Abstract
In recent years, polymeric hydrogels are widespread in the field of biological materials such as wound dressing and wound care. In this work, we report for the first time the preparation and application of pHEMA nanocomposite hydrogels containing iron oxide nanoparticles as wound dressings. For this purpose, nanocomposite hydrogels based on poly (hydroxyl ethyl methacrylate) (pHEMA) and various amounts of 5, 10, and 15 wt% iron oxide nanoparticles were successfully prepared via radical polymerization. The structure and morphology of nanocomposite hydrogels were determined by Fourier transform infrared spectroscopy (FTIR) and field emission scanning electron microscope (FE-SEM), respectively. The results of gel fraction and the degree of swelling of hydrogels demonstrated that the gel percentage of pHEMA increased, and the degree of swelling decreased with increasing the percentage of nanoparticles. The WVRT and the porosity of hydrogels decreased by increasing the quantity of nanoparticles and were suitable for wound dressing applications. The effect of iron oxide nanoparticles on the mechanical properties of nanocomposite hydrogels was also studied using compression test and hardness shore A durometer. The results indicated that the compression strength, modulus, strain, and hardness are steadily increasing compared to pure hydrogel by adding nanoparticles. The maximum increase was obtained for a hydrogel sample with 15 wt% iron oxide nanoparticles. Antibacterial properties and biocompatibility were determined by the disk-diffusion and MTT assay methods, respectively. Based on the results, nanocomposite hydrogels exhibited higher percentages of cell survival and better antibacterial properties compared to pure pHEMA.
Keywords
Introduction
Hydrogels are three-dimensional polymeric networks with a high capacity to absorb water and biological fluids. They can absorb water up to 1000 times their mass without dissolution. Compared to other synthetic polymers, hydrogels are more similar to living tissues in terms of structure and chemical or physical properties such as wettability, high elasticity, and good biocompatibility.1–3 In general, hydrogels are cross-linked in both chemical and physical ways. 4 High absorption capability of hydrogels is related to the presence of hydrophilic functions in the polymer chain including hydroxyl, amine, amide, carboxylic, and sulfonic groups, while their resistance to dissolution is due to cross-linking agents between chains. 1 The history of hydrogels goes back to 1960 when Wichterle and Lim 5 proposed the use of poly (2-hydroxyethyl methacrylate) (pHEMA) as a hydrophilic hydrogel in contact lenses. Subsequently, the technology of different forms of hydrogels6,7 was developed in various industries including food packaging, 8 pharmaceuticals, 9 sensors, 10 agriculture, 11 electronic devices, 12 tissue engineering, 13 drug delivery systems, 14 wound dressing, 15 artificial skin, 16 scaffolds, 17 cell separation and adsorbents for the removal of pollutants.18,19 The success of a biomaterial depends primarily on its biocompatibility and non-toxicity.15,20,21 Many factors affect the biocompatibility of material, and available sources indicate that pHEMA hydrogels are biocompatible materials.22–24 It is noteworthy that the surface tension between the hydrogels and biological tissue is very low, which reduces protein absorption and cell adhesion. Due to the high-water content of these materials, the hydrogel surface is known as a hydrophilic permeable surface that has high biocompatibility. Despite the benefits of hydrogels, the mechanical properties of most hydrogels are low (especially after swelling), 3 and this is important for the use of hydrogels as a wound dressing. A wound dressing with higher mechanical properties can cover the wound area and reduce the penetration of the environmental microbe into the wound. Nanoparticles such as nano-silver, 25 nano-clay, 26 nano-silica, 27 nano-gold, 28 nano-iron oxide, 29 nano-zinc oxide,20,30 and nano-chitosan15,21 are commonly used to improve the mechanical properties and performance of the hydrogels. The swelling and biological activity properties of silica gel/pHEMA nanocomposites were investigated by Costantini et al. 22 They also used 30 wt% silver nanoparticles and improved the swelling behavior and biological activity compared to pHEMA. Xiang et al. 31 prepared nanocomposite hydrogels of silver/poly (HEMA-PEGMA-MAA). Their results showed that nanocomposite hydrogels not only have a much higher swelling ratio and faster deflation rate but also have good adjustable electrical properties compared to pure hydrogels. Liu et al. 23 successfully synthesized highly ordered nanostructures consisting of Cu (0) nanoparticles in the pHEMA hydrogel via situ optical polymerization and situ chemical recovery. They stated that these compounds had a negative surface charge and were more biocompatible than pure pHEMA hydrogel. According to their reports, Cu (0)/pHEMA compounds are a good choice for thromboresistant applications. Researches have also been carried out on the thermal stability, biodegradability, and swelling of pHEMA hydrogels containing clay and kaolin nanoparticles.32,33 Resmi et al. 34 synthesized and identified gelatin-hydroxypropyl methacrylate hydrogels containing silver nanoparticles and evaluated their applications for wound dressing. They showed that this hydrogel is a copolymer with mechanical stability, moisture retention, and good antibacterial properties. In the last decade, nanocomposite hydrogels, including iron oxide nanoparticles, have been used in medical and biomaterial applications.35–37 Iron nanoparticles have attracted the attention of scientists in biotechnology due to their low toxicity, biocompatibility, low cost, size, conductivity, ease of separation, and detection. 38 These studies attest to the fact that pHEMA hydrogels and iron oxide nanoparticles are suitable choices for preparing a wound dressing. In this work, nanocomposite hydrogels based on pHEMA and iron oxide nanoparticles were presented as new wound dressings, and their antibacterial and mechanical properties were also investigated.
