Abstract
1,1-Bis(3-methyl-4-hydroxyphenyl)cyclohexane-based benzoxazine monomer and 1,1-bis(3-methyl-4-cyanatophenyl)cyclohexane were blended together via in situ polymerization. The chemical blending of benzoxazines with cyanate ester was carried out thermally and the resulting product was analyzed by Fourier transform infrared spectra. The benzoxazine–cyanate ester hybrid polymer matrices were investigated for their thermal and morphological properties. The glass transition temperature, curing behavior, thermal stability, char yield, and flame resistance of the hybrid polymer matrices were studied by means of differential scanning calorimeter and thermogravimetric analysis.
Keywords
Introduction
Polymer matrix materials have gained much importance due to their incredible properties suitable for wide-range industrial applications. Such polymer properties are conceptually departed from those of conventional materials. In recent years, significant efforts have been made to improve the high-performance characteristics of polymer-based matrices by hybridizing compatible polymeric materials. At present, natural and synthetic polymeric materials have been used globally for different industrial and engineering applications. Polymeric materials used for the fabrication of advanced components should exhibit the desired physicochemical properties, mechanical strength and stiffness, thermal stability, and chemical resistance. 1 –5
Cyanate ester resins are well-known among the most important thermosetting resins developed in the recent past. Cyanate ester resins replace the use of epoxies in the high technology area of automobile, aerospace, and electronics industries due to their excellent properties. The outstanding properties of these matrices are low dielectric constant, low moisture absorption and low out gassing, excellent mechanical properties, good dimensional and thermal stability, good flame resistance, good adhesive properties, high glass transition temperature (T g), and good chemical resistance. 6 –11
Polybenzoxazines are another class of versatile phenolic resins that combine both the thermal properties and flame retardance of phenolics with improved mechanical behavior and amenable molecular design flexibility. Polybenzoxazine resins are used as high-performance matrices and composites and are used in aerospace coatings, structural materials, and electronic encapsulating materials due to their efficient bonding, high strength and modulus, excellent chemical resistance, and amenable processing methods. In addition, polybenzoxazine possesses some unique properties that overcome the limitations of conventional phenolics and epoxy resins, including high T g, high char yield, low water absorption, low dielectric constant, inexpensive raw materials, and no volume shrinkage upon curing. 12 –15
The molecular structure of benzoxazines offers excellent design flexibility that allows properties of the cured materials to be controlled for specific requirements. The properties and performances of polybenzoxazine can be improved through structural modification by designing new skeletally modified benzoxazine monomers proceeds through the ring opening of the heterocyclic monomers by thermal treatment, in the absence of any catalysts, without producing by products or volatiles. The inherent molecular nature of benzoxazines facilitates efficient blending with epoxy, poly(imide-siloxane), and polyurethane polymers as well as the reinforcement with nanoclays, carbon nanotubes, metal oxides, and polyhedral oligomeric silsesquioxane. 16 –22
In the recent past, Nair and coworkers prepared bisphenol A-based benzoxazine (BA-a) and cyanate ester blends and investigated its cure chemistry and polymer properties. 23 Thermogravimetric analysis (TGA), differential thermal analysis, and scanning electron microscopic (SEM) behavior of the cured blend ascertained the reaction of benzoxazine with cyanate ester enhanced the thermal stability of polybenzoxazine. Kimura et al. also studied the curing reaction of bisphenol A-based benzoxazine (BA-a) and cyanate ester blends. 24 They further confirmed that the cured benzoxazine and cyanate ester resin showed good thermal stability and high electrical and water resistance. 25 The mechanical and dielectric properties of blends of dicyanate ester and bisphenol A-based benzoxazine were studied by Yan et al. 26
Although so many investigations are being continued on benzoxazine–cyanate ester blends, 27 –30 we attempted on 1,1-bis(3-methyl-4-hydroxyphenyl)cyclohexane-based benzoxazine and cyanate ester hybrid polymer matrices by the way how its compatibility influences thermal and morphological properties of these hybrid polymer matrices. The benzoxazine–cyanate ester hybrid polymer matrices were prepared via in situ polymerization and their molecular structure, cure behavior, T g, thermal stability, char yield, flame resistance, and morphological properties were studied by appropriate analytical techniques. The results obtained are discussed and reported.
