Abstract
A detailed study was conducted on the synthesis, characterizations, and properties of poly (3,4-ethylenedioxythiophene)/Mag-CTA nanocomposites. Poly (3,4-ethylenedioxythiophene) (PEDOT) nanocomposites were developed from a natural clay called Maghnite by improving the dispersion of 3,4-ethylenedioxythiophene (EDOT) matrices in organic clay sheets. However, the PEDOT/Mag-CTA nanocomposites have been characterized by different physico-chemical techniques such as X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FTIR), thermogravimetry analysis (TGA), differential scanning calorimetry (DSC), and give the name of atomic force microscope (AFM), and scanning and transmission electron microscopy (SEM and TEM). The results of the (XRD), (TEM) and (SEM) analysis confirm that the PEDOT has been inserted into the Mag-CTA interlayer, and the presence of clay in the polymer matrix results in a desired increase in thermal stability. Electrical conductivity measurements show a decrease in conductivity with increasing load ratio
Keywords
Introduction
Polymer matrix materials have evolved considerably as a result of their exceptional properties, which can be exploited in a wide range of industrial applications. The properties of these polymers are conceptually different from those of conventional materials. In recent years, many efforts have been made to improve the high performance characteristics of polymer-based matrices by hybridizing compatible polymer materials. Currently, natural and synthetic polymeric materials have been used worldwide for various industrial and engineering applications. Polymer materials for the manufacture of advanced components should have the desired physico-chemical properties, mechanical strength and stiffness, thermal stability, and chemical resistance.1–4 Over the last few decades, there have been an exponential growth in interest in clays, in virtually all scientific fields, for scientific research and industrial applications. 5 Conductive polymers, including poly(3,4-ethylenedioxythiophene) (PEDOT), have attracted particular interest due to their excellent environmental stability, low oxidation potential, 6 high electrical conductivity, low band gap, and good electro-mechanical behavior, which make possible countless potential applications in high technology products and other new devices.7–10 PEDOT has many of the most significant applications, including through-hole plating on printed circuit boards, antistatic coatings that prevent dust attraction on cathode ray tubes (CRTs), 11 primers for electrostatic spray coating of plastics, 12 sensors,13,14 transparent electrodes for inorganic electroluminescent devices 15 in capacitors,16,17 and fuel cell membrane applications. 18 Due to its high stability and considerable conductivity, poly(3,4-ethylenedioxythiophene) (PEDOT) is currently one of the conductive polymers used, with applications in energy conversion and sensing devices as well as in bioelectronics. 19 Xu et al. have recently presented an electroplating procedure for the realization of three-dimensional macro-porous materials (3D-P-PEDOT), composed of micrometer-sized craters and nanometer-sized febrile structures, as well as their use as nitrite and ascorbic acid. 20 EDOT can be polymerized by electrochemical or chemical oxidation. Both processes produce a pure, insoluble material that does not melt below the decomposition temperature.21,22 Recently, PEDOT nanoparticle in aqueous media have been developed from ammonium persulphate and the surfactant dodecylbenzene. 23 Ballav et al. have made an exception by preparing polythiophene/montmorillonite nanocomposites by directly reacting the monomer and montmorillonite-Na+ without solvent. 24 Polymer structures in the nanometer range are also likely to form clay/polymer hybrids. These hybrids have many applications due to their mechanical properties, thermal stability, and reduced gas permeability. 25 Several conductive polymers such as poly (3,4-ethylenedioxythiophene) have been effectively used in bone scaffolds for tissue regeneration. 26 Clays are the most widely available and least expensive natural materials, with high mechanical and chemical resistance. Clay polymer materials have attracted significant attention from researchers, as the interactions between them influence the properties of the clay and the polymer. 27 In this work, we report the preparation of intercalated PEDOT/Mag-CTA nanocomposites using oxidative polymerization of 3,4-ethylenedioxythiophene (EDOT). The physical-chemical, electrical, and electro-chemical properties of the prepared nanocomposites have been measured and compared to those obtained with the pure polymer (PEDOT) obtained under identical experimental conditions. The document is presented as follows: In the next section, we present the methods used and the experimental procedures. After this, the preparation and physical-chemical characteristics of PEDOT/Mag-CTA and PEDOT are examined. Next, electrical and the electrochemical behavior of PEDOT/Mag-CTA nanocomposites and pure polymer (PEDOT) has been studied and compared. The last section summarizes the conclusions concerning the structural, thermal, and electrical properties of the materials obtained. The structural and thermal properties of the obtained materials will be studied by XRD, TGA, DSC, AFM, and scanning and transmission electron microscopy (SEM and TEM) analysis.
