Abstract
Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide (CTS-g-P(AA-co-AMPS)/GO) composite hydrogel is synthesized and used to remove rhodamine 6G (R6G) and methyl violet (MV) from aqueous solutions by adsorption. The composite is characterized by infrared spectroscopy (IR), powder X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscope (TEM), thermogravimetric analysis (TGA), and differential scanning calorimetry (DSC). The effect of the adsorption conditions, including the amount of graphite oxide (GO), the initial concentration of dye solutions, the adsorption time, ionic strength, and the mass of the composite, on the adsorption capacities has been studied in detail. It can be seen that small amount of GO can improve the adsorption capacities of both dyes. It is found that the adsorption capacities of R6G and MV can be increased by 57.26% and 26.39%, respectively, compared with CTS-g-P(AA-co-AMPS graft copolymer. The maximal adsorption capacity of R6G and MV is 625.3 and 326.4 mg/g, respectively. The interaction between GO and dye molecules are speculated. GO acts as crosslinking points and combine with the CTS-g-P(AA-co-AMPS) graft copolymer through hydrogen bonds and electrostatic actions. The adsorption isotherms and thermodynamics are discussed. The Gibbs free energy of R6G and MV is that △GR6GӨ = −2.478 KJ/mol and △GMVӨ = −2.577 KJ/mol and it indicates that the adsorption of R6G and MV on CTS-g-P(AA-co-AMPS)/GO is spontaneous and satisfied the Redlich-Peterson equation. Kinetic studies show that the adsorption is in accordance with the Lagergren pseudo first-order kinetic model.
Introduction
Since the destruction of industrial dye wastewater on the ecological environment in water, dye wastewater treatment has been one of the research hotspots in recent years. Much attention has been paid to remove dyes from printing and dying waster liquid, such as ion-exchange, adsorption, flocculation, electrolysis, biological methods, etc.1–5 Among them, adsorption seems to be the simplest and most effective one owing to its low cost, high efficiency, and good re-use performances.6–9
Chitosan (CTS) is a natural alkaline polysaccharide which is renewable and biodegradable. Because of the functional groups such as amino and carboxyl groups which are able to combine with dye molecules through ion-exchange, chelation and electrostatic attractions, CTS and its derivatives are used widely as cheap but effective adsorbents in wastewater treatment. Fan L 10 fabricated a magnetic beta-cyclodextrin-chitosan/graphene oxide materials through a facile chemical route and used them as excellent adsorbents for dye removal. Schio RDR 11 synthesized a novel polyurethane/chitosan foam (PU/chitosan) using a polyol derived from castor oil and applied to remove food red 17 dye (FR17) from aqueous solutions. The PU/chitosan was able to remove >98% of FR17 dye from the solution. Reghioua A 12 developed a new magnetic Schiff’s base-chitosan-Benzil/zinc oxide/Fe3O4 nanocomposite (Cs-Bz/ZnO/Fe3O4) and used it as a superior, recoverable, and environment friendly biohybrid nanocomposite adsorbent. Jawad AD 13 synthesized crosslinked chitosan-epichlorohydrin/kaolin (Chi-ECH/KA) composite and successfully applied for color removal and chemical oxygen demand (COD) reduction of reactive blue 19 dye (RB19) from aqueous environment. Other chitosan/clay and chitosan/metallic oxide composite adsorbents14–16 were also prepared and used to remove dyes from aqueous solution.
2-acrylamide-2-methylpropanesulfonic acid (AMPS) is an anionic monomer with good salt tolerance and water solubility. In addition, it is superior to acrylamide (AM) in thermostability and acid and alkali resistance due to the large steric hindrance of the sulfonic acid group. 17 Therefore, AMPS is more efficient than AM as water treatment agents.
Graphite oxide (GO) is a flaky carbon nano-material with plenty of oxygen-containing functional groups and good dispersive property. It can improve the thermodynamic properties together with the electrical performances of the polymeric materials. In recent years, GO is used to be incorporated into superabsorbent polymeric adsorbents to improve the network structure and sorption behavior of the products. A porous xylan/poly(acrylic acid)/graphite oxide nanocomposite was prepared by graft polymerization and used for adsorption of cationic ethyl violet dye in wastewaters. 18 Adsorption tests showed that this material had excellent adsorption properties; the maximum adsorption capacity for ethyl violet dye was 273.99 mg.g−1. Deliyanni E A 19 prepared a novel composite material consisting of cross-linked chitosan and GO. The composite presented high adsorption capacity for reactive Black 5 dye.
