Abstract
In the present work, an attempt has been made to develop high-performance polymeric hybrid binary blends of epoxy/benzoxazine and benzoxazine/cyanate ester with varying weight percentages (25/75, 50/50 and 75/25 wt%) of resins, namely, bisphenol-F epoxy resin (DGEBF), benzoxazines [bisphenol–F/aniline (BF-a) and imidazole core-based bisphenol/aniline (IBP-a)] and cyanate ester [bisphenol-F bifunctional cyanate ester (BF-CE)]. The molecular structure, polymerisation temperature/cure behaviour, glass transition temperature (Tg) and thermal stability of the neat polymeric matrices and binary hybrid blends of polymeric matrices were characterised using different analytical techniques, viz. Fourier Transform infra-red spectroscopy (FTIR), Nuclear Magnetic Resonance spectroscopy (NMR), Differential Scanning Calorimetry (DSC) and thermogravimetric analysis (TGA). Among the binary hybrid blends, the lowest polymerisation temperatures (Tp) were noticed in the case of blends of epoxy/benzoxazine were 219°C for DGEBF/BF-a (25/75 wt%) and 170°C for DGEBF/IBP-a (25/75 wt%). Similarly, in the case of blends of benzoxazine/cyanate ester, the lowest values of Tp observed were 155°C and 153°C for BF-a/BF-CE (75/25 wt%) and IBP-a/BF-CE (75/25 wt%), respectively. The highest values of Tg observed for the blends of epoxy/benzoxazine were 175°C and 254°C for DGEBF/BF-a (25/75 wt%) and DGEBF/IBP-a (25/75 wt%), respectively. Whereas, the highest values of Tg observed in the case of blends of benzoxazine/cyanate ester were 234°C and 278°C for BF-a/BF-CE (25/75 wt%) and IBP-a/BF-CE (75/25 wt%), respectively. From the TGA results of blends, the maximum degradation temperature (Tmax) and limiting oxygen index (LOI) value calculated from the char yield, which ascertain that almost all the binary hybrid blends of epoxy/benzoxazine and benzoxazine/cyanate ester possess good flame retardant behaviour.
Introduction
Polybenzoxazines (PBZs) are relatively new class of addition-cure, thermosetting phenolic resins that have recently attracted much attention as matrices for high-performance applications, such as aerospace composites, electronic insulation applications, etc.1–4 Polybenzoxazines are derived from heterocyclic bis-1,3-benzoxazine monomers that undergo cationic ring opening polymerisation at higher temperatures in the absence of catalyst without releasing any by-products, leading to form 3D cross-linked network structure.5–7 The formation of hydrogen bonding interaction results between the phenol and tertiary amine within the network structure contributes to number of useful properties required for different high-performance applications, namely, high glass transition temperature (Tg), high thermal stability, low surface energy, low water absorption, excellent dimensional stability, high flame retardance and good dielectric behaviour.7–12
Conventional bisphenol-A based benzoxazines and epoxy resins available in the market, are toxic in nature.13,14 Hence, researchers are focussing their attention to reduce the toxicity of the industrially valuable benzoxazine and epoxy materials, using preferably non-toxic precursor alternate to the conventional materials. Bisphenol-F is considered to be a less non-toxic material and is a perfect choice to replace the bisphenol-A material.6,15,16 Recently, our research group has developed and reported the different bisphenols, namely, imidazole-based bisphenol 17 and pyrazolidine-based bisphenol, 18 benzaldehyde-based bisphenol and their corresponding benzoxazines. 19
To extend the utility of PBZ for a wide range of applications is a continued effort by reducing the cure behaviour (lowering polymerisation temperature), enhanced process-ability and improved toughness behaviour through modification of PBZ with suitable compatible resins and additives through different methods.1,20,21 The blending of PBZ with similar resinous materials like epoxies,22,23 urethanes,24,25 cyanate esters26,27 through co-polymerisation/hybridization results an improved properties and more suitable for high-performance applications than those of individual PBZs. Our research group has studied the enhancement of thermal behaviour for benzoxazines with the various blends of epoxy, 28 bismaleimide 5 and cyanate ester resins. 29 Also, the thermal behaviour was studied for imidazole core-based benzoxazine, bisphenol-F based benzoxazine and bismalimide blends.17,19 Similarly, a thermal behaviour of cardanol benzoxazine blend with bisphenol-F benzoxazine and bismalimide matrices were studied.1,21
In continuation of earlier studies and impressive results obtained from the work, made us to develop non-toxic bisphenols (bisphenol-F and imidazole core bisphenol) based benzoxazine blends with low temperature cure, thermally stable and flame retardant behaviour using industrially valuable bisphenol-F epoxy (DGEBF) resins and bisphenol-F based dicyanate ester with varying weight percentage compositions. Hence, the binary hybrid blends developed in the present work can be used in the form of high-performance sealants and insulation materials, where application warrants high thermal stability and good flame retardancy.
