Abstract
Bacterial cellulose (BC) is a promising option for ecologically viable processes. In this work, BC was produced using starch hydrolysate (rice and corn flakes) as carbon sources. The starch was hydrolyzed by fungus for glucose production. A maximum yield of 2.80 ± 0.43 g/L of BC was obtained with the hydrolyzed rice medium. Moreover, BC produced in the rice and corn media had crystallinity of around 50%. Fourier-transform infrared spectrometry confirmed the functional groups in BC as well as the absence of contaminants from production process. The thermogravimetric analysis revealed that the polymers formed with alternative media had greater thermal stability. Scanning electron microscopy revealed a morphological structure with the random arrangement of nanofibrils in a non-uniform manner distributed in the weave of both polymeric films. The biofilms produced in different media had similar properties to those of films obtained with a conventional medium, revealing that the polymeric characteristics are not modified.
Introduction
Bacterial cellulose (BC) is a biodegradable, non-toxic polysaccharide with considerable liquid retention capacity and important properties for applications in the medical field and tissue engineering.1–3 This biopolymer has similar characteristics to cellulose polymer of a vegetable origin, but it differs in terms of the degree of polymerization and purity and has more stable, resistant nanofibers with an ultra-fine reticular structure, high crystallinity, tensile strength, elasticity, and durability.4,5
Cellulose can be produced by algae, fungi, protozoa, and prokaryotic organisms. 6 Among the producers of BC, bacteria of the genus Komagataeibacter (recently reclassified from the genus Gluconacetobacter based on the phylogenetic, phenotypic, ecological, and chemotaxonomic characteristics of the 16S rRNA sequence) are gaining prominence in the production of this biopolymer of considerable scientific value.7,8 The production of pure cellulose nanofibrils by bacteria of the genus Komagataeibacter occurs through the consumption of glucose in the medium, which is metabolized into cellulose by the microorganisms. 9 Glucose can be obtained from different sources; it may be a free sugar or may be obtained through the hydrolysis of polysaccharides, such as starch. In the microbial hydrolysis of starch, filamentous fungi are the most adaptable to growth on solid substrates, can grow in environments with low humidity, and have high enzymatic activity for the hydrolysis of starch in glucose molecules.10–13
During the metabolic process, the components of the medium are assimilated by the microorganisms either through degradation or polymerization, resulting in the synthesis of specific metabolites. This metabolic process requires carbon, hydrogen, oxygen, nitrogen, and elements such as iron and zinc, which can be found in various agro-industrial byproducts. Considering their nutritional value, these byproducts can be used as substrate or raw material for microbial growth and the formation of cellulose biopolymers.14,15
Several substrates are reported in the literature as important sources of carbon and nitrogen for use in the production of BC. Costa et al. 16 used industrial waste to obtain polymers with desired properties. There are countless sources for BC production, such as olive oil residue, sago byproducts, coconut water, apple pomace, sugarcane, acerola residue, and potato starch.16–19 Considering the need to overcome the challenges of industrial-scale BC production, the use of renewable carbon sources and the optimization of bioprocesses constitute a way to reduce the high costs involved. 20
Starch stands out among the most abundant carbon sources and belongs to the class of polysaccharides that occur naturally in roots, corn, wheat, rice, barley, and potatoes. This carbohydrate is of paramount importance to the development of biodegradable films and consists of two types of glucose polymers: amylose and amylopectin.21,22 Most importantly, agro-industrial byproducts containing large concentrations of starch are accessible and inexpensive. Furthermore, grains such as rice and corn may also provide micro and macro nutrients useful for the BC production, favoring the development of new and clean biotechnologies. 15
The main motivation of this work was to evaluate two biotechnological processes. The first is the fermentation of a medium containing corn and rice starch by Aspergillus oryzae for the production of a saccharified medium. The second process is to use this saccharified medium to produce BC by Komagataeibacter rhaeticus.
Therefore, the aim of the present study was to use rice and corn starch hydrolyzed by Aspergillus oryzae in the production of BC.
Materials and methods
Microorganisms
The fungus Aspergillus oryzae was acquired from Probióticas Brasil® and used in the starch hydrolysis process. The bacterium Komagataeibacter rhaeticus was obtained from the culture collection of the Department of Antibiotics of the Federal University of Pernambuco, Brazil, and used for the production of BC.
