Abstract
Dodecylbenzenesulfonic acid (DBSA) doped polyaniline and carbon black (CB) composites (PANI-DBSA/CB) were synthesized by in-situ polymerization of aniline in the aqueous dispersion of carbon black (CB) in the presence of ammonium persulfate (APS) as an oxidant. Various composite samples with varying amounts of CB (1 wt% and 2 wt%) were synthesized and were characterized by using X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), and scanning electron microscopy (SEM). XRD studies confirmed the formation of the well-organized and semi-crystalline structure of the composites. SEM analysis revealed the increase in grain size and the granular structure of the synthesized composites. DC conductivity was measured in the temperature range from 300 K to 393 K using two probe methods. An increase in DC conductivity was found on the inclusion of carbon black at all temperatures and the most probable model of charge transport was found to be three-dimensional variable range hopping (3D-VRH). As the concentration of CB in the composites increased, activation energy was found to be decreased. The density of states, hopping length, and hopping energy were calculated, respectively, and were found to be influenced by CB incorporation.
Introduction
Conducting polymers (CPs) are analogous to those of metals or semi-conductors having conjugated backbone of enriched electrons with the ability to conduct. 1 The existence of conjugated backbone within the conducting polymers (CPs) provides them with the property of electrical conductivity. 2 Ever since their discovery, CPs are of significant interest due to their ease of synthesis, good process ability, lightweight, less toxicity, and high thermal conductivity.3,4
Among other conducting polymers, polyaniline has attracted much attention in the last two decades due to their potential applications in various hi-tech aspects, such as in microelectronics, 5 active electrodes in batteries, 6 sensors, 7 as electrochromic material for displays,8,9 as well as electromagnetic shielding. 10 Moreover, PANI has considerable tunable chemical structure.11,12 Carbonous materials (CB, graphene and carbon nanotube) include non-faradaic categories, which have good electrical conductivity, eco-friendliness, and good chemical stability. 13 However, the utilization of PANI has been limited because of its poor mechanical properties, solubility, and processability. In order to enhance its solubility and processability, a number of methods have been studied. The most adopting methods is to dope these conducting polymers with organic acids possessing long alkyl-chain sulfonic acids such as dodecylbenzenesulfonic acid (DBSA) and camphorsulphonic acid (CSA). 14 CB particles also have much greater tendency to form a conductive network due to their aggregate structure, 15 which is necessary to provide a high electrical property in comparison to the other conductive fillers, not only enhances its heat but chemical resistance as well. 16
PANI composites are attractive because they can combine with the properties of other compounds, and the composites of PANI with carbon nanotubes,17,18 graphite, 19 graphene, 20 metal powders, 21 and carbon black (CB)22,23 have been accepted to effectively improve the electric conductivity.
Among the conductive organic fillers, carbon black has several advantages due to its particular ordered nanostructure, low density, large surface area, high thermal stability, good electrical conductivity, lower cost, corrosion resistance, less material requirement for percolation, and ease of processing into host polymers. 24
The present study investigates the structural, morphological, and electrical properties of PANI-DBSA/CB composites. To understand the charge transport mechanism in pristine polyaniline (PANI), polyaniline doped in dodecylbenzenesulfonic acid (PANI-DBSA) and its composites with carbon black (PANI-DBSA/CB) to find out the cause of increased electrical conductivity in these composites when compared with pristine PANI. The Mott’s parameters were calculated and were found to be influenced by adding CB in polyaniline.
Experimental part
Materials
Aniline (C6H5NH2) and DBSA (C12H25C6H4.SO3H) were purchased from Sigma-Aldrich. Hydrochloric acid (HCl) and ammonium persulfate (APS) were obtained from Fluka. Carbon black (CB) powder was obtained from Sigma-Aldrich. All the materials were used as obtained except aniline.
Synthesis of dodecylbenzenesulfonic acid doped polyaniline (PANI–DBSA)
The typical synthesis of pristine PANI was performed as follows. In 30 mL distilled water, 4.7 mL aniline was added dropwise for about 1 h and solution was left stirring for 6 h. To maintain pH between 0 and 1, HCl was added into it dropwise. After this, 11.63 g of ammonium persulfate (APS) was added dropwise for about period of 30 minutes. Whereas keeping monomer to oxidant ratio 1:1. Addition of APS into the solution turned it into greenish black color, indicating the start of organic polymerization reaction. The solution was left for 24 h to complete the polymerization process, and the next day solution was filtered with methanol and plenty of distilled water until the filtrate became colorless. Then obtained paste was kept in a vacuum oven for 24 h at 60°C. For comparative purposes, PANI-DBSA was synthesized under similar conditions with reprotonation in 1M aqueous DBSA at molar ratio 1:1 and used for synthesis of the composites using CB.