However, there are few studies on CTS/polymer/GO ternary complexes. In this study, chitosan graft poly (acrylic acid-co-2-acrylamide-2- methylpropanesulfonic acid)/graphite oxide (CTS-g-P(AA-co-AMPS)/GO) composite hydrogel has been synthesized and used to adsorb rhodamine 6G (R6G) and methyl violet (MV) in aqueous solutions. The composite is characterized by infrared spectroscopy (IR), powder X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscope (TEM), thermogravimetric analysis (TGA), and differential scanning calorimetry (DSC). The effect of the adsorption conditions on the adsorption capacities are studied in detail. The interaction between GO and dye molecules are speculated. The adsorption thermodynamics and kinetics are also discussed.
Experimental
Materials
The properties and chemical structures of R6G and MV.
Preparation
Preparation of GO
GO is prepared according to the modified Hummers method as stated in literature. 20 The GO dispersion (0.002 g/mL) is prepared by dissolving 2 g of dry graphite oxide powder in 1000 mL of deionized water as shown in the literature. 21
Preparation of CTS-g-P(AA-co-AMPS)/GO composite hydrogel
AA (7.2 g, 0.1 mol) is put in a three-necked flask equipped with a condenser, a thermometer, and a nitrogen line. Sodium hydroxide solution (12.8 g, 25wt%) is dropped into the flask and the mixture is stirred vigorously for 30 min in an ice-water bath. Thus, the AA solution with a neutralization degree of 80% is obtained. Then, CTS (0.45 g) and a certain amount of GO dispersion are added into the flask consecutively. The reaction mixture is stirred continuously for 30 min before AMPS (0.5 g) was added to the flask. After the water bath is slowly warmed to 56°C, MBA solution (0.005 g/mL, 5 mL) is dropped into the flask under nitrogen atmosphere. After stirring for 40 min, the water bath is heated up to 65°C and KPS solution (0.05 g/mL, 10 mL) is slowly dropped into the flask. The mixture is stirred at 65°C for about 1 h until a black gel is formed. The gel was taken out of the flask, washed with deionized water, cut into small pieces, and then dried in an oven at 60°C to a constant weight.
Characterization
IR spectra are recorded with KBr tablet on a Nicolet’s 380 Fourier transform infrared spectrometer (Nicolet Instrument Co., Ltd., Switzerland). XRD patterns are obtained from the Rigaku Corporation Ulti X-ray diffractometer (Japan Science Corporation), the scanning range is 5–60°, the speed is 8°/min, the current is 30 mA, and the voltage is 40 kV. SEM and TEM photos are taken on a SU8000 scanning electron microscope and a JEM-2100 transmission electron microscope, respectively. DSC curves are achieved on DSC1 differential scanning calorimeter (Mettler Toledo Company, Switzerland) with a scan rate of 10°C/min. TGA is performed on a TGA2 thermogravimetric analyzer (Mettler Toledo Company, Switzerland) at a heating rate of 10°C/min using dry nitrogen purge.
Adsorption
Dry CTS-g-P(AA-co-AMPS)/GO powder (0.2 g) is put in a beaker with 150 mL of dye solutions at room temperature. After a certain period of time, the mixture is filtrated and the filtrate is obtained for future determination of its volume and the absorbency at the maximum absorption wavelength (λmax). The equivalence adsorption capacity (qe) is calculated on basis of equation (1)
Desorption
The reuse performances of adsorbents are of great significance in industrial production. A certain amount of CTS-g-P(AA-co-AMPS)/GO composite hydrogel which has adsorbed dyes with an equivalence amount is suffered to be dried, ground, and immersed in 50 mL of hydrochloric acid solution (5%). After stirring for 18 h, the mixture is filtered and the filtrate is obtained for the determination of its absorbance. The desorption rate (D%) is calculated according to equation (2)
Results and discussion
IR spectra
The IR spectra of GO (a), CTS-g-P(AA-co-AMPS) (b), and CTS-g-P(AA-co-AMPS)/GO (c) are shown in Figure 1. As for GO, see Figure 1a, the broad band from 3200 to 3500 cm−1 is due to the stretching vibration of -OH. Those at 1640 and 1080 cm−1 are the absorption peaks of C=C and-C-O-C-, respectively. The peak at 731 cm−1 is due to the bending vibration of -OH.
