Abstract
Anti-infection ability and desirable air permeability (AP) are the important aspects of wound dressings that should be regulated with regard to primary polymers and antibacterial agents. This study aims to fabricate and optimize a wound dressing with antibacterial properties. For this purpose, polycaprolactone (PCL) as a hydrophobic polymer and polyvinylpyrrolidone (PVP) as a hydrophilic polymer were electrospun with Lawsonia inermis (Henna) extract as an antibacterial component based on the experiments proposed by Design-Expert software. The morphology and properties of the wound dressing were studied by scanning electron microscopy, Fourier transform infrared, and AP. The response surface method (RSM) was used to determine the optimal fiber diameter and AP of the samples as a function of the polymer concentration and feed rate. The optimal PCL/PVL/Henna wound dressing has antibacterial properties against Gram-positive and Gram-negative bacteria while being biocompatible according to the MTT assay. These fibrous structures can be used as a wound dressing to prevent infection and accelerate wound healing; thanks to proper Henna release, breathability, swelling ratio, and mechanical performance.
Introduction
Electrospinning is a manufacturing technique for producing submicron fibers from natural, biological, and synthetic polymers for various applications. The effective parameters in electrospinning are classified into three main groups, including factors related to spinning solutions, the electrospinning process, and the operating environment. 1 The method has attracted more interest and attention in recent years due to its adaptable applications in various fields such as electronics, 2 protective textiles, 3 food packaging, 4 as well as medicine such as tissue engineering, 5 and wound dressing. 6
Nanofibrous wound dressings are effective structures because of their advantages, especially in their porosity and high surface area. 5 These structures can be as simple as a fibrous structure made of a polymer or a complex engineered composite containing layers of different polymers and reactive drug materials. An ideal dressing material should accelerate wound healing, reduce the loss of proteins, electrolytes, and fluid from the wound, and help minimize pain and infection. Some new ways of obtaining an effective wound dressing are tetracycline hydrochloride-loaded electrospun nanofibrous mats based on PVA and chitosan developed by Alavarse et al., 6 curcumin-loaded electrospun wound dressings developed by Mutlu et al., 7 and long-acting and broad-spectrum antimicrobial PCL/gelatin wound dressings developed by Shi et al. 8
Biocompatible polymers have been widely used in electrospun wound dressings due to their biodegradability 9 and compatibility. 10 One of the important biocompatible polymers is polycaprolactone (PCL). 8 The low biodegradation rate and low hydrophilicity of PCL are the main drawbacks of this polymer in biomedical fields. Researchers overcome this limitation by electrospinning PCL mixed with polyvinylpyrrolidone (PVP).11,12 Janus antibacterial nanofibers were prepared by phase separation of PCL/PVP-Ag nanoparticles. 13 Shitole et al. 14 fabricated novel core/shell wound dressings from poly(vinylpyrrolidone) iodine (PVPI) and PCL using a coaxial electrospinning method followed by surface modification with poly-l-lysine. They reported that the resulting structures exhibited enhanced biocompatibility, showed a selective bio-interaction pattern, and improved cell adhesion and proliferation while conferring an antimicrobial property. Hu et al. 15 also fabricated bimetallic zinc oxide/silver nanocapsules to improve the antibacterial activity of PVP/PCL nanofibrous wound dressings.
The antibacterial function as a desirable property in wound dressings can also be produced by Lawsonia inermis (Henna). 16 Researchers focus on this herbal medicine because of its fewer side effects and faster healing rate than commonly used synthetic drugs. 17 Vakilian et al. 18 fabricated a hybrid nanofibrous scaffold of poly‐L‐lactic acid and gelatin into which Henna was incorporated. They confirmed the antibacterial activity of Henna against E. coli and S. aureus. They also proved the biocompatibility of the scaffold on 3T3 fibroblast cells. Yousefi et al. 20 studied the antibacterial activity, cell biocompatibility, and wound healing activity of chitosan nanofibers containing Henna. They found that these nanofibrous structures have great potential for skin tissue engineering due to its significant synergistic antibacterial activity, cell viability, and wound healing activities. It has been demonstrated that Henna enhances the cell attachment, proliferation, and collagen secretion of gelatin-oxidized starch nanofibers. 16
Against this backdrop, this study aimed to fabricate and optimize antibacterial wound dressings using PCL, PVP, and Henna. The fiber diameter and air permeability (AP) of PCL/PVP/Henna were optimized using the response surface method (RSM) as a function of polymer concentration and feed rate. The biological and physicochemical properties of the optimal wound dressing were also investigated.