Experimental
Materials
Cetyl trimethylammonium bromide (98%), 3,4-ethylenedioxythiophene (99%), and potassium peroxydisulfate ( 80%) were marketed by Sigma-Aldrich Corporation Algeria and used in this work without further purification. All these reagents were used as received from Sigma-Aldrich. The montmorillonite clay used in this study was supplied by ENOF Maghnia society situated in the west of Algeria and used as received
Preparation process for sodium maghnite (Mag-Na) and maghnite-cetyltrimethylammonium (Mag-CTA)
After removing the coarse material by sedimentation, the purified crude maghnite was brought into contact with sodium chloride solution (25 g of clay in 120 mL) with stirring for 4 h. Subsequently, the elaborated maghnite was collected by siphoning, knowing that the montmorillonite crosses a distance of 10 cm every 6 h. Then, it was washed several times with distilled water, filtered through a sintered glass with a porosity of 5 μm, dried in an oven at 120°C, pulverized with a mortar, and finally stored in a desiccator. 28 The transition from maghnite-Na+ to Mag-CTA corresponds to a substitution of Na ions present on the surface of the montmorillonite layers by alkylammonium cations. 30 mmol of CTAB (C16H33)N(CH3)3Br, 3 mL of hydrochloric acid, and 250 mL of water at 72°C were introduced into a beaker, then 10 g of maghnite-Na+ were added. The mixture is placed under ultrasonic irradiation for ten minutes to obtain a white precipitate. Following filtration, the maghnite is washed several times with water to remove the sodium bromide formed. When the rinsing phases are completed, the Mag-CTA is dried at 120°C, ground and stored in a desiccator.29,30
Preparation of PEDOT/Maghnite-CTA nanocomposites
Different poly(3,4-ethylenedioxythiophene)-clay nanocomposites have been prepared using different amounts of modified clays corresponding to 3, 5,7, and 10% by weight based on the initial amount of 3,4-ethylenedioxythiophene and 3,4-ethylenedioxythiophene without clay has also been prepared under the same conditions as the standard. The PEDOT/Mag-CTA nanocomposites containing different amounts of Mag-CTA were obtained by oxidative polymerization of 3,4-ethylenedioxythiophene (EDOT). In containers, different amounts of CTAB-Mag (3, 5, 7, and 10%) were previously heated under vacuum at 100°C for 20 min to remove all traces of moisture, and then each dried amount was added to an aqueous solution containing the same amount of oxidant (K2S2O8(0.05 M)). The mixture was stirred at room temperature for 2 h with an ultrasonic generator at 400 rpm to obtain a better dispersion. The EDOT monomer was added drop by step with stirring, then the polymerization was carried out at 45°C for 4 h. The precipitate was filtered and washed with distilled water. After drying at 65°C under reduced pressure for 6 h, the black powder was obtained (Scheme 1). These nanocomposites will then be referred to as PEDOT/Mag-CTA3wt%, PEDOT/Mag-CTA5wt%, PEDOT/Mag-CTA7wt%, and PEDOT/Mag-CTA 10wt% according to the clay they contain.
31
Experimental conditions for the preparation of PEDOT/Mag-CTA nanocomposites are presented in Table 1. Schematic representation of the synthesis of PEDOT/Maghnite-CTA intercalated nanocomposites. Experimental conditions for the preparation of PEDOT/Mag-CTAwt% nanocomposites.