7
For CTS-g-P(AA-co-AMPS) graft copolymer, see Figure 1(b), the broad band from 3200 to 3600 cm−1 is due to the stretching vibration of –OH in the carboxyl groups of poly acrylic acid. That at 1739 cm−1 is ascribed to the stretching vibration of C=O. Those at 1610 and 1414 cm−1 are because of the bending vibration of N-H and C-N in CTS. The peaks at 1115 and 1047 cm−1 are related with the asymmetric and symmetric stretching vibration of S=O in AMPS. When it comes to CTS-g-P(AA-co-AMPS)/GO composite (Figure 1(c)), there are the characteristic absorption peaks of -C-H in the polymeric side chain (2856 cm−1), the stretching vibration of C=C in GO (1649 cm−1). It can be seen there is a red shift of C=C in GO after being combined with CTS-g-P(AA-co-AMPS). (a) Infrared spectroscopy spectra of GO, (b) CTS-g-P(AA-co-AMPS), and (c) CTS-g-P(AA-co-AMPS)/GO. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
Powder XRD analysis
Figure 2 shows the XRD patterns of GO (a), CTS-g-P(AA-co-AMPS) (b), and CTS-g-P(AA-co-AMPS)/GO (c). It can be seen clearly in Figure 2(a) that there is a diffraction peak at 2θ = 12.04°. The calculated slice distance is 0.7351 nm according to the Bragg’s formula 2dsinθ = λ. However, both CTS-g-P(AA-co-AMPS) (b) and CTS-g-P(AA-co-AMPS)/GO (c) exhibit amorphous state. It indicates that GO has been peeled off and dispersed into the CTS-g-P(AA-co-AMPS) evenly after intercalation polymerization.
22
(a) X-ray diffraction patterns of GO, (b) CTS-g-P(AA-co-AMPS), and (c) CTS-g-P(AA-co-AMPS)/GO. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
SEM photos
It can be seen in Figure 3(a) that GO shows a clear flaky structure. While CTS-g-P(AA-co-AMPS) graft copolymer exhibits uneven granular structure (see Figure 2(b)). However, CTS-g-P(AA-co-AMPS)/GO composite has porous three-dimensional networks. Therefore, it is easier for small molecules to penetrate into the inside of the composite rather than the CTS-g-P(AA-co-AMPS) graft copolymer. (a) The scanning electron microscopy photos of GO, (b) CTS-g-P(AA-co-AMPS), and (c) CTS-g-P(AA-co-AMPS)/GO. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
TEM photos
The TEM photos of GO (a), CTS-g-P(AA-co-AMPS) (b), and CTS-g-P (AA-co-AMPS)/GO (c) are shown in Figure 4. Obviously, GO shows a thin and flaky structure. While CTS-g-P(AA-co-AMPS) graft copolymer is granular (see Figure 4(b)). When it comes to CTS-g-P(AA-co-AMPS)/GO composite (Figure 4(c)), it has both the thin slice of GO and particles of CTS-g-P(AA-co-AMPS). There is a uniform dispersion of particles around the sheets of GO. (a) The transmission electron microscope photos of GO, (b) CTS-g-P(AA-co-AMPS), and (c) CTS-g-P(AA-co-AMPS)/GO. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
DSC analysis
