Abstract
Dyes removal from wastewater of industries is considered the prime demand of the environment. Three trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels, previously reported as novel antimicrobial agents, were investigated for the first time in this work as adsorbents for Basic Red 12 (BR 12) cationic dye. These hydrogels behave as polyanionic materials. Their adsorption capacity for the BR 12 dye increased significantly with increasing both the temperature, the solution pH and their cross-linking content. Their adsorption fitted to the pseudo-second-order kinetic model. Their adsorption isotherms conform to the Langmuir model suggesting that the adsorption nature is a monolayer having an optimum sorption capability of 38.27 mg g−1 and removal efficiency of 97.38%. The results proved that the adsorption process is remarkably proceeded by chemisorption phenomenon. The data of adsorption thermodynamics indicate that the adsorption is an endothermic process. The regeneration and reuse of the hydrogels were possible. Thus, incorporation of the functionality of the cross-linker into chitosan within the same structure efficiently enhanced its features. It is a good manner for achieving appropriate series of efficient adsorbents which are considered as favorable nominees in the field of basic dyes removal.
Keywords
Introduction
High-quality water is critical in the physiology for the living organisms. Hundreds of synthetic non-biodegradable dyes, discharged to the aquatic resources after the different industrial coloring processes, are poisonous even at very low concentrations. This leads to water contamination and can be accumulated in the organisms causing the skin irritation, cancer, allergies, jaundice, heart defects and mutations. 1 Thus, the dyes removal becomes environmentally very necessary and represents a large challenging in industrial wastewater treatment. The adsorption processes are considered extremely efficient techniques for dyes removal because they are rapid, low cost, simple concerning the design of the treatment operation unit and their by-products are nontoxic. 2 Despite the widespread use of the activated carbon in various cleaning processes, it stays costly. 3 Thus, its substitution by non-expensive alternate adsorbents has recently been the interest of many researchers. 4
The biopolymer, chitosan, has attracted a great significance in the biotechnological field because of its antimicrobial activity 5 and the super ability for adsorbing the anionic dyes 1 due to its polycationic character. Chitosan is also abundant, hydrophilic, biologically compatible, biologically degradable, renewable and nontoxic. 6
Chitosan easily dissolves in acidic media, and this is considered as its worst drawback, particularly in case of its employment as adsorbent for dyes removal because the effluent is commonly acidic. Consequently, controlling the solubility of chitosan is considered a desirable property for such applications. Thus, it is essential to develop stable and durable biopolymeric materials, based on chitosan, via controlling their solubility characteristics through grafting copolymerization7,8 or polymer blending. 9 The chemical cross-linking process is a promising alternative for stabilizing chitosan in acidic solutions.10,11 Therefore, modification of chitosan via cross-linking is used to improve its properties such as enhancing its mechanical properties, increasing its chemical stability by lowering its solubility, inhibiting its degradation rate and increasing the life span of its products in different media.
On the other hand, chitosan’s polycationic character limits its application for cationic dyes removal and represents a weak point. It is well established that the cationic dyes are more poisonous than the anionic ones. 12 Thus, it is necessary to provide chitosan with suitable anionic groups to act as binding sites for the cationic dyes. 13
Accordingly, the present work was directed toward using the previously prepared chitosan hydrogels 14 as promising adsorbents for cationic dyes because they possess carboxylic groups that can act as anionic binding centers for cationic dyes. BR 12 dye was chosen, as a representative example of cationic dyes, because of its high toxicity. The kinetics, isotherms and thermodynamic of the adsorption process for the cationic BR 12 dye onto these hydrogels were studied. Some parameters that affect the adsorption process like temperature, period, pH of the medium and cross-linking moiety content of the hydrogels were also studied.
Experimental section
Materials
Three hydrogels, H1, H2 and H3, utilized in present work, were prepared (Figure 1 and Table 1) following our procedure illustrated previously. 14 H3 in present study was reported as H4 in our previous work. 14 Fabrication of the hydrogels into particles of various sizes (0.4 mm, 0.8 mm, 1.5 mm and 3 mm) were done according to method described in our previous work. 15 Preliminary investigations showed a greater adsorption capacity for BR 12 dye onto the hydrogels of 0.4 mm particle size relative to those of larger sizes. Thus, the adsorption process was confined on the hydrogels of 0.4 mm particle size. BR 12 dye (Figure 2) was obtained from Hoechst (Germany).

