Abstract
In this study, 2-{[(2H-1,3-benzodioxol-5-yl)methyl]amino}-2-oxoethyl 2-methylprop-2-enoate (BMAOME) monomer was synthesized, and copolymers were prepared with glycidyl methacrylate (GMA). Structural characterizations of the compounds were performed using FTIR, 1H-, and 13C-NMR techniques. Monomer reactivity ratio values were calculated by Finemann–Ross (FR) and Kelen–Tudos (KT) methods. The Tg value of the polymers was determined by differential scanning calorimetry (DSC) and their thermal stability was determined by thermogravimetric analysis (TGA). The molecular weights (w and n) and polydispersity index of the polymers were determined by gel permeation chromatography. The Ea value of thermal decomposition was determined by using the Ozawa and Kissinger methods. The photo-stability of the copolymers was investigated. Furthermore, the photo-stability of the copolymers and the biological activity of polymers against different types of bacteria and fungi were investigated.
Keywords
Introduction
Due to their unique properties and wide range of applications, the interest in functional polymers is increasing day by day both in academia and industry. The properties of such materials are usually determined by the presence of polar or ionic functional groups in the side branch. Functional polymers allow the polymer to adapt to a wide variety of applications beyond its mechanical properties. Also, polymers with desired functions can be easily prepared by using monomers with appropriate functional groups.1–3
Copolymerization offers flexibility with respect to possible monomers and a great diversity of polymeric products, and composition drift, which may need to be minimized or exploited in a controlled way, for specific structure–property relationships. The reactivity ratio is a measure of the tendency for a comonomer to show a preference for insertion into a growing chain in which the last inserted unit was the same, rather than the other comonomer. 4
The benzopiperonyl amine skeleton is an essential template for many biologically active compounds. This molecule and its derivatives are known to be potent antitumor agents, calmodulin antagonists, neurotransmission blocking, and neuroprotective agents. 5 A previous study showed that piperonyl acetate, a piperonyl derivative, has no dermatological effect when used as a toxicological fragrance. 6 Cypermethrin (CY), which contains the piperonyl group, is a frequently used class II pyrethroid pesticide, and piperonyl butoxide (PBO) plays an essential role in the pesticide formulation of synthetic pyrethroids. 7
Poly methyl methacrylate (PMMA) and related acrylic and methacrylic polymers have been widely applied in medicine because of their low cost, straightforward polymerization initiation by free radical or anionic mechanisms, ease of processing, and generally inert quality. Application areas include orthopedics, dentistry, controlled-release systems, cosmetics, and ophthalmology. 8 PMMA has a light transmittance of 92% and is also used to produce transparent nanocomposites reinforced with nano-filler reinforcement. 9
The epoxy group carried by the glycidyl methacrylate (GMA) monomer reacts with various functional groups, leading to the formation of new modified polymers. These functionalized polymers can be used as curing agents to harden engineering plastics or to improve the compatibility of10–13 mixing systems. GMA-based copolymers are of great importance in biotechnological applications. In addition, homogeneous and heterogeneous polymer network structures prepared with GMA are used in coatings, matrix resins, and adhesives. 14
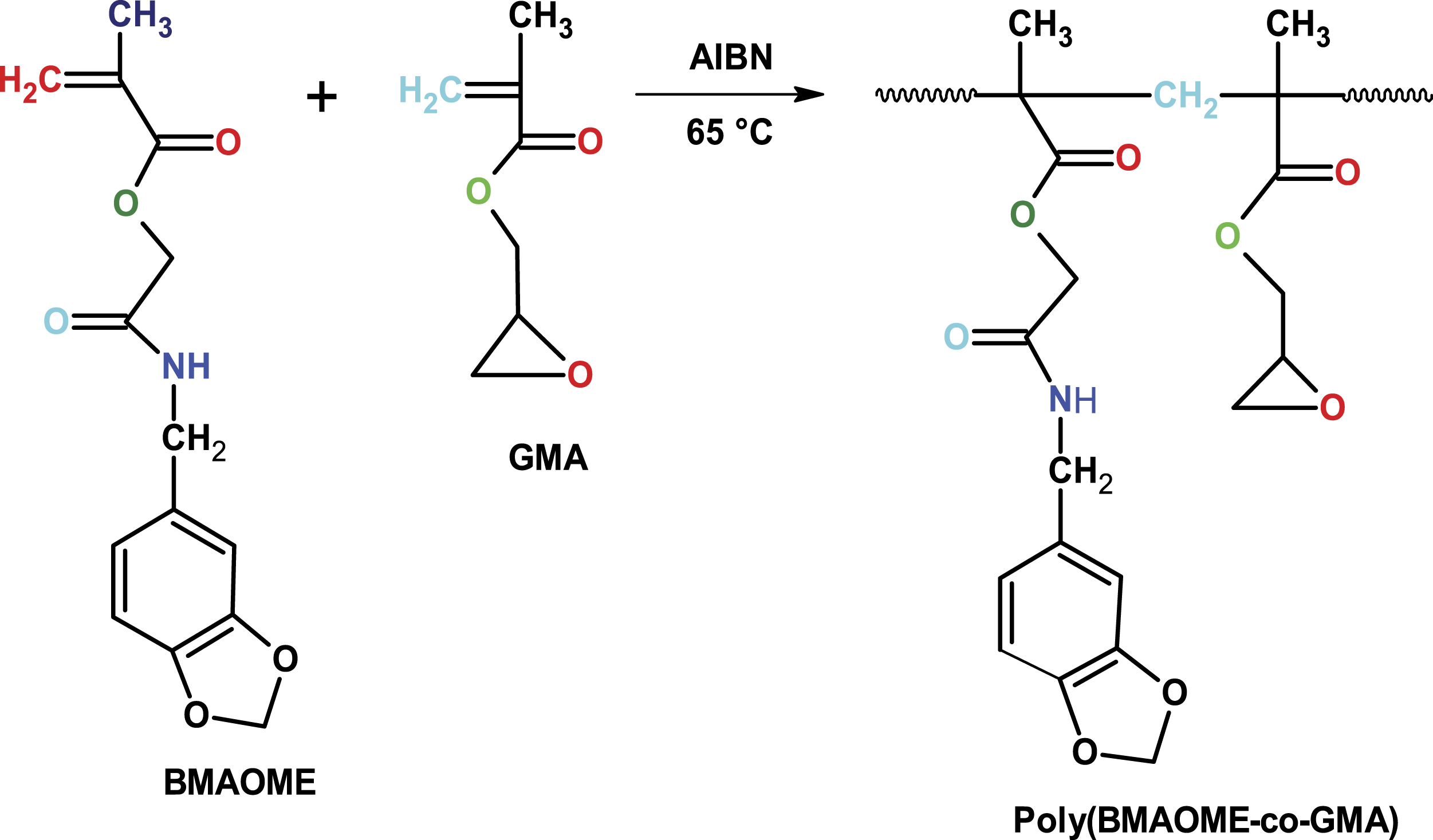
In this study, first, a methacrylate monomer (BMAOME) carrying benzopiperonyl in the side branch was synthesized. The copolymers of BMAOME with GMA were prepared by the free radical polymerization method. Then, the monomer reactivity ratios were calculated and the thermal degradation kinetics and biological activities of the copolymers were investigated. The results were discussed in detail and revealed a relationship between structure and activity.