Experimental
Materials
2-hydroxyethyl methacrylate (HEMA contained >250 ppm mono-methyl hydroquinone ether as an inhibitor) as a monomer with a liquid density of 1.07 g/cm3 (at 25°C), ethylene glycol di-methacrylate (EGDMA) as cross-linking additive with a density of 1.05 g/cm3 and molecular weight of 198.22 g/mol, and 2, 4, 6-trimethyl benzoyl-di-phenyl-phosphine oxide (TPO) as an initiator, were obtained from Sigma-Aldrich Co. Iron oxide nanoparticles with a particle size of 20–40 nm was purchased from Research Nanomaterials Co.
Preparation of nanocomposite hydrogels
To prepare the pure hydrogel, 7 mL HEMA monomer was added to 2 mL deionized water and 10 mL EGDMA in a clean test tube equipped with a nitrogen inlet and magnetic stirrer (800 r/min) under room temperature. After 15 min, the initiator (TPO) was added to the mixture and stirred for 5 min. To complete the gelation process, the mixture was exposed to ultraviolet light for 40 min. The HEMA monomer was mixed with different weight percentages of iron oxide nanoparticles (5, 10, and 15 wt %) and deionized water (for 1 h, 200 r/min), and EGDMA was added to prepare the nanocomposite hydrogel. The preparation process continued, as explained for pure hydrogel.
Characterization
Fourier-transform infrared spectra (FTIR)
The interaction between pHEMA and nano iron oxide particles was investigated by Fourier-transform infrared spectra (FTIR, Tensor-27, Bruker Co, USA) in the range of 400–4000 cm−1.
Morphology
A field of emission scanning electron microscope (FE-SEM, MIRA3-TESCAN, Czech) equipped with an energy dispersive X-Ray analyzer (EDAX) was used to observe the morphology and distribution of nanoparticles in hydrogels. The samples were coated with a thin layer of gold to obtain higher resolution images.
Mechanical properties
Mechanical properties of nanocomposite hydrogels, including compression strength, compression strain, and modulus, were determined with a Teclock tester (Teclock Co, Japan) at room temperature. The test performed was based on the ASTM D695-02a, and the samples were prepared with diameter and height of 19 mm and 38 mm, respectively. The hardness of samples was determined using Shore A durometer made by Teclock Co. Japan. Regular samples were cut according to standard ASTM D-2240-95 with a thickness of 5–6 mm. The tests were carried out at 25°C, and the indention force was recorded 15 s after the pressing probe touched the specimen. These mechanical tests (compression and hardness) were repeated three times, and the average value of tests was finally reported for each sample.
Degree of swelling
Water absorption of the hydrogels is one of the most crucial parameters in applications of hydrogels as wound dressing, which is determined using the swelling test. The degree of swelling of the samples was obtained by measuring the initial and swollen weights. The prepared samples were immersed in double-distilled water for 48 h at room temperature. Then the swollen samples were brought out of the water to the surface and were dried as well as weighed. These samples were later dried in a vacuum oven at 60°C and were reweighed (see equation (1))
Gel fraction
Three pieces of the pre-weighed samples were dried under vacuum at room temperature to check for changes in weight. The samples were soaked in water for 4 days and then were extracted from water and were dried under vacuum at room temperature until the weight of the samples became fixed. The gel fraction of the hydrogels was calculated using the equation
Water vapor transmission rate
A standard method was used to measure the water vapor transmission rates (WVTR) of the hydrogels.