Grains
Two grains were used as alternative carbon sources. Zea mays (corn flakes) and Oryza sativa (parboiled rice—type II) were ground in a low-speed industrial blender for 5 min and sieved with a 14 mesh, to homogenize the granulometry. Next, 120.0 g/L of the grains were transferred to two 1000-mL Fernbach flasks. The fungus Aspergillus oryzae was inoculated at a concentration of 1.8 × 106 spores/mL for 72 h at 28°C and sterilized in a Prismatec-CS autoclave for 20 min at 121°C and 1 atm.
Conservation medium
The Hestrin–Schramm (HS) culture medium used for the conservation and control of the bacterium was prepared with the following composition: glucose (C6H12O6; 30 g/L), peptone (5 g/L), yeast extract (5 g/L), disodium phosphate (Na2HPO4. H2O; 5.1 g/L), citric acid (C6H8O7; 1.15 g/L), and water. The solid preservation medium had the same composition with the addition of agar (18 g/L).
Starch hydrolysis
For glucose production through the hydrolysis of starch, two inoculums of 50 mL were prepared, both containing the Czapek-Dox (CZ) medium modified with corn flakes or rice as a carbon source. The CZ medium had the following composition: 3.0 g/L of NaNO3, 0.01 g/L of FeSO4.7H2O, 0.5 g/L of KCl, 1.0 g/L of K2HPO4, 0.5 g/L of MgSO4.7H2O, 120.0 g/L of ground corn flakes or rice, and distilled water. The two modified CZ media were autoclaved for 20 min at 121°C and 1 atm. The fungus at a concentration of 1.8 × 106 spores/mL was then inoculated and incubated for 72 h at 30°C to obtain the inoculum.
Each inoculum was added to a 1-L Fernbach flask containing 60 g of previously autoclaved ground corn flakes or rice. After 72 h of fermentation at 30°C, 500 mL of distilled water was added to each fermentation system. The systems were then heated at 45°C in a water bath for 60 min to optimize the starch hydrolysis process. The saccharified starch undergoes heating at 121oC for 20 min to sterilize it, as well as to deactivate the enzymes. Then, it was filtered and used to prepare the must for cellulose production.
Bacterial cellulose production
The alternative hydrolyzed media were both supplemented with Na2HPO4 at a concentration of 2.7 g/L. The media were also supplemented with glucose up to a concentration of 20 g/L, if necessary, thereby obtaining the cellulose production medium.
To prepare the inoculum, 0.6 g/L of microbial cells were used and the medium was incubated for 3 days at 30°C. At the end of this period, the inoculum (10% v/v) was transferred to each of the Roux flasks containing 400 mL of the production medium and incubated for 30 days at 30°C.
For the purposes of comparison, 400 mL of the HS liquid medium (control) was used for the culturing of standard BC. The media were sterilized in a vertical autoclave for 20 min at 121°C and 1 atm. The control medium (10% (v/v) of inoculum) was incubated for 30 days at 30°C. Once produced, the pellicle was washed under tap water. The 3,5-dinitrosalicylic acid (DNS) method was used to identify the remaining glucose concentration in the fermented HS, rice, and corn flake media.
Purification and drying
The BC pellicles were washed with distilled water and a 0.1 M solution of NaOH at 80°C for 20 min in a water bath to eliminate all retained bacterial cells. The pellicle was washed in deionized water until neutral pH was achieved. The hydrated BC pellicles were placed on a non-stick plate, dried in a freezer at −2°C for 24 h and weighed. For the remaining media, glucose concentrations were measured using the DNS method.
Characterization of BC films
Fourier-transform infrared spectrometry—The dried BC membranes were mounted on a support and analyzed in the Shimadzu IR Tracer-100 spectrometer. The spectrum was obtained in the wavelength range of 400–4000 cm−1 to characterize the chemical structure of the biomembrane through the identification of functional groups.
Thermogravimetric Analysis (TGA)—Readings were performed using a simultaneous thermal analyzer (5000, PerkinElmer) on samples of about 5 mg. Each sample was scanned over a temperature range 30–600°C with a heating rate of 10 °C/min.