Experimental
Materials
Chloroacetyl chloride, sodium methacrylate, and piperonylamine (Sigma) were used as in their original form. Ethanol, dichloro methane, chloroform, and n-hexane were freshly purified in molecular sieves before their use. 1,4-dioxane, acetonitrile, and potassium carbonate (Sigma) were used as in their original form. Azobisisobutyronitrile (AIBN) was recrystallized from a chloroform–methanol mixture (v/v:3/1).
Characterization techniques
FTIR spectra were measured on a Perkin–Elmer Spectrum BX FT-IR spectrometer. 1H- and 13C-NMR spectra were recorded in DMSO with tetramethylsilane as the internal standard using a Bruker GmbH DPX-400 400 MHz spectrometer. The glass transition (Tg) temperatures were determined by DSC with a Shimadzu DSC60H. Samples of about 5–8 mg held in sealed aluminium crucibles and the heating rate of 10oC/min under a dynamic nitrogen flow (5 L/h) were used for the measurements. The thermal stabilities of the polymers were investigated by TGA with a Shimadzu TG60H in a nitrogen stream at a heating rate of 20oC min−1. Molecular weight characteristics of the polymers were determined by e waters 410 GPC equipped with a differential RI detector and calibrated with polystyrene standards.
Synthesis of the BMAOME monomer
Monomer synthesis was carried out in two steps. In the first step, the piperonylamine compound was reacted with chloroacetyl chloride as described in the literature and the N-(2H-1,3-benzodioxol-5-yl)-2-chloroacetamide compound was prepared. 15
In the second step, N-(2H-1,3-benzodioxol-5-yl)-2-chloroacetamide (0.004 mol, 0.98 g), sodium methacrylate (0.004 mol, 0.47 g), and 30 mL acetonitrile at 75–80°C was kept in a reflux condenser for 24 h and thus the BMAOME monomer was obtained. After the reaction, the mixture was cooled and filtered, and the solvent was removed by the evaporator. The monomer obtained was purified by crystallization of ethanol (yield 78%). Scheme 1 shows the reaction flow. Synthesis of BMAOME monomer.
IR (neat), cm−1: 1682 (C=O for amide), 1724 (C=O for ester), 1633 (CH2=C-), 1600 (C=C), 3235 (-NH), and 3100 (ArC-H).
1H-NMR (δ, ppm from TMS in CDCl3): 6.6–7.1 (aromatic protons, 3H); 5.6 (CH2=, 1H); 6.2 (CH2=, 1H); 8.6 (-NH-, 1H); 1.9 (CH3-, 3H); 4.4,4.6(OCH2, 2H, 2H); 6.0(OCH2, 2H in piperonyl ring).
13C-NMR (δ, ppm from TMS in CDCl3): 165, 168 (
Synthesis of poly(BMAOME-co-GMA)
The copolymerization of BMAOME with GMA using different feed ratio was carried out in glass ampoules under N2 atmosphere in 1,4-dioxane solution with AIBN (1 wt. % concerning the total monomer weight) at constant monomer concentration (2 mol/L) in an oil bath at 65 ± 0.1oC for a given reaction time. After the polymerization was complete, all copolymers were precipitated in ethanol. The copolymers were then dissolved in CH2Cl2 and reprecipitated in ethanol for purification. The resulting copolymer was dried to constant weight in a vacuum oven at room temperature. The BMAOME monomer was polymerized under the conditions in which the copolymers were obtained.
Antimicrobial activity of the polymers
The antimicrobial effects of compounds on various microorganisms were investigated in DMSO solution at 100 μg concentration. To do this, we used Staphylococcus aureus ATCC 29,213, Escherichia coli ATCC 25,922, Pseudomonas aeruginasa ATCC 27,853, Proteus vulgaris, Salmonella enteritidis, and Klebsiella pneumoniae as bacteria, and Candida albicans CCM 31 as fungi. The results were standardized against penicillin G under the same conditions. The medium cell culture of Yeast Extract Peptone Dextrose (YEPD) was prepared according to the methods in the literature. 16 The standard deviation (SD) values of the detected biological activity results were calculated. The mean ± standard deviation (SD) values of the results were obtained by one-way analysis of variance using SPSS software, where the level of significant difference was determined as p <0.05.
Photo-stability of the copolymers
A series of studies were conducted to determine the photo-stability of the synthesized copolymers. Solutions of each polymer with a specific concentration in dimethyl sulfoxide (DMSO) were prepared and used for the photo-stability experiments. A quartz cuvette was used as a photoreactor. Two Philips black lamps (8 W) at 365 nm were used as the UV light source. The photo-stability of the polymers was characterized using the UV-visible spectrometer (UV-1700 PC Shimadzu device, Japan) for different time intervals.