Synthesis of PANI-DBSA/CB composites
1wt% and 2wt% of aniline and CB powder 0.047 g and 0.095 g, respectively, were dispersed into 15 mL of aqueous solution of HCl. 4.7 mL aniline was added dropwise to the above solution and stirred for 1 h at room temperature to form an aniline–HCl solution containing CB nanoparticles. Then aqueous APS was added dropwise to the above solution and allowed to react for 6 h with constant stirring. A dark green precipitate was obtained and washed with methanol and distilled water several times until the filtrate was colorless. The precipitate was finally dried at 60°C under a vacuum for 24 h to obtain the nanostructured composites. Reprotonation in 1M aqueous DBSA solution was done at molar ratio 1:1. 24
Measurements
X-ray powder diffraction analysis was carried out using an automated diffractometer, Panalytical X'Pert PRO equipped with Cu Kα radiations (λ
Results and discussions
X-ray diffraction analysis
XRD pattern of different samples were studied by using Bragg’s law
Figure 1 shows the XRD pattern of the PANI, PANI-DBSA, and composites of the PANI-DBSA/carbon black composites. The X-ray diffraction pattern of the pure PANI, which has a broad diffraction peak between 10° and 30°, centering around 25.251° due to amorphous nature of polymer (PANI) chain (Figure 2(a)).
25
While in case of PANI-DBSA, the peaks appeared at position 9.81°, 19.94°, 25.16°, and 26.90° corresponding to d-values 9 Å, 4.45 Å, 3.54 Å, and 3.31 Å.
26
However, the pattern of carbon black shows two broad features at 2θ–24° and 44°. These are due to graphitic crystallites of carbons and correspond to the (0 0 2) and (1 0 0/1 0 1) lines, respectively, of the graphite structure with d-values 3.074 Å and 2.056 Å, respectively.27,28 X-ray diffraction patterns of PANI, PANI-DBSA, PANI-DBSA/1% CB, and PANI-DBSA/2% CB. XRD, X-ray diffraction; PANI, polyaniline; DBSA, dodecylbenzenesulfonic acid; CB, carbon black. (a) Plot of ln(σ) versus reciprocal of the temperature for PANI, PANI-DBSA, PANI-DBSA/1% CB, and PANI-DBSA/2% CB and (b) ln(σ) vs T−1/4 for PANI, PANI-DBSA, PANI-DBSA/1% CB, and PANI-DBSA/2% CB. PANI, polyaniline; DBSA, dodecylbenzenesulfonic acid; CB, carbon black.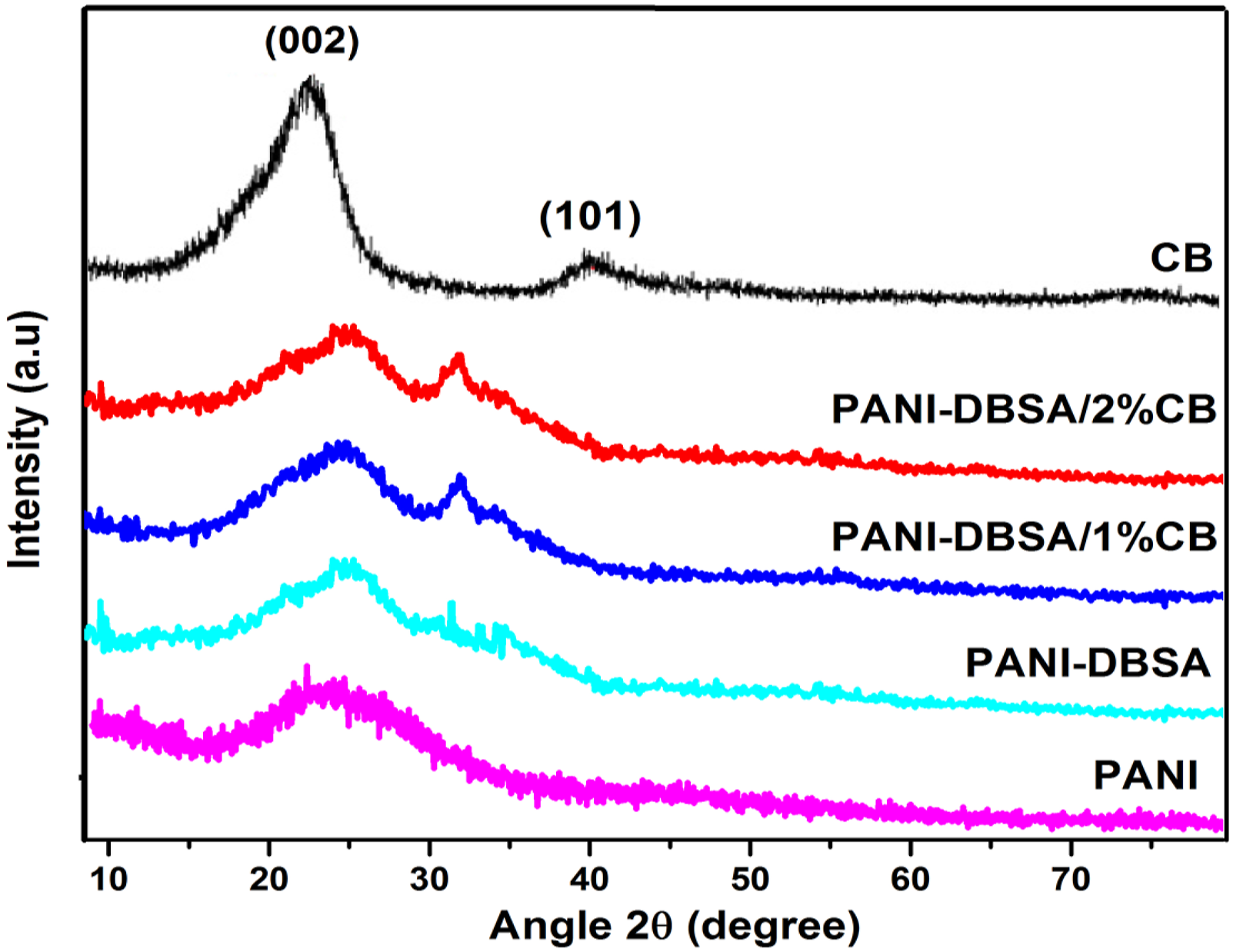