Preparation of the hydrogels.
Synthesis of the cross-linked chitosan hydrogels and their elemental analyses.


Chemical structure of BR 12 dye.
Measurements
The stock BR 12 dye solution was prepared via dissolving the dye in double distilled water at 0.001 mol/l concentration. The adsorption kinetics and equilibrium were performed by batch experiments. The dye concentration was calculated via determining the absorbance at λmax = 485 nm using UV-1650 PC Shimadzu spectrophotometer.
Adsorption studies
50 mg of the adsorbent were introduced into a glass conical flask, then 50 ml of BR 12 solution at 1 × 10−4 mol/l concentration were added. The flask was shaken in a water bath at a selected temperature and at a speed of 80 rpm to reach equilibrium. Various adsorption process variables like temperature (25, 35, 45 and 55°C) and dye solution pH (4, 7 and 10), regulated using sodium hydroxide or hydrochloric acid solutions, have been investigated.
The adsorption capacity of adsorbent for the dye at equilibrium time (qe (mg/g)) and at time t (qt (mg/g)) was quantified by the relationships of balance of weight (Eqs. 1 and 2, respectively).


where Co, Ce and Ct are the initial dye concentration, the dye concentration at equilibrium time and the dye concentration at time t (mg/l), respectively, V is the volume of the dye solution (l), and W is the mass of adsorbent (g). 16
The removal efficiency percent of the dye in an equilibrium state is determined by Eq. 3. 17

where Co and Ce are the initial concentration of the dye and the concentration of the dye at equilibrium time (mg/L), respectively.
Adsorption kinetic studies
The pseudo-first order (Eq. 4) and the pseudo-second order (Eq. 5) represent the two kinetic models that are usually utilized for exploring the adsorption mechanism for a dye onto an adsorbent.


where qe and qt are the quantities of the dye adsorbed onto the adsorbent at equilibrium time and at different time intervals (mg/g), respectively, k1 and k2 are the adsorption rate constants of the pseudo-first order (min−1) and the pseudo-second-order (g/mg min) kinetic models, respectively.16,18,19
Adsorption isotherm studies
In the adsorption isotherm experiment, 10 mg of adsorbent were added with 10 ml of different concentrations of BR 12 dye solution (1–10 × 10−5mol/l) at pH 7 and temperature 55°C and agitated till equilibrium time in a thermostat water bath.
The experimental data were illustrated in the format of the Langmuir and Freundlich isotherms. The Langmuir isotherm is presented by Eq. 6.

where qe is the quantity of dye adsorbed at equilibrium time (mg/g), Ce is the equilibrium concentration of dye in solution (mg/l), qmax is the monolayer sorption capacity (mg/g) and KL is a constant related with the energy of the adsorption (l/mg).
The fundamental feature of the Langmuir isotherm could be expressed in terms of a dimensionless equilibrium factor, such as the separation factor or equilibrium factor (RL) (Eq. 7).

The Freundlich isotherm is presented using Eqs. 8 and 9:


where qe is the quantity of the dye adsorbed at equilibrium time (mg/g), Ce is the dye equilibrium solution concentration at a constant temperature (mg/l), KF and n are empirical constants that can be related to the adsorption capacity and the adsorption intensity, respectively. 18
Adsorption thermodynamic studies
Free energy change ΔG° (KJ/mol) was calculated using Eq. 10, enthalpy change ΔH° (KJ/mol) and entropy change ΔS° (J/mol K) were calculated from the slope and the intercept of the plot of ln KC versus T−1 (Eq. 12), respectively.



where R is the gas constant (8.314 J/K mol), T is the temperature (K) and KC is the equilibrium constant 19
Desorption studies
The restoring of the adsorbents is carried out firstly by washing the dye-saturated hydrogels with distilled H2O to get rid of the non-adsorbed dye. Then, the hydrogels were immersed in 20 ml of absolute ethanol as a desorbing medium under constant shaking at 80 rpm at 25°C for 24 h. The amount of the desorbed dye was calculated based on Eq. 13.