Results and Discussion
Characterization of the poly(BMAOME-co-GMA)
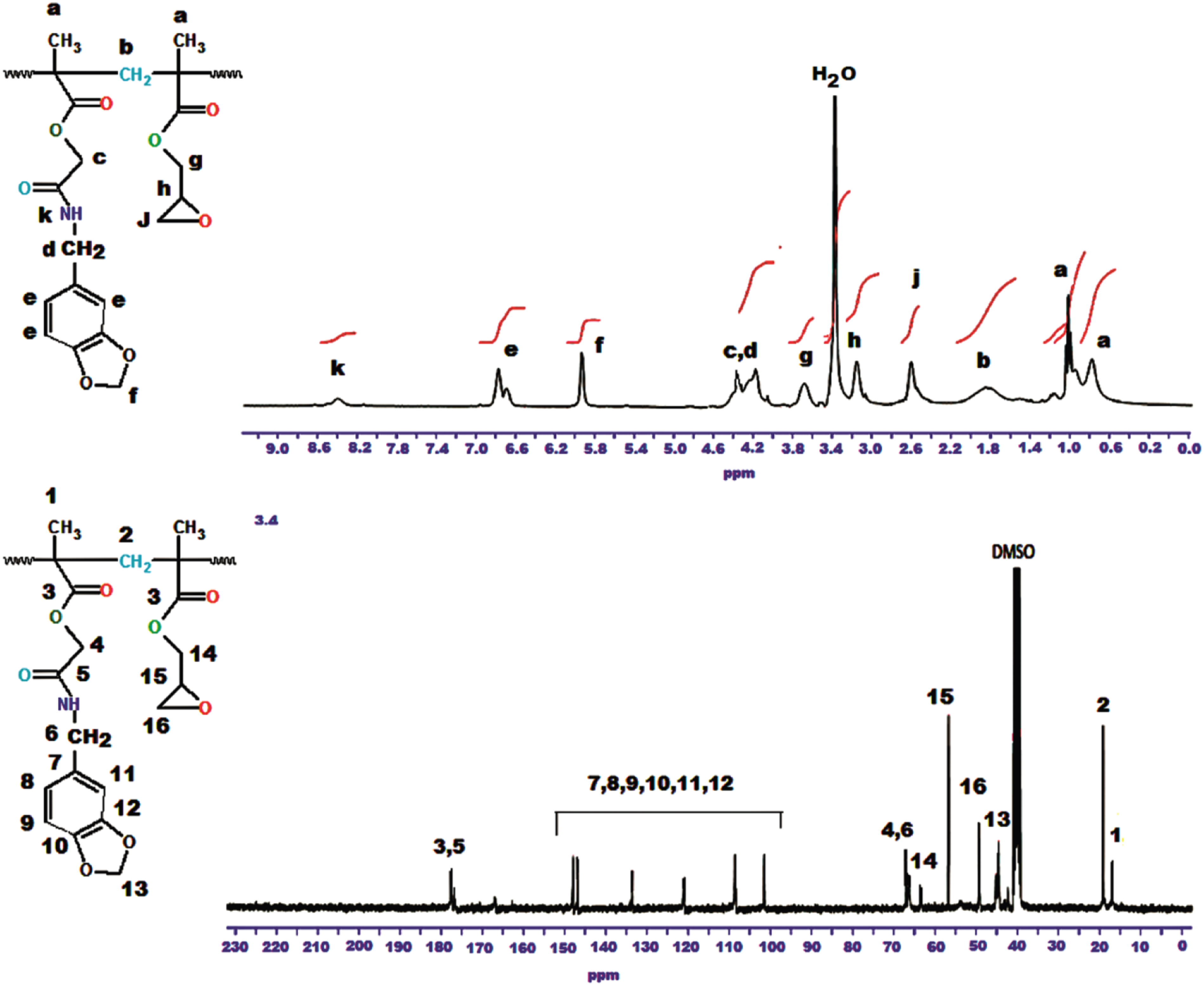
Figure 1 shows the 1H-NMR spectrum and peak evaluation of the BMAOME monomer. Table 1 gives the experimental data for six BMAOME-co-GMA copolymers of different compositions, and Scheme 2 illustrates the formula of poly(BMAOME-co-GMA). 1H-NMR spectrum of poly(BMAOEM). Feed and composition ratio of monomers in the copolymer. Formation of the copolymer.

Copolymers were characterized using FTIR, 1H- and 13C-NMR techniques. Figure 2 shows the FTIR spectrum of poly(BMAOME-co-GMA) [46:54]. The peaks corresponding to aromatic C-H stretch were observed to be at 3058 cm−1. The asymmetrical and symmetrical stretching due to the methyl and methylene groups were observed at 2995, 2948, and 2892 cm−1. The shoulder at 1728 cm−1 and peak at 1682 cm−1 were attributed to the ester and amide carbonyl stretching of BMAOME and GMA units. Figure 3 shows 1H- and 13C-NMR spectra of poly(BMAOME-co-GMA) [46:54] and characteristic peaks in both spectra corresponding to the copolymer formula. The peaks were consistent with the molecular structure in both spectroscopic curves. FTIR spectrum of the poly(BMAOME-co-GMA) (0.46:0.54). 1H-NMR and 13C-NMR spectrum of the poly(BMAOME-co-GMA) (0.46:0.54).
Determination of monomer reactivity ratios
The compositions of the poly(BMAOME-co-GMA) samples were determined by elemental analysis. Table 1 presents the obtained results.
FR and KT parameters for the poly(BMAOME-co-GMA) system.

α = (Hmax.Hmin)1/2=1.7416. M1 and M2 are molar fractions of the monomers in the feed. ml and m2 are the molar fractions in the copolymer composition.
