Abstract
Biopolymers, in particular polysaccharides, have attracted considerable interest in the field of drug delivery due to their biodegradable and biocompatible nature. This study is focused on the preparation and characterization of drug delivery devices based on sodium alginate (SA) composite films with poly(sodium 4-styrenesulfonate) (PSS). The prepared composite films were characterized for the determination of physiochemical properties, molecular interactions, and drug release behavior. The possible intermolecular hydrogen bonding between SA and PSS was determined by ATR-FTIR spectroscopy. Surface characterization was done using AFM. Polymeric films consisted of pristine SA and PSS exhibited relatively uniform and flat surfaces. However, the composite films showed phase separation that became more prominent as the concentration of PSS in the composite films was increased up to 40% (w/w). The contact angle (CA) values, using deionized water as a function of time (s), were ranging from 74° to 90°, and a decrease in CA (64° to 76°) was recorded for each composite film till 40 s. These CA values revealed that all the composite films were hydrophobic. It was observed that as the concentration of PSS in the films increased, hydrophobicity slightly varied as compared to the blank films of SA and PSS. Maximum CA (89°) was shown by a composite film having SA/PSS (90/10). Ciprofloxacin hydrochloride monohydrate (CPX), a model drug, loaded in a suitable composite film (cross-linked with 0.3 M CaCl2 solution) and drug release was evaluated in pH 1.2 simulated gastric fluid (SGF) and pH 7.4 phosphate buffer saline (PBS) solution. In SGF, around 90% of the model drug was released in 110 min that was approximately 77% in the case of PBS. Therefore, it was concluded that a sustained drug release behavior was exhibited in SGF as compared to PBS solution. These results suggest that these films are a promising and may potentially be subjected to study further their drug delivery behavior in applications like wound dressing.

Keywords
Introduction
In recent years, several polymeric films have been used as a drug carrier in the field of biotechnology, agriculture, gene therapy, wound healing and controlled drug delivery applications.1-4 The applications of biodegradable films and hydrogels are emerging in the area of drug delivery and tissue engineering due to their biocompatible and non-toxic nature.5-13
Sodium alginate (SA), a natural polysaccharide, consists of a linear chain of (1,3)-α-D-guluronic acid and (1,4)-β-D-mannuronic acid. SA is usually obtained from marine brown algae. 14 It has been widely used in controlled drug delivery and pharmaceutical dosage materials due to their biocompatible, biodegradable, 13 high mechanical strength, and high hydrophilic nature. 15 It was investigated that SA based microspheres were used for drug delivery applications due to their flexibility in size, shape and their ability to entrap cells as well as biomolecules. These microspheres were proposed as a system for the controlled release of Netrin-1. 16
Since a single polymer appears suitable with respect to one property, however, it may lack the other desirable properties. Therefore, there is a trend to combine different polymers together, having distinct property, to achieve the desired results. 17 For example, a pure SA (99.99%) membrane show unsatisfactory results due to their poor flexibility and higher sensitivity toward degradation process. Such issues may be resolved by mixing, cross-linking, and modification the SA with other polymeric materials.18-21 Similarly, there are many reports of mixing SA with other polymers such as poly(ethylene oxide), 14 chitosan, 22 poly(vinyl alcohol), 23 Poly(decylviologen), 24 poly (methacrylic acid), 25 and gelatin 26 to improve its properties. Bioadhesive pills, for intraoral drug delivery using SA/chitosan mixtures, were prepared to evaluate release behavior of ketoprofen. 27 A promising drug delivery vectors based on amide functionalized alginate nanoparticles were prepared using emulsion solvent evaporation methods and sustained release of the anti-viral drug, zidovudine, was observed in PBS. 28