Experimental
Materials
Cyanogen bromide, triethylammonium bromide, paraformaldehyde, and triethylamine were purchased from Aldrich (St Louis, Missouri, USA). 4,4′-Diaminodiphenylmethane (DDM) (HT972, Ciba-Geigy, amine equivalent = 49.5 eq/kg) was supplied by CDH Chemicals (Chennai, Tamil Nadu, India). Sodium hydroxide, anhydrous sodium sulfate, cyclohexanone, o-cresol, 1,4-dioxane, acetone, and chloroform were obtained from SRL (Chennai, Tamil Nadu, India). Hydrochloric acid and glacial acetic acid were purchased from Merck (Germany). All the solvents used were extrapure analytical reagent grade and used as received.
Synthesis of benzoxazines
1,1-Bis(3-methyl-4-hydroxy phenyl)cyclohexane, DDM-based 1,1-bis(3-methyl-4-hydroxy phenyl)cyclohexane benzoxazines (CBDDM), and 1,1-bis(3-methyl-4-cyanatophenyl)cyclohexane (BMCPC) were prepared according to the reported procedure. 19,20,31,32 The chemical structures of CBDDM and BMCPC are presented in Figure 1.

The chemical structures of 1,1-bis(3-methyl-4-hydroxy phenyl)cyclohexane, CBDDM, and BMCPC.
Benzoxazine–cyanate ester polymer hybrid matrices
Benzoxazine and cyanate ester monomers were mixed (by solution method) at various weight ratios in 1,4-dioxane, and the solution mixture was stirred at 25°C for 30 min. 33 The blend solutions were cast onto the glass plates that were pretreated with dichlorodimethylsilane, dried at 60°C for 4 h for evaporating the solvent, and then thermally treated at 100, 140, 180, 200, 220, 250°C for 1 h each to obtain the hybrid films. The color of the films after curing up to 250°C was dark red with high benzoxazine content and became brown with higher content of cyanate ester (Figure 2).

Synthesis of benzoxazine–cyanate ester hybrid polymer matrices.
Instrumentation
Fourier transform infrared (FTIR) spectra were recorded on a Perkin-Elmer 6X FTIR spectrometer. Optical-grade potassium bromide (KBr; approximately 100 mg) was ground in a mortar with a pestle, along with enough solid samples to make a 1 wt% mixture for making KBr pellets. After the sample was loaded, a minimum of 16 scans were collected for each sample at a resolution of 4 cm−1. The T g of the samples were analyzed using a DSC 200 PC differential scanning calorimeter (Netzsch Gerateban GmbH, Germany) in the temperature range between 30°C and 300°C at a heating rate of 10°C min−1 under nitrogen atmosphere. The flow rate of the purge gas (nitrogen) is 50–80 mL min−1. The purpose of the purge is to vent corrosive volatiles and to prevent the entering of water into the DSC cell. TGA was carried out using a DSTA 409 PC analyzer (Netzsch Gerateban GmbH) in the temperature range between 30°C and 850°C at a heating rate of 10°C min−1 under nitrogen atmosphere. The flow rate of the purge gas (nitrogen) is 50–80 mL min−1. The surface morphology of the fractured surface of the samples was performed using field-emission scanning electron microscope (JEOL JSM Model 6360, Japan). Auto Fine Coater (JEOL JFC-1600); platinum sputtered on the fractured surface of the samples at vacuum pressure 8 Pa/20 mA for 95 s. The fractured surface of the samples was coated with platinum before scanning.