Characterizations
The Fourier transform infrared (FTIR) spectra were recorded on a Perkin Elmer FTIR spectrometer under dry air flux using 128 scans with a resolution of 4 cm1 in order to investigate the inclusion of PEDOT in the interlayer of montmorillonite (MMT). The powder X-ray diffraction (XRD) patterns of the samples were recorded using a Philips XRD X’PERT PRO II diffractometer (2goniometers, French) using Cu-Kα radiation and (λ = 1.5404 Å) produced at 40 kV and 40 mA. (TEM) Samples were prepared by ultra-microtomy under cryostatic conditions, producing sections approximately 20 nm thick. The sections were examined using the HRTEM Jeol-2011 electron microscope under an accelerating voltage of 200 kV (TEM model JEM-2011, JEOL, Japan). SEM images were taken using a scanning electron microscope (JEOL 6610 LA) at an accelerating voltage of 30 kV. A thermo-gravimetric analysis (TGA) was performed using Setaram LabsysTM DTA, DSC, and TGA instruments between 20 and 1600°C (nominal, 1400°C) with a heating rate of 10°C/min; it was carried out for pure PEDOT and their nanocomposites based on Mag-CTA. The electrical measurements have been carried out using an ohmmeter (Advantest, Digital Electrometer, TR8652) (four-point method), and the electrical resistances of the pellets were evaluated using a two-point probe technique using a digital multimeter (Fluke 8845A) in dry air. The AFM topographic images were produced using a PicoSPM molecular imaging system equipped with a NanoScope IV controller under ambient conditions. The mean effective roughness (R) was determined using the statistical application of Nanoscope software, whose nanometric measurements were obtained under ambient conditions at 50% humidity and 20-25°C. The system was installed on a vibration isolation active table with minimal acoustic disturbance.
Results and discussion
FT-IR analysis
Figure 1 shows the infrared spectra (FT-IR) of (A) PEDOT, (B) Mag-CTA, and (C) PEDOT/Mag-CTA. The FT-IR spectra of PEDOT, organic clay, and PEDOT/Mag-CTA nanocomposite are shown in Figure 1. As can be seen from the Figure 1(a), the infrared spectra of PEDOT shows vibrations around 1309 and 1524 cm−1 which are assigned to the thiophene ring. The two frequencies of vibrations are attributed to C-C and C=C stretching of the quinoidal structure of the thiophene ring. The absence of peaks at 1490 and 1190 cm−1 indicates that the polymerization taken place. The vibrations at 1183, 1141, and 1080 cm−1 are assigned to the stretching of the C-O-C bonds in the ethylenedioxy group and the C-S bonds in the thiophene ring. In Figure 1(b), the band at 3629.4 cm−1 is assigned to the OH group linked to the aluminum in octahedral coordination. In addition, the two bands at 1012 and 481 cm−1 are associated to the deformation vibrations of Si-O-Si. In Figure 1(b), the IR spectra of the surfactants modified maghnite, revealed the presence of new bands at 3011, 2931, 286, 1490, and 1446 cm−1, assigned, respectively, to N-(CH3)+3, asymmetric CH2, symmetric CH2, CH3+, and CH3. The IR spectra in Figure 1(b) show some characteristics that combine the specific groups of the montmorillonite and the CTAB molecule. The FT-IR spectra of PEDOT/Mag-CTA composite (Figure 1(c)) exhibited a first band related to the thiophene ring passing from 1309 to 1320 cm−1, and a second band corresponding to the C-O-C bond passing from 1183 cm−1 to 1205 cm−1. The reason for this shift can be referred to the interaction between the PEDOT polymer and the maghnite-CTA. On the other hand, it can be seen from Figure 1(c) that the Si-O stretching vibration at 1048 cm−1 reveals the vertical attenuation of the spacer layer. This leads to the conclusion that EDOT has been effectively polymerized and inserted in the interlayer of organoclays, resulting in an increase in the interlayer space and a decrease in the number of Si-O. The small bands between 2854 and 2925 cm−1 can be assigned to the C-H elongations of ethylene in the thiophene ring. The FT-IR bands observed in the nanocomposites, when compared to the unmodified clays and those of PEDOT, demonstrated the presence of PEDOT in the nanocomposites. The FT-IR bands observed in the nanocomposites, when compared with Mag-CTA bands and PEDOT bands, demonstrated the presence of PEDOT in the nanocomposites. Kvarnstrom et al. have found the same results in the synthesis of PEDOT.32,33 FTIR spectrums of (A) PEDOT, (B) Maghnite-CTA ,(C) PEDOT/Mag-CTA (% ) nanocomposites.
X-ray diffraction analysis
Figure 2 shows the X-ray diffraction patterns for (a) sodium maghnite (Mag-Na+) and (b) modified maghnite (Mag-CTA). Mag-Na+ has a peak at 2θ = 7.0 corresponding to spacing d001 = 15.549 Å. The incorporation of the alkylammonium ions caused the appearance of two peaks. The first peak at 2θ = 4.5 and the second peak at 2θ = 2.3, which may be due to a heterogeneous organization (dispersion) of the maghnite sheets, resulting in the possibility of a group of sheets separated by a distance of d = 19.34 Å and another group of sheets separated by a distance of d = 37.87 Å (Figure 2).