Experimental
Material
Bisphenol-F was received from Anabond Ltd, Chennai. Bisphenol-F epoxy resin was purchased from Roto Polymers Ltd, Chennai, India. Aniline, cyanogen bromide, benzil, acetic acid, ammonium acetate, acetone, triethyl amine, ethyl acetate and sodium hydroxide, p-aminophenol and p-hydroxybenzaldehyde were purchased from SRL, India. Paraformaldehyde and anhydrous sodium sulphate were purchased from Sigma-Aldrich, India.
Synthesis of imidazole-core-based bisphenol
A mixture of (0.1 mol) benzil, p-aminophenol, and p-hydroxybenzaldehyde and 0.2 mol of ammonium acetate were taken in a 500 mL round-bottomed flask and kept for stirring at 110°C for 8 h in the presence of acetic acid. Then, the content was cooled to room temperature and washed with deionised water until the acetic acid was removed. The obtained white solid material was filtered, washed several times with deionised water and dried under vacuum overnight. The product imidazole-core-based bisphenol (IBP) obtained was analysed for its molecular structure by Fourier Transform infra-red spectroscopy (FTIR) and 1H-Nuclear Magnetic Resonance spectroscopy (NMR) spectroscopies. 17
Synthesis of the benzoxazine monomer
The bisphenol-F and IBP and aniline-based benzoxazines were synthesised separately as per the reported procedure. 17 In brief, 0.4 mol of paraformaldehyde was mixed with 0.2 mol of bisphenols (bisphenol-F, imidazole core bisphenol) separately under vigorous stirring and 0.2 mol of aniline was added in drop-wise and temperature was raised to 110°C. The stirring was continued for more than 6 h until the formation of the homogeneous product. The progress of the reaction was monitored through thin layer chromatography. The obtained crude product was filtered and washed three times with 2N NaOH followed by distilled water. The organic phase was dried over anhydrous Na2SO4 and the solvent was evaporated in a rotary evaporator (Yield: 92%).
Synthesis of the cyanate ester monomers
0.05 mol of bisphenol-F and 0.1054 mol of cyanogen bromide were dissolved in 150 mL of dry acetone in a 250-mL round-bottomed flask. The flask was placed in an ice bath, and 0.1054 mol of dry triethylamine was added in drop-wise over 30 min with constant stirring. The reaction solution was allowed to spontaneously warm to room temperature; afterwards, it was stirred for 24 h. Then, the reaction product obtained was filtered, then filtrate was evaporated and then the product was poured into water and extracted using dichloromethane, washed with deionised water several times, and then finally washed with brine solution. After it was dried over sodium sulphate, the solvent was removed by rotary evaporation, and light, yellow crystals obtained were preserved for further study. 29
Preparation of homo polymers
Benzoxazines (BF-a, IBP-a), epoxy resin and cyanate ester (BF-CE) resins were separately taken in a silane pre-treated glass petri plate and heated at 120°C for an hour, in order to expel the moisture and air bubbles. Then, the samples were used for the preparation of varying weight percentages (25/75, 50/50 and 75/25 wt%) of binary hybrid epoxy/benzoxazine and benzoxazine/cyanate ester blends.
