Abstract
Chitin, a cell wall polysaccharide, extracted from Nelumbo nucifera rhizome (NNR), was subjected to microwave treatment to modify its physical and functional characteristics. The NNR flour was irradiated at different levels of the microwave treatment period (1, 2, 3, 4, and 5 min). Chitin was extracted from the native and microwave-treated samples and analyzed for physical and functional characteristics. The microwave treatment resulted in some variations in the extract yield, structure, morphology, and composition of chitin that were directly correlated with its functional properties. Regression analysis of the data showed a significant (p < 0.05) time-dependent linear decrease in extract yield, polynomial decrease in water-holding and swelling capacities, an exponential increase in oil holding, and an exponential decrease in iron-binding capacity of chitin extracted from microwave-treated flour. These variations in the studied functional properties may be due to microwave-induced hydrolytic degradation of chitin, structural rearrangements, and exposure of some lipophilic functional groups on the surface of chitin. The data would be a valuable contribution to the literature regarding microwave-induced modification in physical and functional characteristics of chitin present in N. nucifera rhizome and other plant-based biomaterials of industrial importance.
Introduction
Chitin is the second most abundant natural polysaccharide after cellulose in the universe. Chemically, it is a polymer of N-acetyl-D-glucosamine units and plays a significant role in the synthesis and function of the cell wall in higher plants and exoskeleton in crustaceans, crabs, shrimps, and arthropods. It is an under-utilized waste of agriculture and fishing industries.1–3 Recent studies have confirmed that chitin and its derivatives may preferably be used in medicine, dentistry, biotechnology, agriculture, pharmaceutical, food processing, textile production, and environmental protection.3–6 Chitin from plant sources plays a significant role in human nutrition. Chitin and chitosan are used as a dietary fiber to facilitate the digestion process. Chitin and chitosan are dissolved in the stomach and form an emulsion with intra-gastric oil droplets, precipitate at pH 6.5–6.8 in the small intestine, entrap oil in their matrixes, pass through the lumen, and empty into the feces. 7 These also play a significant role in the structure and texture of various food products. The chitin and its derivatives also possess various biological activities including antioxidant, antibacterial, wound healing, and anticancer activities.2,8 Chitin is easily degraded by microorganisms and can be a valuable candidate for the synthesis of environment-friendly biodegradable polymers of commercial importance. It could be an important renewable source for medicinal, industrial, and food applications after some modifications.
Recent investigations are focusing on the novel methods of modification of chitin and its derivatives to enhance its industrial and biomedical applications. 9 The structural and functional quality of chitin and its derivatives can be improved by various physical, chemical, and enzymatic modifications methods including ultra-sonication-assisted supercritical CO2 treatment, ionic-liquid, cyclic acid anhydride and polypyrrole treatment, oxidative polymerization with poly (3-hexylthiophene), polystyrene grafting, and photo crosslinking with methacrylic anhydride.10–20 The high-frequency microwave irradiation technique has been also used for the extraction, isolation, and modification of chitin from shrimp and arthropods skeleton.8,22,23 The microwave treatment has been found to change the extraction yield, molecular structure, crystalline patterns, physical properties, and functional quality of chitin and other polysaccharides from different sources.22,24–26
Nelumbo nucifera rhizome (NNR) is an important source of dietary fiber, nutritionally important biochemical constituents, bioactive phytochemicals, and industrially applicable polysaccharides. 27 Along with cellulose, pectin, guar gum, chondroitin sulfate, dextran, and inulin, it also contains chitin as a major polysaccharide. 28 Previously, studies have been reported on the structural and functional modification of chitin extracted from the cytoskeleton of plants and fungi and the exoskeleton of some animals using physical, chemical, and enzymatic methods.11,18,20,21 Studies have been also reported on the physical modifications in the structure, and the physical and functional characteristics of starches and other polysaccharides present in N. nucifera seed and rhizome.26,28,29 However, the literature lacks data regarding the microwave-induced modifications in physical and functional properties of chitin present in NNR. Therefore, the present study was designed to investigate the effect of microwave treatment on the physical and functional characteristics of chitin extracted from NNR for its industrial, environmental, and biomedical applications. The data would be a significant contribution to the literature regarding the modification of structural and functional quality of chitin from plant sources to increase its nutritional, pharmaceutical, biomedical, and industrial importance.