According to the FR method, the monomer reactivity ratios can be obtained as follows


Alternatively, the reactivity ratios can be obtained with the KT method, which is based on the following equation

And α is a constant equal to (Hmax.Hmin)1/2, Hmax and Hmin being the maximum and minimum H values, respectively, from the series of measurements. From a linear plot of η as a function of ξ, the values of η for ξ = 0 and ξ =1 can be used to calculate the reactivity ratios according to the following equations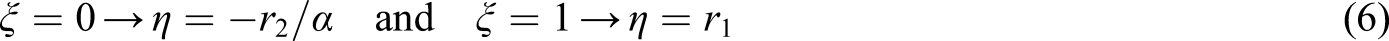
The graphical plots concerning the methods previously reported are given in Figures 4(a) and (b), whereas the reactivity ratios are summarized in Table 3. (a) KT plots and (b) FR plots for determining the monomer reactivity ratios in the copolymerization of poly(BMAOME-co-GMA). Monomer reactivity ratios for free-radical copolymerization of the BMAOME-GMA system. aReactivity ratio of BMAOME. bReactivity ratio of GMA.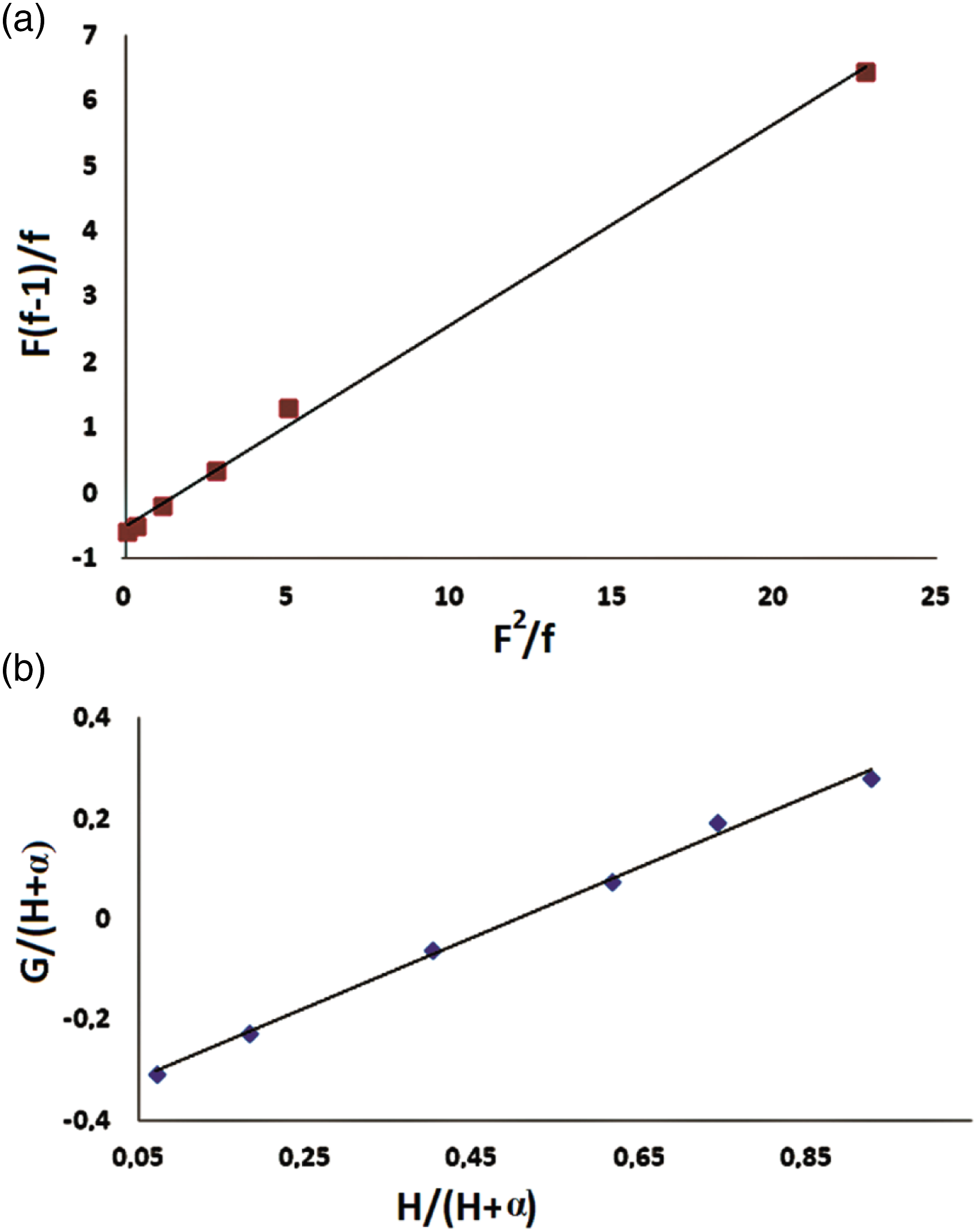

Calculated r1 and r2 values by FR and KT methods were less than one. The rGMA value was greater than the rBMAOME value. This result indicates that GMA had higher reactivity compared to BMAOME. Calculated r1 and r2 values close to zero means that this type of copolymer has an alternative tendency. In the BMAOME-GMA system, monomers that are the active centers always tend to add other types of monomers. The monomers were neatly lined up in the polymer chain. The entry of monomer molecules into the copolymer chain does not depend on the starting composition. In such copolymeric systems, alternating copolymers containing predominantly methacrylate monomer were formed. 19
Thermal stability of the polymers
Figures 5 and 6 shows TGA and DTG curves for poly(BMAOME) and three copolymers, respectively. The thermal degradation of poly(BMAOME) took place in one step. Decomposition started at 285°C and ended at about 490°C. The temperature value at which 50% mass loss occurred was 360°C. Also, the amount of residue left at 600oC was around 11%. The initial decomposition temperature of poly(GMA) was 184oC, and the residue left at 600oC was measured as 2%. The high thermal stability of poly(BMAOME) is the functional group diversity caused by the piperonyl group in the side branch. In particular, dipole–dipole interactions of ester carbonyl groups create secondary forces between the polymer chains, increasing thermal stability. The initial decomposition temperatures of the copolymers were observed to be between the homopolymers’ values as expected.