Poly(sodium 4-styrenesulfonate), also named as polystyrene sulfonate, is a water-soluble polymer. It has sulfonate groups and mostly used as ion-exchange resins to remove ions such as potassium, calcium, sodium from solutions in biomedical applications. 29 PSS is commonly used as potassium ions binder to cure chronic and acute kidney disease in people suffering hyperkalemia. 30 PSS may have side effects when it is blended with sorbitol, causing intestinal necrosis.31,32 PSS has also been integrated with other polymers to get good results, such as a stable and homogenous dispersion of graphene oxide could be achieved through in situ reduction/extrafoliation, using negatively charged PSS. 33 PSS and Poly(allyl amine) hydrochloride, a polyelectrolyte multilayers blend, is employed as pH-responsive drug delivery system when it is coated with hollow mesoporous silica. 34 Hydrolytically degradable films are prepared, which can be used as drug delivery and controlled released materials. These fabricated films contain 20 to 40 base layers of heparin, followed by a set of barrier layers such as PSS (a degradable polymer), Poly(diallyldimethylammonium chloride) (a non-degradable polymer), a thermally cross-linked poly(allylamine hydrochloride), and poly(acrylic acid). 35
The chemical name of ciprofloxacin hydrochloride monohydrate (CPX) is 1-cyclopropyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,4-dihydroquinoline-3-carboxylic acid hydrochloride. CPX, a concentration-dependent broad-spectrum antibacterial drug, is very effective against infections such as in eyes, nose, ear and on skin. In these cases, drug formulations such as polymeric gels or polymers e.g. lactic acid, carbopol gel, hydroxypropyl methylcellulose, and layered hydroxides, are designed for controlled drug release. CPX is mostly used in the UK for the treatment of soft tissue infections and skin infections because it is very effective against gram-positive bacteria. When other first-line therapies are failed or inappropriate for other patients, CPX is the best choice.36,37 Several studies have been reported in the literature investigating the controlled drug release from different hydrogels, films,
The objective of this work was to study the drug release behavior of SA augmented with PSS. To achieve a combination of properties, SA was mixed with PSS that ultimately demonstrated an improvement in the film formation and drug release device. The drug release behavior in the selected films was evaluated using ciprofloxacin hydrogen chloride monohydrate (CPX) as a model drug. To the best of our knowledge, there is no report in the literature presenting the controlled drug release behavior of SA and PSS composite films using CPX as a model drug.
Materials, methods and characterization
Materials
Poly(sodium 4-styrenesulfonate) (Mw 70,000 g mol−1, Sigma Aldrich, USA), Alginic acid sodium salt from brown algae (ALG, medium viscosity, Brookfield viscosity 4 to 12 cps, Sigma Aldrich, USA) and ciprofloxacin hydrochloride monohydrate was collected from Tokyo Chemical Industry Co. Ltd (TCI, Portland, OR 97203, USA). All of the other chemicals and reagents were of analytical grade and used as received without further purification.
Preparation of composite films
Sodium alginate (SA) and their composite films with poly(sodium 4-styrenesulfonate) (PSS) were prepared using solution casting/solvent evaporation technique. 14 SA/PSS composite films were prepared by mixing in different ratios (100/0, 90/10, 80/20, 70/30, 60/40) and each film was labeled as APSS-0, APSS-1, APSS-2, APSS-3, APSS-4, respectively. Specific amounts of SA were first dissolved in deionized water at room temperature using a magnetic stirrer for 2 h. PSS, in different amounts, was dissolved in deionized water at room temperature. Then aqueous solutions of PSS were added to SA solutions dropwise, and mixture were stirred for 2 h at room temperature. These blended solutions were poured on polystyrene Petri dishes (100 × 15 mm) and dried at room temperature for films formation. These composite films were kept in a vacuum desiccator for 3 days and weighed until a constant mass was obtained. Finally the prepared films were placed in airtight plastic bags and stored at room temperature until further analysis.
Attenuated total reflectance-Fourier transform infrared spectroscopy
Intermolecular interactions among polymers chains were confirmed by attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spectroscopic analysis using a spectrometer (Thermo Electron Corp., Madison, WI, USA) in transmittance mode. The spectra were recorded from 4000 to 400 cm−1 at a resolution of 4 cm−1 and 256 scans per sample.
Surface morphology
Surface morphology of the prepared films were measured by atomic force microscopy (AFM) using Nanoscope IIIa Multimode microscope (Veeco Instruments Inc., Santa Barbara, CA, USA), equipped with silicon RTESP7 cantilever (Veeco Nanoprobe, Camarillo, CA, USA), in tapping mode. A nominal spring constant of 40 N/m was used for silicon tip. All experiments were performed in air at ambient conditions, and images were recorded as 10 µm × 10 µm at scan rate of 0.5 Hz.