Experimental
Sampling and preparation of flour
N. nucifera rhizomes (2 kg, about 15–20 pieces of an average length of 15 ± 5 cm) were collected from the local fields, cultivated during the crop season spring 2018. The samples were washed first with fresh water and then with distilled water to remove mud and other contamination. The fine slices of rhizomes, cut with a sharp knife, were dried in airflow under the shade, ground using pastel and mortar, and sieved through a fine muslin cloth. The flour, thus obtained, was subjected to microwave treatment.
Microwave treatment
The NNR flour was irradiated at five levels of microwave treatment period (MWTP) (1, 2, 3, 4, and 5 min) in open glass jars at the low-medium intensity (200 W) using a microwave oven (Microwave-Orient-OMW-720-ADL). After each minute, the treatment was discontinued for 30 s and the sample was mixed thoroughly to avoid burning. The native and the microwave-treated flours were preserved in light-protected, moisture-free, and airtight glass jars at sterile and thermally controlled (25 ± 2°C) laboratory conditions to minimize the microbial contamination during the study period.
Extraction of chitin
Chitin was extracted from the native and microwave-treated NNR flour by the previously described method.
30
NNR flour (10 g) was soaked in 0.68 M HCl (100 mL) for 6 h at room temperature. The sample was washed with dilute acid until the appearance of bubbles and color change was stopped. The sample was then washed with distilled water until neutral pH and dried till constant weights. Then, the dried sample was soaked in 0.62 M NaOH (1:10 w/v) for 16 h at room temperature. The sample was washed again with distilled water until neutral pH and dried till constant weight. The yield of chitin was calculated as
The overall procedure for microwave treatment of flour and extraction and analysis of chitin is presented in Figure 1. Overall procedure for microwave treatment of flour and extraction and analysis of chitin. *SEM: Scanning electron microscopy, EDX: Energy-dispersive X-ray spectra, FTIR: Fourier-transform infrared, TGA: Thermogravimetric analysis, WHC: Water-holding capacity, OHC: Oil-holding capacity, SC: Swelling capacity, IBC: Iron-binding capacity.
Physical characterization
Scanning electron microscopy/energy-dispersive X-ray analysis
The structure, morphology, and elemental composition of chitin extracted from the native and microwave-treated NNR flour were observed by taking the scanning electron microscopic (SEM) images and energy-dispersive X-ray (EDX) spectra. SEM analysis was performed using a scanning electron microscope (Model: JEOL-JSM-6480) by previously reported protocols. 31 The dried powder of chitin was suspended in anhydrous ethanol, transferred to a cylindrical microscope stub, and covered with a gold-coated carbon strip, and scanned under an electron microscope at 200, 50, and 20 μm. The elemental composition of chitin was determined by an EDX analyzer equipped with an electron microscope.
Fourier transform infrared analysis
The KBr pellets of chitin were prepared by mixing potassium bromide with thin films of chitin (sample: KBr ratio 1:100 w/w). The chitin-KBr pellets were scanned in the IR region (wavenumber 4000–650 cm−1) at a resolution of 2 cm−1 on a Fourier transform infrared (FTIR) spectrophotometer (Model: FTIR-8100A, manufactured by Shimadzu, Japan) using previously reported protocols. 32
Thermogravimetric analysis
The thermal stability and decomposition patterns of chitin extracted from the native and microwave-treated NNR flour were determined by thermogravimetric analysis using a thermogravimetric analyzer (STA449F3, Netzsch, Germany). The powdered sample of chitin (2 mg) was heated in the sample pan from 30–800°C at a rate of 10°C/min under a protective nitrogen atmosphere (flow rate 40 mL/min).