where qd is the quantity of dye desorbed from the dye-saturated adsorbent (mg/g) and qa is the quantity of the dye adsorbed onto the adsorbent (mg/g). 20
Results and discussion
The mechanism of BR 12 dye removal by the investigated hydrogels could be proposed via studying the effect of both the temperature and the pH of the dye solution on its adsorption process as follows:
Effect of temperature
Temperature could be considered as one of the most efficient factors for the adsorption processes of the dyes. Figure 3 shows the temperature influence (25, 35, 45 and 55°C) on the adsorption capacity for BR 12 dye onto H1–H3 as a function of time (dose of the hydrogel = 50 mg, initial dye concentration = 1 × 10−4 mol/l, dye solution volume = 50 ml and pH 7). It could be noted that a relatively slight increase in the adsorption capacity (qt, Eq. 2) for BR 12 dye with increasing the temperature was observed (Figure 3). The removal efficiency (%) (Eq. 3) for BR 12 dye by H1, H2 and H3 increased from 20.90% to 47.09%, from 62.28% to 67.52% and from 89.52% to 96.33%, respectively, with increasing the temperature from 25 to 55°C (Online Supplemental File-1).
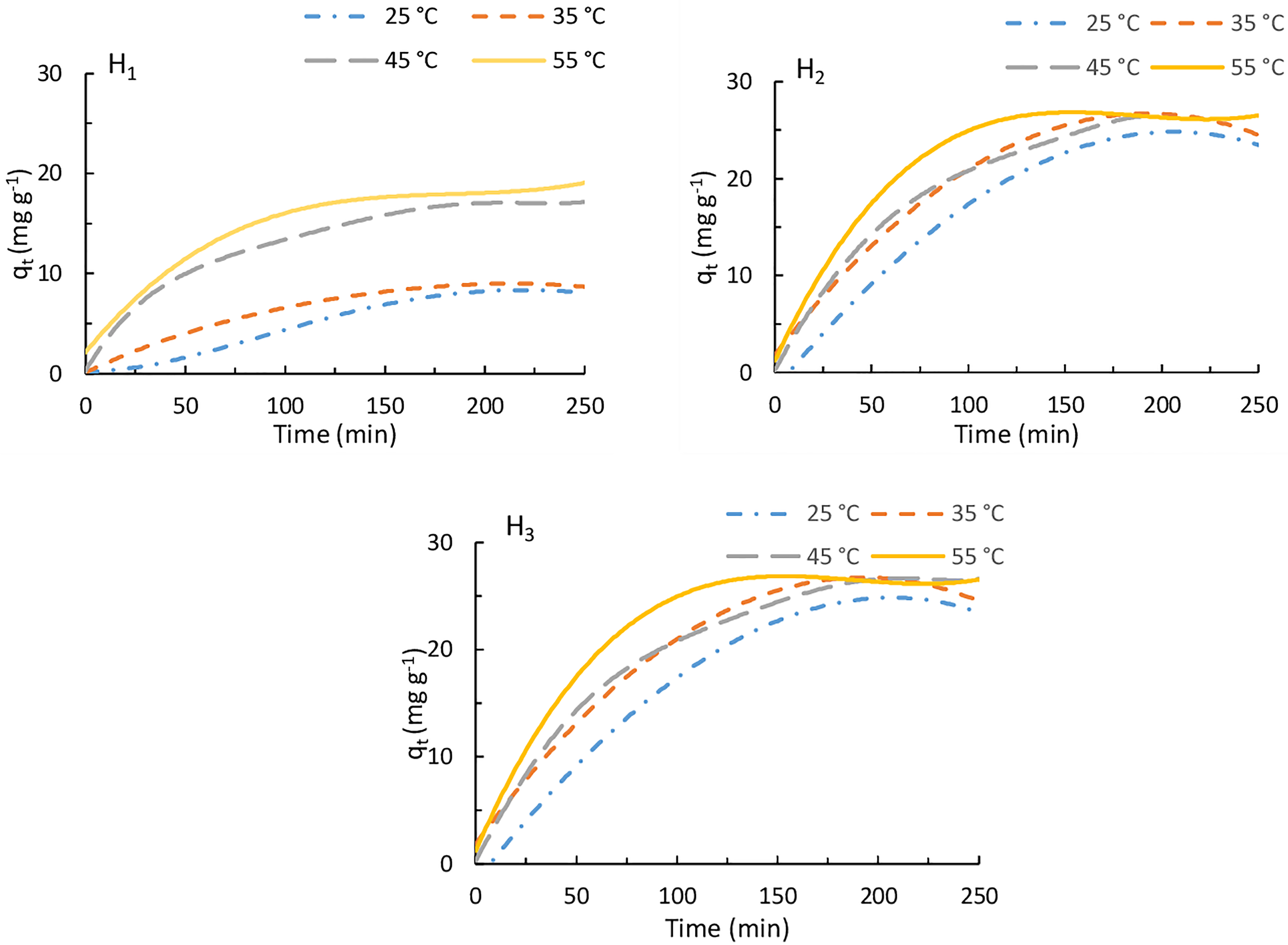
Effect of temperature on adsorption for BR 12 dye onto cross-linked chitosan hydrogels at various time intervals (adsorbent dose = 50 mg, initial dye concentration = 1 × 10−4 mol/l, volume of dye solution = 50 ml and pH 7).
The enhancement of the adsorption capacity and consequently the percent removal efficiency for BR 12 dye onto the investigated hydrogels by increment of the temperature refer to the improvement of the kinetic energy of the dye and water molecules as well as the increase in swelling capability within the interior structure of H1, H2 and H3, which improve the breakthrough of the dye molecules into the space of the hydrogel interlayer. 21 Also, the temperature increment results in enhancement of the diffusion rate of the dye molecules across the border of the exterior layer and into the interior porosity of the hydrogel matrix. Finally, at equilibrium, the capability of a hydrogel for a dye will be changed with changing the temperature. 22