34
As shown in the Figure 2, the increase in this distance indicates that there is an intercalation of cetyltriméthylammonium ions in the Mag-Na+ interfacial galleries by a simple cation exchange and flocculation of the intercalated sheets due to interactions between the maghnite sheets. XRD patterns of the Maghnite-Na+, Maghnite-CTA.
Figure 3 shows the XRD spectra of the PEDOT/Mag-CTA (3, 5, 7, and 10%) nanocomposites obtained, with PEDOT/Mag-CTA nanocomposites prepared from 3wt% giving exfoliated morphologies. It is indicated at the low angles by the absence of the diffraction peak; nanoleaves are characterized in an individual way by the distance of several dozen angströms between layers. The preparation of PEDOT/Mag-CTA nanocomposites of 5 and 7% by weight showed the displacement of the basal peak d001 at small angles between 2Ɵ = 2.6-2.5°, including distance d001 = 35.3 Å and 33.5 Å, respectively. The second peak at 2θ = 5.1° and 4.9° present at the following distances d001 = 18.83 Å, 18.52 Å, and thus confirms their interlaced/exfoliated mixture morphologies. With the exception of nanocomposites prepared with 10% Mag-CTA d001 = 25.5 Å. The polymerization of PEDOT reveals morphology of intercalated/exfoliated clay mixture with low yield due to the closure of the clay galleries leading to tortuous diffusion pathways. The increase in the basal spacing of Mag-CTA, measured by (XRD), proved the existence of polymerization at the intermediate layers of the clay. XRD patterns of Mag-CTA and PEDOT/Mag-CTA nanocomposites.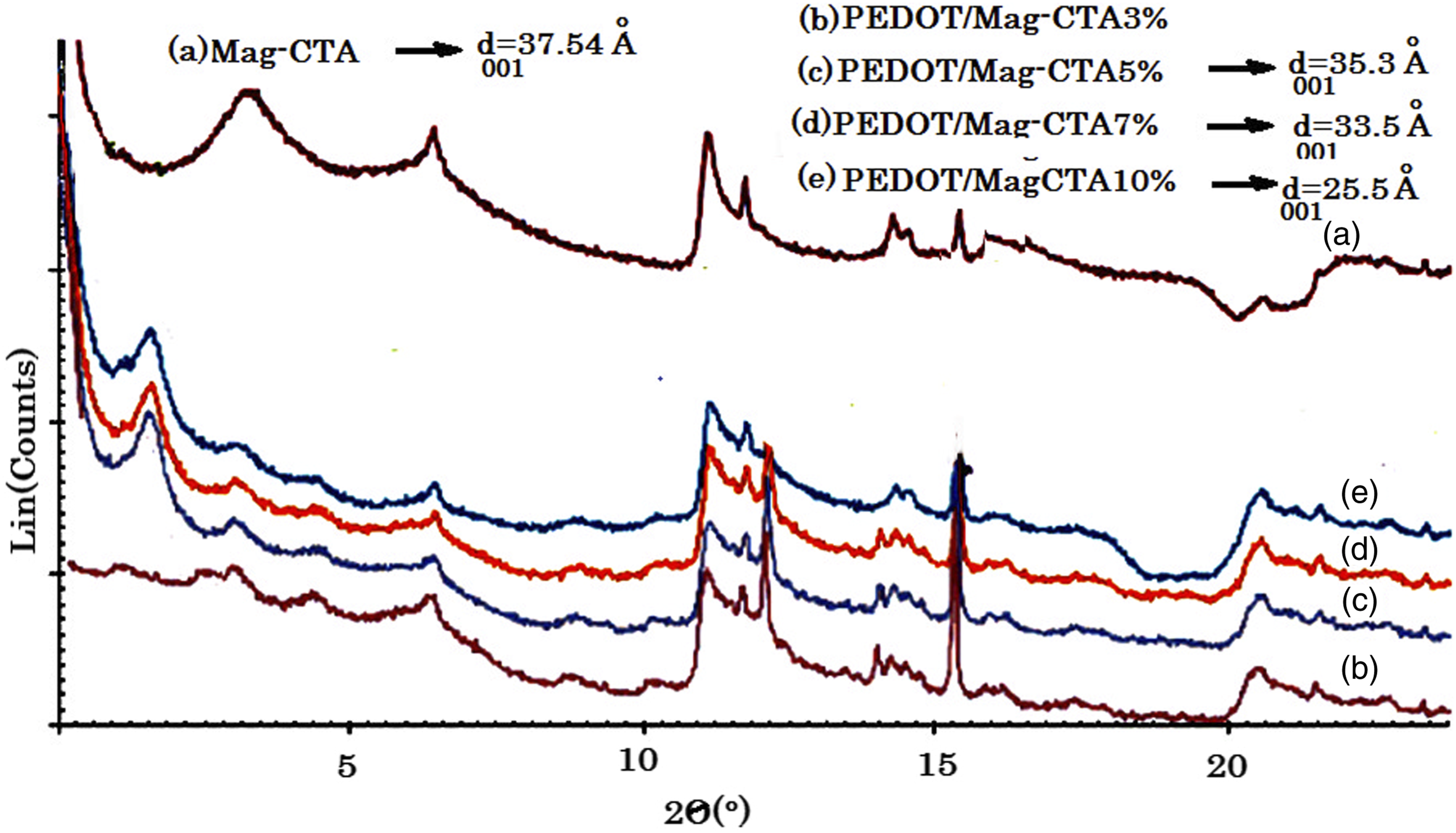
Transmission electron microscopy analysis
In order to confirm the relevance of the X-ray diffraction results, Na-Mag and its organo-modified counterpart were subjected to transmission electron microscopy (TEM) analysis, as shown in Fig. s1. The TEM images taken on Na-Mag show structural homogeneity in terms of interlayer distance. In contrast, CTA-Mag revealed a striped pattern at the nanoscale, revealing an intercalated structure where surfactants are interspersed between maghnite layers. These results are consistent with those obtained by XRD analysis.
