Abstract
Immobilized metal ion affinity chromatography is one of the methods used for the adsorption of proteins. In this study, poly(glycidyl methacrylate) cryogel discs were prepared by free radical polymerization. The metal chelating groups were polymeric chain of poly-(
Introduction
Lysozyme, which is essential component of cell walls of certain bacteria is an enzyme that catalyzes the hydrolysis of polysaccharides (Mehra et al., 1975). It has a small molecular weight (14.4 kDa) and used commonly for different purposes. The first discovery by Fleming in 1921 had been from human mucosal secretions (Fleminger et al., 1990). Lysozyme is generally located in animals, plants, and microorganisms in breast milk, tears, and saliva (Chandran and Ereifej, 1981; Wardlaw, 1962). Lysozyme has many applications in food and pharmacological technology and it is used in eye drops, wound healing creams, and as food preservative (Juneja et al., 2012; Mehra et al., 1975). Recently, use of lysozyme as an anticancer drug and in the treatment of HIV infection has been investigated (Hartono et al., 2011; Ye et al., 2008). In the literature, the different methods for lysozyme purification were reported such as precipitation (Chang et al., 2000), centrifugation (Guerin-Dubiard et al., 2005), ultrafiltration (Wan et al., 2006), and chromatography (Bai et al., 2011).
Immobilized metal ion affinity chromatography (IMAC) is used as a group-specific affinity method for the purification of therapeutic proteins, histidine-modified proteins, nucleic acids, hormones, phosphopeptides, and enzymes (Andersson, 1984; Bendinskas et al., 2011; Shu et al., 2004). The basic principle of IMAC is formation of the coordinate covalent bonds between the electron donor residues on a protein surface (histidine, tryptophan, cysteine) and chelated transition metal ions (Porath, 1988; Porath et al., 1975; Porath and Olin, 1983). In the IMAC, variety of metals could be used (Lönerdal and Keen, 1982; Porath and Olin, 1983) for interactions with proteins. The most popular transition metals used in IMAC are Cu(II), Zn(II), Ni(II), and Co(II). In the IMAC, metal ions are immobilized by binding covalently to the hydrophilic support with metal chelating functionalities. The transition metals can form stable complexes with electron-rich compounds and may coordinate molecules containing oxygen (O), nitrogen (N), and sulfur (S) by ion dipole interactions and dative bond formation (Porath et al., 1975). IMAC applications have been used in separation of amino acids, nucleotides, and small molecules such as amines and in purification of various proteins (Berna et al., 1997; Liu and Yu, 1990; Sulkowski, 1989). IMAC is also used to determine surface topography of proteins (Boden et al., 1998) and offers many advantages compared with chromatographic methods such as high binding capacity and high recovery yield, nondenaturing elution conditions beside cheapness and stability (Brena et al., 1994; Ordaz et al., 2000).
Cryogels are gel matrices of the monomers or initiators which are prepared in partially frozen solvents. The basic characteristic of cryogels is composed of interconnected macropores and they are used in various biological applications as matrix owing to their osmotic, chemical, and mechanical features (Lozinsky et al., 2003). Cryogels are also cheap materials and don't cause any environmental pollution. Porous cryogels also have many advantages such as large pores, short diffusion path, and low pressure drop (Andaç et al., 2012; Bereli et al., 2008; Tamahkar et al., 2011). Cryogels are formed by the low density polymerization of monomers (<%10 (w/v)) in an aqueous solution (Önnby et al., 2012) and frozen ice crystals formed interconnected pores with three-dimensional polymeric structure (Dispinar et al., 2012; Hajizadeh et al., 2013). Because of high biocompatibility, biocatalysis and affinity chromatography with cryogel are the most common applications. Cryogels have been used in biomedical, cosmetics, and pharmaceuticals (Dispinar et al., 2012; Gun'ko et al., 2013; Kumar et al., 2006; Bereli et al., 2013).