For AFM, the solutions of polymers were spin-coated on newly cleaved mica surface using spin coater (Laurell Technologies Corporation, USA). The spinning rate was kept around 1500 rpm for 2 min. All the prepared samples were placed in oven at 48 ± 2°C for 2 days to obtain the complete dried films. 42
Contact angle analysis
For the measurement of contact angle (θ) in degree, the dried films were attached to the microscope glass slides using double-side tape. A drop of water (8μL) was placed on each film by a micro-injector syringe (Hamilton Company, USA). Different images were recorded till 40 s using video contact angle instrument VCA optima, AST Products Inc. An automatic CA measurement, from both the right and left-hand side of the drop, was done by VCA-optima software. Triplicate measurements were performed per film which was cut from three different sites till 40 s.
Drug delivery exploration
Preparation of simulated gastric fluid (SGF) and phosphate buffer saline (PBS) solution
A pH 1.2 simulated gastric fluid (SGF) was prepared by dissolving 1 g NaCl with 4 mL of HCl and diluted up to 500 mL using deionized water. 40
Phosphate buffer saline (PBS) solution was prepared by dissolving 8 g NaCl, 0.2 g KCl, 1.44 g Na2HPO4, 0.24 g KH2PO4 in 800 mL deionized water. The pH of PBS solution was adjusted as 7.4 using 0.1 M NaOH and 0.1 M HCl solution. Finally, the solution was diluted up to 1000 mL with deionized water. 43
The pH of SGF and PBS solution was measured using pH meter (Fisher Scientific accumet, Singapore).
Method of drug loading
To evaluate the controlled drug release, 20 mg of ciprofloxacin hydrochloride (CPX) was dissolved in deionized water. The drug solution was added dropwise simultaneously after the addition of PSS in the blended solution having SA to PSS ratio 90:10 (as described in the “Preparation of composite films” section) and stirred at room temperature for the next 2 h. Then this solution was poured on the polystyrene petri dish for film formation at room temperature. The drug-loaded film was peeled off, kept in a vacuum desiccator unless a constant weight is recorded, and stored in a refrigerator for further analysis.
In vitro drug delivery measurements
The controlled drug release mechanism was studied using a smart dissolution tester (USP4, Sotax AG, CH-4123, Allschwil 1, CE 7smart, Basel, Switzerland). 44 Film was cut from six different regions for triplicate measurement in SGF and PBS solution. The average weight and average thickness of films were 0.0346 g and 63 to 93 μm, respectively. Film thickness was determined using a micrometer (Mitutoyo Corporation, model PK-0505CPX, Japan) by performing at least three to four measurements from different sites. These drug-loaded pieces of films were immersed into 0.3 M calcium chloride (a cross-linker) solution for around 3 to 5 s. 23 Then put these bits of films separately in cells above the glass beads. Each cell consisted of cylindrical portion and the filter head. The lower cone of a cell contained glass beads and these glass beads were placed above a spherical ruby stone. The dissolution medium circulated through filter head which had a filtered paper (Whatman® membrane pore size; 0.45 μm) on the top. The dissolution medium (SGF and PBS) was circulated automatically by pumping it through each cell at the rate of 5 mL/min. Conical flasks, having magnetic stirrer, filled with 500 mL of dissolution medium and temperature was maintained at 37 ± 0.2°C by placing conical flasks on hotplate with external temperature probe.45,46 After every 10 min, 5 mL solution was collected separately from each SGF and PBS solution using a pipette. Fresh 5 mL of SGF and PBS solutions were added back every time to keep the volume up to 500 mL. This procedure was repeated for the next 3 h for SGF and 2 h for PBS solution. The absorbance of collected solutions was done at λmax 277 nm for SGF and 270 nm for PBS using UV-visible spectrometer (Agilent Technologies, Cary 60, UV-vis). The standard drug solutions, 100 ppm of CPX in SGF and PBS, was taken as reference. 40
Results and discussion
Initial characterization
According to the initial test results, all-composite films were transparent and slightly yellow in color as shown in Figure 1. An average films thickness, ranging from 63 to 97 μm, was determined using a micrometer (Mitutoyo Corporation, model PK-0505CPX, Japan) by performing at least three measurements per film from different sites as shown in Table 1. The expected interactions between SA and PSS are shown in Figure 2.