Preparation of binary blends
The binary blends of BF-a and IBP-a benzoxazines were separately prepared by mixing of varying weight percentages, such as 25:75, 50:50 and 75:25 wt% of DGEBF resin and BF-CE resin (Schemes 1-4). Then, the blend was mixed thoroughly till it became homogeneous. The resulting homogenous blend was taken in a silane pre-treated glass petri dish and heated at 120°C for an hour, in order to remove the moisture and air bubbles. Then, the samples were heated to 210°C at the heating rate of 20°C per hour and post-cured at 230°C for 3 h.
The prime objective of the present work is to lower the polymerisation temperature of benzoxazine by way of blending with other industrially valuable resins, like epoxy resin and cyanate ester. In addition, it is also exploited to derive the benefits of high-performance properties of benzoxazines, epoxies and cyanate ester. In order to enhance the thermal stability and to improve the glass transition temperature of reactive blends, the synthesised BF-a and IBP-a benzoxazines were separately blended with DGEBF and bisphenol-F based bifunctional cyanate ester. Then, curing behaviour (Tp) and glass transition temperature (Tg) of the blended resins were studied by Differential Scanning Calorimetry (DSC) technique. The thermal stability of thermally cured reactive blends was studied by TGA technique.
Characterisation
The molecular structure of BF-a, IBP-a benzoxazines and bisphenol-F bifunctional cyanate ester was confirmed by FTIR and 1H-NMR spectral analyses. Fourier Transform infra-red spectroscopy spectra measurements were carried out with Agilent Cary 630 FTIR Spectrometer. 1H-Nuclear Magnetic Resonance spectroscopy spectra were obtained with Bruker (400 MHz) using dimethylsulfoxide (d6-DMSO) as a solvent and tetramethylsilane (TMS) as an internal standard. Thermal behaviour of BF-a and IBP-a benzoxazines and corresponding polymeric blends were studied by DSC and TGA techniques. Differential Scanning Calorimetry measurements were recorded using NETZSCH STA 449F3 under N2 atmosphere (60 mL min−1) at the heating rate of 10°C min−1. Thermogravimetric analysis measurements were recorded using NETZSCH STA 449F3 under N2 atmosphere (60 mL min−1) at heating rate of 20°C min−1.
Results and discussion
Bisphenol-F and imidazole core bisphenol-based difunctional benzoxazines were synthesised based on Mannich condensation reaction using aniline with paraformaldehyde. After work-up, respective benzoxazines obtained were isolated and characterised with appropriate techniques. Further, binary reactive hybrid blends were made with benzoxazines, epoxy and cyanate ester. Then, the neat matrices and binary blends were subjected to appropriate studies.
Thermal behaviour
Neat polymeric matrices
The polymerisation behaviour of different benzoxazine monomers was studied by DSC analysis at the heating rate of 10°C per minute. The peak maxima of the exothermic peak obtained for the benzoxazine systems BF-a and IBP-a are 235°C and 231°C, respectively. Similarly, the peak maxima of the exothermic peak for the DGEBF and bisphenol-F cyanate ester (BF-CE) are 375°C and 131°C, respectively (Figure 1). DSC thermograms of bisphenol-F epoxy (DGEBF) and bisphenol-F cyanate ester (BF-CE). DSC: Differential Scanning Calorimetry.
Thermal behaviour of neat polymer matrices.
