Abstract
In this work, we compare how the properties of polycaprolactone (PCLO) nanocomposites are affected by the method of nanocomposite synthesis and the differences in graphene oxide (GO) properties. PCLO nanocomposites with freeze-dried or hot-dried graphene oxide (FGO/HGO) were synthesized by
Introduction
Polycaprolactone (PCLO) is an aliphatic polyester, whose biodegradability made it popular for biomedical applications. 1 Specifically, many PCLO-based products were commercialized for tissue engineering such as nerve guidance conduits, the monofilament sutures or the root canal filling materials.2–4 However, further PCLO application potential is limited by its poor mechanical performance. This deficiency can be suppressed by addition of various nanofillers, which opens new possibilities in biomedicine and beyond. Recently, graphene-based nanofillers were reported to improve not only the mechanical properties of various polymers 5 but also several biomedical properties of PCLO.6–8 A combination of good biomedical and mechanical properties is promising for future medical applications. However, it is important to balance the individual properties, which are heavily influenced by synthesis procedure.
Two commonly used procedures to synthesize PCLO nanocomposites are
The major aim of this work is to compare the properties of PCLO/GO nanocomposites prepared by
Experimental
Materials
Graphite (325 mesh, GK Graphite Týn CR 12 995; Czech Republic). Sulfuric acid (96%, Penta s. r.o.; Czech Republic). Sodium nitrate, potassium permanganate (Lachema; Czech Republic). Hydrogen peroxide (30%, Penta s. r.o.; Czech Republic). ε-Caprolactone (97%, Sigma-Aldrich) was dried over powdered calcium hydride (Sigma Aldrich) and distilled under reduced pressure (40 Pa), water content determined by Karl-Fischer titration was less than 30 ppm. Stannous octoate (95%, Sigma-Aldrich). Toluene and methanol (p.a., Sigma-Aldrich).
Synthesis of the graphene oxide
The GO was synthesized using the modified Hummers method according to the work of Rajaura et al. 15 The main steps of GO synthesis are discussed in Electronic Supporting Information (ESI). The obtained dispersion was then dried by freeze-drying or hot-drying. During the freeze-drying, the water was sublimed from the frozen sample (−70°C) at reduced pressure (50 Pa). In case of hot drying, Petri dishes with dispersion were placed in a vacuum oven (60°C) and the water was evaporated at reduced pressure (800 Pa). Both GOs were ground in the knife mill. The freeze-dried GO will be further denoted as FGO, whereas the hot-dried GO as HGO.
Synthesis of the nanocomposites
The PCLO nanocomposites were prepared by
All steps of the
The conditions for the synthesis of PCLO matrix used for the melt mixing were analogous to the
Characterization
The elemental composition of GOs was determined by combustion analysis using a Vario EL Cube (Elementar, Germany) and by X-ray photoelectron spectroscopy (XPS) on an ESCAProbeP (Omicron Nanotechnology Ltd.; Germany). The ESCAProbeP used an aluminium anode with energy 1486.7 eV as a source of monochromatic light. The data were analysed with CASA XPS software using Shirley background subtraction.
The interlayer distance of GO layers and the level of GO dispersion in polymer matrix were measured by X-ray diffraction (XRD) on an X´Pert
3
Powder diffractometer (PANanalytical; United Kingdom) with Bragg-Brentano parafocusing geometry and a CuKα tube providing wavelength of λ = 0.154 nm. Peak profiles were obtained using HighScore 4.0 software (PANAlytical). The calculations of GO interlayer distance were based on Bragg equation (1) (
The surface area of graphite and GOs was measured using a Coulter SA 3100 sorption analyzer, (Beckman Coulter). Before the sorption experiments, samples were degassed for 4 h at 95°C under high vacuum. Detector cooled by liquid nitrogen (−196°C) was used to evaluate the results using Kelvin and Brunauer, Emmett and Teller equations, respectively.
To obtain transmission electron microscopy (TEM) images, the powder nanofillers were ground in an agate mortar and dispersed in isopropanol. Then, 7 μl of the dispersion was dripped on a copper grid (Ted Pella Inc. mesh 300, lacey carbon) and isopropanol was evaporated. The measurements were carried out on an EFTEM 2200 FS (Jeol, Ltd.; Japan) operating at an accelerating voltage of 200 kV.
The polymer yield of nanocomposites was determined gravimetrically after extraction of low-molecular compounds, performed three times for 30 min in methanol (50 mL per 0.5 g of sample). To calculate the polymer yield, the equation (2) was used (
The average values of molar masses were determined using a chromatographic system Breeze (Waters; USA) with a refractive index detector and multi-angle laser light scattering detector miniDawn TREOS (Wyatt; USA). The separation was performed on a PSS Lux LIN M 5 μm column (7.8 × 300 mm) at 35°C in tetrahydrofuran at an elution rate of 1 mL/min using polystyrene standards for calibration.