The DSC curves of GO (a), CTS-g-P(AA-co-AMPS) (b), and CTS-g-P(AA-co-AMPS)/GO (c) are shown in Figure 5. It can be seen that there is an obvious exothermic peak at 225°Cin GO. Both CTS-g-P(AA-co-AMPS) and CTS-g-P(AA-co-AMPS)/GO exhibit endothermic peaks. As for CTS-g-P(AA-co-AMPS) graft copolymer, the glass transition temperature is 60–80°C, while that of CTS-g-P(AA-co-AMPS)/GO is 70–90°C. And both CTS-g-P(AA-co-AMPS) and CTS-g-P(AA-co-AMPS)/GO have good thermal stability and the degradation temperature is about 400°C. (a)Differential scanning calorimetry curves of GO, (b) CTS-g-P(AA-co-AMPS), and (c) CTS-g-P(AA-co-AMPS)/GO. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
TGA curves
The TGA curves of GO (a), CTS-g-P(AA-co-AMPS) (b), and CTS-g-P(AA-co-AMPS)/GO (c). As can be seen in Figure 6(a), there are two weight loss stages in GO. The first one is below 150°C, due to the evaporation of the adsorbed moistures. The second one is between 200 and 250°C, which is ascribed to the decomposition of the oxygen-containing functional groups. As for CTS-g-P(AA-co-AMPS) graft copolymer (Figure 6(b)) and CTS-g-P(AA-co-AMPS)/GO composite, they have similar weight losses at 360–450°C, mainly because of the decomposition of the polymeric backbones. However, the residue of CTS-g-P(AA-co-AMPS)/GO is obviously more than that of CTS-g-P(AA-co-AMPS) during the whole thermal decomposition process. Therefore, it proves that the thermal stabilities of CTS-g-P(AA-co-AMPS)/GO composite is better than that of the CTS-g-P(AA-co-AMPS) graft copolymer. (a)Thermogravimetric analysis curves of GO, (b) CTS-g-P(AA-co-AMPS), and (c) CTS-g-P(AA-co-AMPS)/GO. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.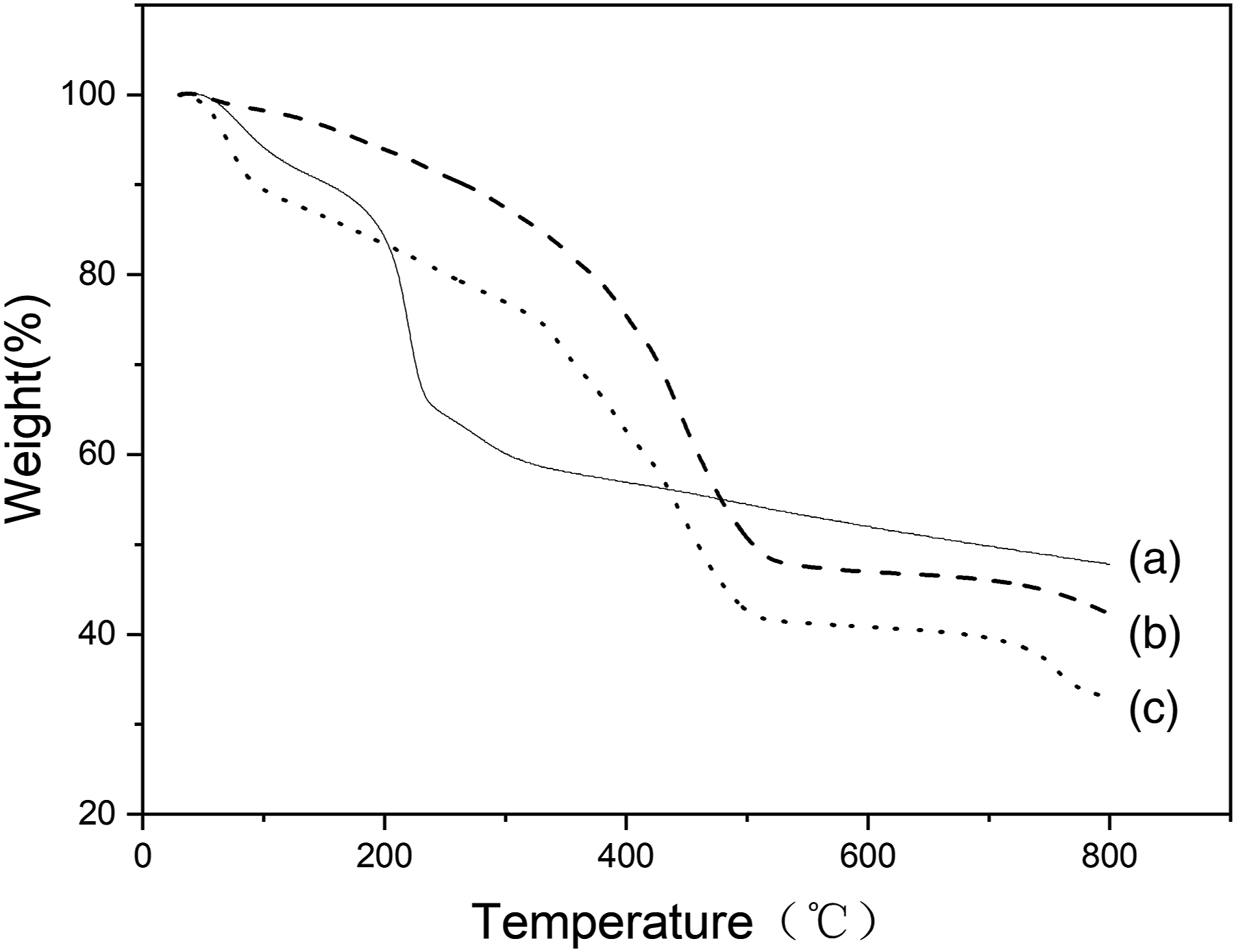
Effect of GO content on adsorption
The effect GO content on the adsorption capacities is shown in Figure 7. It can be seen that small amount of GO can improve the adsorption capacities of both dyes. As the content of GO rises from 0 to 0.03 g (0.002 g/mL, 15 mL), the adsorption capacities of R6G and MV go up from 397.6 and 289.9 mg/L, to 625.3 and 326.4 mg/g, respectively. Further increase of GO content leads to a decrease of adsorption capacity. It is known that GO works as cross-linking points and helps to form three-dimensional networks thereby improving the adsorption properties of the resin. However, the excessive GO will hinder the extension of the polymeric chains. Thus, proper amount of GO is 0.03 g. Effect of content of GO on adsorption capacities (CTS-g-P(AA-co-AMPS)/GO) 0.05g, adsorption time 18h, dye concentration 500mg/L). CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
Effect of mass of the composite on adsorption
The dosage of the adsorbent is an important parameter in wastewater treatment for it reflects the efficiency of the technology.