39
The samples were prepared with a diameter of 40 mm and a thickness of 3 mm. The hydrogel sample was placed like a cap on the mouth of a test tube with a diameter of 35 mm containing 25 mL of water. The test tube was transferred to an adjustable temperature-humidity camber (Giant Force Instrument, GTH-072TR, Taiwan) with a temperature of 35°C and humidity of 35% for 24 h. The water vapor transmission rate was calculated according to the following equation
39
Porosity measurement
The samples were prepared in a cylindrical shape, and their dimensions were measured to calculate the volume. The samples were placed in ethanol until saturated, and the weights of samples were measured before and after saturation. Porosity was determined using the equation
40
Biocompatibility
MTT powder was prepared in PBS at a concentration of 5 mg/mL and kept at 4°C in the darkness. Samples with various codes were placed on a sterilized 24-well plate and rinsed with 70% ethanol three times. Each time the samples were exposed to ultraviolet radiation for half an hour after washing with ethanol. After sterilization of the samples, human Caucasian fetal foreskin fibroblast cells (HFFF2) (obtained from the Pasteur Institute of Iran) were used to investigate the cell behaviors and their biological evaluations. Cells with a concentration of 1×104 cell/well were cultured in 24-well plates containing the test specimens and were stored in the incubator for 24 and 72 h at 37°C and 90% humidity (one plate for 24 h and another plate for 72 h). In the next step, the cell culture medium was extracted, and 100 mL culture medium containing 10% MTT solution was added to each well. The prepared sample was incubated for 4 h in the dark at 37°C until blue formazan crystals were visible. The MTT medium was removed, and the blue crystals were dissolved in 100 mL isopropanol. After 15 min the existed solvent in each well was pipetting and was read by Elisa reader at 570 nm. The percentage of cell survival was calculated using the equation
Antibacterial properties (microbe penetration)
The microbe penetration test was performed to prove the resistance of nanocomposite hydrogels to bacteria. The antibacterial properties of the prepared hydrogels were evaluated using the bacteria Escherchia coli (E.coli, Gram-negative) and Staphylococcus aureus (S. aureus, Gram-positive). For this purpose, 1.5 × 108 CFU/mL of half-McFarland (CFU/mL) of selected bacteria as a suspension was cultured on Luria broth (LB) agar medium. The samples were placed into the plates and were incubated for 48 h at 37°C. The antibacterial properties were investigated as an inhibition zone of bacteria around the samples.
Results and discussion
Fourier-transform infrared spectra (FTIR)
Figure 1 shows the FTIR spectra of synthesized hydrogel (pHEMA) and nanocomposite hydrogel (pHEMA+15% nano Iron). The broad peak in the range of 3200–3600 cm−1 was ascribed to the hydroxyl group stretching vibrations of the hydrogels. Stretching vibrations of C-H were observed at 2925 cm−1. The peaks at 1437 cm−1 and 1474 cm−1 indicated the scissoring vibrations and bending of CH2 groups. C=O stretching vibration bond appeared at 1735 cm−1. The peaks at 1175 and 1087 cm−1 were also assigned to the stretching vibration of C-O.41,42 The characteristic peaks of nano iron oxide appeared at 3407, 1623, and 586 cm−1
43
that were covered by stronger peaks of hydrogel (see Figure 1(b)). These results show that the hydrogel structure is formed in samples. Also, the peaks of nanocomposite hydrogel (pHEM+15% Nano) are stronger than that of pure hydrogel (pHEMA). It seems that the presence of nano iron oxide increased the number of functional groups (per unit volume) in the molecular structure of nanocomposite hydrogel, indicating a high density of polymeric chains per unit volume of sample, high chain entanglements, high gel concentration, and good interactions between nanoparticles and chemical structure of the polymer. The Fourier transform infrared spectroscopy (FTIR) spectra of (a) pure pHEMA and (b) pHEMA containing 15 wt% of iron oxide nanoparticles.
Morphology of nanocomposite hydrogels (FE-SEM and EDAX)
The FE-SEM images of the pure hydrogel (pHEMA) and nanocomposite hydrogels containing 5, 10, and 15 wt% iron nanoparticles are shown in Figure 2. Despite the few clusters of iron oxide nanoparticles in images (Figure 2(b)–(d)), good dispersion and distribution of the nanoparticles were observed, which can be attributed to the suitability of the preparation method. As can be seen, the pure hydrogel has a porous structure and is free of nanoparticles (Figure 2(a)). The porosity of the hydrogel has an important role in wound dressing application. Having proper porosity can affect the wound healing rate due to the diffusion of oxygen.
