Abstract
One of the storage conditions affecting quality of food stuffs due to short shelf life is temperature. Thermal insulation can be achieved by adding phase change materials (PCMs) to packaging materials. PCMs store and release latent heat of phase change during melting and crystallization operations, respectively. Thus, they can provide thermal protection for packaged foods. The aim of this study is to prepare new food packaging materials poly (ethylene terephthalate)–poly (ethylene glycol) (PET–PEG) multiblock copolymers as solid–solid phase change materials (SSPCM) as potential food packaging materials with thermal energy storage (TES) property. Polyesterification was carried out with PEG at different average molecular weights (1000, 4000 and 10,000 g/mol), ethylene glycol (EG) and terephthaloyl chloride (TPC). Synthesized PET–PEG multiblock copolymers were characterized using Fourier transform infrared (FT-IR) spectroscopy and differential scanning calorimetry (DSC) methods. The crystal structures of PET–PEG multiblock copolymers were characterized by polarized optical microscopy (POM) and their surface properties were determined by performing contact angle tests. TES capacity of the PET–PEG multiblock copolymers was found in range of 26.1–150.5 J/g. Consequently, this study demonstrates the potential of PET–PEG multiblock copolymers suitable for effective thermal preservation in packaging material applications to maintain the quality of packaged food stuffs.
Keywords
Introduction
Some of phase change materials (PCMs) so called functional materials are applied as packaging materials to preserve foods without using petroleum fuel or electricity. Temperature changes in the food transport chain can have negative consequences for product quality and safety. Packaging systems containing PCMs can keep product quality under control during transportation and storage. Considering energy and environment, thermal energy storage (TES) is renewable, that is, TES system stores heat and uses it when necessary. Thus, it protects the environment and benefits economically.1,2 When it comes to food packaging, plastic, cardboard or wooden packaging usually comes to mind. These packages have limited insulation properties. In order to keep the products at desired temperature, products can be stored using packaging with PCM properties. PCMs are substances that exhibit phase transition (e.g. fusion/crystallization) at a given temperature or a narrow temperature range. Latent heat energy was stored during the phase transition and then this energy was released back into the environment. 3 Heat rise can be absorbed by material through phase transition, thereby limiting the increase in product temperature. 4 Limited insulation properties and low thermal storage capacity of known packaging materials need further improvements for protection in the cold chain. There are ideas for imparting TES to the thermal storage capacity of a package by the addition of PCMs.5,6 The basis of this idea is that PCMs are one of the most preferred energy storage methods with their high energy storage capability and obtainable isothermal heat storage properties. 7 PCMs can be used for the protection of such as solid and liquid food products and biological products in storage and distribution that are important for logistics.
Food safety should be ensured in the protection of food stuffs (microbiological, chemical and physical) and in temperature fluctuations. PCMs are generally paraffins, salt hydrates, fatty acids, organic and non-organic eutectic mixtures. PCMs can be divided into three main groups depending on TES phase transition temperatures: (i) PCMs with capable of phase change below 15°C; (ii) PCMs with a phase transition temperature in the range of 15–90°C and (iii) High temperature PCMs with phase transition temperatures above 90°C. PCMs can also be classified as gas–liquid, solid–gas, solid–liquid and solid–solid phase transition materials. 8
Cold storage (2–8°C) is used to prevent microbial, chemical and/or enzymatic activities. In case of a problem in cold system, heat wave can occur causing cold chain microbial or chemical change in products. This situation can cause a decrease of expiration date of foods. Therefore, there are many studies on reducing cold chain temperature fluctuations. In this sense, functional packages can be important keeping temperature of the food within the desired limits thus increasing the quality and safety of the products. In cases where the heat source is inadequate or interrupted, packaging materials that can store latent heat energy are required. For example, cardboard packages have limited thermal insulation. It withstands temperature rise up to an average of several hours, which can cause food to deteriorate in packaging. 9
Significant food storage temperatures are stated to be as above 60°C, 18°C, 0–7°C, -2-(2) °C and −18-(-30)°C. 10 On the other hand, double-stuffed transport containers inside polyurethane foam for using hot and cold catering (Thermo Box) have been produced in recent years; however, it cannot be suitable for logistics due to its large volume. These synthesized solid–solid phase change materials (SSPCMs) have high latent heat energy storage capacity and they are crystalline plastics that undergo small volume change at phase change temperature.