Figure 3 and Online Supplemental File-1 also demonstrate the effect of the cross-linking moiety content on the adsorption process. At a particular temperature, both the removal efficiency (%) and the adsorption capacity for BR 12 dye by the hydrogels significantly improved with the increase of their cross-linking moiety content. This is due to the increment in the number of the carboxylic groups, from H1 to H3, acting as anionic binding sites for the cationic BR 12 dye.
For a comparison, the adsorption of BR 12 dye onto parent chitosan was determined (dose of the chitosan = 50 mg, initial dye concentration = 1 × 10−4 mol/l, dye solution volume = 50 ml, temperature = 55°C and pH 7). It was found that chitosan does not show any adsorption for BR 12 cationic dye indicating the role of carboxylic groups of the inserted cross-linking linkages in the adsorption process.
Effect of dye solution pH
pH could be considered as one of the main factors affecting adsorption process via a set of various mechanisms through acquirement or loss of protons from the adsorbents and also from the molecules of the dyes. 13
Figure 4 illustrates the results of the impact of the pH of the media (4, 7 and 10) on adsorption capacity for BR 12 dye onto H1, H2 and H3 hydrogels as a function of time (dose of the hydrogel = 50 mg, initial dye concentration = 1 × 10−4 mol/l, dye solution volume = 50 ml and temperature = 55°C). The results referred that the adsorption capacity (qt, Eq. 2) of the hydrogels for BR 12 dye increases significantly with the pH increment (Figure 4). The percent removal efficiency (Eq. 3) of BR 12 dye by H1, H2 and H3 increased from 0.00% to 70.67%, from 0.00% to 97.33% and from 0.00% to 97.38%, respectively, with increasing the pH of the dye solution from 4 to 10 (Online Supplemental File-2). The observed no removal of BR 12 dye by all the hydrogels in a high acidic solution (pH 4) may be ascribed to the high concentration of H+ ions which severely vie the cationic BR 12 dye for adsorbing onto the hydrogels. H+ ions are efficiently adsorbed onto the hydrogels due to their small size forming a high repulsive force between the molecules of the cationic BR 12 dye and the adsorbent surfaces carrying positive charges leading to no adsorption of BR 12 at low pH.