23
The effect of the mass of the composite on adsorption is shown in Figure 8. It is clear that the equivalent adsorption capacities of both dyes first decrease rapidly and then tend to be constant with the increase of the dosage of the composite. The increase in adsorbent dosage leads to an overlapping of the adsorption sites on the composite. So it is more efficient for batch delivery of adsorbents during industrial wastewater treatment. Effect of adsorbent dosage on adsorption capacity (GO 0.03 g, adsorption time 18h, initial dye concentration 500 mg/L). GO: Graphite Oxide.
Effect of contact time on adsorption
The effect of adsorption time on adsorption is shown in Figure 9. The adsorption capacities of both dyes first increase fast and then approach equivalence with the extension of contact time. As for R6G, the adsorption capacity increases greatly within the first 4 h and then approaches equilibrium. It is obvious that the equilibrium adsorption amount of R6G is much higher than that of MV. The molecular chains of the adsorbent stretch more rapidly at the first stages of adsorption. Thus, it is convenient for the hydrophilic functional groups to contact with the dye molecules.
[24]
Besides, the osmotic pressure is great at the initial stages, which is a benefit for adsorption. With the extension of time, the adsorption approaches equivalence. The equivalent adsorption time for R6G and MV are about 12 h. Effect of adsorption time on adsorption capacity (CTS-g-P(AA-co-AMPS)/GO 0.05g, GO 0.03g, initial dye concentration 500 mg. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
Effect of initial dye concentration on adsorption
The effect of initial dye concentration on adsorption is shown in Figure 10. It can be seen that the adsorption capacities of both dyes first increase fast and then approach constant values with the enhancement of initial concentration of dye solutions from 50 to 1000 mg/L. At high concentration of the dye, a greater number of the dye molecules will be available in the bulk solution. As a result, more dye molecules can be captured by the active adsorption sites on the surface of adsorbent.14,25 The diffusion of dye molecules to the composite accelerates with the rise of the initial concentration of dye solutions. Thus, the interactions between dye molecules and the composite are enhanced and the adsorption capacities are improved.
26
Finally, the adsorption sites on the composite gradually approach saturation with the continuous growth of the concentration of dye solutions. High concentrations of dyes enlarge the gradient between the bulk solution and that in the adsorbent, which strengthens the driving forces for dye molecules to diffuse onto the surface of the adsorbent.