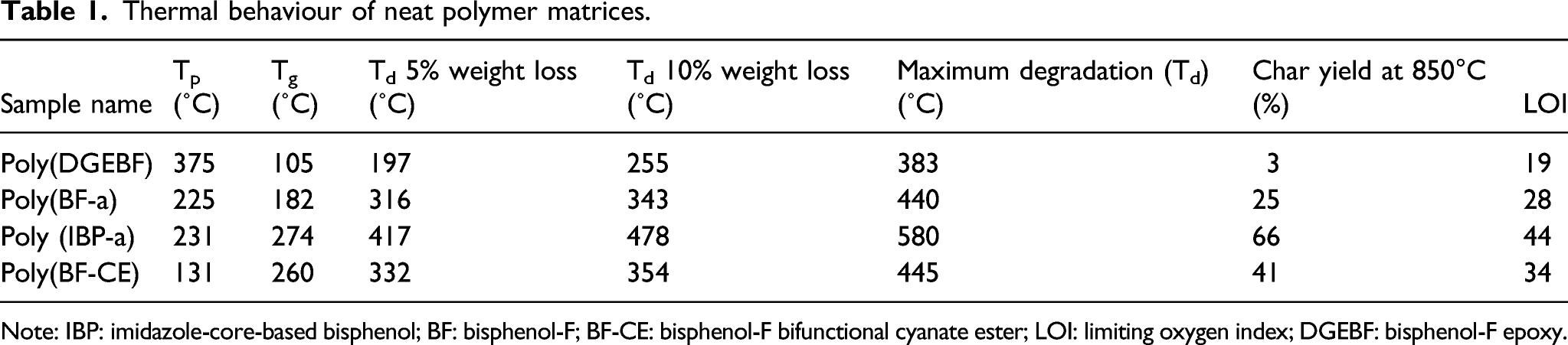
Note: IBP: imidazole-core-based bisphenol; BF: bisphenol-F; BF-CE: bisphenol-F bifunctional cyanate ester; LOI: limiting oxygen index; DGEBF: bisphenol-F epoxy.
Binary reactive blends of benzoxazine and epoxy matrices
The chemical blending of BF-a, IBP-a benzoxazines separately carried out with conventional DGEBF epoxy resin offer an enhanced thermal stability (Figures 2 and 3). The thermal characteristics of the 75:25, 50:50 and 25:75 wt% of the prepared DGEBF epoxy and BF-a benzoxazines blends were studied and the results obtained indicate that the curing temperature are 253, 231 and 219°C, respectively (Figure 2(a)). In the case of DGEBF epoxy and IBP-a benzoxazines 75:25, 50:50 and 25:75 wt% blends, the curing temperature observed for all the blends are about 170°C (Figure 2(b)). It is considered as lower cure temperature than that of neat IBP-a and DGEBF matrices, the lowering curing temperature obtained may be explained due to the contribution of the activation of imidazole core of the benzoxazine molecule. (Schemes 1 and 2) DSC thermograms of (a) bisphenol-F epoxy (DGEBF) and bisphenol-F benzoxazine, (b) bisphenol-F epoxy (DGEBF) and IBP-a benzoxazine blends. DSC: Differential Scanning Calorimetry. TGA thermograms of (a) bisphenol-F epoxy (DGEBF) and bisphenol-F benzoxazine, (b) bisphenol-F epoxy (DGEBF) and IBP-a benzoxazine blends. TGA: thermogravimetric analysis. Preparation of bisphenol-F epoxy and bisphenol-F benzoxazine hybrid blend. Preparation of bisphenol-F epoxy and imidazole core bisphenol benzoxazine hybrid blend. Preparation of bisphenol-F cyanate ester and bisphenol-F benzoxazine hybrid blend. Preparation of bisphenol-F cyanate ester and imidazole core bisphenol benzoxazine blend.





Oxirane ring is highly reactive than that of oxazine ring,31,32 which may be plausibly explained using primary, secondary and tertiary amines. Amines are highly reactive with oxirane ring functionality than that of oxazine ring.33,34 Hence, it was observed that the curing temperature of benzoxazine/epoxy blends is lower than that of homo polymerisation of neat individual matrices. The oxirane ring forms a less stable intermediate of zwitterion formation with tertiary amine than that of oxazine ring, this influences to open the ring of benzoxazine with zwitterion intermediate. Hence, the polymerisation of benzoxazine blends occurs at lower temperature due to the influence of hydroxyl group attached with oxirane intermediate, and in turn also contributes to form three-dimensional cross-linked network structure (Scheme 5). Proposed mechanism for hybridization of epoxy and benzoxazine blend.
Thermal behaviour of epoxy/benzoxazine blends.