Experimental
Materials
Polycaprolactone (Aldrich, Mn = 8 × 104), polyvinylpyrrolidone (Aldrich, Mn = 5.5 × 104), dimethylformamide (DMF, Merck, ρ = 0.94 g/cm3), chloroform (Merck, ρ = 1.48 g/cm3), ethanol (EtoH, Merck, ρ = 0.790–0.793 g/cm3), and Henna were used in this study.
Methods
Experimental design
The range and code level of the variables.


Samples proposed by Design-Expert software and their response factors, including the fiber diameter and AP.
Electrospinning
The solutions for electrospinning consist of the mixed solutions (50/50 v/v) of PCL (10%, 12.5%, and 15% w/v in dymethylformamide/chloroform (50/50)), PVP (25%, 30%, and 35% w/v in ethanol), and Henna (10% W/V)).16,18 The mixed solution was stirred at medium speed for 5 h before electrospinning.
The solution was injected via an infusion pump (WPI Inc, AL 1000) and a high voltage power supply. The roller collector was placed at a distance of 15 cm from the steel nozzle. The applied voltage for collecting all the samples was set at 17 kV, and the range of the feed rate was set at 1–2 mL/h. All the samples were electrospun at a relative humidity of 50% and ambient temperature.
Characterization
The chemical composition of the fabricated nanofibers contains PCL and PVP with Henna, and the structures were analyzed by Fourier transform infrared spectroscopy (FTIR) (Nicolet, Nexus 670) in attenuated total reflectance (ATR) mode. Attenuated total reflection Fourier transformed infrared spectra were recorded from 4000 to 500 cm−1 with a resolution of 2 cm−1 and five scans.
A scanning electron microscope (CamScan MV2300, the Czech Republic and England) was used to study the morphology of the electrospun nanofibers. An average of 100 random measurements of fiber diameters present in each sample was reported as fiber diameters via Image J software.
For an evaluation of the amount of airflow permeation through the nanofiber layers, the AP test was performed according to ASTM D737 under the constant preset air pressure of 200 Pa using an AP tester (MO21S, SDL, USA). The test was carried out on different locations of the prepared layers, and an average was reported as the AP of each layer. To eliminate the influence of the thickness parameter on the results, in the end, all the results were divided by the thickness of the layer. The thickness of the layers was measured using a micrometer (Yuri, India).
The antibacterial tests of the wound dressings were performed using the AATCC 147 method. The samples of the test materials were placed in close contact with the agar surface, which was previously coated with a 0.1% inoculum suspension of a test bacterium. After incubation at 37°C for 24 h, a distinct zone of inhibition was formed around the samples and recorded as the antibacterial activity of the sample. Escherichia coli (E. coli) (Gram-negative) and Staphylococcus aureus (S. aureus) (Gram-positive) were used to evaluate the antibacterial properties of the nanofibers.
A Franz diffusion cell was used to study the drug delivery of Henna-containing wound dressings using a phosphate buffer solution with a pH of 7.4 at a temperature of 37°C. 1 mL of the buffer solution was withdrawn after a predetermined time of 1–7 h and replaced with a fresh solution. High-Performance Liquid Chromatography analyzed the withdrawn solution.
