physical barrier, which can not only prevent the escape of decomposition gas, but also prevent the diffusion of oxygen into the material.26,27
Ablation and heat insulation performances of samples
The ablation resistance of the composite can be characterized by linear ablation rate and mass ablation rate, and the thermal insulation performance can be characterized by back temperature. When the ablation flame temperature is 1800°C, the sample of PI-BE40-GF reacted to form procelain at high temperature. As shown in Figure 6, compared with PI-GF, the surface of PI-BE40-GF with glass powder and albite is hard after ablation, which is due to the porcelain reaction between BE effectively fix the short cut high silica fiber and carbon generated by resin pyrolysis, and prevent the further erosion of high temperature and high-pressure gas flow on the material. The microstructure of the samples in Figures 6(b) and 7(b) showed a dense ceramic structure after ablation of PI-BE40-GF at 1800°C compared with the microstructure of PI-GF in Figures 6(a) and 7(a). At 1800°C, a large number of white liquid beads spontaneously and uniformly distribute in the carbon. These small balls tend to indulge and diffuse together under the driving of surface tension, which can be used as the adhesive to connect the porous carbon. Obviously, no cracks, pits or cracks were observed. The microstructure densification of coke results in significant shrinkage of the sample. This phenomenon can be explained by the densification mechanism that larger liquid volume allow faster carbon rearrangement.26,27 Therefore, compared with PI-GF (Table 4), the mass ablation rate of PI-BE40-GF is reduced by 83%, the linear ablation rate is reduced by 168%, and the back temperature is reduced by 70°C after 10 min of ablation (Figure 8), which indicates that the introduction of BE component can meet the requirements of heating and oxygen enrichment thermal protection, and can significantly improve the ablation and thermal insulation properties of polyimide composites.
28
Macroscopic of polyimide samples after ablation. (a) PI-GF; (b) PI-BE40-GF. Microstructure of polyimide samples after ablation. (a) PI-GF; (b) PI-BE40-GF. Ablation properties of polyimide samples. Ablation back temperature curves of samples.



Conclusion
The polyimide composites were prepared by using glass powder and albite as filler and chopped high silica fiber as reinforcement. The results showed comparison with polyimide, the tensile strength and flexural strength of the composites increased with the addition of albite, glass powder and high silica fiber, while the breaking elongation first decreased and then increased. Thermogravimetric analysis showed that the initial decomposition temperature and maximum decomposition temperature of the composites in air atmosphere were increased in different degrees, and the char yield of the composite material increased from 0% to 70% when compared to polyimide, and the increase rate was 70%. The results of ablation test showed that albite promote the formation of a covering layer by combining with silica and other components on the ablated surface. The linear ablation rate and mass ablation rate were reduced by 22.5% and 18.2%, respectively. These results showed that albite and glass powder fillers are beneficial to form a dense covering layer, which delays the diffusion of hot oxygen into the inner layer and effectively reduce the carbon loss of the composites during the ablation process in the air. Compared with the polyimide/high silica composite, the ablation back temperature of the albite/glass powder doped polyimide/high silica composites decreases about 70°C, which showed that the material has excellent ablation and heat insulation properties. It is expected to be used in ablative heat protection system.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Government Funded Projects by Department of Science and Technology of Jilin Province (20190302001GX); Jilin Provincial Department of Education (JJKH20200758KJ, JJKH20200761KJ); and Changchun University of Science and Technology Innovation Fund (XJJLG-2018-12).