The aim of this study is to produce PET–PEG multiblock copolymers as food packaging materials to create a thermal preservation effect for a period. PET-based polymers in the food sector, insoluble in common solvents and even affected by many other chemicals are inert featured food packaging materials. PEGs are nontoxic antigenic PCM substances. PEGs of different molecular weights (100–20,000 g/mol average), capable of storing latent heat between 3.2°C and 68.7°C provide a considerable amount of heat transfer during phase change, they are very stable. And they could be validated in SSPCM material production due to hydroxyl chain ends. 11
PET–PEG multiblock copolymers obtained using the esterification reaction are estimated to be used as food packaging materials. 12 PET–PEG copolymers were synthesized by polycondensation at 270–280°C using the conventional DMT route of PET production and have SSPCM characteristics at 10–60°C without obvious liquid substance appearing. 13 Polyester-based packaging materials were developed for foods stored in the temperature range of 3–80°C. Creating isolated environments where sudden temperature fluctuations are present is aimed to maintain stability of food stuffs. In this study, PET–PEG multiblock copolymers were synthesized as new kinds of polymeric SSPCMs from different average molecular weights of 1000, 4000 and 10,000 g/mol for PEGs to capture target temperature ranges for transportation of food and biological products at 18–60°C. PET–PEG multiblock copolymers were characterized by FT-IR and polarized optical microscopy (POM) and differential scanning calorimetry (DSC). Also, free energy of surfaces was calculated to polyester polymers from contact angle values.
Materials and methods
Materials
PEGs at average molecular weights of 1000, 4000 and 10,000 g/mol and ethylene glycol (EG) were obtained from Merck company and they were used as received. terephthaloyl chloride (TPC), N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMAc); formamide and diiodomethane were obtained from Aldrich company and they were also used directly.
Synthesis of PET-PEG multiblock copolymers
Experimental codes for PET–PEG multiblock copolymers.


PET–PEG multiblock copolymer synthesis scheme.
Characterization of PET-PEG multiblock copolymers
FT-IR spectroscopy analysis
Analyses of copolymers synthesized were carried out using FT-IR spectroscopy method. The FT-IR spectra of PET-PEG multiblock copolymers were taken on a KBr disk at 4000–4400 cm−1 wavelength using a JASCO 430 FT-IR spectrophotometer.
DSC investigations
The thermal properties of the synthesized PET-PEG multiblock copolymers solid–solid phase transition temperatures and enthalpy were determined by commonly used temperature modulated DSC analysis method (Perkin Elmer Jade, DSC). DSC analyses were performed in a flow rate of 60 mL min−1 and a heating rate of 5°C min−1 in inert nitrogen atmosphere.
Crystalline properties of PET–PEG multiblock copolymers: Polarized optical microscopy (POM) analysis
POM analyses were monitored using a Leica DM EP microscope equipped with a computer monitored digital camera. In this study, samples of PET–PEG multiblock copolymer films were heated on a coverslip at 60°C and then allowed to cool room temperature. The coverslips at both side of phase change temperature were investigated by using POM at ×40 magnification at 500 px. Surface of each sample was photographed using an integrated camera. The surface structures of the synthesized polyester multiblock copolymers were compared not only to pristine PEG polymers but also to other SSPCMs in the literature.