40
The porosity of the nanocomposites decreased by increasing the nanoparticles from 5 to 15%. These results are in agreement with that of the porosity test (see Table 1). The FE-SEM images of (a) pure pHEMA and nanocomposite hydrogel containing (b) 5, (c) 10, (d) 15 wt% of iron oxide nanoparticle. WVTR and porosity of hydrogels containing 0, 5, 10, and 15% wt nano iron oxide.

The qualitative presence of the Fe element was evaluated via EDAX (Figure 3). It can be seen, pHEMA is free of iron oxide nanoparticles, and carbon, nitrogen, and oxygen were observed for this hydrogel. For nanocomposite samples, the Fe element was observed on the surface. As seen, the percentage of Fe element on the surface of the samples increases by adding the nanoparticles into pHEMA, which is visible by the increase in peak intensity in the EDAX results. EDAX results of (a) pure pHEMA and its nanocomposite hydrogels containing (b) 5, (c) 10, and (d) 15 wt% of iron oxide nanoparticles.
Gel fraction and degree of swelling of hydrogels
The dependency of the gel fraction and degree of swelling on the amount of nano iron oxide are shown in Figure 4. Addition of 5, 10, and 15% iron oxide nanoparticles to pHEMA increased its gel percentage (from 75% for pure hydrogel to 88% for nanocomposite hydrogel containing 15% nano iron oxide), and a direct relationship between the gel percentage and amount of nanoparticles was evident. The nanoparticles embedded in the network of hydrogel samples act as cross-linking points and enhance the entangled structure and gel density of hydrogels. It can be expected that higher gel density or gel fraction improves the mechanical properties of hydrogels due to stronger interactions between the polymer chains (see results of mechanical properties). FE-SEM images (Figure 2) clearly showed that the textures of hydrogel samples containing nanoparticles were finer than the pure hydrogel, which indicates a higher gel fraction with increasing of cross-links. The swelling percentage of hydrogels is directly proportional to the amount of water absorbed and is an important feature of the hydrogels in different applications. Water storage capacity in hydrogels decreases with increasing the number of hydrophobic groups and network density.1,2,7 This property demonstrates the ability of the wound dressing to absorb the wound fluids. In Figure 4, the swelling percentage of pHEMA decreased somewhat with increasing the percentage of nanoparticles. There seems to be a reasonable relationship between the percentage of swelling and the percentage of gel. When the cross-linking density and gel fraction of samples increased, swelling capacity decreased. For this reason, with increasing the percentage of nanoparticles as cross-linking points, swelling of hydrogels has decreased. However, according to the percentages of swelling of nanocomposite hydrogels compared to pure pHEMA, the swelling capacity of hydrogels was suitable for wound dressing applications. Gel fraction and degree of swelling of pHEMA and nanocomposite hydrogels containing 5, 10, and 15 wt% of iron oxide nanoparticles.
Water vapor transmission rate
One of the factors in using hydrogel as a wound dressing is the ability to maintain proper moisture for the wound. Low or high WVRT may result in drying of the wound and eschars or accumulation of exudates and infections, respectively. Both of these conditions cause the retardation of the healing process. In Table 1, it can be seen that the WVRT of hydrogels decreased from 53 (g/m2/hr) for pHEMA to 37 (g/m2/hr) for pHEMA+15% nanoparticle. The results obtained from the WVRT test are in agreement with that reported range for other wounds (36–56 g/m2/hr). 39
Porosity of hydrogels
The porosity of hydrogels is investigated as an important item that can increase the speed of wound healing (Table 1). As can be seen in Table 1, the porosity decreased from 64 to 48 by increasing the quantity of nanoparticles. This result shows that the hydrogels can absorb a large amount of exudate from the wound surface and also allow the wound to exchange oxygen easily with the environment. The results are acceptable compared to other reports. 40 Therefore, the prepared hydrogels were suitable for wound dressing applications.