Scanning Electron Microscopy—The dried BC pellicles were mounted on a copper stub using double adhesive carbon conductive tape and coated with a layer of gold for 30 s (SC-701 Quick Coater, Japan). The SEM photographs were obtained using a scanning electron microscope (TESCAN, MIRA 3, Czech Republic) operating at 10 kV at room temperature.
X-Ray diffractometry—All BC membranes were analyzed using a diffractometer (Rigaku) with Cu Kα radiation. The crystallinity index was determined using Segal’s equation given below
Statistical analysis
The data obtained from the experiments, especially those related to yields, were analyzed using descriptive statistics techniques and analysis of variances to compare the BC production of the media from hydrolysates between them and in relation to the HS control media. All the experiments relative to the production of cellulose were carried in triplicate. All the data analysis was realized in Microsoft Excel ®.
Results and discussion
Bacterial cellulose production
Mass and yield of dehydrated BC films produced in alternative and standard media according to concentration of glucose produced from hydrolysis.
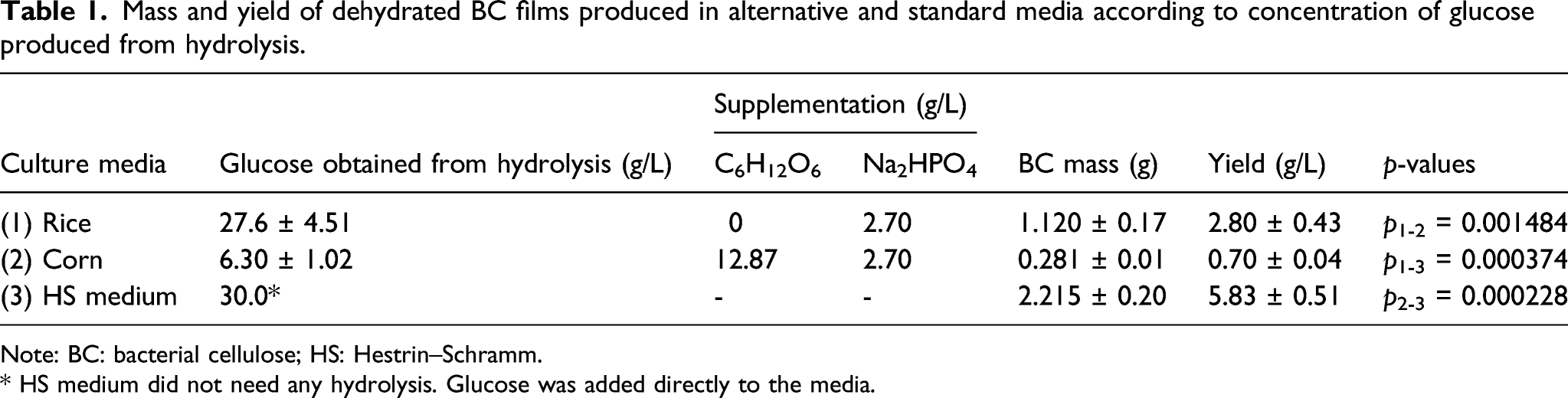
Note: BC: bacterial cellulose; HS: Hestrin–Schramm.
* HS medium did not need any hydrolysis. Glucose was added directly to the media.
A comparison of the production of biopolymers reveals the influence of starch in the process. Due to the difficulty filtering the medium containing the corn hydrolysate, part of the starch remained in the composition of the medium, leading to a lower yield even with glucose supplementation.
Compared to the control medium, with which the yield of BC was 5.83 ± 0.51 g/L, the alternative, low-cost production involving the fermentation of rice had an efficiency was around 50%. It can also be seen, analyzing the p-values, that the cellulose production means were statistically different between them at a 95% confidence level. In the literature, average BC production using the standard HS culture medium is 3.26 g/L at the end of 14 days of culture.24,25 BC can be produced from residual fiber sludge hydrolysates, which is waste originating from cellulose factories consisting mainly of cellulose and hemicellulose. Hydrolysis was performed with the fungus Trichoderma reesei and BC was produced from the resulting hydrolysate using the microorganism K. xylinus, with yields of up to 11 g/L after 7 days of cultivation. 26 It is also possible to produce hydrolysates from fruit peels, such as durian peel, which is a typical fruit from Asia, through hydrolysis with mineral acids. The durian peel was hydrolyzed with diluted sulfuric acid and the product was used as a culture medium for BC growth (using K. xylinus), achieving a maximum yield of 2.67 g/L after 8 days. 27
Another study used orange peel as a raw material, in which two enzymes (cellulase and pectinase) were used for hydrolysis, obtaining BC films with a yield of 3.16 g/L after 8 days in an aqueous medium using K. xylinus as the producing microorganism. 28 Extending this work, citrus peels (lemon, tangerine, grapefruit, and orange) were used for the obtainment of hydrolysates to produce BC, resulting in yields ranging from 2.06 to 3.92 g of BC/g of peel after 21 days of culture with the microorganism K. hansenii 29 Thus, there is a good equivalence of yields between the hydrolysates produced in the present study with those described in the literature in recent years.