Visual appearance of the prepared composite films; SA/PSS in different ratio such as 100/0 (APSS-0), 90/10 (APSS-1), 80/10 (APSS-2), 70/30 (APSS-3), 60/40 (APSS-4), 0/100 (APSS -5).
Films thickness data.


The expected interactions between the incorporated components of the prepared composite films.
Attenuated total reflectance-Fourier transform infrared spectroscopy
Attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spectroscopy is used to unfold intermolecular or intramolecular interactions, especially hydrogen bonding between functional groups in polymeric materials. 12 Commonly, intermolecular or intramolecular interactions can prompt band broadening and cause a shift in the absorption frequency of specific functionality on a polymeric materials. 47 This technique provides information about the conformational transformations that take place in compatible polymeric blends. 48 In this study, ATR-FTIR spectroscopy was employed to confirm the intermolecular interactions between sodium alginate and poly(sodium 4-styrenesulfonate). We explored the possibility of envisioned interactions of SA molecules with the functionalities on the polymeric backbone of PSS in the blend membranes.
ATR-FTIR spectra of pristine SA (APSS-0) exhibited characteristic peaks around 3650–3000 cm−1 (O–H, stretching vibrations), 2910 cm−1 (C–H, aliphatic, stretching), 1594 cm−1 (COO−, asymmetric stretching), 1405 cm−1 (COO−, symmetric), and 1025 cm−1 (C-O-C, stretching). The assigned absorption bands were used to characterize ingredients of the films. In the case of blend membranes APSS-1, APSS-2, APSS-3 and APSS-4 a broadband from 3650–3000 cm−1 was attributed to the overlapping of O–H stretching vibrations of –COOH (carboxylic acid) group and O–H (alcoholic) group. 49 Two strong peaks at 1594 and 1405 cm−1 were corresponded to the asymmetrical and symmetrical stretching vibrations of the carbonyl groups (–C=O), respectively.28,40,50 The bands at 2910 cm−1 and 2853 cm−1, corresponded to CH2 stretching vibrations. A peak at 681 cm−1 attributed to alkyl groups in the polymeric chain, which was not observed in the case of pristine SA. These findings indicated the existence of blend constituents. 51 A strong asymmetric stretching vibration absorption band centered at 1204 cm−1 was due to the O=S=O unit of PSS. The phenyl ring substituted with a sulfonic group vibrations displayed the peak at 1003 cm−1. A peak, located at 1126 cm−1, exhibited broadening and overlapping with the phenyl ring attached to the sulfonic anion which indicated the deprotonation of the sulfonic acid peak in APSS-1, APSS-2, APSS-3 and APSS-4. Furthermore, the vibrational peaks at 1160 cm−1 and 1040 cm−1 was assigned to sulfonate groups in case of PSS film (APSS-5). We observed shift of these peaks from 1160 cm−1 to 1180 cm−1 and 1040 cm−1 to 1033 cm−1 in composite films. We inferred that these characteristic shifts were prominent as the %age of SA (w/w) was increased, (Figure 3). This trend confirmed the hydrogen bond interactions between sulfonate group of PSS and –OH group of SA in the composite films. 52

ATR-FTIR study of SA, PSS and their composite films.
Surface morphology
Atomic force microscopy (AFM) is an excellent tool to explore the surface morphology and texture of the films. 1 AFM micro-images of the SA, PSS, and their composites films demonstrated (Figure 4) that the surface morphology varied significantly with PSS contents. Composite films with SA/PSS composition 100/0 (APSS-0) and 0/100 (APSS-5) exhibited relatively uniform and flat surfaces. On the contrary, composite films exhibited phase separation. There was a micro-separation in case of APSS-2. This separation was more prominent in the films having 40% (w/w) PSS (APSS-4). 14 These results were in good agreement with contact angle measurements.