To prepare the specimens for tensile tests and dynamic mechanical analysis (DMA), the nanocomposites were ground to chips, which were hot pressed in a mould with dimensions 60 × 50 × 0.7 mm using this procedure: (I) 5 min preheating, (II) 5 min hot-pressing at 75°C (degassing after 1 min), (III) 5 min cooling. Finally, dog bone specimens were cut from the pressed foil (distance between shoulders – 20 mm, width – 4 mm, thickness – 0.7 mm) and stored in a desiccator before the measurement. Dynamic mechanical behaviour of nanocomposites was studied using DMA DMX04T instrument (RMI; Czech Republic). The measurements were realized in bending mode in the temperature range: –110–50°C at a heating rate of 2 °C/min, frequency 1 Hz and force amplitude 500 mN. The tensile tests were carried out on an Instron 3365 (Instron; USA) at a rate of 1 mm/min until the proportional elongation exceeded 1%, then the rate was increased to 50 mm/min.
The crystalline properties of nanocomposites were obtained using differential scanning calorimeter DSC Q100 (TA Instruments; USA). The measurements were carried out in a 0–100°C range (heating – cooling – heating) at rate of 10 °C/min. To calculate the crystalline content of nanocomposites, the equation (3) was used
17
(
The optical micrographs of nanocomposites were obtained using a Leitz DMRXE microscope equipped with objective HC PL Fluotar (10x/0.3 PH1). Nanocomposite chips on microscopy slides were placed on Linkamcell Scientific LTS 350 plate that was heated to 80°C.
Scanning electron microscopy was carried out on TESCAN Vega 3 LMU apparatus in Univac mode.
Results and discussion
The synthesis of graphene oxide
The chemical composition of freeze dried graphene oxide and hot dried graphene oxide.

aOxygen content was calculated with the assumption that material does not contain any other element.

X-ray photoelectron spectroscopy survey spectra (lefthand side) and deconvoluted C1s spectra (righthand side) of FGO (top) and HGO (bottom).