Cellular viability was determined by the MTT method. For this purpose, primary human skin fibroblast cells were incubated for 48 h, and a Tecan Sunrise™ microplate reader was used (492 nm). Cells were cultured in Dulbecco’s modified Eagle’s medium supplemented with fetal calf serum (10%) and l-glutamine (2 mM). Cells were placed in physiological conditions at °C in 5% CO2 for 24 and 48 h. Scaffold pieces measuring 0.5 cm × 0.5 cm were used for cell studies (soaked in a 2-ml culture medium for 24 h). 5
For assessing the breathability of wound dressings, water vapor permeability (WVP) was performed according to ASTM E 096. In this method, the sample was placed on a cup containing 50 ccs of water and sealed. The weight loss of the cup in a day is divided by the surface area of the sample and is considered the samples’ breathability. The pre-weighed samples (W0) were immersed in the pseudoextracellular fluid (PECF) as simulated wound fluid to determine the swelling ratio. At equilibrium (when the samples stopped absorbing PECF), the mats were removed, the excess water was wiped off with a filter paper, and then weighed (Wt). The swelling ratio was calculated using equation (3)
Mechanical performance was measured using a uniaxial tensile tester (model No. 5567, Instron Co., USA) equipped with a 10-N load cell at a strain rate of 0.5/min. Rectangular specimens 3 mm long and 5 mm wide were cut from nanofibrous mats. The average of five specimens is given.
Fourier transform infrared spectroscopy
The FTIR analysis for prepared samples was performed to identify the components in the prepared substrate. The chemical structure of the three components and the FTIR spectrum of the fabricated structures are shown in Figure 1. As shown in this figure, a peak in 3428 cm−1 is assigned to the OH bond of Henna.
16
The peak in 2946 cm−1 is related to the stretching vibration of CH2, which is present in both PCL and PVP [31]. The sharp peak at 1725 cm−1 is represented by the C=O group present in PVP and PCL. The characteristic peak of PVP is related to the C−N group, which occurs at 1663 cm−1 and 1288 cm−1. The absorption peak in 1462 cm−1, 1424 cm−1, and 1369 cm−1 is also related to the C=C aromatics of Henna. At 1188 cm−1 and 1045 cm−1, the identification peaks of PCL were deleted, which are related to the C-O-C group. There is a peak at 961 cm−1 associated with the vibration of the PVP ring. Since the identification peaks of all three components are present in the FTIR spectra of the sample,11,16, it can be concluded that the blend structure was successfully formed. Chemical structure and FTIR spectra of the electrospun PVP/PCL/Henna mat.
Morphology of the samples
Table 2 presents the fiber diameter of the samples in different proposed runs. Also, typical SEM images and histogram of the samples are shown in Figure 2. In most of the samples, the structures were successfully fabricated although in some cases, beads were observed (run 4, 5, 18, and 19). There seems to be a critical limit to the PVP concentration, below which the structure of the compound differs from the uniformity of the fibers and the beads appear in the structure. In all bead-like structures, the PVP concentration is in the low 25% range, and it appears that PVP concentration is a dominant factor (compared to the PCL concentration and feed rate) in the morphology of the fibers. Typical SEM images of (a) sample of run 3 with a minimum fiber diameter of 175 nm, (b) its fiber diameter distribution histogram, (c) sample of run 16 with a maximum fiber diameter of 342 nm, (d) its fiber diameter distribution histogram, and (e) and (f) sample of run 4 and 19 with beaded morphologies.
As shown in Figure 2, the minimum fiber diameter can be attributed to the dressing of run 3 with a diameter of 175 nm, and the maximum was reached in run 16 with a diameter of 342 nm. Polymer concentration is one of the most important factors influencing the fiber diameter in the electrospinning process. As shown in Table 2, the synergistic effect of the PCL and PVP maximum concentration and the maximum feed rate leads to the maximum fiber diameter in this series of experiments. Sample 3 is in the opposite fabrication condition with a low polymer concentration and feed rate, resulting in the minimum fiber diameter.