20
According to the TGA data, the thermal stability of the copolymers was observed to be between 220 and 262oC depending on the composition. Table 4 shows some thermal properties of the polymers. TGA curves for poly(BMAOME) and some copolymers. DTG curves for poly(BMAOME) and some copolymers. TGA data of the some copolymers. aInitial decomposition temperatures.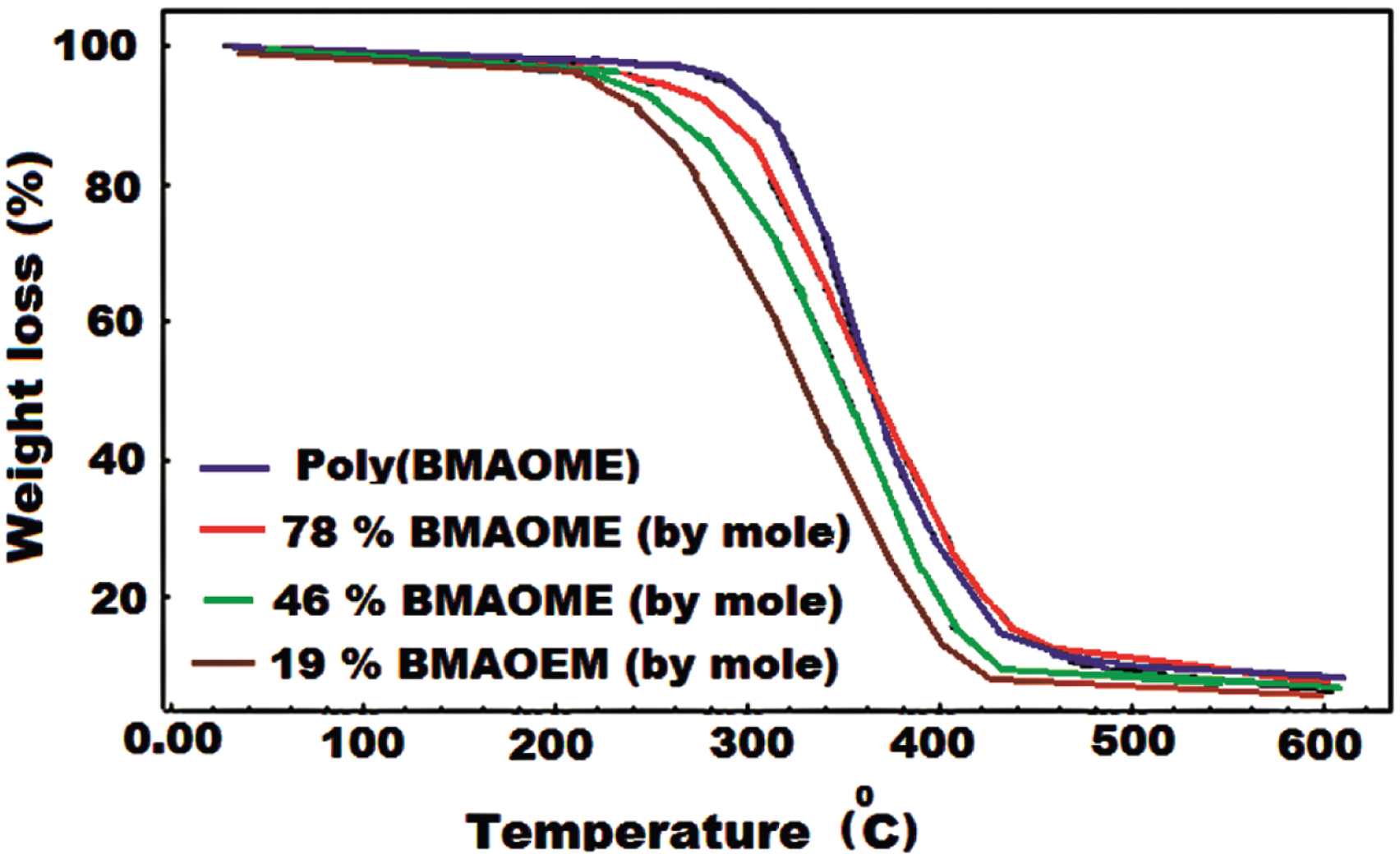


Thermal degradation of the methacrylate polymers takes place in the form of breakage in the main chain, which is called depolymerization; degradation continues with ester decomposition at high temperatures, eliminating small molecules, and the cyclic anhydride structure develops as the temperature rises further. 21
Tg’s of the polymers
DSC curves are shown in Figure 7 for certain copolymers. The pure poly(BMAOME) and poly(GMA) showed single glass transition temperatures, at about 112oC and 74oC, respectively. The glass transition temperature values of copolymers increased with the increase of BMAOME units in the copolymer. H bonds formed between highly electronegative O atoms and NH groups and strong dipole–dipole interactions between polar ester and amide groups cause a decrease in the free volume of the BMAOEM monomer and an increase in the Tg value. The values of Tg for copolymers are between the values of the homopolymers in question. DSC curves of the polymers heating rate of 10 0 C/min.
The experimental Tg value in copolymers can be determined with the Gibbs–Di Marzio
22
equation (7)

Comparison of observed Tg value of copolymers.

aWeight fraction of BMAOME in the copolymer.
bWeight fraction of GMA in the copolymer.
cTg Calculated by the Gibbs–Di Marzio equation.
dTg Calculated by the Fox equation.
eTg Observed in the DSC curve.
Thermal degradation kinetics
Non-isothermal methods are based on the ratio of the temperature difference between the sample and the reference material receiving equal heat to the temperature of the reference material. 24 The most widely used non-isothermal methods for determining the thermal activation energy are known as Kissinger and Ozawa. For kinetic studies, thermograms of all polymer samples were obtained at four different heating rates (5.0, 10.0, 15.0, and 20.0oC min−1) in an inert N2 atmosphere.
The Kissinger method
The Kissinger method 25 has been used in the literature to determine the activation energy of solid state reactions from plots of the logarithm heating rate versus the inverse of the temperature at the maximum reaction rate in constant heating rate experiments.