Results and discussion
Structural evaluation of PET-PEG multiblock copolymers
FT-IR analysis results of PET–PEG multiblock copolymers are given in Figure 2 (a) SSPCM1, (b) SSPCM2 and (c) SSPCM3. As it can be seen in the spectra, there were vibration bands of the C–H group between 2500 cm−1 and 3000 cm−1. PET group had a vibration peak at 1100 cm−1 due to the –CH2–O–CH2– skeletal structure. Also, it can be seen that vibration peak at 1240 cm−1 was due to C–O–C as the peak at 3510 cm−1 were due to hydrogen bonding interactions of PEG groups. Besides, the characteristic carbonyl peak was observed at around 1710 cm−1 in any of the FTIR spectra of SSPCMs. These results showed that copolymerization reaction was realized successfully. FT-IR spectra of PET–PEG multiblock copolymers 
Heat storage capacity of PET-PEG multiblock copolymers
Thermal properties of the synthesized SSPCMs such as solid–solid phase change onset temperature for heating and cooling process, respectively, T s–s, heating (°C) and T s–s, cooling (°C), phase change enthalpy for heating and cooling process ΔHs–s, heating (J/g), ΔHs–s and cooling (J/g) were measured by using the DSC analysis method. The DSC curves of the SSPCMs are demonstrated in Figures 3–5 and the thermal properties determined from these curves are also illustrated in Table 2. Especially, SSPCM2 and SSPCM3 have the highest latent heat storage capacities. Latent heat values were measured as 26–150.5 J/g for heating and as −62.6 – (−)133.0 J/g for cooling of SSPCM1, SSPCM2 and SSPCM3, respectively. In a similar study, synthesized β-cyclodextrin/4,40-diphenylmethane diisocyanate/poly (ethylene glycol) copolymer (CD–CD/MDI/PEG) were synthesized
13
and according to DSC results, phase transition enthalpy and melting temperature of the PEG 8000 decreased in the modified PEG state. That is, the concentration of the crystal region increased with increasing percentages of mass of PEG segments in structure. Qian synthesized a PET/PEG copolymer and reported two different phase transition temperatures at 55°C and 220°C for solid–solid and melting, respectively. DSC curves of SSPCM1. DSC curves of SSPCM2. DSC curves of SSPCM3. Thermal properties of PET–PEG multiblock copolymers.



Surface morphology of PET-PEG multiblock copolymers
The synthesized SSPCMs were composed of the hard part PET and the soft part PEG at the copolymer backbone. PEG units with low melting temperatures cannot be processed like PET at high temperatures in thermomechanical processes. This resulted in micro-phase separation with increasing temperature effect. Soft units (PEG blocks) were distributed into the matrix of hard units (PET blocks). However, presence of this hard unit adversely affected formation of a PEG crystal structure. Figure 6 demonstrates POM images of PEG 10000 and SSPCM3, PEG 4000, SSPCM2 and SSPCM1. PEG 10000 and PEG 4000 had typical spherical crystalline morphology with an apparent cross-extinction pattern at room temperature (Figure 6(a),(c), respectively). The morphology of the spherulite crystal was observed below the solid–solid phase change temperatures from the POM images of SSPCM1, SSPCM2 and SSPCM3 (Figures 6(b), (d), and (e)). The crystalline phase of PEG appeared as an indeterminate phase in SSPCMs. PEG groups in SSPCMs transformed from crystalline phase to amorphous phase and then PET chain of SSPCMs retained solid state and mechanical strength.