Note: IBP: imidazole-core-based bisphenol; BF: bisphenol-F; LOI: limiting oxygen index; DGEBF: bisphenol-F epoxy.
The DGEBF was blended with varying weight percentages (75:25, 50:50 and 25:75 wt%) of benzoxazines (BF-a and IBF-a) and cured at appropriate temperatures. The thermal degradation behaviour for the blends of BF-a and IBF-a with DGEBF was analysed using thermogravimetric analysis and the results obtained are presented in Figure 3. As shown in Figure 3, the thermal stability of blends was found to be gradually improved with increasing weight percentage of benzoxazines. The maximum degradation temperatures of 75:25, 50:50 and 25:75 wt% poly(DGEBF/BF-a) (Figure 3(a)) were noticed at 392°C, 399°C and 404°C, respectively. The char yield of 75:25, 50:50 and 25:75 wt% of blends of poly(DGEBF/BF-a) were found to be 22%, 26% and 31%, respectively.
Similarly, Figure 3(b) shows the TGA profile for the binary blends of IBP-a with varying weight percentages (75:25, 50:50 and 25:75 wt%) of DGEBF. The values of percentage char yield of binary blends of poly (DGEBF/IBP-a) with ratios of 75:25, 50:50 and 25:75 wt% are 34%, 39% and 42%, respectively, and the values of maximum degradation temperature of the blends observed are 424°C, 434°C and 456°C, respectively. From the obtained results, 25:75 wt% of poly(DGEBF/IBP-a) possesses better thermal behaviour than that of other blends. The curing temperature (Tp) of DGEBF/IBP-a was observed at 170°C, which exhibits lower curing temperature than that of the rest of the epoxy blends (DGEBF/BF-a). This may be explained due to the presence of imidazole core group which influences to reduce the curing temperature. The values of glass transition temperature (Tg) 254°C, maximum degradation temperature (Tmax 456°C) and char yield (42%) observed for 25:75 wt% of poly(DGEBF/IBP-a) are higher than those of other epoxy blends, due to the presence of tetra aryl substituted imidazole core containing benzoxazine. The tetra aryl substituted imidazole is a rigid unsymmetrical molecule, which in turn contributes to enhanced thermal stability of poly(DGEBF/IBP-a) matrices. Specifically, 25:75 wt% of poly(DGEBF/IBP-a) containing higher concentration of IBP-a, which helps to improve the thermal stability than that of the other epoxy blends.
Binary reactive blends of cyanate ester and benzoxazine
Bisphenol-F-aniline benzoxazine (BF-a) and imidazole core bisphenol-aniline benzoxazine (BPI-a) were blended with bisphenol-F based cyanate ester (BF-CE) (Schemes 3 and 4). The polymerisation temperature prepared blends were studied using DSC technique and it was observed that the curing temperature of 75:25, 50:50 and 25:75 wt% BF-CE and BF-a blends were found to be 163°C, 158°C and 155°C, respectively (Figure 4(a)). Similarly, the curing temperature were observed for 75:25, 50:50 and 25:75 wt% BF-CE and IBP-a blends at 153, 189 and 192°C, respectively (Figure 4(b)). DSC thermograms of (a) bisphenol-F cyanate ester and bisphenol-F benzoxazine, (b) bisphenol-F cyanate ester and IBP-a benzoxazine blends. DSC: Differential Scanning Calorimetry.
Benzoxazine and cyanate ester resins can also be cured in the absence of any hardener and accelerator. The curing temperature of benzoxazine/cyanate ester blends was lower than that of homo polymerisation of individual neat matrices. The cyanate ester and benzoxazine undergo formation of ring with a less stable intermediate of zwitterion and oxonium ions. Hence, the polymerisation of benzoxazine occurs at lower temperature due to the influence of cyanate ester group and in turn also contributes to form three dimensional cross-linked triazine network polymer structure (Scheme 6). Proposed mechanism for hybridization of cyanate ester and benzoxazine blend.
Thermal behaviour of polybenzoxazine and cyanate ester blends.