The interlayer distances and surface areas of freeze dried graphene oxide and hot dried graphene oxide.

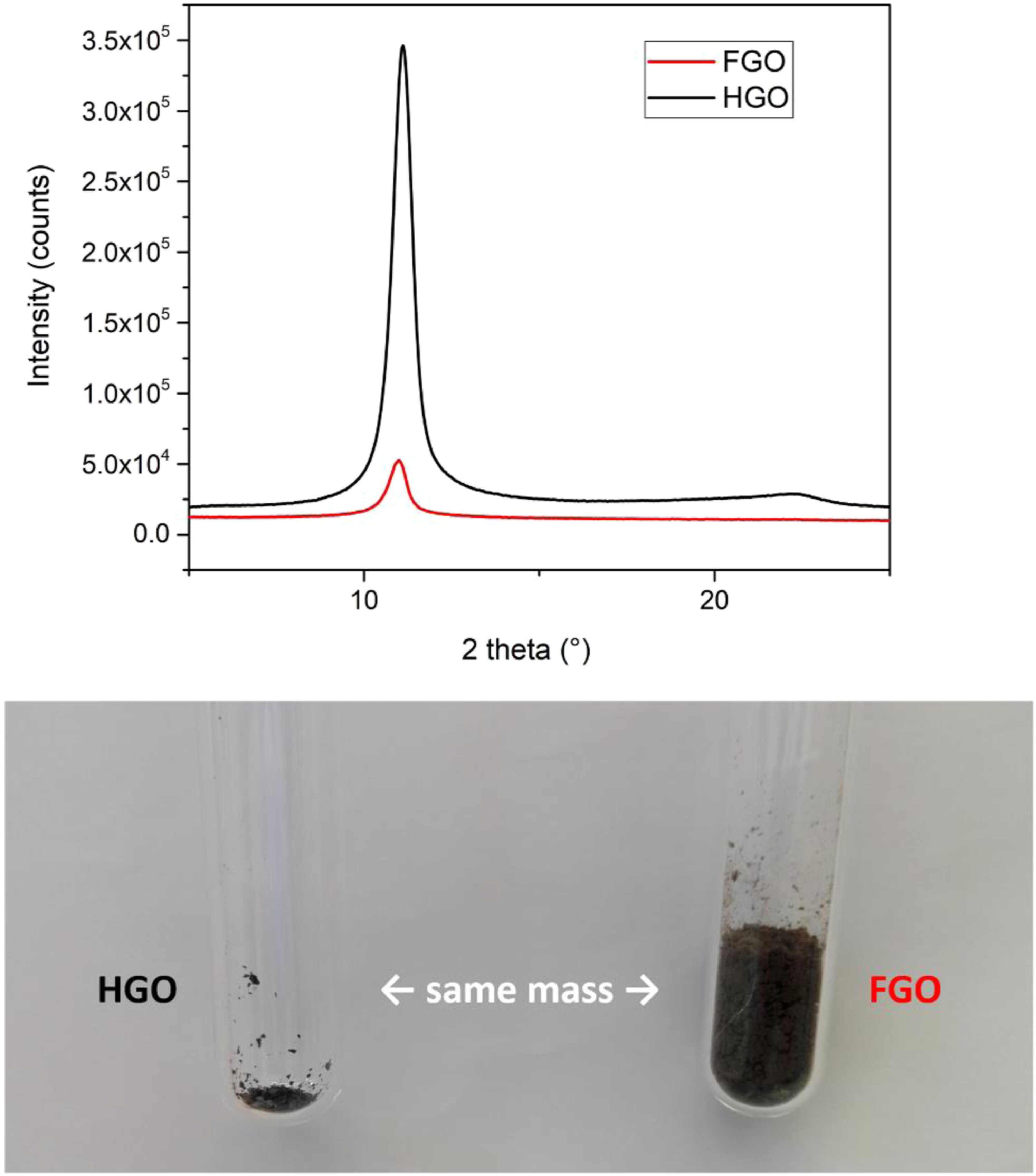
The X-ray diffraction patterns of FGO and HGO (top), the comparison of bulk densities of FGO and HGO (bottom). FGO: Freeze dried graphene oxide; HGO: Hot dried graphene oxide.
Sorption analysis proved that freeze-drying slightly increased the surface area of the product, whereas hot-drying decreased the surface area by an order of magnitude (Table 2). It is noteworthy that while the GOs significantly differ in their surface areas, their interlayer distances are similar. This discrepancy was resolved via TEM that indicate the presence of single sheets and multiple layers in both GOs (Figure 3). However, FGO micrographs show mostly flat surfaces with low level of folding, while HGO sheets exhibit much more folding and wrinkling. Such distortion of HGO can strongly decrease the accessibility of its surface to nitrogen atoms used in sorption analysis which can explain the difference in surface areas. We also believe that the difference in surface area is responsible for the extremely high bulk density of FGO compared to HGO (Figure 2). The transmission electron microscopy images. (a) FGO single sheet, (b) FGO multiple sheets, (c) HGO single sheet and (d) HGO multiple sheets. FGO: Freeze dried graphene oxide; HGO: Hot dried graphene oxide.
The synthesis of nanocomposites
Molar masses of nanocomposites
Polymer yield, molar masses and dispersity of polymer matrix from polycaprolactone/graphene oxide nanocomposites.

acontent in polymerization mixture (
bweight average molecular weight obtained from SEC.
cnumber average molecular weight obtained from SEC.
ddispersity
emolar masses of virgin PCLO before melt mixing.
molar masses of virgin PCLO after melt mixing.
gNA = material not available.
Thermal properties of nanocomposites
Thermal properties of nanocomposites.