Work presented in this paper is based on the purification of lysozyme (selected model protein) by IMAC with poly(glycidyl methacrylate) (PGMA) cryogel discs. In the first step, PGMA cryogel discs were prepared using GMA as basic monomer which was selected because its functional epoxy groups allow to attach the ligand easily via free radical polymerization under frozen conditions. Then, poly-(
Experimental
Materials
Lysozyme (chicken egg white, EC 3.2.1.17), poly(
Preparation of PGMA cryogel discs
Production of PGMA cryogel disc was performed using the Çimen et al.'s (2015) procedure. PGMA cryogel disc was polymerized with free radical polymerization by using water and MBAAm as the pore-former solvent and cross-linker, respectively. In the polymerization, APS and TEMED have been used as the redox initiator couple. A typical preparation procedure is as follows: GMA (1.0 ml) and HEMA (4.2 ml) were dissolved in 3.7 ml of deionized water. MBAAm (0.283 g) was dissolved in 10 ml of deionized water. Second solution was mixed with previous one and stirred until obtaining homogeneous solution in ice bath. TEMED (25 µl) and APS (20 mg) were added on the mixture to initiate the polymerization reaction. Then, the reaction mixture was poured between two glass plates separated with spacer having thickness as 1.5 mm. The polymerization mixture was frozen at 16℃ for 24 h and then thawed at room temperature. After washing with 200 ml of water, the cryogel was cut into circular disks (0.8 cm in diameter) with a perforator and stored in buffer containing 0.02% sodium azide at 4℃ until use.
P-His@PGMA cryogel discs
PGMA cryogel discs were washed several times with 15 ml of deionized water. After washing they were treated with 10 ml of acetate buffer (pH 5.0). Then, 10 ml of poly-(
Chelation of Cu(II), Zn(II), and Ni(II) ions with P-His@PGMA cryogel discs
A 1000 ppm atomic absorption standard solution (containing 10% HNO3) was used as a source of Cu(II), Zn(II), and Ni(II) ions. The P-His@PGMA cryogel discs were separately mixed with 10 ml of Cu(II), Zn(II), and Ni(II) solution (100 ppm, pH 5.0) at room temperature and stirred magnetically for 2 h. The concentration of Cu(II), Zn(II), and Ni(II) ions in the resulting solution was determined with a graphite furnace atomic absorption spectrophotometer (AA800, Perkin-Elmer, Bodenseewerk, Germany). The amount of chelated Cu(II), Zn(II), and Ni(II) ions was calculated by using the concentrations of the Cu(II), Zn(II), and Ni(II) ions in the initial solution and those in the equilibrium.
Characterization of PGMA cryogel discs
The swelling behavior of the membrane (S) was defined by the following procedure: the membrane sample was washed on porous filter paper until washing was clear. Then, water was dried by suction and weighed (mwet gel). It was dried to constant mass weight in the oven at 60℃ and the mass of fully dried sample was measured (mdry gel). The swelling ratio was calculated as follows

The characteristic functional groups of cryogel discs were analyzed by using a Fourier transform infrared spectrophotometer (FTIR, Perkin Elmer, Spectrum One, USA). The samples were prepared by mixing 2 mg of dried/crushed cryogel discs and 98 mg of powdered KBr (IR Grade, Merck, Germany) and pressed into a pellet form. The FTIR spectrum was then recorded in the wave number range of 400–4000 cm−1. The morphology of a cross-section of the dried cryogel discs was investigated by SEM. Cryogel disc samples were dried at room temperature for seven days and then coated with gold–palladium (40:60) and examined using a JEOL JSM 5600 (Tokyo, Japan) SEM.
The surface area of the cryogels was determined with the Brunauer–Emmett–Teller (BET) equation using single point analysis and a Flowsorb II 2300 from Micromeritics Instrument Corporation, Norcross, USA.
Lysozyme adsorption and elution studies from aqueous solution
In a typical adsorption experiment, lysozyme was dissolved in 25 ml of appropriate buffer solution and cryogel discs were subsequently added. The adsorption was followed by monitoring the decrease in UV absorbance of lysozyme at 280 nm. Effects of concentration of lysozyme, pH of the medium contact time, and ionic strength on the adsorption capacity were examined. To observe the effects of the concentration of lysozyme on adsorption, it was changed between 0.1 and 2.0 mg/ml. pH was varied between 4.0 and 8.0. Ionic strength was adjusted by using sodium chloride (NaCI) while being varied in the range of 0.0–1.0 M. Lysozyme desorption from the cryogel discs was performed with 1.0 M NaCI. The desorption ratio was determined for 1 h contact time. The reusability of the adsorption medium was tested for five adsorption–desorption cycles. After each adsorption–desorption cycle cryogel discs were washed several times with deionized water.