TEM images of PEDOT/Mag-CTA nanocomposites containing 3, 5, 7, and 10% by weight of Mag-CTA are shown in Figure 4. In Figure 4(a), exfoliated structures were observed in the matrix PEDOT, showing a high degree of exfoliation and that the clay layers are dispersed in the matrix. The PEDOT/Mag-CTA nanocomposites at 3% by weight show some disorder in the morphology of the clay. Contrary to the results in Figure 4(b–d), the TEM images of the PEDOT/Mag-CTA nanocomposites show intercalated/exfoliated mixture morphology characterized by the exfoliation of some structures and the intercalation of others structures. TEM images of: (a) PEDOT/Mag-CTA-3wt% (Exfoliated), (b) PEDOT/Mag-CTA-5 wt% (exfoliated/intercalated structures), PEDOT/Mag-CTA-7 wt% (exfoliated/intercalated structures), and (d) PEDOT/Mag-CTA-10% (exfoliated/intercalated structures) nanocomposites.
Scanning electron microscopy analysis
The scanning electron microscopy images of the Na-Mag and organo-modified clays are shown in Fig. s2. Indeed, the SEM images showed that the morphology of the maghnite was preserved following the modification carried out by the CTAB. The granulometry of the clays obtained is between 1 and 10 μm. It is established that the intercalation process takes place exclusively in the interlamellar space. This justifies the obtaining of a similar morphology between the maghnite-Na and the organo-modified clay (CTA-maghnite).
Figure 5 shows the SEM micrographs for the synthesized PEDOT/Mag-CTA nanocomposites. The analysis of nanocomposites by scanning electron microscopy (SEM) has shown that as much as the quantity of organic clay dispersed in the PEDOT matrix is large, the nanocomposites become more irregular and porous. The micrographs in Figure 5(b–c) show more ordered and denser structures. Smaller aggregates are observed with high aggregate density at loading rates of 3, 5, and 7% while these aggregates become larger with lower density per unit area at a loading rate of 10%. This shows that increasing loading rates in nanocomposites increases the compactness of the samples which become denser. The PEDOT is in the form of non-uniformly distributed agglomerates; in the case of 10% clay, these organically modified clay particles are more dense. The white portion of the sample observed (Figure 5(c)) shows a rougher surface and a covering of the montmorillonite surface by the PEDOT. SEM images of: (a) PEDOT/Mag-CTA-3wt% , (b) PEDOT/Mag-CTA-5wt% , PEDOT/Mag-CTA-7wt% and (d) PEDOT/Mag-CTA-10% nanocomposites.