Results and discussion
FTIR spectral analysis
The CBDDM/BMCPC hybrid polymers are obtained through the formation of co-reacted network comprising triazine ring and polybenzoxazine (Figure 3). The reaction between phenolic –OH of polybenzoxazine and cyanate group of cyanate ester occurs through iminocarbonate intermediate which further induces the curing of cyanate ester and benzoxazine to form polybenzoxazin–polycyanurate network. 34,35 The absorption peaks for benzoxazine and cyanate ester appeared at 947 cm−1 indicating the presence of oxazine ring and peaks at 2237 and 2270 cm−1 ascertain the presence of cyanate groups. The appearance of peaks at 1370 cm−1 (–C=N stretching vibration), 1570 cm−1 (triazine ring), and 3380 cm−1 (hydroxyl group of polybenzoxazine) confirms the formation of CBDDM/BMCPC hybrid polymer (Figure 4).
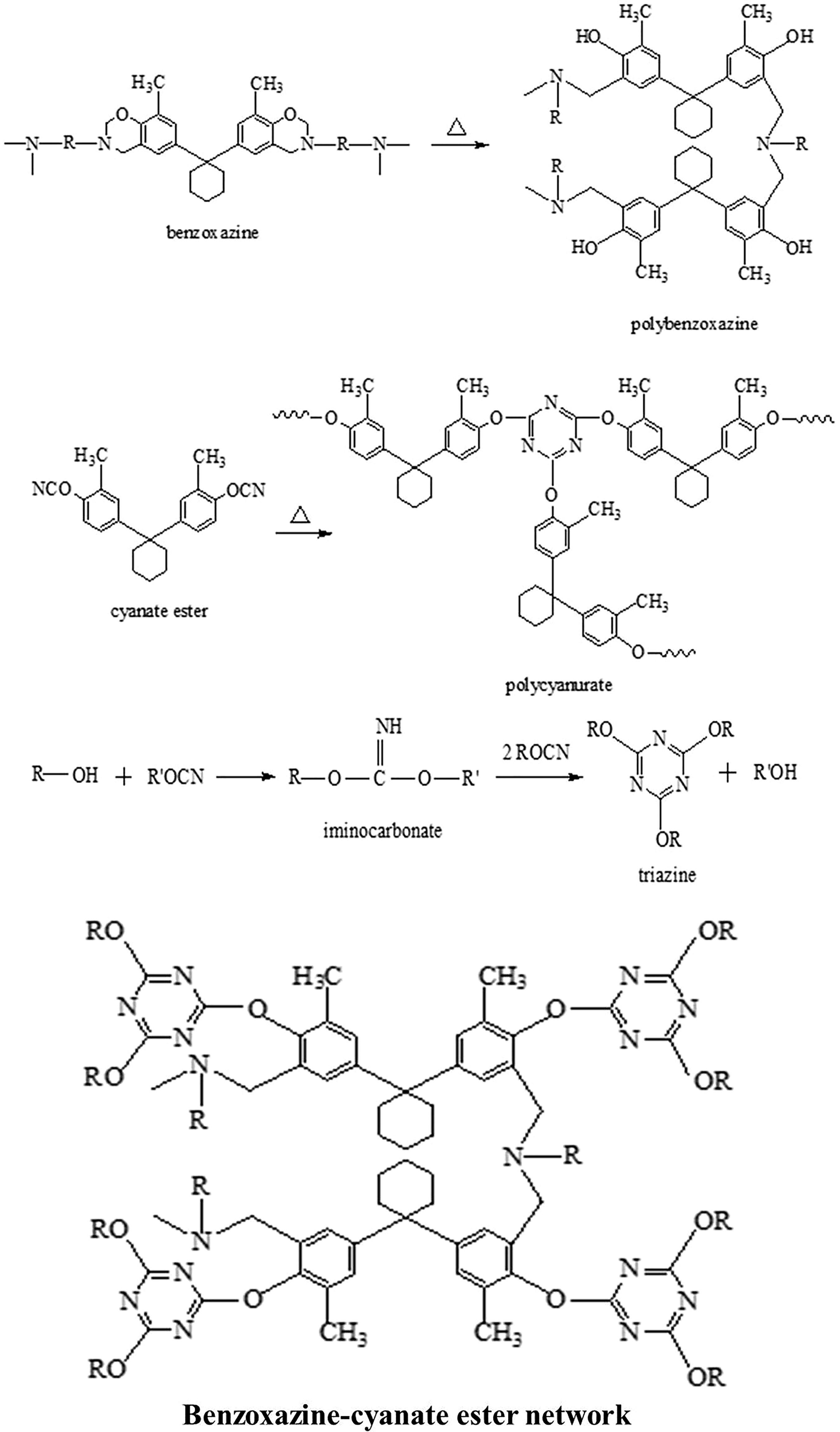
Schematic representation and curing reaction of benzoxazine–cyanate ester polymers.