Purification of lysozyme from chicken egg white
To check efficiency of PGMA cryogel discs to purify lysozyme from natural source, the purification studies were conducted from chicken egg white. Egg white samples were separated following the method reported elsewhere (Köse and Denizli, 2013). Briefly, fresh chicken egg white was separated from fresh eggs. The egg white was filtered through the cheese cloth in order to remove particulate matter such as chalazae and then it was diluted to 50% (v/v) with phosphate buffer (100 mM, pH 7.0). The diluted egg white was homogenized in an ice bath at 4℃ and centrifuged at 5000 r/min for 30 min. This solution was transferred in a flask including the PGMA cryogel discs. The mixture was stirred in a rotary mixer at room temperature for 2 h. Lysozyme adsorption was periodically checked by taking a sample from the adsorption media. Thereafter, the PGMA cryogel discs were removed from the adsorption media by lysozyme purification and resuspended in a washing buffer (acetate buffer, pH 4.0). Adsorbed lysozyme was eluted using 1.0 M NaCI solution. The purity of the eluted fraction was analyzed with SDS-PAGE gel electrophoresis.
Results and discussion
Characterization of PGMA cryogel discs
Supermacroporous cryogel discs were synthesized by free radical polymerization in the frozen state of monomers GMA with MBAAm as a cross-linker in the presence of APS/TEMED as initiator/activator pair. After, poly-( FTIR spectra of (a) PGMA and (b) P-His@PGMA cryogel discs. The nitrogen isotherm of the cryogel discs.

Physical properties for PGMA cryogel discs.

Determined using single-point Brunauer–Emmett–Teller method.
Barrett, Joyner, and Halenda (BJH) cumulative desorption pore volume of pores between 20 and 245 Å.
Internal morphology of the cryogel discs obtained by SEM is given in Figure 3. In the figure, it is clearly seen that PGMA cryogel discs have large supermacroporous and wide flow channels having thin polymeric walls. Interconnected flow channels and supermacroporous provide easy diffusion of lysozyme into the structure and effective interactions with binding domains. The pore size of the cryogel discs is much larger than the size of the lysozyme, which leads to diffusion without limitations and plugging during adsorption process. These large pores were formed as a result of thermally induced separation of both phases, liquid and solid (Kirsebom et al., 2010). Also, rough surface of pore walls was due to the addition of surfactant into cryogelation recipe.
SEM micrograph of PGMA cryogel discs.
Poly-(l )-histidine immobilized PGMA cryogel discs
In order to evaluate the amount of metal chelating ligand (poly-( Adsorption capacity of PGMA cryogel discs for poly-
Chelation of Cu(II), Zn(II), and Ni(II) ions onto P-His@PGMA cryogel discs
Additionally, the chelating capacity of cryogel discs for Cu(II), Zn(II), and Ni(II) ions was investigated and the amount of immobilized Cu(II), Zn(II), and Ni(II) on the PGMA cryogel discs was calculated. The amount of chelating capacity onto the PGMA cryogel discs was shown in Figure 5. It should be noted here that after chelation and washing steps leaching of Cu(II), Zn(II), and Ni(II) was not observed from any of the Cu(II), Zn(II), and Ni(II) immobilized P-His@PGMA cryogel discs in all the solvent media used for adsorption–desorption studies. It should be noted that the amount of chelating capacity increased with contact time.