With the Kissinger method, the activation energy can be determined using equation (6) given below 
where β is the heating rate, Tp is the temperature corresponding to the rate peak maximum position, and R is the ideal gas constant. Using the Kissinger method and from the recorded experimental data, the activation energy of polymers was calculated from a straight line fit of a plot of ln(β/T
p
2
) versus 1000/Tp. The activation energy value of poly(BMAOME) was determined to be 137.37 kJ/mol. The Kissinger graphs for poly(BMAOME) and poly(BMAOME 46%-co-GMA) are shown in Figure 8, while the values for the copolymers are given in Table 6. From the TGA data, it was determined that the thermal stability of poly(BMAOME) was higher than that of poly(GMA). This may be due to strong dipole–dipole interactions between poly(BMAOME) units and strong H bonds caused by amide groups. These are the reasons why thermal stability increases as the amount of BMAOME in the copolymer increases. The Ea values of the copolymers were between 128.49 and 105.67 kJ/mol calculated by using the Kissinger method. Kissinger graphs of (a) poly(BMAOME) and (b) poly(BMAOME-co-GMA) (0.46:0.54). Activation energies (Ea) obtained by the Kissinger equation for polymers.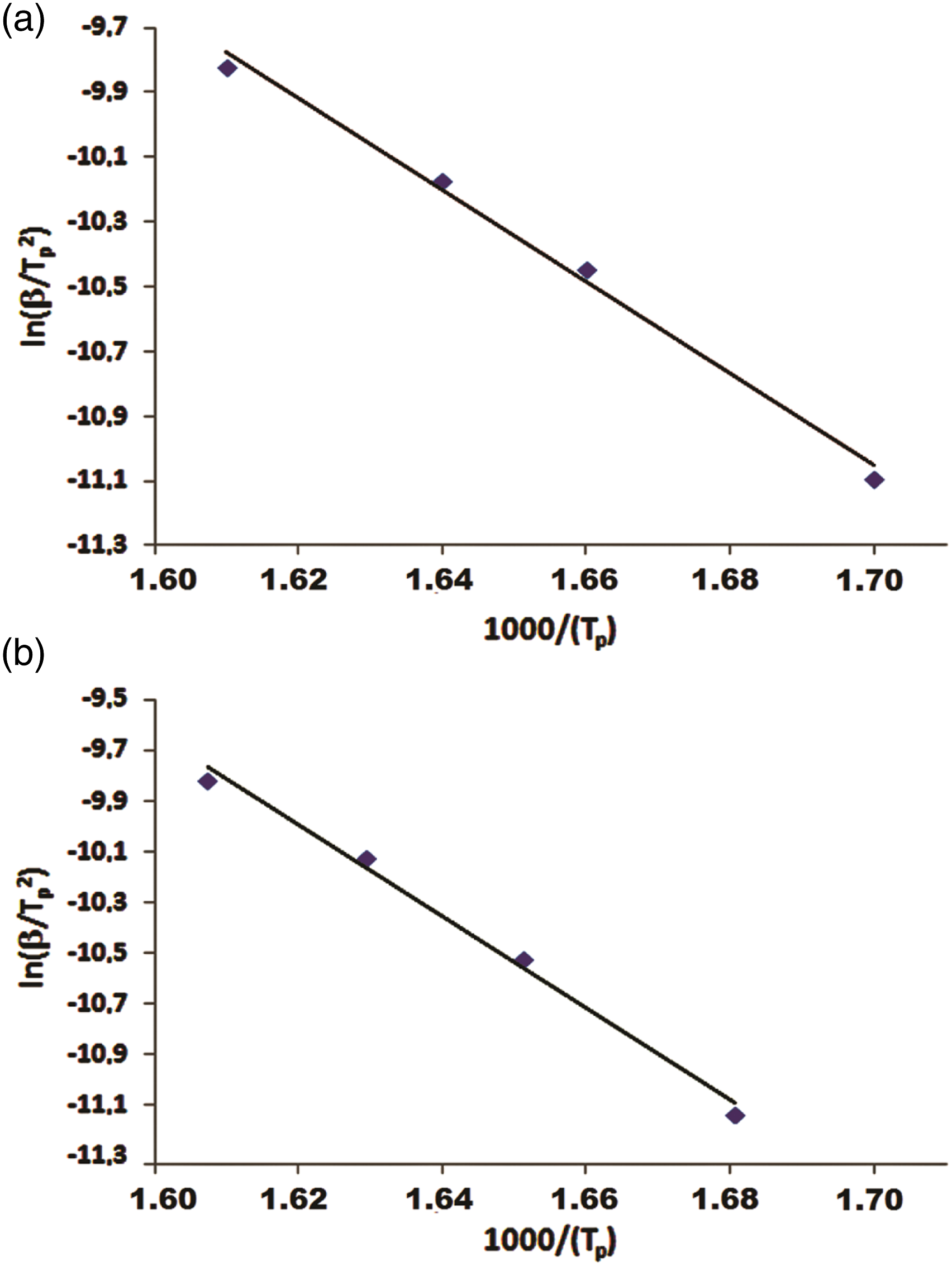

The Ozawa method
Activation energy (Ea) of the obtained polymers was found according to the equation of Ozawa,
26
whose equation is given below
When the results of the Ozawa analysis for poly(BMAOME 46 %-co-GMA) were plotted, the data consisted of parallel lines (Figure 9), confirming the accuracy of the method. The calculated Ea values of the polymers at different conversions are listed in Table 7. The average Ea values of the poly(BMAOME) were determined to be 133.26 kJ/mol by the Ozawa method. The average Ea values of the copolymers increased with the increase of the mole fraction of BMAOME in the copolymer. This