FTIR spectra of CBDDM/BMCPC hybrid polymer matrices.
Thermal properties
Tg and curing behavior
Table 1 presents the data of DSC thermograms of CBDDM/BMCPC hybrid matrices and its homopolymers. The curing behavior of the self-curing precursors and self-cured polymers of CBDDM/BMCPC hybrid matrices was examined by DSC analysis. The DSC curves obtained for these hybrid matrices are shown in Figure 5. The onset of the curing exotherm for CBDDM and BMCPC starts at 165 and 168°C, respectively, with respective peak maxima at 203 and 211°C. The onset of the exotherm for homopolymers, PCBDDM and PBMCPC starts at 228 and 222°C, respectively, with respective peak maxima at 244 and 263°C. The increase in onset temperature is due to the ring-opening polymerization of benzoxazine and self-polymerization of triazine ring of cyanate ester resins. The onset temperature for cured CBDDM/BMCPC hybrid matrices with 0.5/1, 1/1, and 1/0.5 wt% starts at 234°C, 232°C, and 230°C, respectively, with respective maxima at 258°C, 254°C, and 249°C (Table 1). The T gs for CBDDM/BMCPC hybrid matrices shifted to higher temperature, according to the increase in the concentration of BMCPC content into CBDDM, suggesting that the incorporation of BMCPC to benzoxazine improves significantly the performance of polybenzoxazine. The appearance of a single glass transition peak in between those of the two homopolymers which implies that the CBDDM/BMCPC hybrid matrices are homogeneous and single chemical entity with no phase separation. The increase in the value of T g for cured CBDDM/BMCPC hybrid matrices is due to the formation of highly cross-linked network structure.
DSC data of CBDDM/BMCPC hybrid matrices.

Tg: glass transition temperature; DSC: differential scanning calorimeter; CBDDM: 1,1-bis(3-methyl-4-hydroxy phenyl)cyclohexane benzoxazine; BMCPC: 1,1-bis(3-methyl-4-cyanatophenyl) cyclohexane.

DSC traces of CBDDM/BMCPC hybrid polymer matrices (a) PCBDDM, (b) PBMCPC, (c) CBDDM/BMCPC (0.5/1 wt%), (d) CBDDM/BMCPC (1/1 wt%), and (e) CBDDM/BMCPC (1/0.5 wt%).
Thermal stability
The thermal stability of the CBDDM/BMCPC hybrid matrices and its homopolymers were analyzed by TGA. Figure 6 shows the TGA curves of these hybrid matrices. The TGA data of CBDDM/BMCPC hybrid matrices blended at various weight ratios are presented in Table 2. The initial degradation temperature of the homopolymers, PCBDDM and PBMCPC starts at 309°C and 394°C, respectively. The cured CBDDM/BMCPC hybrid matrices with 0.5/1, 1/1, and 1/0.5 wt% starts to degrade at 369°C, 352°C, and 341°C, respectively.

TGA curves of CBDDM/BMCPC hybrid polymer matrices (a) PCBDDM, (b) PBMCPC, (c) CBDDM/BMCPC (0.5/1 wt%), (d) CBDDM/BMCPC (1/1 wt%), and (e) CBDDM/BMCPC (1/0.5 wt%).
Thermal stability of CBDDM/BMCPC hybrid matrices.

CBDDM: 1,1-bis(3-methyl-4-hydroxy phenyl)cyclohexane benzoxazine; BMCPC: 1,1-bis(3-methyl-4-cyanatophenyl) cyclohexane; LOI: limiting oxygen index.