Adsorption capacity of metal ion on P-His@PGMA cryogel discs. Initial metal concentration: 100 ppm, pH 5.0, contact time: 24 h.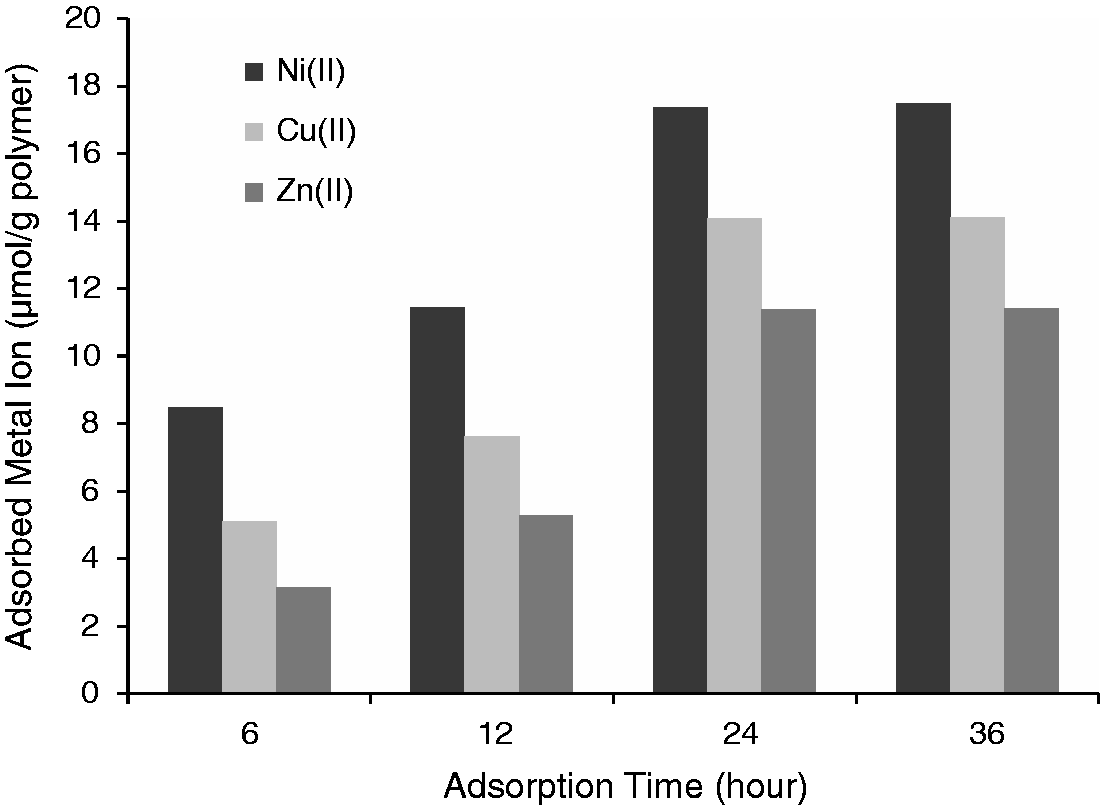
Adsorption of lysozyme from aqueous solutions
Effect of pH
The amount of lysozyme adsorbed onto the PGMA and P-His@PGMA cryogel discs as a function of pH was shown in Figure 6. In the present study, we varied the pH value between 4.0 and 8.0. In this group of experiments, the initial concentration of lysozyme was kept constant as 0.5 mg/ml. In Figure 6, it was shown that the pH of solution effects adsorption of lysozyme on all Me(II)@P-His@PGMA cryogel discs greatly. It was observed that the adsorption capacities, with increasing pH, reached to maximum values at pH 6.0 for Zn(II) and at pH 7.0 for Cu(II) and Ni(II) chelated cryogel disc, respectively. The result may be dependent on the difference of coordination spheres of heavy metal ions. Therefore, pH 7.0 was used in further experiments. Lysozyme adsorption amount onto the Ni(II), Zn(II), and Cu(II) ions chelated P-His@PGMA cryogel disc shows a maximum value at pH 7.0 (17.9, 10.9, and 14.3 mg/g polymer), respectively. Specific interactions such as electrostatic and coordination, between lysozyme and chelated Cu(II), Ni(II), and Zn(II) ions at pH 7.0 may result both from the ionization states of several groups on amino acid side chains in lysozyme structure and from the conformational state of lysozyme molecules at this pH. As seen in Figure 6, the adsorbed amount of lysozyme decreased at pH values lower and higher than optimal pH values. This could be due to the ionization state of lysozyme and could be caused by repulsive electrostatic forces between adsorbed lysozyme molecules at the chelated Cu(II), Ni(II), and Zn(II) ions via P-His@PGMA. Increase in conformational size and the lateral electrostatic repulsions between adjacent adsorbed lysozyme molecules may also cause a decrease in adsorption efficiency. In addition, the potential competition between lysozyme and hydroxide ions at higher pH values caused the significant decrease in adsorption capacity.