AFM analysis
Average thickness (ȴ’) and average RMS roughness (R) of PEDOT and PEDOT/Mag-CTAwt% nanocomposites.


AFM images of PEDOT pure (a) and PEDOT/Maghnite-CTA 5wt% (b) films.
Thermogravimetric analysis
Analysis thermogravimetry (TGA) and differential thermal analysis (DTG) of the samples have been performed under a N2 atmosphere between room temperature up to 900°C. The results are shown in Figures 7 and 8. The TGA curve for PEDOT/Mag-CTA in Figure 7 shows a typical two-step weight loss process: the first weight loss for PEDOT is at about 305°C, but increases to 375°C for PEDOT/Mag-CTA after EDOT has been intercalated. The second weight loss at 600 to 700°C is associated with the dehydroxylation of the silicate structure. The TGA also shows that increasing the weight percentage of Mag-CTA results in a considerable improvement in the thermal stability of the PEDOT matrix. This improvement in thermal stability can be explained by the fact that montmorillonite has a barrier effect. In other words, the presence of Mag-CTA (% by weight) slows down the decomposition of nanocomposites. TGA curves of PEDOT pure and PEDOT/Mag-CTA- wt% nanocomposites. DTG curves of PEDOT/Mag-CTA- wt%, nanocomposites.

According to Figure 7, the thermal stability of the nanocomposites is important at a rate of 5wt% by weight of Mag-CTA, while the nanocomposites at 7 and 10% by weight of Mag-CTA decompose at temperatures lower than that of the nanocomposite PEDOT/Mag-CTA at 5wt% by weight. The decrease in thermal stability of the PEDOT/Mag-CTA nanocomposites (7 and 10% by weight) can be attributed to the absorption of excessive amounts of cetyltrimethylammonium bromide (CTAB).
Degradation temperatures of pure PEDOT and nanocomposites with different ratios of Mag–CTA.

Thermal analysis
The analysis has been carried out to study the influence of the loading rate on the glass transition temperature of the matrix. Figure 9 shows that the Tg of PEDOT/Maghnite-CTA nanocomposites is higher than that of pure poly(3,4-ethylenedioxythiophene), which is 390°C. This increase is due to the introduction of the clay into the matrix which makes it more rigid. The thermogram of PEDOT/Maghnite-CTA clay content (5wt%) nanocomposite shows the presence of a first endothermic peak at 82.5°C, which indicates the initial release of the molecules water. A 2nd endothermic peak around 290°C is attributed to the decomposition of CTAB. The third peak around 400°C is associated with the reticulation of the polymeric chains which is retarded relative to that of poly (3,4-éthylènedioxythiophène). Finally, we can observe a peak around (700-900°C) which represents the final decomposition of the nanocomposite. DSC analysis graphs for nanocomposites of PEDOT/Maghnite-CTA 5%(byweight) nanocomposites.
Conductivity
Variation of the electrical conductivity of nanocomposites as a function of the amount of Maghnite-CTA.

Conclusion
The influences of the amount of organo-clays (Mag-CTA) prepared and used on the properties of PEDOT nanocomposites/clay Algerian were investigated. PEDOT/Mag-CTA nanocomposites were synthesized by oxidative polymerization of the EDOT monomer in the presence of modified montmorillonite. X-ray diffraction and electron transmission microscopy analysis revealed (intercalation/exfoliation) structures for the PEDOT/Mag-CTA nanocomposites. The improvement in thermal stability of nanocomposites is affected by the presence of clay in the polymer matrix which delays its decomposition. The results obtained show that the amount of organic clay of 5wt% is recommended for the preparation of a nanocomposite with high thermal stability. Electrical conductivity measurements showed a decrease in conductivity with increasing charge rate since montmorillonite inhibits the movement of electrons on polymer chains and limits their inter-strand mobility. In addition, it should be noted that the introduction of special chemical modifications in MMT shows that higher properties could be obtained from clay, as proven by Algerian bentonites.
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911221080302 – Supplemental Material for Synergistic effect of organic clay fillers based on cetyltrimethylammonium bromide in the synthesis of poly (3,4-ethylenedioxythiophene) nanocomposites
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911221080302 for Synergistic effect of organic clay fillers based on cetyltrimethylammonium bromide in the synthesis of poly (3,4-ethylenedioxythiophene) nanocomposites by Lahouari Mrah in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