Functional properties of starch
The water-holding, oil holding, swelling, and iron-binding capacities of chitin extracted from the native and microwave-treated NNR flour were determined by the methods described in literature.26,32
Statistical analysis
The results of extract yield and functional properties of chitin were presented as mean ± SD of three replicates. The significance of microwave-induced variations at different levels of treatment time was analyzed by one-way analysis of variance (ANOVA) and the means were separated using Tukey’s multiple range test at 95% confidence level (p ≤ 0.05). Regression analysis of the data was performed to find out the trend and extent of variation in the studied parameters under the influence of microwave treatment.
Results and discussion
Extract yield
The microwave treatment has been known to affect the structure and activities of chitin and other plant-based polysaccharides. It has been found to affect the physical, functional, and antioxidant properties of soluble and insoluble polysaccharides of root, stem, leaves, and fruits of various plants.25,26,32–35 It has been also found to significantly affect the extract yield, morphology, crystalline structure, and functional properties of N. nucifera seed and rhizome starch.26,32 The present study reports the effect of microwave treatment on the structural, physical, and functional properties of cell chitin present in N. nucifera rhizome.
Experimental values of extract yield and functional properties of chitin extracted from the native and microwave-treated Nelumbo nucifera rhizome flour.
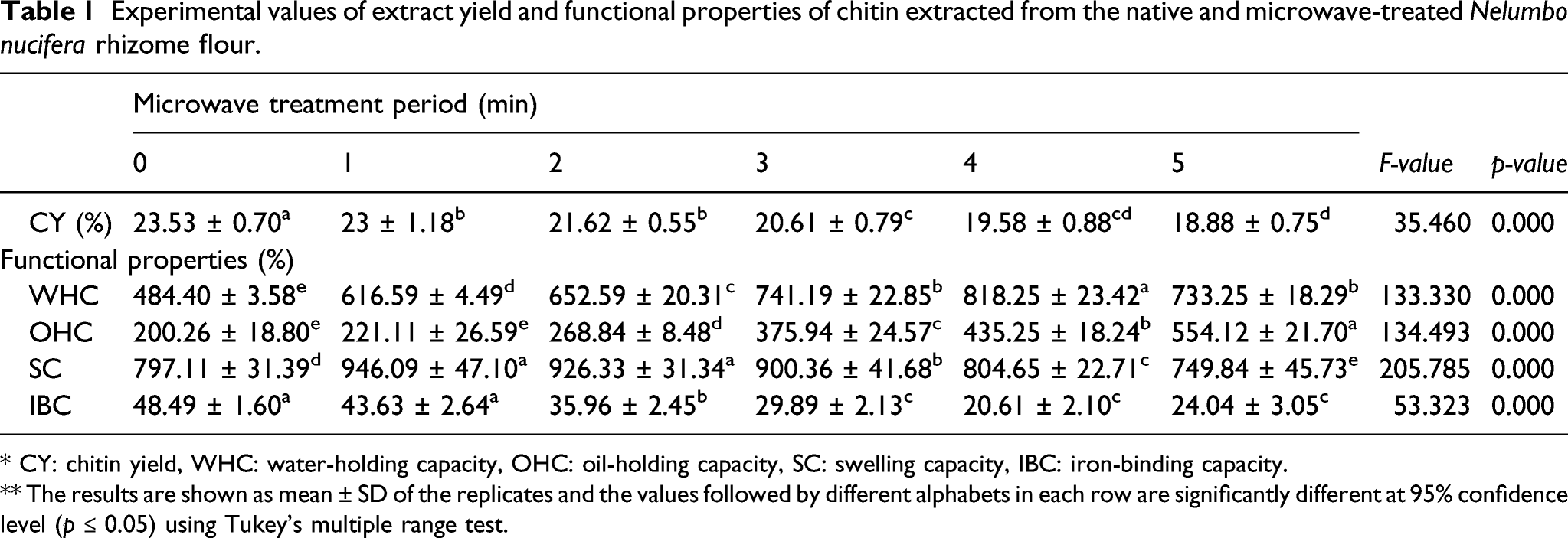
* CY: chitin yield, WHC: water-holding capacity, OHC: oil-holding capacity, SC: swelling capacity, IBC: iron-binding capacity.