Effect of pH on adsorption of lysozyme on the Me(II)@P-His@PGMA cryogel discs. Initial concentration of lysozyme: 0.5 mg/ml; Ni(II) content: 17.4 µmol/g, Cu(II) content: 14.1 µmol/g, Zn(II) content: 11.4 µmol/g T: 25℃. Adsorption kinetics of lysozyme by Me(II)@P-His@PGMA cryogel discs. Initial concentration of lysozyme: 0.5 mg/ml; Ni(II) content: 17.4 µmol/g, Cu(II) content: 14.1 µmol/g, Zn(II) content: 11.4 µmol/g T: 25℃..

Effect of adsorption kinetics
Adsorption kinetics of lysozyme of all the cryogel discs was determined. High adsorption rates were observed at the beginning of adsorption and then saturation values (i.e. adsorption equilibrium) were gradually achieved within 60 min (Figure 7). The order of adsorption capacity observed was Ni(II)@P-His@PGMA > Cu(II)@P-His@PGMA > Zn(II)@P-His@PGMA > P-His@PGMA > PGMA. This very fast adsorption is probably due to the high binding rate and strong affinity interactions between electron donor residues on the lysozyme molecules and metal ions on the surface.
Effect of lysozyme concentration
Adsorption capacities of Me(II)@P-His@PGMA cryogel discs are shown in Figure 8. The lysozyme adsorption amount increased with increasing concentration of the lysozyme and a saturation value was reached at a concentration of 1 mg/ml. At initial part of the curves, the rapid increase in capacity was observed, which emphasized a high affinity between analyte and adsorbent. Then, those curves started to decline and became constant when the lysozyme concentration was greater than 1 mg/ml. A negligible amount (2.04 mg/g) of lysozyme molecules adsorbed onto PGMA cryogel discs. It may be due to the physical entrapment into pores and/or chemical attachment through epoxy rings of PGMA groups. Ni(II), Cu(II), and Zn(II) chelation onto the cryogel discs significantly increased the lysozyme adsorption capacity of the cryogel discs up to 36.4, 26.8, and 17.3 mg/g polymer. It is clear that this increase is due to the specific interaction between chelated Ni(II), Cu(II), and Zn(II) ions and lysozyme molecules.
Adsorption capacity of Me(II)@P-His@PGMA cryogel discs for lysozyme. Ni(II) content: 17.4 µmol/g, Cu(II) content: 14.1 µmol/g, Zn(II) content: 11.4 µmol/g T: 25℃.
Jain et al. (2010) used porous nylon membranes modified with poly(2-(methacryloyloxy)ethyl succinate) brushes and their derivatives. NTA-Ni(II) was used as the ligand and reported the adsorption capacity of 120 mg/g in 1 mg/ml lysozyme solutions. Suna et al. (2011) prepared monodispersed and superparamagnetic carboxymethyl chitosan magnetic nanoparticles. Then, treated with Zn(II), Cu(II), and Fe(III) ions solutions to obtain immobilized metal affinity magnetic nanoparticles and the lysozyme adsorption capacity of the nanoparticles were 200, 185.19, and 232.56 mg/g, respectively. Bayramoğlu et al. (2002) synthesized interpenetration networks (IPNs) in membrane form with 2-hydroxyethyl methacrylate (HEMA) and chitosan (pHEMA/chitosan) via UV-initiated photopolymerization. Procion Brown MX-5BR was covalently attached onto IPNs membrane as a metal chelating dye–ligand. The adsorption capacities of the dye–ligand, dye–ligand–Fe(III), and dye–ligand–Cu(II) immobilized IPNs membranes were 79.1, 147.4, and 128.2 mg/ml, respectively. In another study, Bayramoğlu et al. synthesized composite membranes with HEMA and chitosan (pHEMA/chitosan) via an ultraviolet-initiated photopolymerization technique and A triazidine dye (Procion Green H-4G) was used to covalently immobilized as a ligand (Bayramoğlu et al., 2013). The lysozyme adsorption capacity of the pHEMA/chitosan dye, pHEMA/chitosan dye-Zn(II), and pHEMA/chitosan dye-Fe(III) membranes were 2.54, 2.85, and 3.64 mg/cm2, respectively.