Studies used glucose from starch hydrolysis to produce BC. Uzyol & Saçan (2017) used glucose from seaweed starch hydrolyzed by commercial alpha-amylases, as well as acid hydrolysis, obtaining a yield of 1.104 g/L. 30
Other researchers sought to optimize BC production using low quality organic rice starch (red jasmin, Homnil rice, Riceburry rice, brown jasmine rice, and jasmine rice). The study showed that milled rice powder requires enzymatic hydrolysis with alpha-amylases and glucoamylase, both of commercial origin, to completely convert starch to reduced sugar. This research offers evidence for the potential of organic low-grade rice to be used as an effective carbon substrate for producing cellulose by Komagatacibacter nataicola. 31
Based on the yield results, some factors are decisive to the production of BC. During the hydrolysis of rice by the fungus, several substances are produced from its metabolism, such as sorbitol, glycerol, and xylitol, in addition to amino acids that arise from the proteases synthesized by A. oryzae. 32 These alcohols can directly promote the fermentation of bacteria as well as the amino acids produced. Therefore, with these substances, it is possible that bacteria in the medium containing rice have more resources for BC synthesis.33,34
Figure 1 shows the polymers before drying. The amount of biofilm produced with the rice medium was greater and the film was more homogeneous throughout its surface. With the corn medium, part of the polymer was not formed as a homogeneous film. This may be explained by the presence of starch in the culture medium, and, consequently, in the structure of the formed BC, which may have hindered polymerization and, consequently, the formation of a uniform film. BC films produced in HS (a) corn, (b) rice, and (c) media. Note: BC: bacterial cellulose; HS: Hestrin–Schramm.
Characterization of BC films
Thermogravimetric analysis
Figure 2 shows the curves of the TGA/DTG analysis of the BC produced from the HS (Figure 2(a)), corn starch hydrolysate (Figure 2(b)), and rice hydrolysate (Figure 2(c)) media. The samples of the media with the corn and rice hydrolysates obtained similar results, with two stages of decay during the analysis. In the rice sample, the loss of about 18% of mass around 100°C was due to the loss of water related to cellulose dehydration. This loss continued to a temperature of 170°C, reaching 25% of the total mass. For corn, the loss was only 12%. In second decay (at 230–440°C), the loss was related to the degradation of cellulose due to the dehydration, depolymerization, and decomposition of glycosidic units,
33
with only 33% of the original mass remaining in each biofilm. This behavior has been reported for similar materials analyzed in the literature, with mass losses of 9–12% in the initial phase and 70% in the second decay.35,36 TGA and DTG curves for BC films produced in HS (a) corn, (b) rice, and (c) media. Note: BC: bacterial cellulose; HS: Hestrin–Schramm; TGA: thermogravimetric analysis.
The degradation process that occurred in the polymers produced with fermented rice and corn was similar to that of pure cellulose. Figure 2(c) shows a slight loss of moisture around 100°C due to the evaporation of the small amount of water remaining after the drying process. In the range of about 280°C–350°C, there was the loss of mass characteristic of the thermal decomposition of cellulose due to the depolymerization, dehydration, and decomposition of the glycosidic units, with the subsequent formation of carbonaceous residues, and, above 350°C, there was thermo-oxidative degradation.37,38 The mass losses related to each event were obtained through the graphs created in the OriginPro 8.5 software (OriginLab Corporation, USA).