27
Effect of initial dye concentration on adsorption (CTS-g-P(AA-co-AMPS)/GO) 0.05g, GO 0.03 g, adsorption time 18h). CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.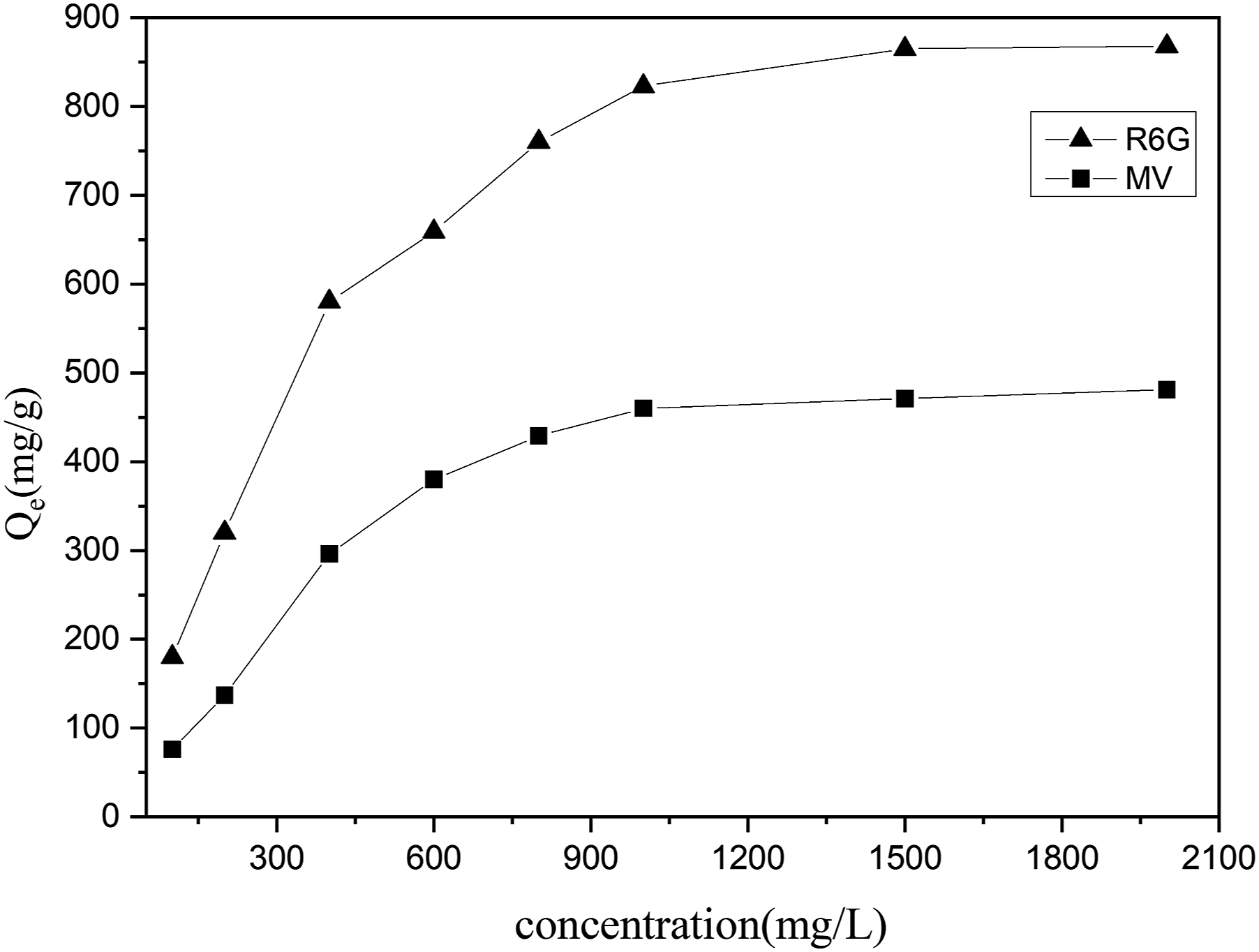
Effect of ionic strength on adsorption
The effect of ionic strength on adsorption is shown in Figure 11. As can be seen, there is a sharp decrease in adsorption capacities of both dyes as the concentration of NaCl solution rises from 0 to 0.02 mol/L. The competitive adsorption of Na+ onto the composite is enhanced with the increase of concentration of NaCl solution, which goes against the adsorption of dye molecules. With further increase of NaCl concentration, the competitive adsorption slowly approaches equilibrium, and the adsorption capacities reach equilibrium. Effect of ionic strength on adsorption (CTS-g-P(AA-co-AMPS)/GO) 0.05g, GO 0.03 g, adsorption time 18h, initial dye concentration 500 mg/L), CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.