Effect of ionic strength
The effect of NaCl concentration on lysozyme adsorption was also investigated. As seen in Figure 9, in binding experiments, high ionic strength weakened the binding of lysozyme onto cryogel disc when increasing amounts of NaCl were added to the adsorption solution. Therefore, with the increasing NaCl concentration, lysozyme adsorption capacity decreased. The explanation to this phenomenon is that the counter salt ions interact with the lysozyme molecules via charge–charge interactions and mask the binding sites on adsorption media. In addition, coordination of chelated heavy metal ions by anion setting as a shield/barrier between heavy metal ions and lysozyme molecules inhibited the interaction of electron donor residues on lysozyme molecules with those of Lewis acidic site.
Effect of ionic strength on lysozyme adsorption onto the Me(II)@P-His@PGMA cryogel discs. Lysozyme concentration: 0.5 mg/ml; Ni(II) content: 17.4 µmol/g, Cu(II) content: 14.1 µmol/g, Zn(II) content: 11.4 µmol/g T: 25℃.
Evaluation of adsorption isotherm
Two models, Langmuir and Freundlich, were tested to describe the relation between equilibrium concentration of lysozyme solution and adsorbed lysozyme on Me(II)@P-His@PGMA cryogel discs. The linearized forms of Langmuir and Freundlich models were used, as given below

The equation given below was used for Freundlich model

n is Freundlich constant. The value of 1/n ranges between 0 and 1 and it is a measure of surface heterogeneity, becoming more heterogeneous as its value gets closer to zero.
Langmuir and Freundlich isotherm parameters for Me(II)@P-His@PGMA cryogel discs.

In order to calculate thermodynamic parameters, the Langmuir isotherm at different temperatures was also examined based on the equation

Thermodynamic variables for adsorption process.

According to the ΔG° (kJ/mol) value in Table 3 the adsorption process was spontaneous. During the adsorption process, this result showed an increase in total irregularity and made ΔS° (J/mol K) value positive. These increases occur with the combination of the changes in the conformation of biomolecules and the regular separation of solvent molecules (Finette et al., 1997). ΔH° value was negative because it depends on nature of adsorption process. The effect of contact time between the adsorbent and adsorbate on adsorption capacity was examined. Due to the high affinity between lysozyme and matrix the process was fast for initial 50 min. The low back pressure and the mass transfer by convection to the matrix were the other factors causing the process to occur fast when batch experiments were performed.
To decide whether the diffusion or chemical binding process determined the adsorption rate, pseudo first-order and pseudo second-order kinetic models were applied to data.
Lagergrens modified equation for pseudo first-order change is given by the equation below

The equation

Rate constants, calculated and experimental adsorbed amounts of Me(II)@P-His@PGMA cryogel discs.

Adsorption of lysozyme from egg white
Lysozyme adsorption from egg white (diluted with phosphate buffer; T: 25℃).