X-ray diffractometry
For the samples produced in the rice medium, the three characteristic peaks of BC in 2θ are indicated in the graph, with values of 15.06°, 17.28°, and 23.20°. The samples produced in the corn medium, however, present a different diagram than expected, with peaks at 15.52°, 17.92°, 21.28°, and 23.34°. This distinction is due to the presence of starch in the culture medium, and, consequently, the structure of the polymer produced. Thus, the low intensity peaks express the presence of starch in the polymeric structure during the analysis (Figure 3). DRX diagrams for BC films produced in corn, rice, and HS media. Note: BC: bacterial cellulose; HS: Hestrin–Schramm.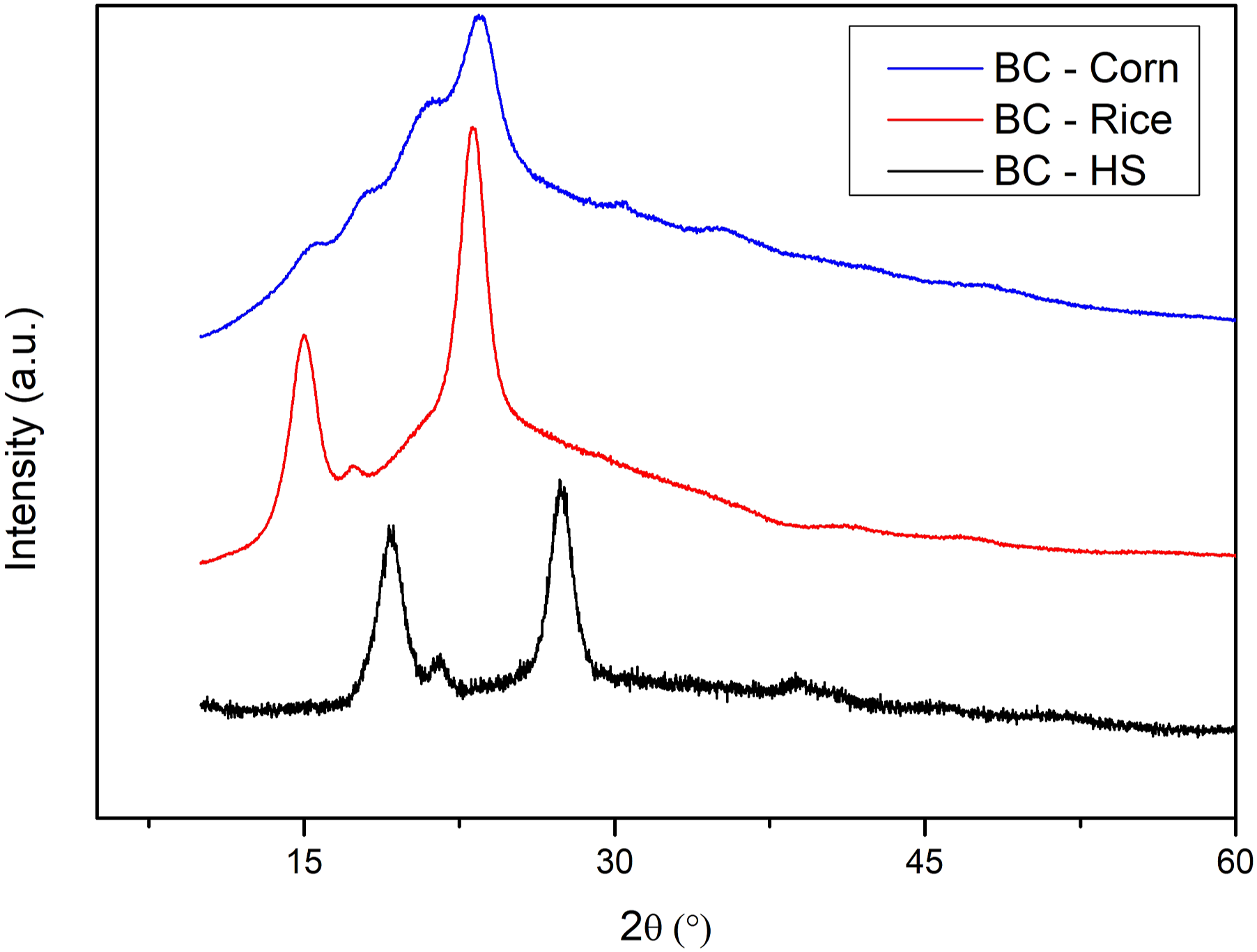
The peaks of the samples obtained from the corn medium express behaviors similar to those found in the literature, according to which, peaks found at 15°, 17°, 18°, and 23° are characteristic of native corn starch samples, which indicates the adherence of the starch present in the culture medium to the biofilm structure. The peaks for the rice samples are similar to those for other biofilm samples obtained using different methods,35,36,39 indicating peaks of pure BC, which are highlighted by the values of approximately 15°, 17°, and 22°. Thus, it is possible to confirm the composition of the film produced from the rice hydrolysate.