Effect of solution pH on adsorption for BR 12 dye onto cross-linked chitosan hydrogels at various time intervals (adsorbent dose = 50 mg, initial dye concentration = 1 × 104 mol/l, volume of dye solution = 50 ml and temperature = 55°C).
On the other hand, at high pH of the BR 12 dye solution, the great number of the hydroxyl ions assist the deprotonation of the carboxylic groups of the cross-linking linkages into the carboxylate anions (-COO−). Thus, the molecules of the cationic BR 12 dye are electrostatically attracted to the adsorbent surfaces carrying negative binding sites resulting in an increment of the dye adsorption. 23 Moreover, the repulsion between the carboxylate anions forces the polymeric chains to be more extended and shows higher swelling ability that enhances the diffusion of the dye molecules into the interlayer space. 24 Thus, the investigated hydrogels act as polyanionic adsorbents (Figure 5). It is worth mentioning that adsorption of BR12 dye onto the cross-linked chitosan hydrogels increases with increment of cross-linking linkages; i.e. from H1 to H3. This is ascribed to the increment in the number of the carboxylic groups that are available for adsorbing the dye.

Schematic demonstration of the interaction mechanism of BR 12 dye with the hydrogels at pH 10.
Kinetics of adsorption
Study of the kinetics of adsorption is essential to suggesting appropriate adsorption systems and determining optimum operating parameters for adsorption conditions.
The rate of a dye adsorption affects by both the soaking time of adsorbent into the dye solution and the diffusion process. 25 The adsorption process includes mainly three steps; first, the molecules of the dye migrate from its aqueous solution to the adsorbent boundary layer, second, they penetrate into the surface of the adsorbent, and third, they distribute into the internal pores through the penetration of the pores. 25
To identify the adsorption process for BR 12 dye by H1–H3, kinetic models including pseudo-first-order (Eq. 4) and pseudo-second order (Eq. 5) at different temperatures and pH were used.
At different temperatures
The different kinetic parameters of the adsorption process for BR 12 dye onto H1–H3 at various temperatures (25, 35, 45 and 55°C) were summarized in Table 2. The adsorption rate constant k1 for pseudo-first-order kinetic model (Eq. 4) was established from the slope of the plot log(qe − qt) versus t (Online Supplemental File-3). Whereas, the adsorption rate constant k2 for pseudo-second-order kinetic model (Eq. 5) was determined from the intercept of the plot of t/qt versus t (Figure 6). The results in Table 2 indicate that the pseudo-second-order kinetic model describes best the adsorption process of BR 12 dye by all the adsorbents H1, H2 and H3 as illustrated from R2 values. For example, R2 of the pseudo-second-order model for the adsorption of BR 12 dye by H1, H2 and H3 at 55°C were 0.927, 0.946 and 0.988, respectively, which are higher than 0.899, 0.861 and 0.983, respectively, for the pseudo-first-order model (Table 2). From Table 2, for a particular hydrogel, the adsorption rate constant k2 for BR 12 dye rose by rising temperature. This suggests that the adsorption process is an endothermic reaction. 19
Kinetic model constants and correlation coefficients for adsorption of BR 12 dye onto cross-linked chitosan hydrogels at different temperatures.


Pseudo-second order for BR 12 dye adsorption onto the cross-linked chitosan hydrogels at different temperatures.
For H3, as a representative example, the maximum adsorption of BR 12 dye was reached after contact time of 200 min at 25°C and after 170 min at 55°C. This indicated that the equilibrium time lowered with rising temperature due to an increase in both the adsorption rate and the pores size due to a greater swelling effect, leading to a greater diffusion of the dye molecules onto the adsorbents. 18
At different pH values
Table 3 lists the different kinetic parameters of the adsorption process for BR 12 dye by H1–H3 at different pH (4, 7 and 10). The adsorption rate constant k1 for pseudo-first-order kinetic model (Eq. 4) was established from the slope of the plot log(qe − qt) versus t (Online Supplemental File-4). The adsorption rate constant k2 for pseudo-second-order kinetic model (Eq. 5) was determined from the intercept of the plot of t/qt versus t (Figure 7).
Kinetic model constants and correlation coefficients for adsorption of BR 12 dye onto cross-linked chitosan hydrogels at different pH values.