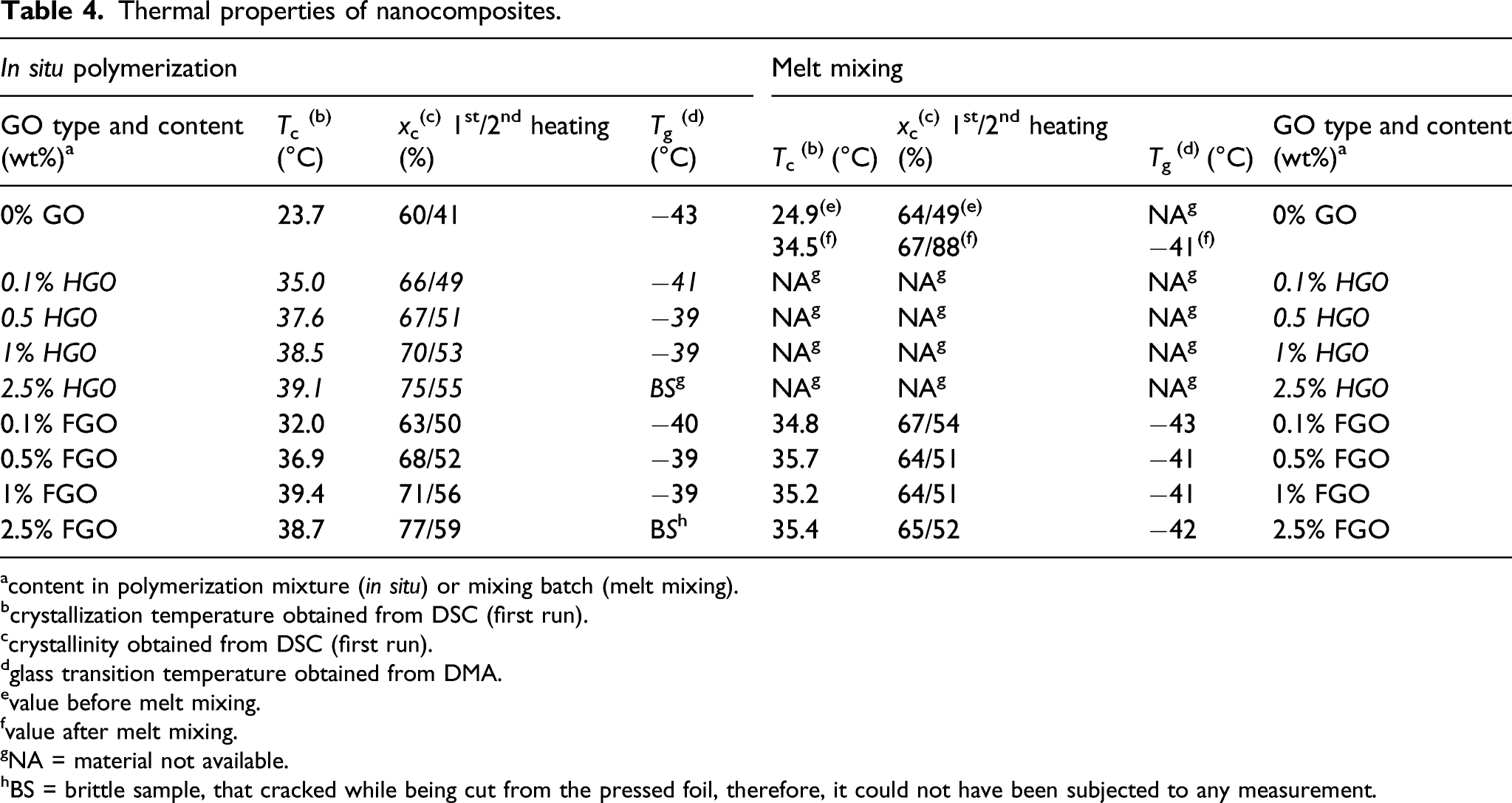
acontent in polymerization mixture (
bcrystallization temperature obtained from DSC (first run).
ccrystallinity obtained from DSC (first run).
dglass transition temperature obtained from DMA.
evalue before melt mixing.
fvalue after melt mixing.
gNA = material not available.
hBS = brittle sample, that cracked while being cut from the pressed foil, therefore, it could not have been subjected to any measurement.
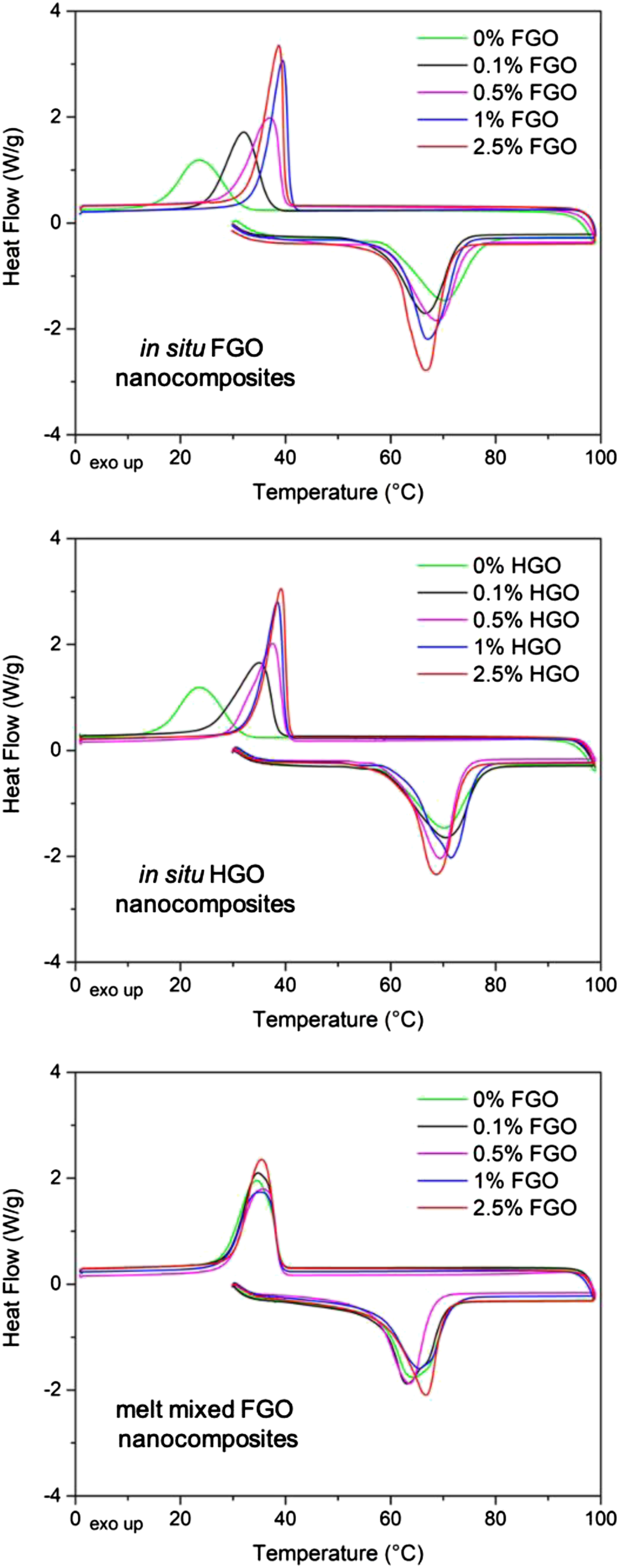
Differential scanning calorimeter curves of polycaprolactone nanocomposites.
Second, dynamic mechanical analysis provided an additional information about the polymer-nanofiller interactions, because nanofillers can restrain the mobility of polymer chains resulting in an increased glass transition temperature (
Morphology of nanocomposites
X-ray diffraction pattern of melt mixed nanocomposite with 2.5 wt% of FGO shows a peak at 12° that can be ascribed to FGO aggregate with a slightly reduced interlayer distance (Figure 5, detailed explanation in ESI). Nevertheless, X-ray diffraction patterns of virgin polycaprolactone, melt mixed (MM) and in situ polymerized (IS) nanocomposites with 2.5 wt% of freeze dried graphene oxide and hot dried graphene oxide; insets show the areas in range of x: 10–20° and y: 0–20,000 counts.
The optical microscopy indicates that although the agglomeration of GO sheets appear to some extent in all samples, Optical micrographs of polycaprolactone/graphene oxide nanocomposites; red scale bar – 100 μm.
Mechanical properties
The choice of the synthesis method and the type and concentration of GO considerably influenced the mechanical properties of the nanocomposites (Figure 7, Table 5, Figure ESI 6 and 7). In case of the Typical stress-strain curves of polycaprolactone nanocomposites. Mechanical