Abstract
To prevent microbial contamination in foods and medical tools, we prepared a novel antibacterial material nano-silver loaded poly(styrene-co-acrylic) (nAg-PSA) composites by in situ reductions. The nAg-PSA was characterized by scanning electron microscope, transmission electron microscopy, energy dispersive X-ray spectroscopy and thermogravimetric analysis. The antibacterial properties of nAg-PSA against Escherichia coli and Staphylococcus aureus were systematically evaluated, and the underlying mechanism was investigated. Results showed that the silver nanoparticles were successfully loaded onto PSA microspheres, highly dispersed on the surface of nAg-PSA nanoparticles. The nAg-PSA nanoparticles all showed significant antibacterial effects, among which nAg-PSA4 had maximum diameters of inhibition zones against the Gram-negative E. coli (1.18 mm) and the Gram-positive S. aureus (1.29 mm), respectively, which closely related to the size and density of silver nanoparticles covered on the surface.
Keywords
Introduction
Recent years, with a great concern on food safety and the fast development of nanotechnology and materials science, a large number of research studies on antibacterial materials have emerged.1,2 Nanoparticle materials are of interest due to their wide surface area and reactive properties. Metallic nanoparticles such as silver, with high antibacterial activity and minimal interference with human cells, are excellent antibacterial material.3,4 The specific “nano-effect” of silver nanoparticles had easy penetration in microbial cells and destructive contact on the surface of cell membranes. It is now widely used for antibacterial textile products, polymer film for food packaging, filters for water, and so on.5–8
However, the aggregation of silver nanoparticles easily causes a loss of surface area and reduction in antibacterial activity. 9 To overcome this problem, Wei, Luo, and Liu used the electrostatic force-driven layer-by-layer self-assembly and synthesized an antibacterial coating with nattokinase-nano-silver and polyethyleneimine. 10 Cong et al. prepared polystyrene/Ag nanocomposite particles through a polydopamine-assisted electrolysis Ag metallization procedure and successfully enhanced the antibacterial activities. 11 Food packaging could be made with antibacterial effect by directly incorporating an antibacterial material in the surface coating and could greatly improve the quality, safety, and shelf life of food.12,13 The existing synthetic methods such as self-assembly and polydopamine-assisted electrolysis have problems such as multiple preparation steps and complicated operations, and the repeatability is difficult to control. Thus, a simple, repeatable, and controllable in situ reduction deposition method is more favorable. 14 In addition, compared with pure silver nanoparticles and pure copolymer, nano-silver loaded poly(styrene-co-acrylic) composites can increase the affinity between silver and the nitrile group on the surface of the microspheres, thereby depositing on the porous polymicrospheres with surface functions. 15 Composite microspheres can effectively prevent the accumulation of metal nanoparticles and improve the catalytic stability and have a wider range of applications. 16 It is believed that the carboxyl group in acrylic acid leads to an increase in the mobility of silver ions in the copolymer, resulting in enhanced antibacterial surface activity. Since Ag+ ions have a high affinity with thiol groups in cysteine residues, they can interact with respiratory enzymes and lead to the disruption of mitochondrial respiratory chain. 17 Mitochondrial dysfunction can interrupt ATP synthesis, leading to DNA damage and cell death. 18
In this work, a novel nano-silver composite material with great potential of antibacterial properties was designed. We used four different sizes of PSA microspheres with plenty of dissociated carboxyl groups as the core, used positively charged silver ions as shell, and combined the core with the shell by in situ reduction method. 19 The antibacterial properties of the new composites were systematically evaluated. It was expected that this nano-silver loaded poly(styrene-co-acrylic) (nAg-PSA) composites would be a satisfactory antibacterial material. This new antibacterial material would prevent microbial contamination in food industry, as well as providing an excellent precursor material for food antibacterial packaging.
Experimental
Materials
Styrene was obtained from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan), acrylic acid was purchased from Shanghai Chemical Reagent First Factory (Shanghai, China), potassium persulfate and silver nitrate were purchased from Tianjin Komiou Chemical Reagent Development Center (Tianjin, China); sodium hydroxide and sodium borohydride (NaBH4) were obtained from Tianjin Guangfu Fine Chemical Research Institute (Tianjin, China). Lauryl sulfate tryptose broth, violet red bile agar, and yeast extract were purchased from Beijing Aoboxing Biotechnology Corporation (Beijing, China). Escherichia coli (E. coli, ATCC 25922) and Staphylococcus aureus (S. aureus, ATCC 25923) bacterial strains were purchased from China General Microbiological Collection Center (Beijing, China). All reagents were of analytical grade without further purification.