15
Losing spheroids image of SSPCM1, SSPCM2 and SSPCM3 polyesters is an evidence that PEG polymers were connected to PET successfully. ×500px, ×40 magnification POM images of 
Contact angle and surface energy measurements
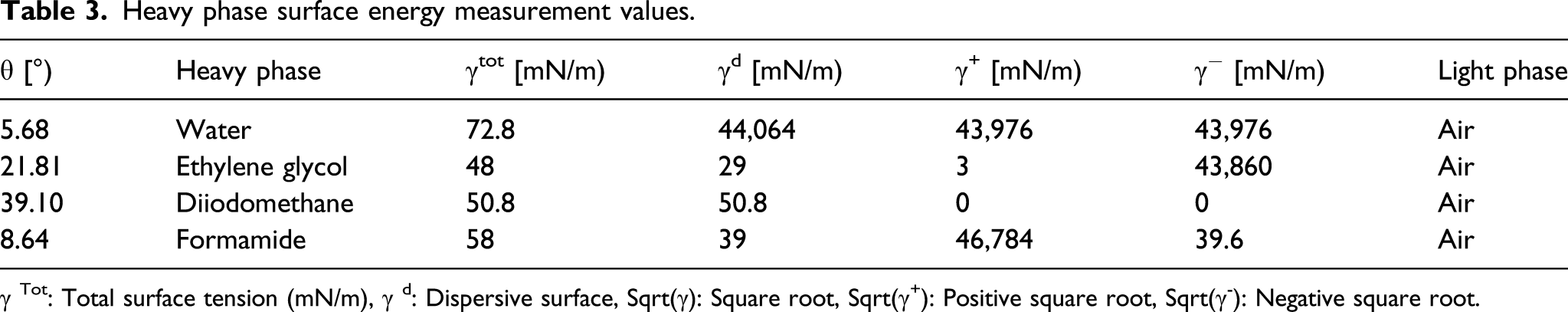
Polymer surface photographs were taken by using a computer camera system for contact angle measurements and surfaces of the PET–PEG multiblock copolymers were measured using distilled water (a) SSPCM1, (b) SSPCM2, (c) SSPCM3 respectively right, left and average contact angle values change graph of distilled water phase, ethylene glycol phase, formamide phase, diiodomethane phase of polymers. Heavy phase surface energy measurement values. γ Tot: Total surface tension (mN/m), γ d: Dispersive surface, Sqrt(γ): Square root, Sqrt(γ+): Positive square root, Sqrt(γ-): Negative square root. Contact angle test properties of PET–PEG multiblock copolymers. γ: Surface tension (N/m), θ: Contact angle, γ tot: Total surface tension (mN/m), γ aDispersive surface tension (mN/m), γ bPolar surface tension (mN/m), Sqrt(γ): Square root, Sqrt(γ+): Positive square root.


Conclusions
In this study, PET–PEG multiblock copolymers were synthesized as a new type of SSPCMs by using a simple esterification technique. Copolymers were confirmed by FT-IR spectroscopy analysis. POM studies showed that SSPCM in solid–solid phase change, the crystalline phase of the soft segment (PEG) was transformed into an amorphous phase. DSC showed that SSPCMs could store the thermal energy at the appropriate solid–solid phase transition temperature in the range 18–57°C and 26–150.5 J/g latent heat enthalpy. SSPCM phase transition enthalpy was observed to be increased by increasing the PEG mass ratio. Results obtained from DSC analysis also showed that the energy storage properties of SSPCMs were consistent. This packaging material can be environmentally friendly for minimizing use of petroleum fuel providing cooling and heating during transportation. PCMs can keep the storage temperature within the set point for an average of 150 min. 16 When SSPCM1 provides food preservation at 18°C, it can prevent temperature fluctuations and provide the quality and assurance of fresh fruits, vegetables and packaged foods that need to be stored below room temperature. SSPCM2 can maintain product temperature by storing heat energy at 57°C for hot food transportation, which is important in the catering industry. When these synthesized PET–PEG multiblock polyester SSPCM polymers are used with other packaging materials, they can be more effective due to their thermal barrier properties. The new generation packaging material produced in this study can be shaped at room temperature with cold press technology. SSPCMs have been shown to be a new generation energy storage material due to the proper phase transition temperature and reversible phase transition behaviour. Some of them can be a latent heat storage material in hot and cold food logistics and storage isolation applications below room temperature (18°C) (fruits, vegetables, etc.) and for hot transport (40–60°C) in food industry.
Footnotes
Acknowledgements
We would like to thank the Giresun University BAP Fund for its financial support for this study (Project no: FEN-BAP-A-160317-31).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