The degradation temperature of the cured CBDDM/BMCPC hybrid matrices for 20%, 40%, and 50% weight loss gradually shifted to higher temperature with the increase of BMCPC content, indicating that the addition of BMCPC enhanced the thermal stability of PCBDDM. The degradation temperatures of the cured CBDDM/BMCPC hybrid matrices are in between those of two homopolymers PCBDDM and PBMCPC. This is due to the formation of highly cross-linked network structure resulted between the reaction of cyanate ester and benzoxazine which contributes to an enhanced thermal stability to the CBDDM/BMCPC hybrid matrices.
Furthermore, the char yield of PCBDDM and PBMCPC is 39.2% and 41.8%, respectively (Table 2). The char yield of the cured CBDDM/BMCPC hybrid matrices with 1/0.5, 1/1, and 0.5/1 wt% was increased from 39.8% to 41.5% which is higher than that of the char yield of PCBDDM. The increase in the concentration of BMCPC content in CBDDM/BMCPC hybrid matrices increases the char yield which in turn improved the flame-retardant behavior of the hybrid matrices. The increase in char yield infers the reduction of the polymer’s flammability and ascertains their good thermal stability.
Flame retardancy
The flame-retardant behavior of CBDDM/BMCPC hybrid matrices and their homopolymers PCBDDM and PBMCPC in terms of the value of limiting oxygen index (LOI) was calculated from the char yield obtained from TGA. The data obtained from TGA (Table 2) show that there is an increase in char yield as the BMCPC content is increased in CBDDM/BMCPC hybrid matrices. The value of LOI was increased with the increase in the values of char yield. The flame resistance can be represented by LOI. The LOI values were calculated using the method reported in the literature. 36 –38
The value of LOI for homopolymers PCBDDM and PBMCPC is 33.1 and 34.2, respectively. The value of LOI for CBDDM/BMCPC hybrid matrices with 1/0.5, 1/1, and 0.5/1 wt% are increased from 33.4 to 34.1 which is higher than the value of LOI for PCBDDM. The incorporation of BMCPC content into CBDDM matrices was increased, and the values of LOI in turn improved the flame resistance. The increase in the values of LOI indicates that the CBDDM/BMCPC hybrid matrices exhibit better flame resistance.
Morphology
Scanning electron microscopy
SEM is used to investigate the morphology of CBDDM/BMCPC hybrid matrices systems (Figure 7). The SEM micrographs of the fractured surfaces of unmodified CBDDM (Figure 7(a)) indicated the presence of homogeneous microstructures. Figure 7(b) to (d) shows the CBDDM/BMCPC hybrid matrices systems indicating the presence of homogeneous morphology due to the efficient interaction between CBDDM and BMCPC. The homogeneous morphology of CBDDM/BMCPC hybrid system was further supported by visual observation studies (Figure 8).There are no phase domains separation between the two components which suggest the formation of homogeneous inter-cross-linked network structure.

SEM morphology of CBDDM/BMCPC hybrid matrices hybrid matrices (a) PCBDDM, (b) CBDDM/BMCPC (0.5/1 wt%), (c) CBDDM/BMCPC (1/1 wt%), and (d) CBDDM/BMCPC (1/0.5 wt%).
Visual observation study of CBDDM/BMCPC hybrid matrices hybrid matrices (a) PCBDDM and (b) CBDDM/BMCPC (1/1 wt%).
Conclusion
Polybenzoxazine-based hybrid matrices namely benzoxazines (CBDDM) and BMCPC-modified cyanate ester hybrid polymer matrices were developed via solvent method. The thermal and morphological properties of benzoxazine–cyanate ester-modified hybrid systems have been studied and compared with those of neat martices. DSC results reveal that the CBDDM/BMCPC hybrid matrices exhibit higher Tg s and curing behavior than that of neat hybrid matrices. The CBDDM/BMCPC hybrid matrices yielded higher char yield than those of neat hybrid matrices. The increase in the values of LOI indicates that the CBDDM/BMCPC hybrid matrices exhibit better flame resistance than that of neat hybrid matrices. Data resulted from thermal studies infer that these hybrid matrices can be utilized in the form of thermally stable encapsulation, adhesive, sealants, and matrices for different engineering applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