Microscopic images of composite films having different SA/PSS compositions such as 100/0 (APSS-0), 80/10 (APSS-2), 60/40 (APSS -4), 0/100 (APSS-5).
Contact angle analysis
Contact angle (CA) measurement is a convenient way to determine hydrophilic or hydrophobic nature and the wettability of a surface/material. CA values depends on surface roughness, heterogeneity, and porosity.1,53-55 CA were recorded using deionized water drops on the SA, PSS, and their composite films at different times ranged from 0 to 40 s 56 as shown in Table 2. Generally, it was assumed that high contact angle (θ > 65°) exhibited hydrophobic surfaces while low contact angle (θ < 65°) related to hydrophilic surfaces. 57 In case of hydrophilic surfaces, the liquid droplets spread out completely on the surfaces over time. 58 It was observed that the CA of each film were decreased with the time (Figure 5). 59 The initial CA (0 s) of composite films, SA/PSS compositions such as 100/0 (APSS-0), 90/10 (APSS-1), 80/10 (APSS-2), 70/30 (APSS-3), 60/40 (APSS-4), 0/100 (APSS -5), were 83°, 89°, 84°, 74°, 78° and 90°, respectively. While the final contact angles (40 seconds) of APSS-0 (76°), APSS-1 (74°), APSS-2 (73°), APSS-3 (68°), APSS-4 (64°) and APSS-5 (75°) were recorded. Initial CA results revealed that as the concentration of PSS in blend films increased, the hydrophobicity slightly increased in case of APSS-1 and APSS-2 while decreased in APSS-3 and APSS-4 due to more chances of phase separation. The CA measurements were restricted to 40 s because after 40 s the water drops lost their symmetrical patterns.
Contact angle analysis data of composite films as a function of time having different SA/PSS compositions such as 100/0 (APSS-0), 80/10 (APSS-2), 60/40 (APSS -4), 0/100 (APSS -5).

*SD means standard deviation.
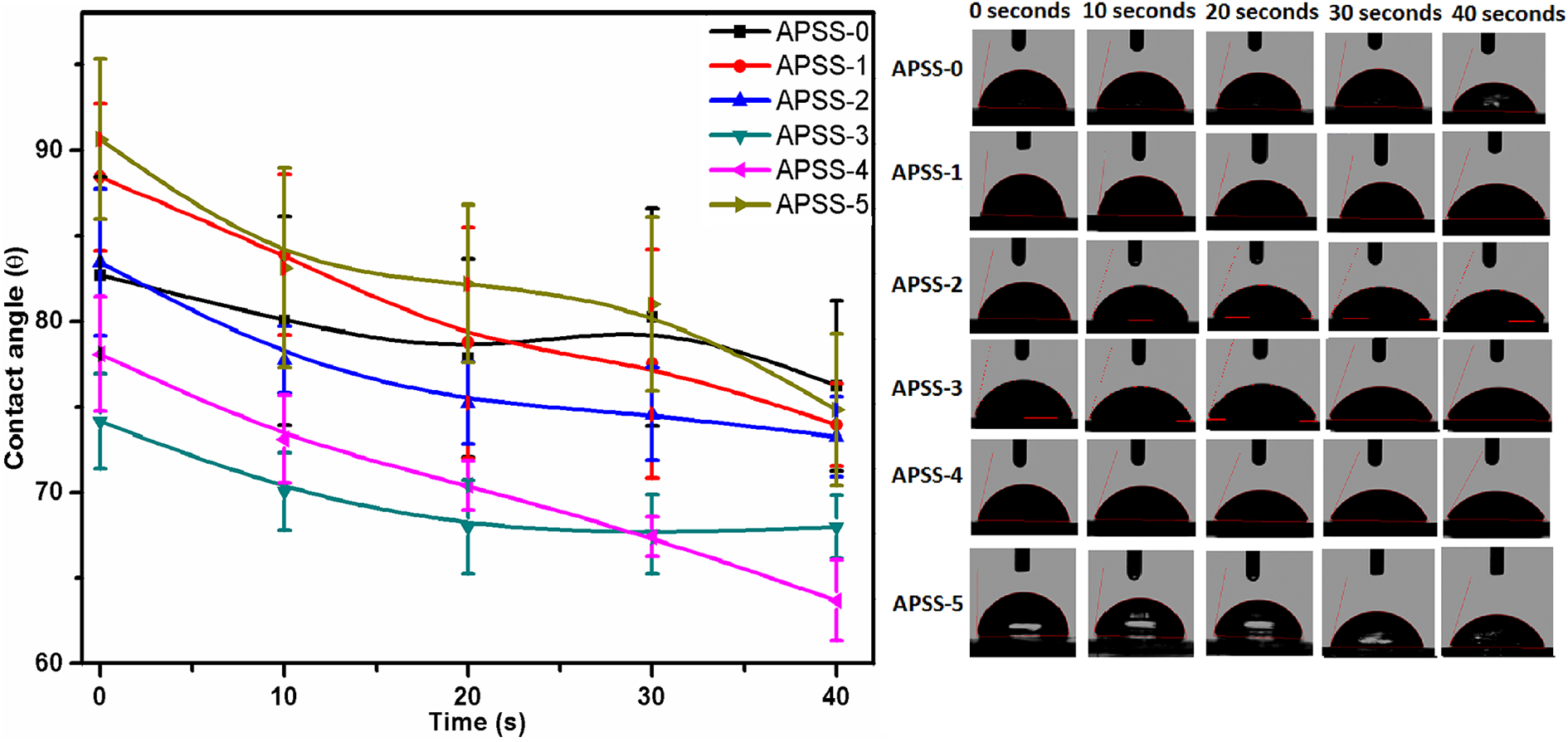
Contact angle analysis of composite films as a function of time having different SA/PSS compositions such as 100/0 (APSS-0), 80/10 (APSS-2), 60/40 (APSS -4), 0/100 (APSS-5).
Drug release analysis