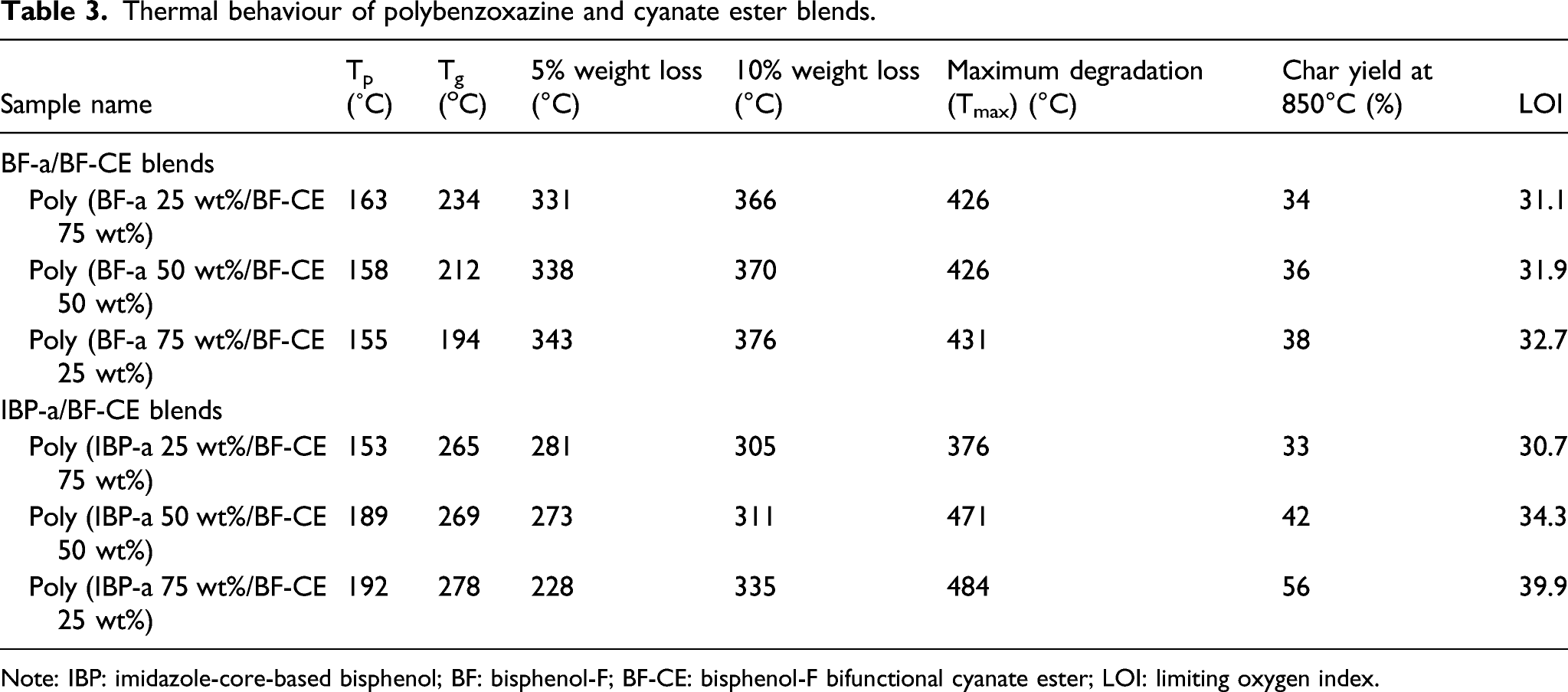
Note: IBP: imidazole-core-based bisphenol; BF: bisphenol-F; BF-CE: bisphenol-F bifunctional cyanate ester; LOI: limiting oxygen index.