** The results are shown as mean ± SD of the replicates and the values followed by different alphabets in each row are significantly different at 95% confidence level (p ≤ 0.05) using Tukey’s multiple range test.

(a) Chitin yield (CY) and (b) agreement between experimental and predicted values of CY of the native and microwave-treated N. nucifera rhizome flour. *MWTP: Microwave treatment period.
Parameters of regression analysis of functional properties of chitin extracted from microwave-treated N. nucifera rhizome flour a .

* WHC: water-holding capacity, OHC: oil-holding capacity, SC: swelling capacity, IBC: iron-binding capacity.
The predicted values of CY were obtained from the above regression equations and plotted against the actual ones to test the applicability of the suggested regression model (Figure 2(b)). The plot showed a good agreement (R 2 = 0.9413) between the actual and predicted values of the CY indicating the suitability of the suggested regression model to study the time-dependent response of CY towards microwave treatment.
The observed decrease in CY of chitin extracted from the microwave-treated NNR flour compared to that from the native flour may be attributed to the conversion of chitin to some chitooligosaccharides by microwave-induced hydrolytic degradation. Similar patterns of extract yield have been reported earlier for starch extracted from microwave-treated N. nucifera rhizome and seed kernel.26,32
Physical characterization
Scanning electron microscopy and energy-dispersive X-ray analysis
The morphological characteristics of chitin extracted from the native and microwave-treated NNR flour observed by SEM are displayed in Figure 3(a) and (b). The chitin extracted from the native NNR flour showed irregular thread-like morphology with no well-defined and measurable shape. Microwave treatment of the flour resulted in a disrupted morphology of chitin with the appearance of small particles. Scanning electron micrographic images of chitin extracted from N. nucifera rhizome flour: (a) Native and (b) microwave treated (MWTP: 5 min).