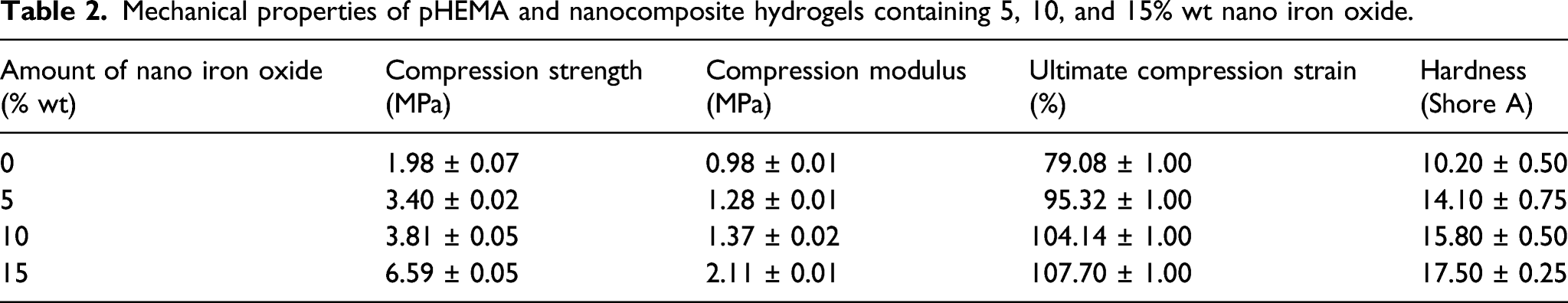
Mechanical properties of hydrogels
Mechanical properties of pHEMA and nanocomposite hydrogels containing 5, 10, and 15% wt nano iron oxide.
Cell survival test of pure hydrogel and nanocomposite hydrogel
The results of cell survival that indicate cytotoxicity of samples are shown in Figure 5. The percentage of cell survival increased by adding nano iron oxide so that the percentage of cell viability increased from 81.84% for pure pHEMA to 99.45% for nanocomposite hydrogel containing 15% nano iron oxide. It can be concluded that the cytotoxicity of the nanocomposite hydrogels decreased with increasing the percentage of nanoparticles. Improvement of the cell viability properties of samples by increasing the nanoparticles is because the nano iron particles have biocompatible properties.29,36,44 Cell survival percent of pHEMA hydrogel and pHEMA containing 15 wt% of iron oxide nanoparticles.
Microbial penetration test
The microbial penetration and antibacterial property of pHEMA and nanocomposite hydrogel were investigated with two types of bacteria, E.coli and S. aureus, as shown in Figure 6. According to the test method, the pHEMA sample did not show antibacterial activity, while the nanocomposite hydrogel containing 15 wt% iron oxide nanoparticles was impenetrable. The diameters of inhibition zones were reported as a measure of antibacterial activity. The results showed that the nanocomposite hydrogels can prevent the growth of bacteria so that the diameters of the inhibition zones around the sample were about 8 mm and 11 mm for E-Coli and S. aureus, respectively. Microbial penetration test (a) pHEMA (with E-Coli), (b) pHEMA +15wt% nanoparticles (with E-Coli), and (c) pHEMA+15% wt nanoparticles (with S. aureus).
Conclusion
In the current study, pHEMA hydrogel and nanocomposite hydrogels with various amounts of iron oxide nanoparticles (5, 10, and 15 wt %) were synthesized and evaluated for wound dressing application. The results of FTIR showed that the synthesis of hydrogels was successful, and there was an adequate interaction between nanoparticles and the polymer hydrogel. Images of FE-SEM and EDAX analyses showed good dispersion and distribution of the nanoparticles in the pHEMA matrix. The results of gel fraction and swelling indicated that with increasing the percentage of nanoparticles, they act as cross-linking points and thus increment in gel fraction, and accordingly, a decrement in swelling with increasing the percentage of nanoparticles in the hydrogel was observed, however, the swelling capacity of hydrogels was suitable for wound dressing applications. The results of WVTR and porosity showed that the values are in agreement range with other reports, and prepared hydrogels are suitable for wound dressing applications. Increasing the percentage of iron oxide nanoparticles in the matrix significantly improved the mechanical properties of the hydrogels, such as strength, modulus, ultimate strain, and hardness. The obtained results from mechanical tests clearly showed that the nanocomposite hydrogels have better mechanical performance than pure hydrogel for wound dressing applications. The cell survival test demonstrated that the cytotoxicity of the nanocomposite hydrogels decreased dramatically with increasing the percentage of nanoparticles as compared with that of the pure pHEMA. The antibacterial properties of hydrogels were examined by using two types of Gram-negative (E-Coli) and Gram-positive (S. aureus) bacteria. According to the results of the antibacterial, the nanocomposite hydrogels were resistant to microbial penetration and can be useful to prevent wound infection. A summary of the results indicates the efficiency of synthesized nanocomposite hydrogels for wound dressing relative to pure pHEMA hydrogel.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.