Ciprofloxacin hydrochloride monohydrate (CPX), a model drug, was loaded in a suitable composite film (APSS-1) and drug release was evaluated in pH 1.2 simulated gastric fluid (SGF) and pH 7.4 phosphate buffer saline (PBS) solution. Drug release behavior from APSS-1 was studied as a function of time as shown in Figure 6. APSS-1 film was selected for drug delivery because this film was found to have high surface hydrophobicity (89º) as compared to the other composite films as confirmed by CA analysis (“Contact angle analysis” section). It was observed that around 90% of drug in 110 min was released in a controlled manner in SGF (Table 3). While in PBS solution, most of the drug was released in half an hour, i.e. 77%. Hence, it was decided, from the graph, that the drug was released in a sustained manner in SGF as compared to PBS solution. The remaining amount of drug might not be determined because film was broken into fragments after 180 min in SGF and 80 min in PBS solution. It is evident from Figure 6 that these films may be used for drug delivery applications like wound dressing.

Drug release (%) in SGF and PBS solution containing film loaded with drug having SA/PSS composition 90/10.
Drug release (%) data in SGF and PBS solution containing film loaded with drug having SA/PSS composition 90/10.

Conclusions
Biodegradable polymeric composite films based on SA/PSS for drug delivery applications were developed by solution casting/solvent evaporation technique. Composite films with SA/PSS composition 100/0 (APSS-0) and 0/100 (APSS-5) exhibited relatively uniform and flat surfaces as compared to composite films. From ATR-FTIR, hydrogen bond interactions between sulfonate group of PSS and –OH group of SA in the composite films was confirmed due to the shift of two peaks from 1160 cm−1 to 1180 cm−1 and 1040 cm−1 to 1033 cm−1. These characteristic shifts were prominent in those films having higher %age of SA (w/w). AFM results indicated that composite films exhibited phase separation which increases as the amount of PSS increases in the films. The contact angles (CA) studies showed that all the films were more hydrophobic in nature. SA/PSS composite films displayed an increase in CA compared to pristine SA. Composite film having 10% (w/w) PSS showed higher value (89º) of initial CA as compared to other composite films. CA of all films decreased over the course of 40 s. The drug release study showed that around 90% of the drug in SGF was released in 110 min while an abrupt release of the drug was observed in PBS i.e. 77% in 30 min. Therefore, a sustained drug release was observed for the composite film APSS-1 in SGF as compared to PBS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