The variation in the elemental composition on the surface of chitin was determined by EDX. The EDX spectra and the numerical data of different elements present in chitin extracted from the native and microwave-treated NNR flour are presented in Figure 4(a) and (b). The EDX spectra showed a decrease in the peak height of carbon and an increase in that of oxygen in chitin extracted from microwave-treated NNR flour as compared to that extracted from the native NNR flour. The microwave treatment resulted in a decrease in the weight % (42.69–27.72) and atomic % (51.94–36.88) and an increase in weight % sigma (1.63–2.39) of carbon present in the chitin extracted from microwave-treated NNR flour. The weight %, weight % sigma, and atomic % for oxygen present in the chitin extracted from microwave-treated NNR flour were found to be increased from 47.73 to 56.91, 1.66 to 2.40, and 43.60 to 56.84, respectively as compared to that present in the native NNR flour. However, a peak of sodium and three peaks of calcium in the native sample, and two peaks of potassium in microwave-treated samples were also observed as impurities. Energy dispersive X-ray spectra of chitin extracted from the native and microwave-treated N. nucifera rhizome flour: (a) Native and (b) microwave-treated (MWTP: 5 min). *MWTP: Microwave treatment period.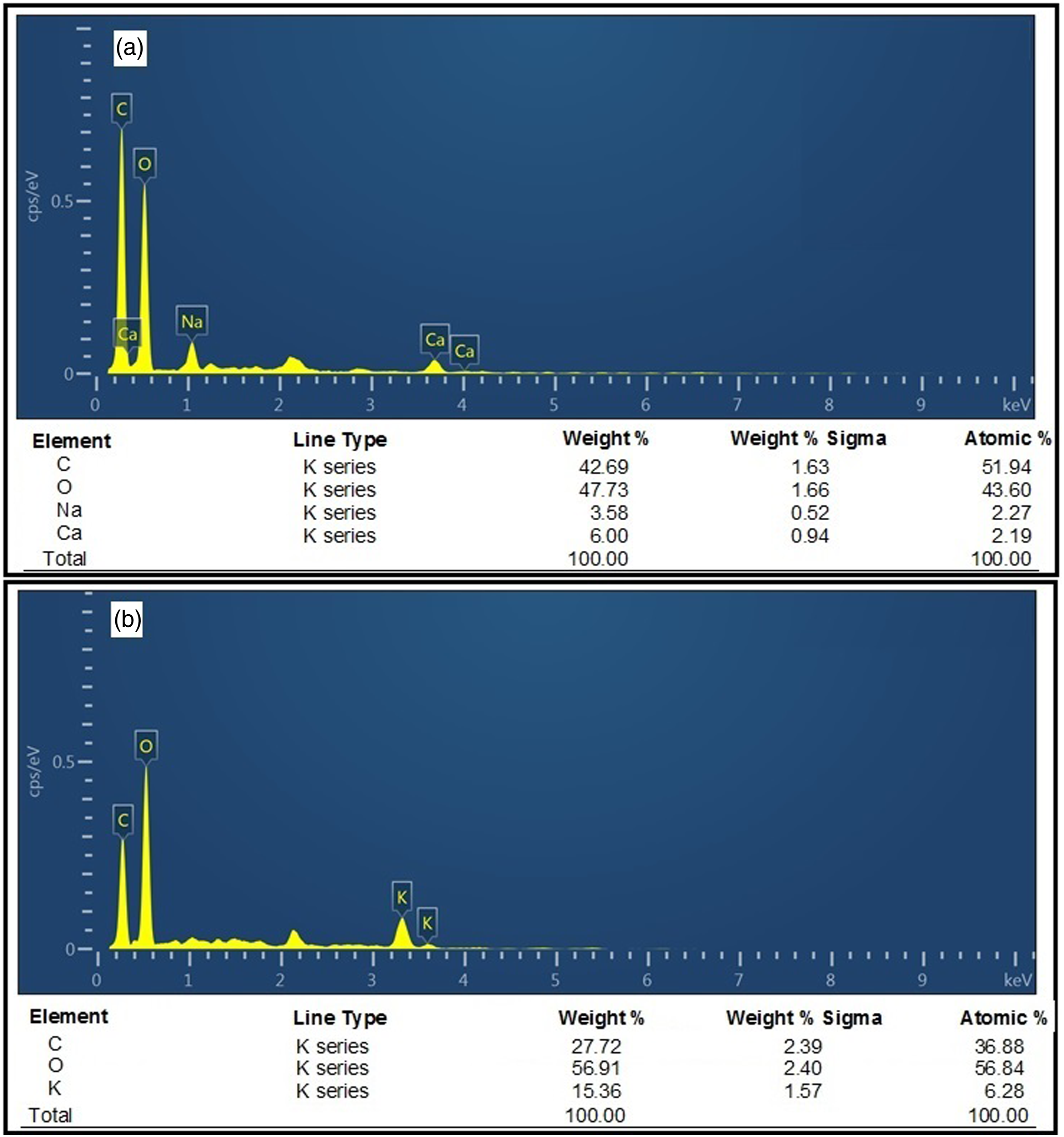
The disruption in the structure and appearance of small particles in the SEM image, decrease in peak height, weight % and atomic % of carbon, and increase in the peak height and atomic % of oxygen provide the evidence for the microwave-induced structural changes in chitin extracted from N. nucifera rhizome. The appearance of small particles in the SEM image indicated the hydrolytic degradation of chitin into some oligosaccharides under the influence of microwave treatment. 36
Fourier transform infrared spectra
The FTIR spectra (wavenumber 650–4000 cm−1) of chitin extracted from the native and microwave-treated NNR flour are presented in Figure 5. The chitin extracted from the native NNR flour showed the characteristic peaks at 898, 1020, 1152, 1320, 1405, 1595, 2920, and 3315 cm−1 indicating the presence of chitin and some ester and amide linkages. The microwave treatment resulted in variations in the transmittance bands of chitin at each of the said wavenumbers from 81.76 to 89.24, 61 to 78.62, 81.61 to 89.96, 83.13 to 89.95, 83.37 to 89.96, 81.10 to 89.20, 88.15 to 92.20, and 82.05 to 89.86 units, respectively. Fourier-Transform Infrared spectra of chitin extracted from the native and microwave-treated N. nucifera rhizome flour. *MWTP: Microwave treatment period.