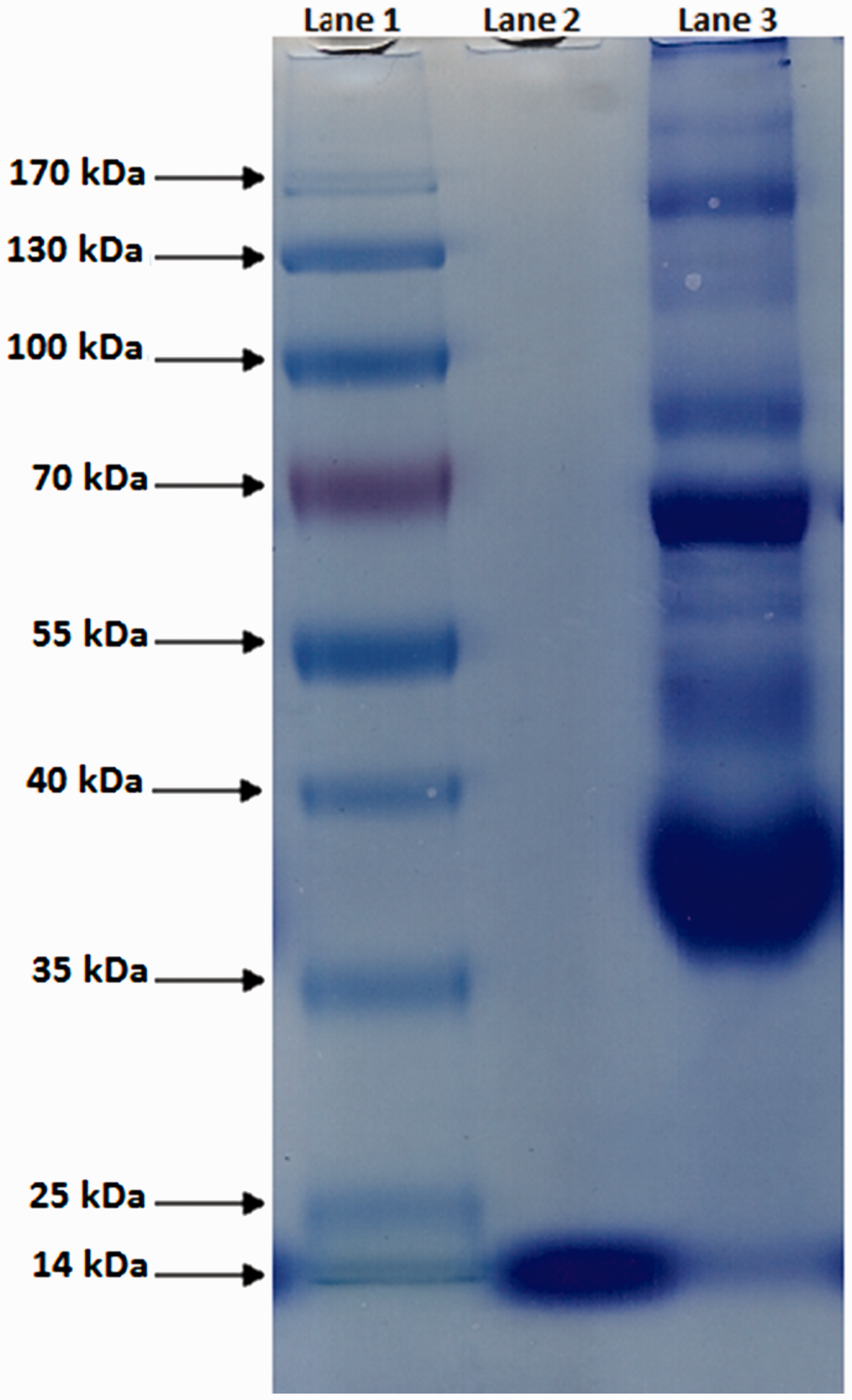
SDS-PAGE of egg white samples. The samples were assayed by SDS-PAGE using 10% separating gel (16 cm × 14 cm). Separating gels were stained with 0.25% (w/v) Coomassie Brilliant R250 in acetic acid. Lane 1, biomarker (fermentas); lane 2, desorption sample; lane 3, egg white before adsorption.
Desorption and repeated use
In order to show the stability and reusability of the PGMA cryogel discs, the adsorption–desorption cycle from aqueous solution was repeated five times using the same cryogel discs. Desorption experiments were performed using 1.0 M NaCI as the desorption agent. As seen in Figure 11, the cryogel disc maintains its adsorption capacity at almost constant value of 94% and it remains very stable during the adsorption studies. No significant decrease in adsorption capacity was observed, after five repeated adsorption–desorption cycles.