properties of polycaprolactone/graphene oxide nanocomposites. acontent in polymerization mixture ( bYoung modulus obtained from tensile test. celongation at break obtained from tensile test. dcomplex modulus obtained from DMA. evalue after melt mixing. fNA = material not available. gBS = brittle sample, that cracked while being cut from the pressed foil, therefore, it could not have been subjected to any measurement.

To assess the influence of different variables on mechanical properties of the PCLO nanocomposites, it is crucial to take into account their thermal and morphological properties. Optical microscopy and XRD suggest that
However, the overall discussion on mechanical properties should not omit the effects of molar masses and crystallinity. We can see a notable difference between mechanical properties of unfilled samples. The Young modulus of unfilled melt mixed sample exceeded the modulus of its
Conclusion
We suggest that the
Supplemental Material
sj-pdf-1-ppc-10.1177_09673911211068923 – Supplemental Material for The effect of nanocomposite synthesis and the drying procedure of graphene oxide dispersion on the polycaprolactone/graphene oxide nanocomposite properties
Supplemental Material, sj-pdf-1-ppc-10.1177_09673911211068923 for The effect of nanocomposite synthesis and the drying procedure of graphene oxide dispersion on the polycaprolactone/graphene oxide nanocomposite properties by Jaroslav Minář, Jan Doležal and Jiří Brožek in Polymers and Polymer Composites
Footnotes
Acknowledgements
The authors would like to thank François Tournilhac from ESPCI Paris for optical microscopy of nanocomposites and Jiří Minář for proofreading.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the specific university research (MSMT No 21-SVV/2019).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