The FTIR spectra confirmed the observed structural and compositional variation in the chitin extracted from the microwave-treated NNR flour. Normally, the absorbance of different functional groups present in chitin extracted from native materials ranges from wavenumber 700 to 3400 cm−1. The absorbance bands at 898 and 1020 cm−1 indicate the stretching vibration for −C−O−C− bonds of the glucosamine ring and 1,4 glycosidic bonds, respectively. The absorbance bands at 2920 and 3315 cm−1 indicate the stretching and vibrations due to the presence of methyl (−C−H) and hydroxyl groups (−O−H) in chitin, respectively. The absorbance bands at 1330, 1405, and 1595 cm−1 may be due to the vibration due to amide (−CN−), amine (−N−H), and some carbonyl (−CO−) groups .37–39 The increase in the transmittance at the observed characteristic peaks may be attributed to the microwave-induced variation in the –C−H stretching and number of −O−H, −CN−, and −CO− groups in the structure of chitin extracted from the microwave-treated NNR flour. These microwave-induced variations in the structure of chitin may be correlated with those observed in SEM/EDX analysis. The results are in agreement with those reported earlier.5,8
Thermogravimetric analysis
The observed weight-loss curves, obtained from the thermogravimetric analysis, showing the thermal behavior of chitin extracted from the native and microwave-treated NNR flour are presented in Figure 6. The chitin extracted from both the native and microwave-treated NNR flour showed almost similar patterns of thermal stability and decomposition with a slight decrease in decomposition temperature (1–2°C) in a range of 150–330°C. Both samples showed multistep decomposition curves resulting in an overall 75% weight loss. Both samples showed similar curves for loss of moisture (up to 15%) at 40–230°C. A fast increase in weight loss indicating the fast decomposition of chitin of native sample (40%) was observed at 230–360°C while that of the microwave-treated sample (36%) was observed at 230–330°C. However, the decomposition rate of chitin started decreasing from 360°C (native) and 330°C (microwave-treated) and became almost similar and constant while heating at 750–770°C. Thermogravimetric curve of chitin extracted from the native and microwave-treated N. nucifera rhizome flour. *MWTP: Microwave treatment period.
Microwave treatment resulted in a decrease in decomposition temperature of chitin extracted from microwave-treated NNR flour as compared to that of the native one. The chitin extracted from NNR flour showed relatively fast decomposition at 230–325°C but a slow weight loss at 326–770°C than that extracted from the native NNR flour. The observed variation in the weight-loss ratio of the native and microwave-treated chitin at different temperature ranges may be attributed to the microwave-induced structural changes in chitin.