Stability and reusability of the Me(II)@P-His@PGMA cryogel discs. Lysozyme concentration: 0.5 mg/ml; Ni(II) content: 17.4 µmol/g, Cu(II) content: 14.1 µmol/g, Zn(II) content: 11.4 µmol/g T: 25℃.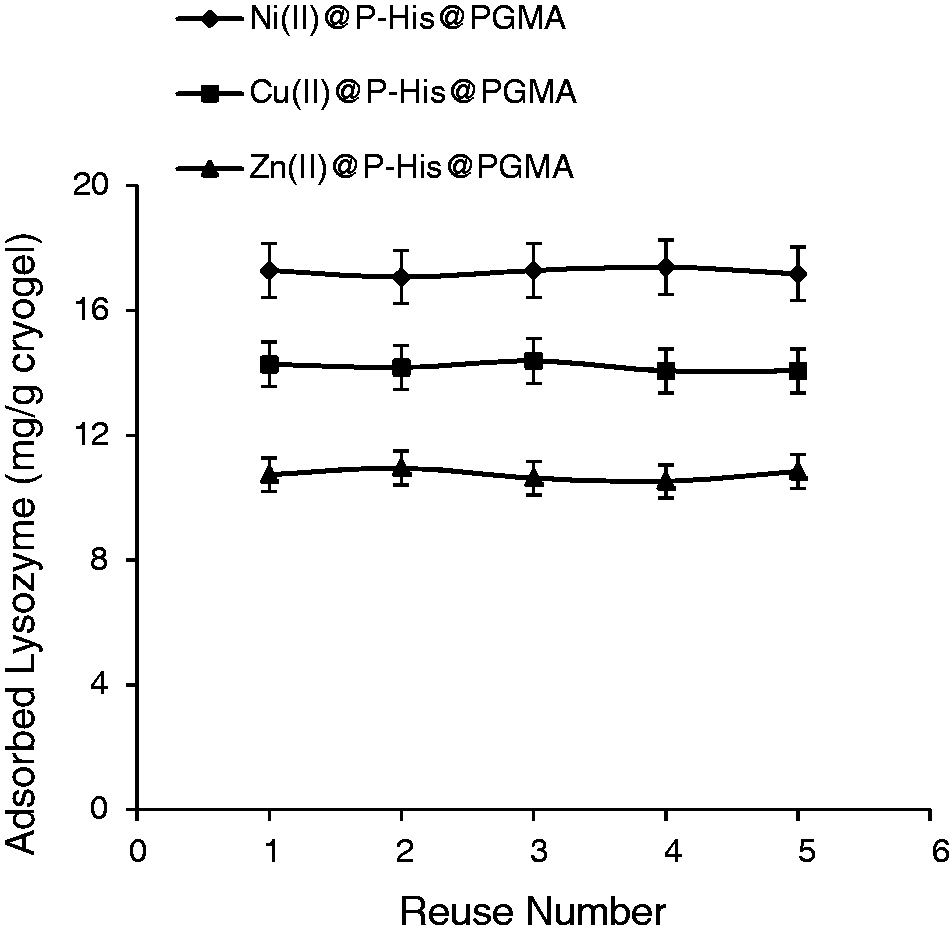
Conclusion
Lysozyme is one of the important industrial enzyme and used as cell disrupting reagent, antibacterial agent, and food additive (Hartono et al., 2011; Ye et al., 2008). In the literature, there are a lot of methods for the purification and separation of lysozyme such as molecule surface imprinting (Gao et al., 2010), affinity ultrafiltration (Ferraris et al., 2011), IMAC (Suna et al., 2011). Recently, a new generation of cryogels separation technique is recognized as the stationary phase (Bereli et al., 2010; Yavuz and Baysal, 2013). Supermacroporous cryogel discs are a good alternative with the many advantages such as flexibility, biocompatibility, physical and chemical stability, and easy preparation for protein purıfication. Bioseparation, biocatalysis, and affinity chromatography are the most common application areas of cryogels (Dispinar et al., 2012). In this study, we also used IMAC based on glycidyl methacrylate monomers, cryogels, and poly-( The chemical structure of poly-
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