Modeling and optimization using RSM
Fiber diameter
Figure 3 shows the effect of polymers concentration on fiber diameters in different feed rates. According to these charts, the maximum diameter in rate 1, 1.5, and 2 mL/h is related to (PCL 15%, PVP 35%), (PCL 12.5%, PVP 35%), and (PCL 15%, PVP 35%), which is equal to 304 nm, 291 nm, and 342 nm, respectively. These are related to the maximum concentration of polymers which confirms that the direct relation between polymer concentrations and final diameters.
20
The effect of polymer component concentrations on the final diameters of fibers at different feed rates: (a) 1 mL.h−1, (b) 1.5 mL.h−1, and (c) 2 mL.h−1.
Equation (4) expresses the relationship between the input parameters and the average diameter of the fibers in response. Here, the non-significant coefficients were excluded from the model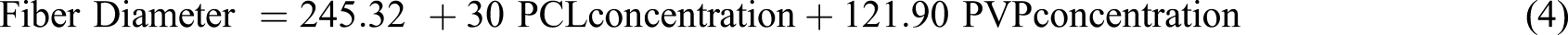
As this model shows, the feed rate has no significant effect on the fiber diameter. This may be due to the narrow range of the feed rate in this study.
The contours of the average diameters in Figure 4 show that an increase in the PVP concentration significantly increases the fiber diameters. Increasing the PCL concentration does increase the average fiber diameter, but the effect is not as significant as in the case of the PVP concentration. (a) Contour plot of PCL and PVP concentration at a feed rate of 1.5 mL/h, (b) contour plot of the feed rate and PVP concentration at a PVP concentration of 12.5%, and (c) 3D plots of the average diameter of fibers as a function of PCL and PVP concentrations at a feed rate of 1.5 mL/h.
Air permeability
Air permeability, as one of the important properties of wound dressings, was performed based on ASTM D 737 (Table 2 and Figure 5). The results of AP showed that as the concentration of components increased, the AP decreased. As the concentration increased, so did the diameter of the fibers,
11
which in turn increased the fiber coverage density and decreased the mean pore diameter, all of which increased the tortuosity of the membranes.
21
Therefore, it is no wonder that the AP showed a decreasing trend.
20
The AP of the non-uniform samples (4, 5, 18, and 19) is maximum because the barrier in front of the airflow is weak. The minimum value of AP refers to samples 15 and 16 prepared under the conditions of 15% PCL, 35% PVP, and a feed rate of 1 mL/h and 2 mL/h, respectively. The concentration of the polymer components according to their effect on the fiber diameter is the most important parameter for the AP of the nanofiber layer (Figure 5).Response surface method was applied to detect the correlation between nanofiber mat AP, polymer concentrations, and the feed rate. Each parameter influences the response factor, and its significance was determined when the p-value is less than 0.05.
21
According to the analysis of the results, equation (5) is the fitted linear equation for the AP of the fibrous wound dressing Air permeability of the nanofibrous wound dressing at (a) different PCL concentrations and (b) different PCL concentrations.

It should be mentioned that this equation is not hierarchical because the p-value of other parameters (such as feed rate) is more than 0.05. Therefore, entering these parameters into the equation reduced the significance of the model. Figure 6 shows the contour and 3D plot of the AP of the nanofiber layer as a function of the three input parameters (PCL, PVP, and feed rate). Contour and 3D plot of AP: (a) the effect of PCL and PVP concentrations at a feed rate of 1.5 mL/h, (b) the effect of the rate and PVP concentration at a PCL concentration of 12.5%, and (c) 3D plots of AP of the fiber layer as a function of PCL and PVP concentrations at a feed rate of 1.5 mL/h.
As shown in Figure 6, the AP of the layer decreases as the concentration of the PVP component increases. The explanation for this result is related to the effect of concentration on the diameter of the fibers. An increase in concentration causes an increase in the diameter of the fibers, while an increase in the diameter decreases the free space between the fibers for airflow so that the AP decreases.