Pseudo-second order for BR 12 dye adsorption onto the cross-linked chitosan hydrogels at different pH values.
The results in Table 3 shows that the pseudo-second-order kinetic model describes best the adsorption process for BR12 dye by all the adsorbents H1, H2 and H3 as illustrated from R2 values. For example, R2 of the pseudo-second-order model for the adsorption of BR 12 dye by H1, H2 and H3 at pH 10 were 0.993, 0.967 and 0.990, respectively, which are higher than 0.916, 0.951 and 0.973, respectively, for the pseudo-first-order model (Table 3). This is in consistent with a study of removal of Methylene Blue dye by a bio-adsorbent based on beads of chitosan-oil palm ash zeolite composite. 26
From Table 3, for a particular hydrogel, it can be noted that k2 rose by rising pH of the medium of BR 12 dye.
For H3, as a representative example, the maximum adsorption of BR 12 dye was reached after contact time of 170 min at pH 7 and after 120 min at pH 10. This indicated that the equilibrium time decreased with increasing pH due to an increase in the adsorption rate and the pores size due to a greater swelling effect, leading to a greater diffusion of the dye molecules onto the adsorbents. 18
Adsorption isotherms
The interaction between the surfaces of the hydrogels and the molecules of the dyes in the equilibrium state could be identified by studying the models of the adsorption isotherms. 25 The isotherms of the adsorption of BR 12 dye onto H1, H2 and H3 hydrogels were performed at the best conditions that attained the optimum efficiency of the adsorption (10 mg of hydrogel, 10 ml of neutral dye solution, and 55°C) and the analysis of the results was done applying the linear Langmuir and Freundlich isotherm models (Eqs. 8 and 11, respectively). The Langmuir isotherm model is represented by the homogeneity along the surface of the adsorbent since each center of the adsorbent is occupied by only one molecule of dye and no further adsorption occurs at these sites. Thus, the nature of the adsorption is a monolayer process. The Freundlich isotherm model proposes the non-homogeneity along the adsorbent’s surface and the adsorption is multilayer process in nature.19,27
Figure 8 and Online Supplemental File-5 represent adsorption isotherms fitted to Langmuir and Freundlich models, respectively. Table 4 lists Langmuir and Freundlich equilibrium constants and their corresponding fitting correlation coefficients (R2). Langmuir isotherm model suits the best, with the highest R2 values for BR 12 dye compared to that of the Freundlich isotherm model. This is in consistent with a study of adsorption of Rhodamine B dye by chitosan and its supported mesoporous adsorbents. 23 The 1/n values of BR 12 dye from Freundlich isotherm model were lower than 1.0 indicating that the cross-linked chitosan hydrogels were suitable adsorbents for BR 12 dye adsorption. This is in consistent with the study previously reported. 13 The BR 12 dye affinity to the hydrogels was also estimated via the separation factor (RL) determined by Eq. 7. The adsorption processes were defined based on RL value. It is irreversible when RL value is equal zero, linear when RL value is equal 1, unsuitable when RL value is higher than 1 or favorable when 0 < RL < 1. 25 In the current study, the adsorption processes of BR 12 dye onto H1–H3 hydrogels are favorable because RL value ranged from 0.005 to 0.337. This is in consistent with the previous finding for removing of Methylene Blue using zeolite modified chitosan. 19

Linear Langmuir isotherm model for the adsorption of BR 12 dye onto cross-linked chitosan hydrogels.
Thermodynamic parameters, Langmuir and Freundlich isotherm constants for the adsorption of BR 12 dye onto cross-linked chitosan hydrogels.

Adsorption thermodynamic
The temperature impact on the adsorption of BR 12 dye was further evaluated via studying the thermodynamic of the adsorption.
Standard free energy change (ΔG°), standard enthalpy change (ΔH°) and standard entropy change (ΔS°) of BR 12 dye adsorption onto H1–H3 were calculated at different temperatures (25, 35, 45 and 55°C). The value of ΔG° were calculated from Eq. 10. The ΔS° and ΔH° values were determined from the intercept and the slop of the plot ln KC versus T−1, respectively, (Eq. 12) and were shown in Figure 9. The values of ΔH°, ΔG° and ΔS° of the adsorption of BR 12 dye onto H1, H2 and H3 are summarized in Table 4.