Figure 5(a), shows the TGA profile for the binary blends of BF-CE with varying weight percentages of BF-a and IBP-a. The values of percentage char yield obtained for binary blends of BF-CE with BF-a in the weight ratios of 75:25, 50:50 and 25:75 wt% are 34, 36 and 38%. respectively. The maximum degradation temperature of the blends observed are 426, 426 and 431°C, respectively. Similarly, the binary blends of BF-CE with different ratios of 75:25, 50:50 and 25:75 wt% of IBP-a possess the char yield values of 33, 42 and 56%, respectively. The maximum degradation temperatures obtained for these blends are 376, 471 and 484°C, respectively. Among the different ratios of blends, the blends with a ratio of BF-CE/IBP-a 25:75 wt% possesses a better char yield and higher degradation temperature than those of neat BF-CE and other blend systems. When increasing the concentration of IBP-a increases the thermal stability of binary system, due to the extension of stable rigid aromatic ring structure. The data resulted are presented in Table 3. TGA thermograms of (a) bisphenol-F cyanate ester and bisphenol-F benzoxazine, (b) bisphenol-F cyanate ester and IBP-a benzoxazine blends. TGA: thermogravimetric analysis.
Flame retardant behaviour
The flame retardant behaviour of the materials can be ascertained from the value of limiting oxygen index (LOI) calculated using char yield obtained from thermogravimetric analysis at 850°C. The value of LOI is calculated using the van Krevelen and Hoftyzer relation.35–37 LOI = 17.5 + 0.4 CR. The percentage char yield (CR) of the sample obtained from TGA analysis at 850°C is presented in Tables 2 and 3. It is well known that the polymeric materials with LOI value greater than 26 may exhibit an excellent flame retardant material. 38 The PBZs blended matrices offer higher char residue and thereby contribute to enhanced values of LOI. The LOI value of poly(DGEBF/BF-a), poly(DGEBF/IBP-a), poly(BF-a/BF-CE) and poly(IBP-a/BF-CE) blends are higher due to the high residual char obtained for these blends. Poly(DGEBF) matrix exhibits a LOI value of 19. It shows the poor flame retardant behaviour than that of polybenzoxazine matrices. On increasing the proportion of benzoxazine ratio in benzoxazine/epoxy blend, the char yield and in turn the value of LOI increased according the weight percentage concentration (Tables 1 and 2). Similarly, the blends of benzoxazine/cyanate ester matrices also indicated the same effect that an increase of benzoxazine ratio increases the values of char yield and LOI. Comparatively, IBP-a/BF-CE (75/25 wt%) exhibits better flame retardant behaviour than that of the rest of the blends (Tables 1 and 3). This may be due to the presence of cross-linked network structure with rich in nitrogen atoms.
Conclusion
In the present work, there are two types of benzoxazines (BF-a and IBP-a), epoxy resin (DGEBF) and cyanate ester (BF-CE) used for the development of binary hybrid blends of epoxy/benzoxazine and benzoxazine/cyanate ester with varying weight percentages (25/75, 50/50 and 75/25 wt%) and the obtained blends were characterised using different analytical techniques. Data obtained from different studies, the lowest polymerisation temperature (Tp of 170°C) for the blend DGEBF/IBP-a (25/75 wt%), the highest glass transition temperature (Tg) of 278°C observed for BF-a/BF-CE (25/75 wt%) and the highest maximum degradation temperature of 284°C observed for IBP-a/BF-CE (75/25 wt%). Further, it was also noticed that all the blends having different weight percentage composition possess very good thermal stability and flame retardant behaviour. It is concluded that the binary hybrid blends developed in the present work can be used in the form of coatings, sealants, adhesives, encapsulants and insulation matrix materials, where application warrants high thermal stability and good flame retardancy.
Footnotes
Acknowledgements
The authors thank the PSG Management, Secretary and Principal, PSG Institute of Technology and Applied Research, Coimbatore-641062, India, for their moral and support. Authors also thank Mrs K. Selvi, and Mr. J. Ezhilarasan, PSG Institute of Technology and Applied Research, Coimbatore, for the lab assistance.
Authors’ contributions
Balaji Krishnasamy: Methodology.Hariharan Arumugam: Conceptualization, Methodology, Investigation, Visualization, Data curation, Validation, Writing - original draft, Writing - review & editing.M. Mohamed Iqbal: Visualization, Data curation, Validation.Muthukaruppan Alagar: Resources, Validation, Supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
All the necessary data are included in the manuscript at appropriate section.