Optimization
Production parameters of the optimized sample and comparison of experimental and theoretical values of reaction parameters.

The optimized sample produced based on the parameters and replicates (6 runs) suggested by the RSM and the average of the six series of data is shown in Table 3. Moreover, the theoretical value of the data was calculated via equations (3) and (4). It is clear that the experimental and theoretical values are close to each other, so it is proved that the theoretical equation is practical and accurate enough for predicting the fiber diameter and AP of PCL/PVP/Henna wound dressings.
Performance testing of optimal PCL/PVP/Henna wound dressings
Biological tests (antibacterial behavior, drug release, and MTT), physicochemical tests (WVP and swelling ratio), and mechanical tests (modulus, tensile strength, and tensile elongation) are critical tests that confirm the successful performance of a wound dressing. The antibacterial results for the optimal sample show that the use of the Henna extract resulted in an antibacterial property of the PCL/PVP mat. The growth inhibition zone for the optimum sample was found to be 4–5 mm in diameter (Figure 7(a) and (b)). The antibacterial performance of electrospun fibrous structures containing Henna has been demonstrated by previous researchers.18,19 Various phenolic compounds such as 2-hydroxy-1,4-naphthoquinone, and flavonoids in Henna extracts may affect the antibacterial activity of Henna-loaded wound dressings. Phenolic compounds could bind with the bacterial cell wall and inactivate its function.
16
Biological and physicochemical testing of the optimal wound dressing: (a) the growth inhibition zone for E. coli, (b) the growth inhibition zone for S. aureus, (c) drug release profile, (d-f) MTT behavior, (g) water vapor permeability, and (h) swelling ratio.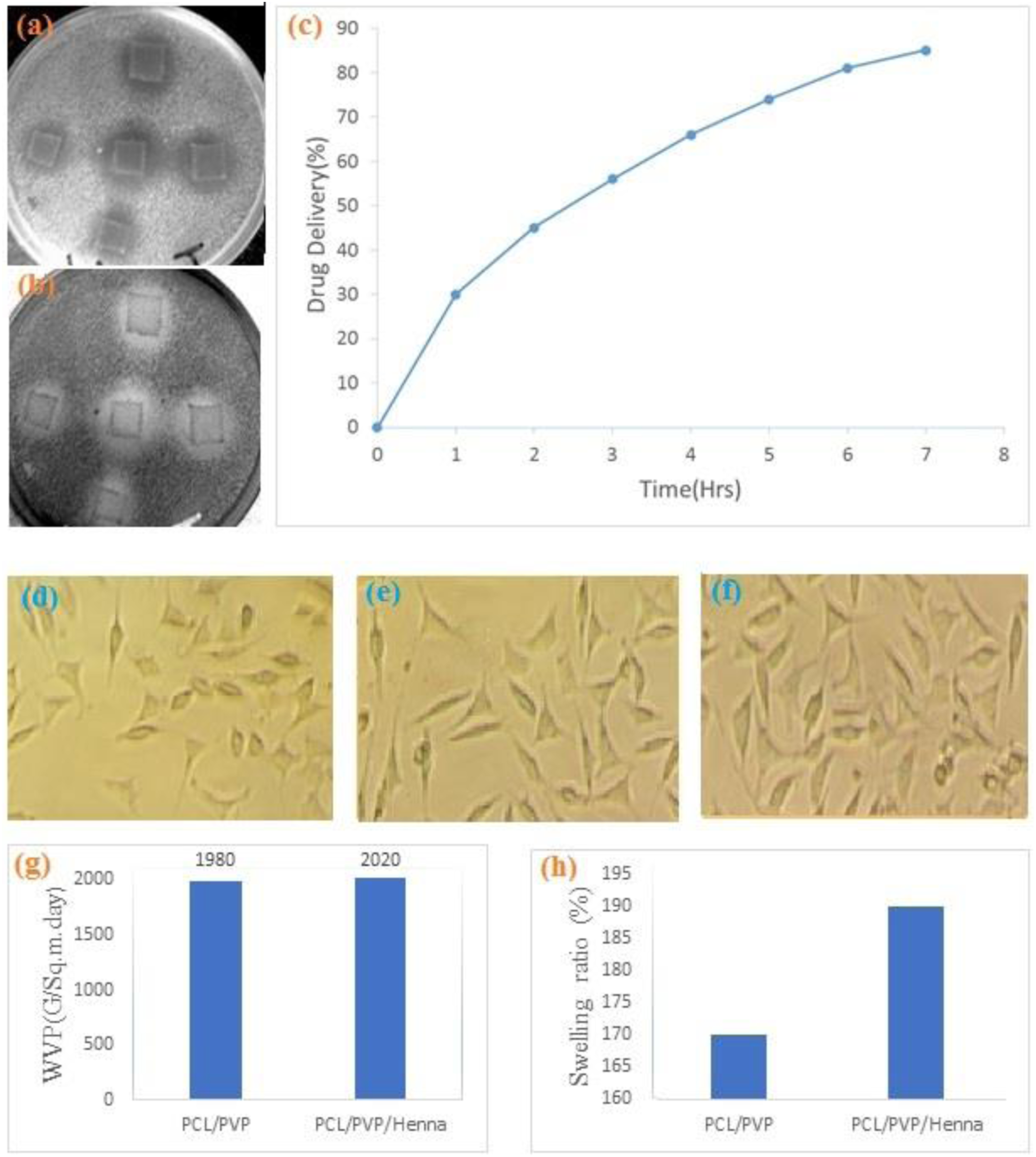
The profile of the drug release from the optimal fibrous layer is shown in Figure 7(c). A high release rate was observed in the initial phase, followed by a slow-release behavior over time. This may be due to the saturation of the buffer medium and reduction of Henna in the dressing as the release process progresses. Due to the high concentration of Henna in the wound dressing and the rapid swelling behavior, there is an abrupt and increased Henna release by pore diffusion release. 17 This rapid release has been reported in other Henna-loaded dressings and can overcome wound infection immediately.17,19