result indicates that BMAOME units increased the thermal stability of the copolymers. Intermolecular secondary interaction forces between the polar groups in BMAOME units contributed positively to the thermal stability. The Ea values of the copolymer are obtained between 126.64 and 106.13 kJ/mol using the Ozawa method. Ozawa plot of poly(BMAOME-co-GMA) (0.46:0.54). Apparent Ea values of the polymers by the Ozawa method.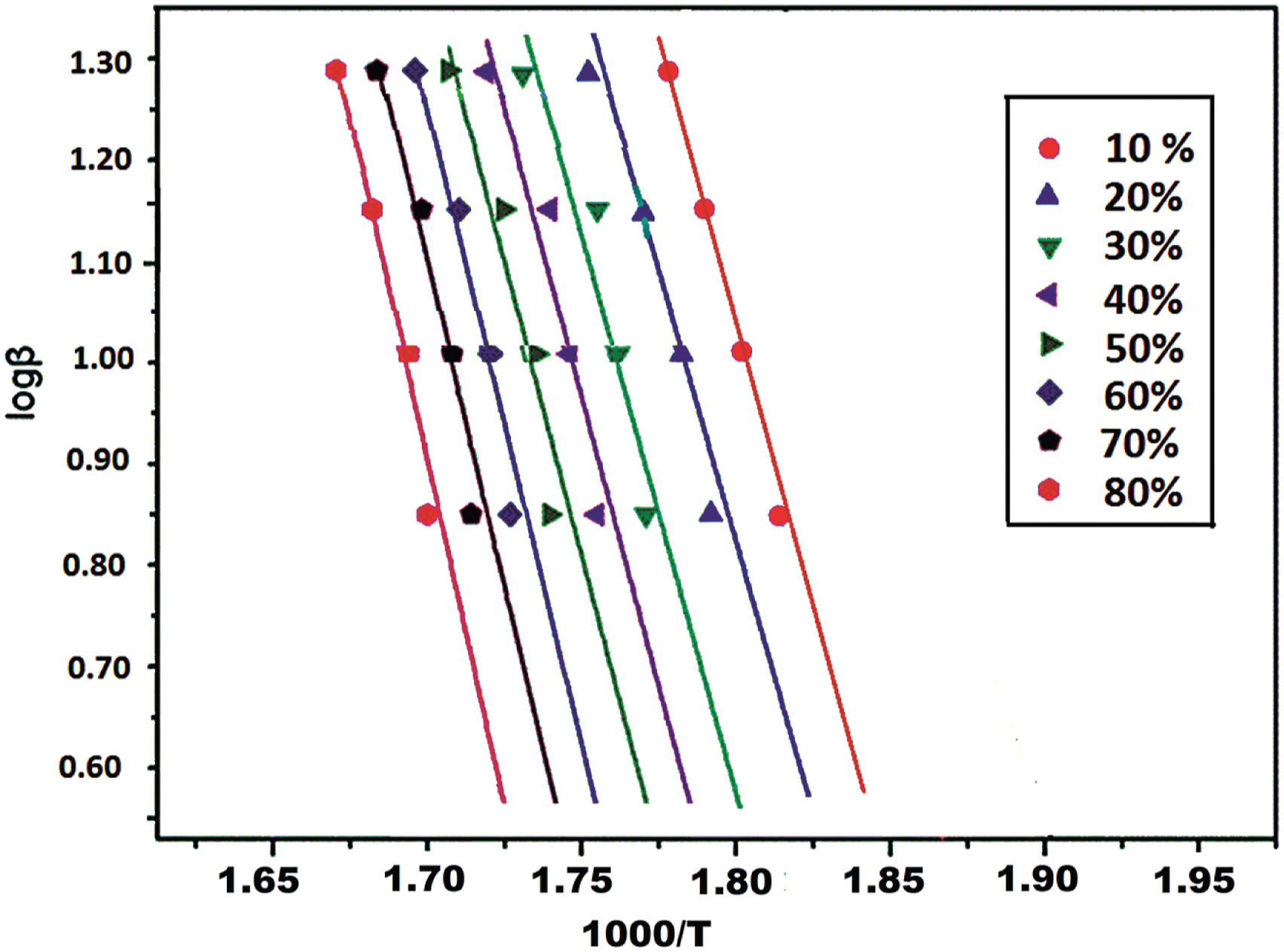

Changes in FTIR spectra during degradation of the polymers
The thermal degradation behavior of polymers has been studied by monitoring changes in FTIR spectra. For this purpose, thin films were prepared on a salt plate using about 5 mg of polymer. The prepared polymer films were heated in a thermostat-controlled oven at a heating rate of 10 C/min, and in N2 inert environment, up to three different temperatures, and FTIR spectra were obtained. The IR spectra of the polymer films heated to 300, 360, and 435oC are shown in Figure 10. It is seen that degradation starts at 320oC in the spectrum of poly(BMAOME). The mass loss at this temperature in the TG of the polymer is about 18%. The mass loss at 360oC reached around 45%, and significant degradation was observed in the FTIR peaks. At 435oC, where the rate of degradation and loss of mass increased, bands of cyclic anhydride structures began to appear at 1805, 1763, and 1018 cm−1, especially in the carbonyl region. The fact that the loss of mass at this temperature is around 80% indicates that the degradation has mainly occurred. The same studies have been done on the copolymer samples, and similar results have been obtained.27,28 FTIR spectra of partially degraded poly(BMAOME) after heating to 300, 360, and 425 0C.
Molecular weights of Polymers
Molecular weights, the polydispersity index and tg values of polymers.
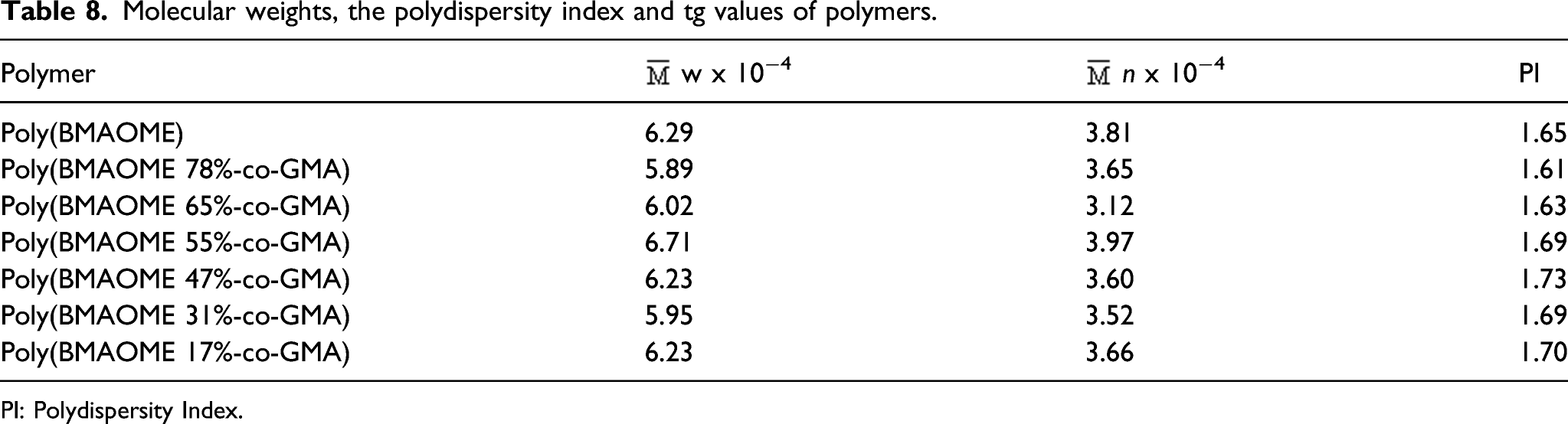
PI: Polydispersity Index.