Adsorption Isotherms
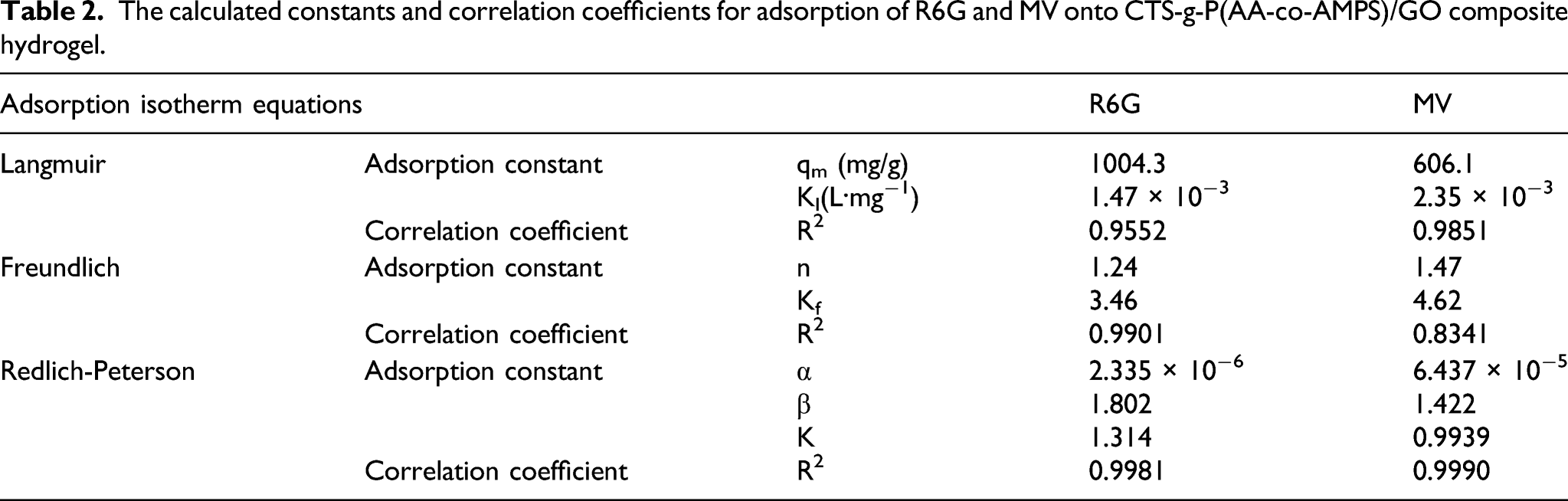
The calculated constants and correlation coefficients for adsorption of R6G and MV onto CTS-g-P(AA-co-AMPS)/GO composite hydrogel.
Langmuir isotherm equation

Freundlich isotherm equation

Redlich-Peterson (R-P) isotherm equation

It can be inferred from the R2 values in Table 2 that the adsorption of R6G and MV confirms to the Redlich-Peterson equation. Since
Adsorption thermodynamics
The equilibrium constant K during adsorption can be calculated according to equation (6), where Kf and n are the constants in the Freundlich equation

The Gibbs free energies of R6G and MV are calculated by substituting the values of K, R(8.314 J/mol/K), and T(298.15 K) into equation (7). The result is that △GR6GӨ= −2.478 KJ/mol and △GMVӨ= −2.577 KJ/mol. Therefore, the adsorption of R6G and MO on CTS-g-PAA/GO/ATP is spontaneous.
Adsorption kinetics
Kinetics parameters for adsorption of R6G and MV onto CTS-g-P(AA-co-AMPS)/GO composite hydrogel.

Pseudo-first-order Equation

Pseudo-second-order Equation

It can be seen that the adsorption of R6G and MV conforms to the pseudo-first-order kinetic model.
The experimental adsorption data for R6G and MV over the hydrogel using the pseudo-first-order and pseudo-second-order rate equations in the linear form were shown in Figure 12. Pseudo-first-order and pseudo-second-order plots for R6G(a and b) and MV(c and d).
Adsorption mechanisms
The interactions between CTS-g-P(AA-co-AMPS)/GO and R6G and MV are shown in Figure 13 and Figure 14, respectively. It can be seen that the interactions between GO and CTS-g-P(AA-co-AMPS) graft copolymer are hydrogen bonds. As for the interactions between CTS-g-P(AA-co-AMPS)/GO and dye molecules, it is speculated that for R6G there are both hydrogen bonds and electrostatic attractions (see Figure 13). However, for MV, there is only electrostatic attraction (see Figure 14). The interaction between CTS-g-P(AA-co-AMPS)/GO and rhodamine 6G. GO: Graphite Oxide, CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide. The interaction between CTS-g-P(AA-co-AMPS)/GO and methyl violet. CTS-g-P(AA-co-AMPS): Chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide.