Functional properties
The experimental values of the studied functional properties including water-holding capacity (WHC), oil-holding capacity (OHC), swelling capacity (SC), and iron-binding capacity (IBC) of chitin extracted from the native and microwave-treated NNR flour are presented in Table 1. The WHC, OHC, SC, and IBC of chitin extracted from the native and microwave-treated NNR flour ranged from 484.40 ± 5.58 to 818.25 ± 23.42, 200.26 ± 18.80 to 554.12 ± 21.72, 749.84 ± 45.7 to 946.09 ± 47.10, and 20.61 ± 2.10 to 48.49 ± 1.60%, respectively. The chitin extracted from the microwave-treated NNR flour showed comparatively higher values of WHC, OHC, and SC but lower values of IBC than those of the native one. Regression analysis of the experimental data showed a significant (p = 0.00) time-dependent microwave-induced exponential increase in OHC, exponential decrease in IBC, and polynomial variation in WHC and SC with relatively higher values of coefficients of determination (R
2
= 0.8513–0.9820) (Figure 7(a)–(d)). The relationship between the functional properties of chitin and MWTP was explained by the following generalized regression equations (a–d) Functional properties and (e–h) Agreement between experimental and predicted values of functional properties of chitin extracted from the native and microwave native and microwave-treated N. nucifera rhizome flour. (a, e) Water-holding capacity, (b, f) oil-holding capacity, (c, g) swelling capacity, (d, h) iron-binding capacity. *MWTP: Microwave treatment period.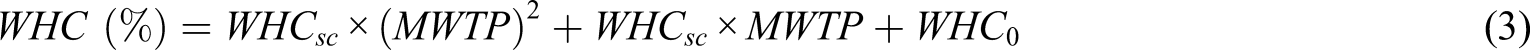



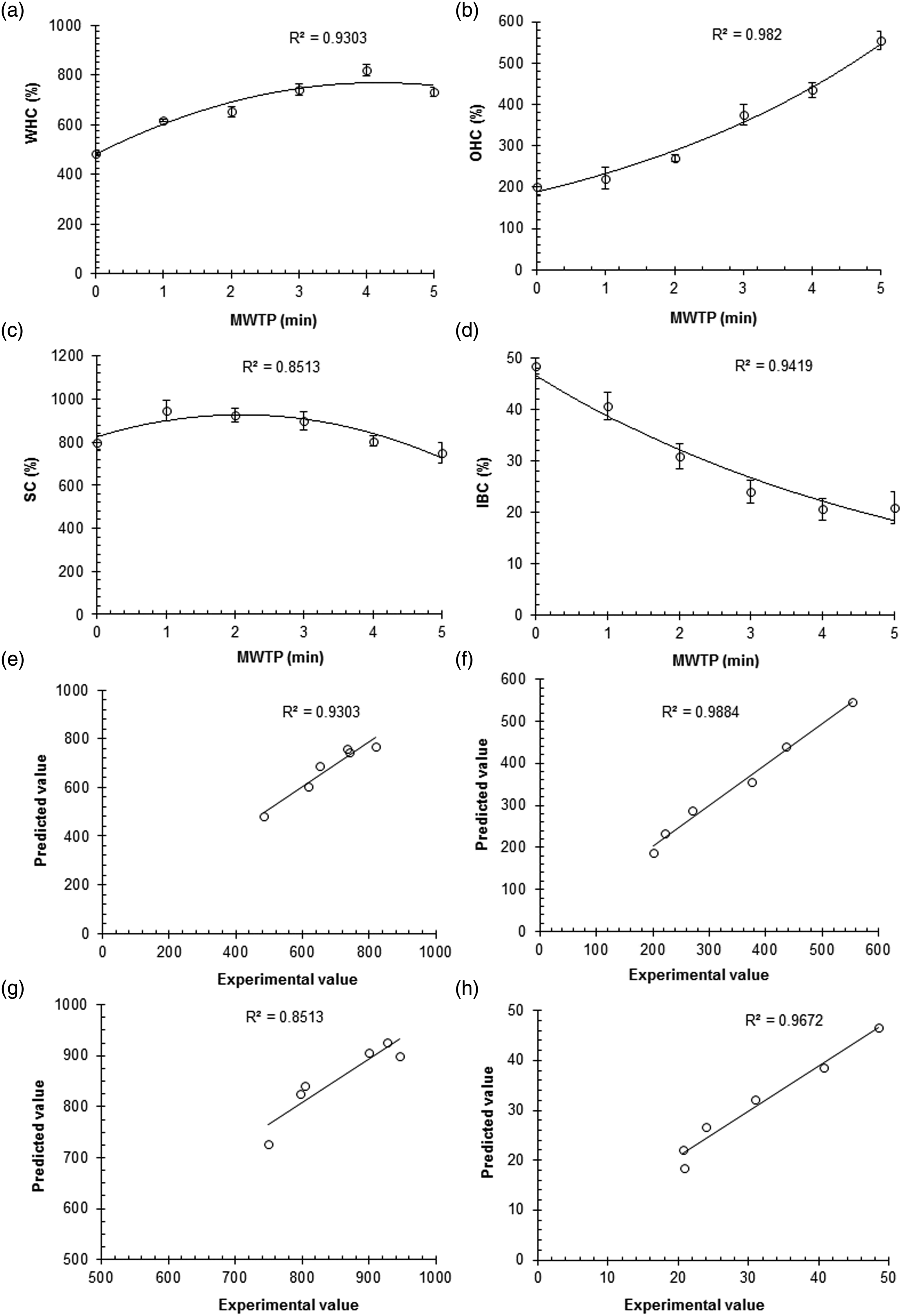
The above equations were used to calculate the predicted values of the studied functional properties of chitin. To test the applicability of the suggested regression model, predicted values calculated from the regression equations were plotted against the actual ones (Figure 7(e)–(h)). The correlation plots showed a good agreement between the actual and predicted values with good signs of correlation coefficients (R 2 = 0.8513–0.9884) indicating the suitability of the suggested regression model to study the time-dependent response of functional properties of chitin towards microwave treatment.