The XRD pattern of PANI/DBSA/1% CB shown as carbon black and PANI have amorphous nature so this composite also shows a very little crystallinity. One broad peak is observed at 25.74° and a shoulder at 31.78° corresponding to 3.45 Å and 2.81 Å d-values, respectively.
The XRD pattern of PANI-DBSA-2% CB shows the presence of partial crystallinity in this sample. The presence of one sharp and broad peak at 25.74° and shoulder at 31.78° shows the presence of PANI, DBSA, and CB. As the concentration of carbon black increases, the peak at 25.75° becomes sharper and its intensity also increases. Because CB has strong interaction peak at 24.9° and its overlapping with the increase of carbon black (CB) content makes peak at 25.75° sharper and its intensity also increases. It shows that with the increase of CB concentration, the composite becomes more ordered and crystalline as compared to pristine PANI.
Electrical properties
The relation between DC conductivity and temperature in polymer samples gives important information concerning the nature of phenomena related to the charge transport in a polymer system.29,30
Figure 2(a) gives the linear plots of ln(σ) versus reciprocal of the temperature, where the Arrhenius behavior is observed. These are the best linear fits to the data. According to Mott’s
31
variable range, hopping mechanism of charge transport the following relation is taken into consideration
The differential of the graph of log vs 1/kT gives activation energy at different temperatures. The Arrhenius exponential law equation is
32
So the negative value of activation energy is calculated after differentiating the graph of log
For strong inter chain coupling and temperature dependence of DC conductivity, the exponent (γ) is 0.25 and the effective dimensionality n = 3 [γ = 1/(1 + n)] and the conductivity data follow 3D VRH model. On other hand for weak inter chain coupling, VRH exponent is 0.5 (n = 1) 35 which can be explained in terms of granular metal model or quasi 1D 35 or the Efros–Shklovskii 36 VRH model.
For polyaniline by plotting log(Ea) vs log T (Figure 3), a straight line of slope {−(γ−1)} equal to 0.76 is obtained, which corresponds to hopping exponent γ ∼ 1/4 and n ∼ 3. This indicates that the 3D variable range hopping (VRH) dominates the charge transport mechanism in pristine polyaniline. Log(Ea) vs log(T) for PANI, PANI-DBSA, PANI-DBSA/1% CB, and PANI-DBSA/2% CB. PANI, polyaniline; DBSA, dodecylbenzenesulfonic acid; CB, carbon black.