Antimicrobial Screening of the polymers
Antimicrobial Effects of the Compounds (mm of zones).
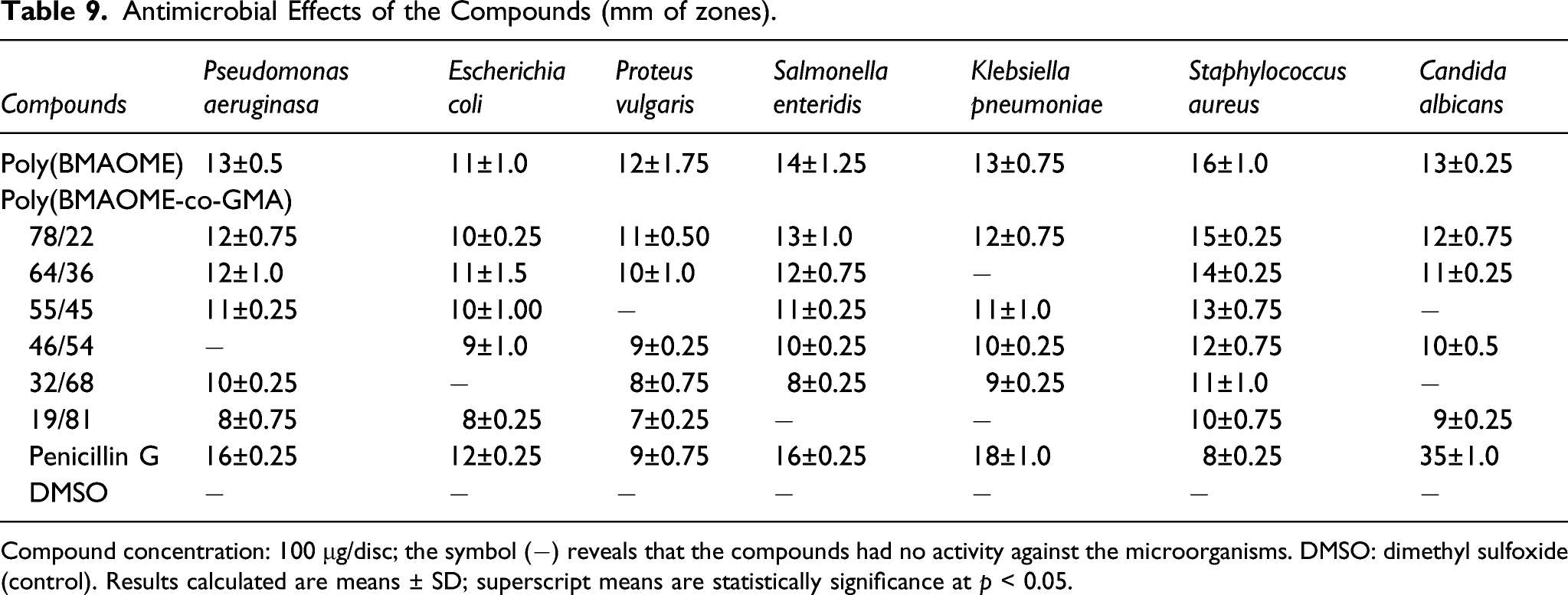
Compound concentration: 100 μg/disc; the symbol (−) reveals that the compounds had no activity against the microorganisms. DMSO: dimethyl sulfoxide (control). Results calculated are means ± SD; superscript means are statistically significance at p < 0.05.
It has been reported in the literature that many amide containing molecules have a wide range of biological activities such as antituberculosis, anticonvulsant, insecticidal, antifungal, and antitumor.31–33 Besides, BMAOME and GMA units in the copolymer structure carry ester groups. Ester compounds have attracted extensive research attention due to their broad biological activities and potential health benefits. In addition, some ester compounds, which are widely used in medicine, food, and cosmetics, have been reported to have various biological activities such as antimicrobial, 34 antiviral, 35 anti-inflammatory, and nematicidal activities. 35 The results show that the analyzed polymers have a mild biological activity similar to conventional penicillin G. All polymers showed strong activity especially against Staphylococcus aureus bacteria.
Photo-stability of the copolymers
Most synthetic polymers are susceptible to UV and visible-light-induced degradation. As the wavelength of the beam becomes smaller, the energy increases. The UV rays from the sun are the most important type of light responsible for the degradation of polymers used especially outdoors. Functional groups in the polymer structure absorb the energy of the UV rays. In the UV-Vis spectra of copolymers, the maximum wavelength of the peaks was about 275 nm. This value corresponds to π-π* transition of C=C and C=O of the pendant aryl and carbonyl chromophore groups in the polymer unit. In the UV-Vis spectra
The piperonyl group in the side branch of poly(BMAOME) was observed to not improve the UV stability of the copolymers. The UV stability of BMAOME-GMA copolymers was similar to other methacrylates in the literature. 39
Conclusions
In our study, a new methacrylate monomer (BMAOME) and its homopolymer containing piperonyl group in the side branch were synthesized. The structure of the compounds was characterized by FTIR and NMR techniques. The copolymers of BMAOME with glycidyl methacrylate (GMA) were synthesized by free radical polymerization at 65oC in 1,4 dioxane. The copolymer composition was determined using elemental analysis. The reactivity ratios of copolymers were determined by the FR and KT methods. It was concluded that the GMA monomer is more reactive, so the copolymers are rich in GMA. The thermal stability of poly(BMAOME) was better than poly(GMA). Thermal degradation activation energy (Ea) values for the polymers were calculated using Ozawa and Kissinger methods. The polymers showed moderate biological activity against some microorganisms. The photo-stability of copolymers was investigated under UV light. However, it was observed that functional side groups did not contribute positively to UV stability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Afyon Kocatepe University Scientific Research Projects Coordination Unit (15-FENBİL-38).

 w x 10−4
w x 10−4 n x 10−4
n x 10−4