Adsorption thermodynamic of BR 12 dye onto cross-linked chitosan hydrogels.
The value of ΔH° is very helpful for determining the kind of adsorption. The positive values of ΔH° for the adsorption processes of BR 12 dye onto H1, H2 and H3 hydrogels indicate endothermic nature of the adsorption process. This is evidenced via increasing of the adsorption of BR 12 dye with increasing the temperature as previously reported work. 19 The negative values of ΔG° (Table 4) indicate the spontaneous nature of the adsorption processes. On the other hand, the positive values of ΔG° indicate the non-spontaneous nature of the adsorption processes (Table 4) as previously reported study. 23 Moreover, it can be noted that the values of ΔG° decreased by rising the temperature which indicate that the adsorption processes are more suitable at elevated temperature. 19 The positive values of ΔS° of the adsorption BR 12 dye onto all hydrogels displayed the increase in the randomness at the solid/solution interface during the adsorption process related to the increase of the heterogeneity of the surface of the adsorbents. This is in consistent with the work previously reported. 19
Comparison between the sorption capacity for BR 12 dye by the investigated hydrogels and other adsorbents
The efficiencies of the hydrogels in the current work, H1, H2 and H3, for removal of BR 12 dye were found to be 70.67%, 97.33% and 97.38%, respectively, which is larger than those of the other previously reported adsorbents (Online Supplemental File-6).28,29 This suggests the possibility for applying these designed hydrogels for removal of BR 12 dye. In the current work, the chemical modification of chitosan has been performed to get hydrogels, H1, H2 and H3, possessing additional characteristic anionic functional groups (-COOH groups) which could effectively adsorb BR 12 dye. Thus, it can be concluded that these polyanionic hydrogels are promising adsorbents for removing of cationic BR 12 dye.
Desorption of cationic BR 12 dye
The regeneration of the adsorbents by removing the adsorbed dyes is essential for their reuse in terms of the costs and economic importance. The BR 12 dye was quantitatively desorbed from the dye-loaded hydrogels as calculated following the Eq. 13. This finding confirms again that the sorption of the dye onto the investigated hydrogels takes place through the electrostatic interactions. The regeneration and the reuse of these hydrogels were possible for three times during which no change in adsorption capacity was observed.
Conclusion
In conclusion, the adsorption capabilities of the trimellitic anhydride isothiocyanate-cross-linked hydrogels based on chitosan for BR 12 dye were found to be impacted by the temperature, pH value, touch time and the cross-linker contents incorporated into chitosan hydrogels. Their sorption capacities remarkably improved with increasing temperature, with increasing adsorption solution pH, with increasing the adsorption time and with the increment of the cross-linker contents introduced into the hydrogels. The results revealed that the capacity of the adsorption for BR 12 dye by H3 is better than that obtained by H2 which in its turn is greater than that observed by H1. The optimum dye removal efficiency of 97.38% was obtained using H3. The results revealed that the Langmuir model described quite the data of the adsorption isotherm suggesting that the interaction between the BR 12 dye and the hydrogels occurs by a chemisorption mechanism. An additional proof on the chemisorption type of the process of the adsorption comes from the data of adsorption kinetics that followed the pseudo-second-order kinetic model. The data of the thermodynamic of the adsorption indicated the spontaneous and the endothermic nature of the process of the adsorption. The regeneration and the reuse of these hydrogels were possible for three times during which the capacity of the adsorption for BR 12 dye onto the hydrogels does not change. Accordingly, the investigated hydrogels could be utilized as efficient alternative adsorbents for removing of BR 12 dye from its aqueous solution for industrial wastewater treatment.
Supplemental material
Supplemental Material, sj-pdf-1-ppc-10.1177_0967391121999364 - Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels
Supplemental Material, sj-pdf-1-ppc-10.1177_0967391121999364 for Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels by Nadia A Mohamed, Nouf F Al-Harby and Mawaheb S Almarshed in Polymers and Polymer Composites
Supplemental material
Supplemental Material, sj-pdf-2-ppc-10.1177_0967391121999364 - Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels
Supplemental Material, sj-pdf-2-ppc-10.1177_0967391121999364 for Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels by Nadia A Mohamed, Nouf F Al-Harby and Mawaheb S Almarshed in Polymers and Polymer Composites
Supplemental material
Supplemental Material, sj-pdf-3-ppc-10.1177_0967391121999364 - Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels
Supplemental Material, sj-pdf-3-ppc-10.1177_0967391121999364 for Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels by Nadia A Mohamed, Nouf F Al-Harby and Mawaheb S Almarshed in Polymers and Polymer Composites
Supplemental material
Supplemental Material, sj-pdf-4-ppc-10.1177_0967391121999364 - Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels
Supplemental Material, sj-pdf-4-ppc-10.1177_0967391121999364 for Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels by Nadia A Mohamed, Nouf F Al-Harby and Mawaheb S Almarshed in Polymers and Polymer Composites
Supplemental material
Supplemental Material, sj-pdf-5-ppc-10.1177_0967391121999364 - Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels
Supplemental Material, sj-pdf-5-ppc-10.1177_0967391121999364 for Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels by Nadia A Mohamed, Nouf F Al-Harby and Mawaheb S Almarshed in Polymers and Polymer Composites
Supplemental material
Supplemental Material, sj-pdf-6-ppc-10.1177_0967391121999364 - Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels
Supplemental Material, sj-pdf-6-ppc-10.1177_0967391121999364 for Effective removal of Basic Red 12 dye by novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels by Nadia A Mohamed, Nouf F Al-Harby and Mawaheb S Almarshed in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