A good fit of conductivity data to 3D VRH model suggests a strong inter chain coupling in this synthesized polyaniline. By plotting Log(Ea) vs log T for PANI-DBSA composite (Figure 3), a straight line with slope {−(γ −1)} equal to 0.76 is obtained, which corresponds to hopping exponent γ ∼ 1/4 and n ∼ 3. This indicates that 3D variable range hopping (VRH) dominates the mechanism of charge transport in the PANI-DBSA.
For PANI-DBSA/1% CB composite, the plot of Log(Ea) vs log T (Figure 3(c) is a straight line with slope {−(γ−1)} equal to 0.73, which corresponds to hopping exponent γ ∼ 1/4 and n ∼ 3 which is again a 3D variable range hopping (VRH). For PANI-DBSA/2% CB composite, the plot of Log(Ea) vs log T (Figure 3(d)) is a straight line with slope {−(γ−1)} equal to 0.74, which corresponds to hopping exponent γ ∼ 1/4 and n ∼ 3 which is again a 3D variable range hopping (VRH).
The activation energy obtained from Arrhenius plot increases with the increase of temperature. Characteristic temperature T was estimated from the slope of graph of log
Mott’s parameters at 300 K.

The characteristic temperature To, hopping length R, and activation energy W were found to decrease for PANI-DBSA and its CB composites which suggests that the band gap between conduction and valence band has reduced thus creating the rise of hopping of charge carriers which give rise to higher dc conductivity with temperature.
The increase in temperature helps to improve segmental mobility by increasing its free volume. Increase in conductivity with rise of temperature is an indication that more ions gain kinetic energy via thermally activated hopping of charge carriers between trapped sites. And CB may act as conducting bridge connecting the conducting domains of PANI. Mott’s parameters are given in the table below Table 1.
Fourier transform analysis
In order to understand the nature of bonding, detect the functional groups, and to confirm the presence of carbon black in the prepared composites, FTIR spectra of the PANI-DBSA and its CB composites are analyzed and are shown in Figure 4. When the FTIR spectra of the PANI-DBSA composite is analyzed, peaks appearing at 580 cm−1, 1295 cm−1, and 1496 cm−1 are associated to C=C stretching vibrations which signaled the oxidized state of PANI, C–H in-plane bending vibrations of secondary aromatic amine and benzoid deformation bands, respectively.23,39 Absorption band present in all three spectrums appearing at 793 cm−1 is due to the sulfonic acid group.
40