The microwave-induced variation in the studied functional properties may also be correlated to the structural and compositional changes in chitin extracted from NNR flour subjected to microwave treatment. The polynomial variation WHC and SC may be attributed to the MWTP-dependent exposure of polar functional groups and degradation of chitin. The exponential increase in OHC may be due to the microwave-induced exposure of some nonpolar functional groups on chitin increasing its lipophilicity. The increase in WHC and SC at microwave treatment for 1–3 min indicates the increased exposure of hydrophilic functional groups. However, the decrease in these functional properties after 3 min of microwave treatment suggests the time-dependent denaturation of the polysaccharide structure. These results are in agreement with the previous studies suggesting the microwave-induced changes in the hydration properties including water-holding and swelling capacities of chitin and other cell wall polysaccharides by increasing their hydrophilicity.35,40,41 The chitin possessing relatively lower values of WHC and SC and higher OHC may be favorable for the preparation of moisture-free products. The exponential decrease in IBC may also be attributed to the microwave-induced structural changes resulting in the decrease of iron loving groups on the surface of chitin. The chitin possessing relatively low IBC is considered a poor adsorbent and favorable for the preparation of metal-free industrial products.42–44 The results for a microwave-induced increase in the studied functional properties are in agreement with those reported earlier for N. nucifera seed starch. 32 However, the results for OHC and SC disagree with those reported for N. nucifera rhizome starch. 26
The results showed a structure–activity relationship for chitin extracted from microwave-treated NNR flour. The decrease in CY and variation in functional properties in response to an increase in MWTP may be correlated with the microwave-induced structural modification in chitin as indicated by the SEM images and EDX and IR spectra. The MWTP-dependent variation in WHC, OHC, SC, and IBC of starch may be attributed to the exposure of some hidden functional groups due to microwave-induced variations in starch structure.
Conclusion
In conclusion, the microwave treatment resulted in a disruption in morphology, hydrolytic degradation of structure, and change in elemental composition, and modification in the functional quality of chitin extracted from NNR flour. The microwave treatment resulted in a time-dependent significant linear decrease in extract yield, an exponential decrease in IBC, an exponential increase in OHC, and polynomial variation in WHC and SC of chitin. These microwave-induced modifications in the structure and properties may improve the functional quality of chitin present in NNR for its use in various industrial, pharmaceutical, and medical preparations requiring materials with relatively low hydrophilic interactions. The data may be a significant contribution to the literature regarding the microwave-induced modification of plant-based biomaterials of industrial importance.
Footnotes
Acknowledgments
The authors are grateful to the Department of Biochemistry, Bahauddin Zakariya University, Multan, Pakistan, and College of Natural and Health Sciences, Zayed University, Abu Dhabi, United Arab Emirates, for providing the facilities for conducting the research work.
Declaration of conflicting interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