The degree of crystallinity of the biopolymers was calculated using equation (1). The result was 55% for the BC produced in the rice and corn media, indicating low crystallinity compared to values commonly obtained for BC, which vary from 80 to 90%. The low crystallinity obtained in this experiment is close to the 61.8% for the BC produced by Salari et al. 24 with cheese whey. The cellulose produced using HS medium had 79.4% crystallinity. The different degrees of crystallinity may be associated with substances in the culture medium that affect the organization of the crystals. Such substances can be present in agro-industrial waste and carbohydrates.35,40
The infrared analysis confirmed the functional groups in the BC and identified that the two samples had coherent results with each other, although there may be the presence of starch in the BC, since the vibrational bands in the Fourier-transform infrared (FTIR) of starch coincide with those of BC, as both are glucose polysaccharides. Figure 4 displays the spectra corresponding to this analysis. FTIR spectra for BC films produced in hydrolyzed corn, hydrolyzed rice, and HS media. Note: FTIR: Fourier-transform infrared; BC: bacterial cellulose; HS: Hestrin–Schramm.
Main characteristic vibrations of FTIR for BC films produced from hydrolysis of corn and rice starch.

Note: FTIR: fourier-transform infrared; BC: bacterial cellulose.
Scanning electron microscopy
The micrographs of the surface of the BC films produced in HS, hydrolyzed rice, and hydrolyzed corn media under the conditions established in the present study are shown in Figure 5. The images show the dispersion of the fibers and the presence of possible starch granules in the BC pellicles produced with the hydrolysate of these carbohydrates. Micrographs of surfaces of BC pellicles produced in HS (a) hydrolyzed rice, (b) hydrolyzed corn, and (c) media. Note: BC: bacterial cellulose; HS: Hestrin–Schramm.
The morphological structure of the BC produced is defined by the random arrangement of nanofibrils not uniformly distributed throughout the weave formed in the film. Areas of denser fibrillar structures are seen, whereas other areas have more dispersed fibers, making the structure more porous. Saska et al. 37 state that this structure is usable for production processes, as the removal of water from the 3D structure would result in the approximation of nanofibers that would be randomly aggregated through the formation of hydrogen bonds.
The presence of starch granules on the surface of the BC produced with hydrolyzed starch shows that the hydrolysis process can be optimized by increasing the concentration of free glucose and, consequently, obtaining a greater yield of cellulose membranes.
Conclusion
The starch hydrolysis process using the fungus Aspergillus oryzae provides a new alternative for the production of BC using grains and tubers, since the glucose in many grains and tubers is not available to cellulose-producing microorganisms and, thus, there is no energy for the production of BC. The hydrolysis of starch makes glucose available as a carbon source in fermentations, enabling alternative means to be used for BC production. The proposed media enable the production of BC while eliminating the need for synthetic glucose, thereby reducing the cost of the production of biopolymers.
Given the results regarding the characterization of BC in this work, several potential applications of BC may be proposed, including its use in active packaging and paper manufacturing, in addition to valuing this material with a nutritional purpose similar to Nata de Coco (coconut cream). Furthermore, other application areas may also be attractive as support material for sensors, active bandages for wounds, among others.
Footnotes
Acknowledgments
The Research and Development Program from Coordination for the Improvement of Higher Level Education Personnel (CAPES) funded this study and Fundação de Amparo a Ciência e Tecnologia de PE (FACEPE).
Author’s contributions
All authors contributed in this work. Karina C. de Souza and Gabriela R. dos Santos carried out the experiments and wrote the manuscript., Felipe C. S Trindade edited and analyzed the data. Andréa F. De S. Costa, Yeda M. B. de Almeida, Leonie A. Sarubbo and Glória M. Vinhas designed the project, analyzed the data and performed manuscript editing and final improvement.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