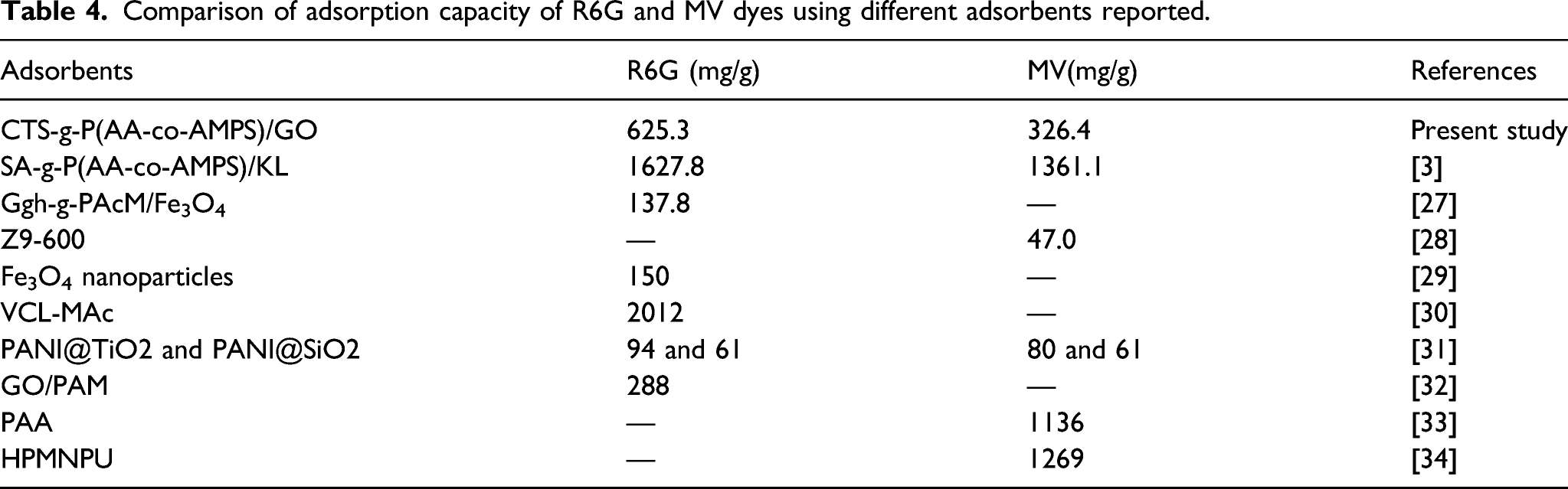
Comparison with previously developed adsorbents
Comparison of adsorption capacity of R6G and MV dyes using different adsorbents reported.
Desorption
According to equation (2), the desorption rate of R6G and MV is 27.5% and 2.6%, respectively. Thus, despite that the desorption efficiency is not the same to different dyes, the CTS-g-P(AA-co-AMPS)/GO composite hydrogel has good reuse performance in wastewater treatment.
Conclusions
In this article, chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid)/graphite oxide (CTS-g-P(AA-co-AMPS)/GO) composite hydrogel is prepared by solution intercalation polymerization. The composite is characterized by IR, XRD, SEM, TEM, TGA, and DSC, before used to remove rhodamine 6G (R6G) and methyl violet (MV) from aqueous solutions by adsorption. The effect of the adsorption conditions, including the amount of graphite oxide (GO), the initial concentration of dye solutions, the adsorption time, ionic strength, and the mass of the composite, on the adsorption capacities has been studied in detail. It is found that the adsorption capacities of R6G and MV can be increased by 57.26% and 26.39%, respectively, compared with chitosan graft poly (acrylic acid-co-2-acrylamide-2-methylpropanesulfonic acid) (CTS-g-P(AA-co-AMPS)) graft copolymer. GO acts as crosslinking points and combine with the CTS-g-P(AA-co-AMPS) graft copolymer through hydrogen bonds. The interactions between CTS-g-P(AA-co-AMPS)/GO and R6G are hydrogen bonds and electrostatic attractions, while those between the composite and MV are only electrostatic attractions. The adsorption isotherms and thermodynamics are discussed. It indicates that the adsorption is spontaneous and satisfied the Redlich-Peterson equation. Kinetic studies show that the adsorption is in accordance with the Lagergren pseudo first-order kinetic model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