The MTT test shows the morphology of the fibroblast cells of the human skin in the control group together with the cells post-seeded on the top of the wound dressing (Figure 7(d)–(f)). There is no significant difference between the PCL/PVP as control wound dressing (Figure 7(d)) and the PCL/PVP/Henna wound dressing at 24 and 48 h (Figure 7(e) and (f)) in terms of cell growth and morphology. With regard to these results, no cell toxicity was observed. This agrees with previous studies that reported no toxicity for the mixture of PCL/PVP.15,19
The physicochemical tests show that Henna loading had no significant effect on the WVP of the fibrous wound dressing than the PVP/PCL control sample (Figure 7(g)). The WVP of mats is mainly dependent on the morphology (number and dimension of pores) of the fibrous structures rather than the composition and additives. 21 This means that the addition of Henna as an antibacterial agent does not alter the permeability properties of PVP/PCL wound dressings. However, the degree of the swelling of wound dressings containing Henna (Figure 7(h)) is significantly higher than that of PVP/PCL dressings, which may be caused by the hydroxyl groups of Henna forming hydrogen bonds with water molecules and improving the swelling properties. 16
Mechanical properties of the Henna-loaded PCL/PVP wound dressing.

Conclusion
The fiber diameter and AP of the PCL/PVP/Henna wound dressing were optimized as a function of the polymer concentration and feed rate. FTIR spectra proved that the blended structure had formed correctly. The fiber diameter varied from 75 to 342 nm, depending on the fabrication conditions. The AP results showed that as the concentration of the components increased, the AP generally decreased because the porosity of the layer directly affected the AP. The optimized dressing was able to kill both E. coli and S.aureus, while it showed no toxicity according to the MTT assay. Due to the good WVP, swelling ratio, and mechanical performance, PCL/PVP/Henna nanofibers are suitable as wound dressings to prevent infection and accelerate wound healing.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