The bands appearing at 1147 cm−1 and 1530 cm−1 are associated to the C–C deformation bands and characteristics absorption bands of PANI due to (N=Q=N) stretching vibrations, respectively.23,41 FTIR spectra of PANI-DBSA, PANI-DBSA/1% CB, and PANI-DBSA/2% CB. PANI, polyaniline; DBSA, dodecylbenzenesulfonic acid; CB, carbon black.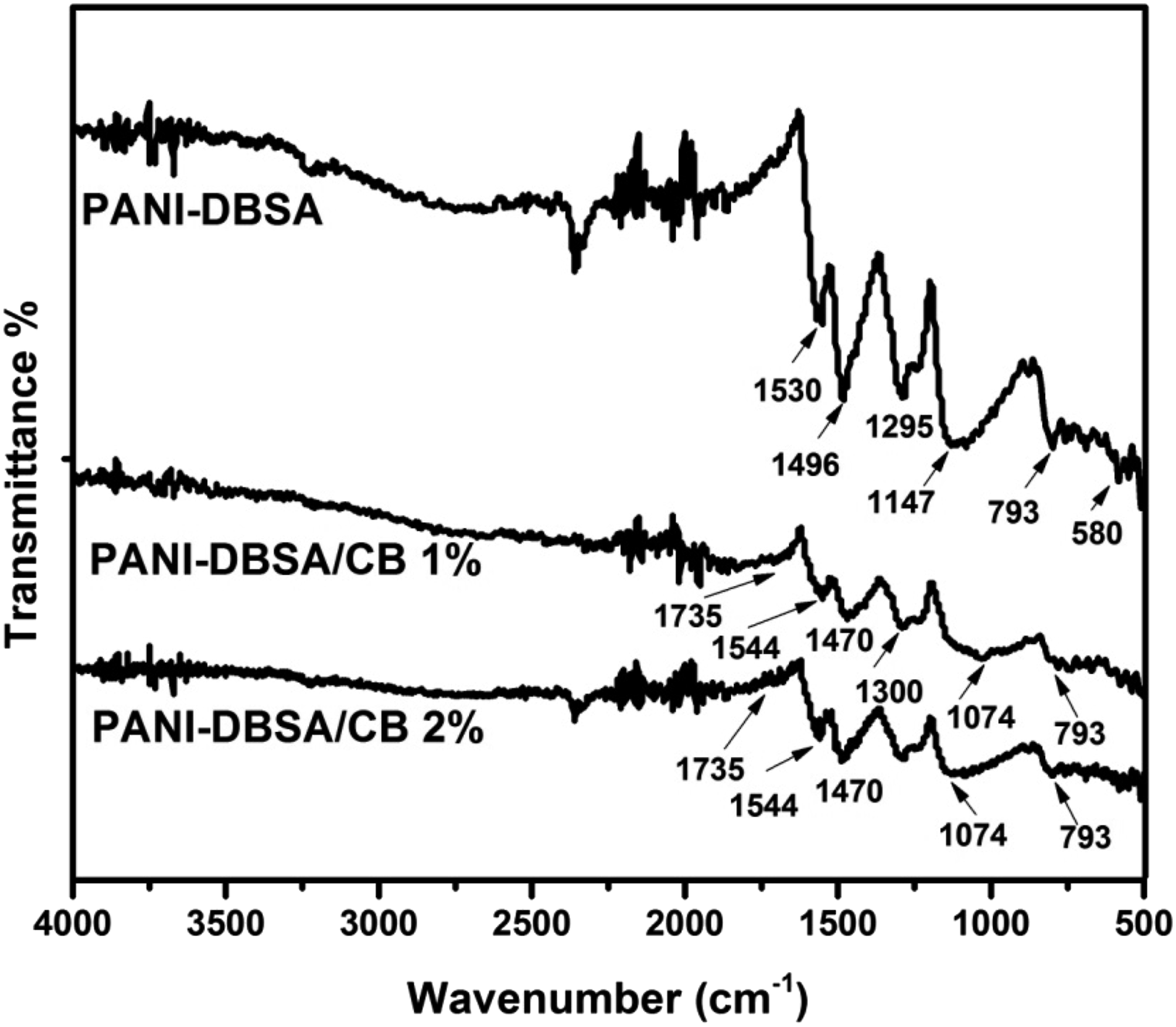
In case of PANI-DBSA composites with CB 1074 cm−1 due to a vibration mode of B–NH+–B or Q=NH+–B. 41 The peak at 1300 cm−1 is due to the stretching vibration. The peaks appearing in the composites at 1544 cm−1 and 1735 cm−1 are due to the C=C stretching vibrations and C–O stretching vibration, respectively. 13 FTIR analysis confirms the successful composites formation of PANI-DBSA with CB. The characteristic peaks of PANI and CB have been present in the composites.
Morphological analysis
SEM of the PANI, PANI-DBSA, PANI-DBSA/1% CB, PANI-DBSA/2% CB and CB is shown in the figure. It is clear that a continuous layer of the conducting polymer is formed on the surface of PANI after adding DBSA (Figure 5(b)). A well-defined conducting network is present in this pattern which is showing further increment in particle size. From XRD and SEM studies, it can be concluded that PANI-DBSA/CB composites are more ordered as compared to pristine PANI. Scanning electron microscopy SEM of (a) PANI (b) PANI-DBSA (c) PANI-DBSA/1% CB (d) PANI-DBSA/2% CB, and (e) CB. SEM, scanning electron microscopy; PANI, polyaniline; DBSA, dodecylbenzenesulfonic acid; CB, carbon black.
These PANI-DBSA/CB synthesized by a very simple process may be helpful for EMI shielding, filler, sensors, and supercapacitors.
Conclusion
Composites of PANI with DBSA and CB were successfully synthesized by in-situ polymerization. Gradual enhancement in crystallinity and grain size was studied by XRD spectra and confirmed by SEM analysis. Study of the charge transport mechanism of PANI-DBSA with CB particles, over the temperature range from 300 to 393 K, was carried out. Electrical conductivity was found to be temperature dependent, indicating that there is three-dimensional (3D-VRH) variable range hopping that is dominating for charge transport mechanism.
This result shows that CB contributes with the electrical conductivity of the prepared composites, creating conductive pathways into the conducting polymer morphologies. Due to low cost and easy preparation routes, these PANI-DBSA composites with CB may prove promising candidates for various applications in electronic devices, such as flexible electrodes, sensors, and charge storage devices.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.