Preparation of PSA nanospheres
Recipes of poly(styrene-co-acrylic acid) composite nanospheres.

St: styrene; AA: acrylic acid; KPS: potassium persulfate; PSA: polystyrene-co-acrylic acid.
Preparation of nAg-PSA nanospheres
We used in situ reductions to attach the silver nanoparticles to the PSA microspheres. 21 First, we mixed 200 mL of PSA (0.3 mM) microsphere dispersion with 10 mL of silver nitrate solution (10 mM) and stirred for 5 h. Then, 10 mL of NaBH4 (10 mM) was added and stirred at 0°C for 2 h. After the reaction, the dispersed liquid was centrifuged at 15,000 g for 20 min. The precipitate was dispersed into 30 mL of distilled water and centrifuged again. After repeating four times, nAg-PSA nanocomposite microspheres were obtained.
Characterization of PSA nanospheres and nAg-PSA nanospheres
The morphology of the PSA and nAg-PSA microspheres was observed by tansmission electron microscope (TEM) (JEOL, JEM-2010). The content of silver particles on the surface of the microspheres was determined by thermogravimetric analysis (TG, Perkin Elmer Pyris 1, Perkin Elmer Co., USA). The crystal structure of silver particles on nAg-PSA was characterized by energy dispersive X-ray spectroscopy (EDX, EDX-7000, SHIMADZU, JPN).
Antibacterial properties
The antibacterial properties of nAg-PSA1-4 solutions against E. coli and S. aureus were evaluated by paper disk diffusion method. 17 The glassware and tools were first sterilized in an autoclave at 121°C for 20 min. The bacterial suspension containing 1×107 CFU/mL was wiped onto a Luria-Bertani solid medium in a sterile workbench. Paper sheets with a diameter of 6 mm were immersed in 0.9% NaCl solution and placed in the center of the medium as control; paper sheets soaked with nAg-PSA solutions (1–4) were placed around. Paper sheets were then incubated at 37°C for 24 h. Three replicate measurements were performed and the average of the repeated measurements was taken.
Second, the bacterial growth curve was obtained according to Wu’s research with some modifications. 22 We mixed the nAg-PSA suspension of 1.5 mL with a nutrient broth of 1.5 mL and added bacterial suspension of 50 μL, vortexed for 5 min, and cultured at 37°C for 24 h. The bacterial culture was extracted with every 3 h interval to monitor the growth of the bacteria. The absorbance value of the extracted bacterial culture at 600 nm was analyzed by an ultraviolet-visible spectrophotometer and the growth curves were plotted accordingly.
The morphology of E. coli and S. aureus interfered with and without nAg-PSA4 was observed by a scanning electron microscope. E. coli and S. aureus were inoculated into LB medium at 37°C for 6 h. Then, the bacterial suspension of 100 μL was added in fresh broth medium; after 6 h, 500 μL of the nAg-PSA4 solution was added. Then, the bacterial solution was washed three times with a 0.9% NaCl solution, and the precipitate was collected after centrifugation at 3000 g for 6 min. The bacteria were fixed with 2.5% (v/v) glutaraldehyde for 12 h and were quickly washed twice with sterile water. The cells were then subsequently dehydrated in ethanol solutions with concentrations of 25%, 50%, 70%, 80%, 90%, and 100%, respectively. The bacteria were dried and observed.
Results and discussion
Preparation and characterization of nAg-PSA microspheres
In EDX mapping (Figure 1(a)), the C and Ag elements were evenly distributed, confirming that the particles on the surface of the PSA microspheres were silver nanoparticles, in good accordance with the TEM image of nAg-PSA1-4 nanospheres (Figure 2). The content of silver nanoparticles was determined by thermogravimetric analysis (Figure 1(b)). As shown in Figure 1(b), the PSA microspheres almost lost the whole weight at 550°C, while the nAg-PSA microspheres remained with certain weight at the same temperature. As a result, the extra weight remained with the nAg-PSA microspheres after thermal decomposition was considered to be the weight of the silver nanoparticles composited on the surface of the PSA microspheres.
23
The possible mechanism of formation of silver nanoparticles on PSA sphere was discussed in our previous work.16,19 EDX mapping of nAg-PSA1 composite nanospheres (a), TG curves of PSA1 nanospheres and nAg-PSA1-4 and the density of silver nanoparticles calculated accordingly (b). PSA: polystyrene-co-acrylic acid. Transmission electron microscope of nAg-PSA1-4 nanocomposites. PSA: polystyrene-co-acrylic acid.
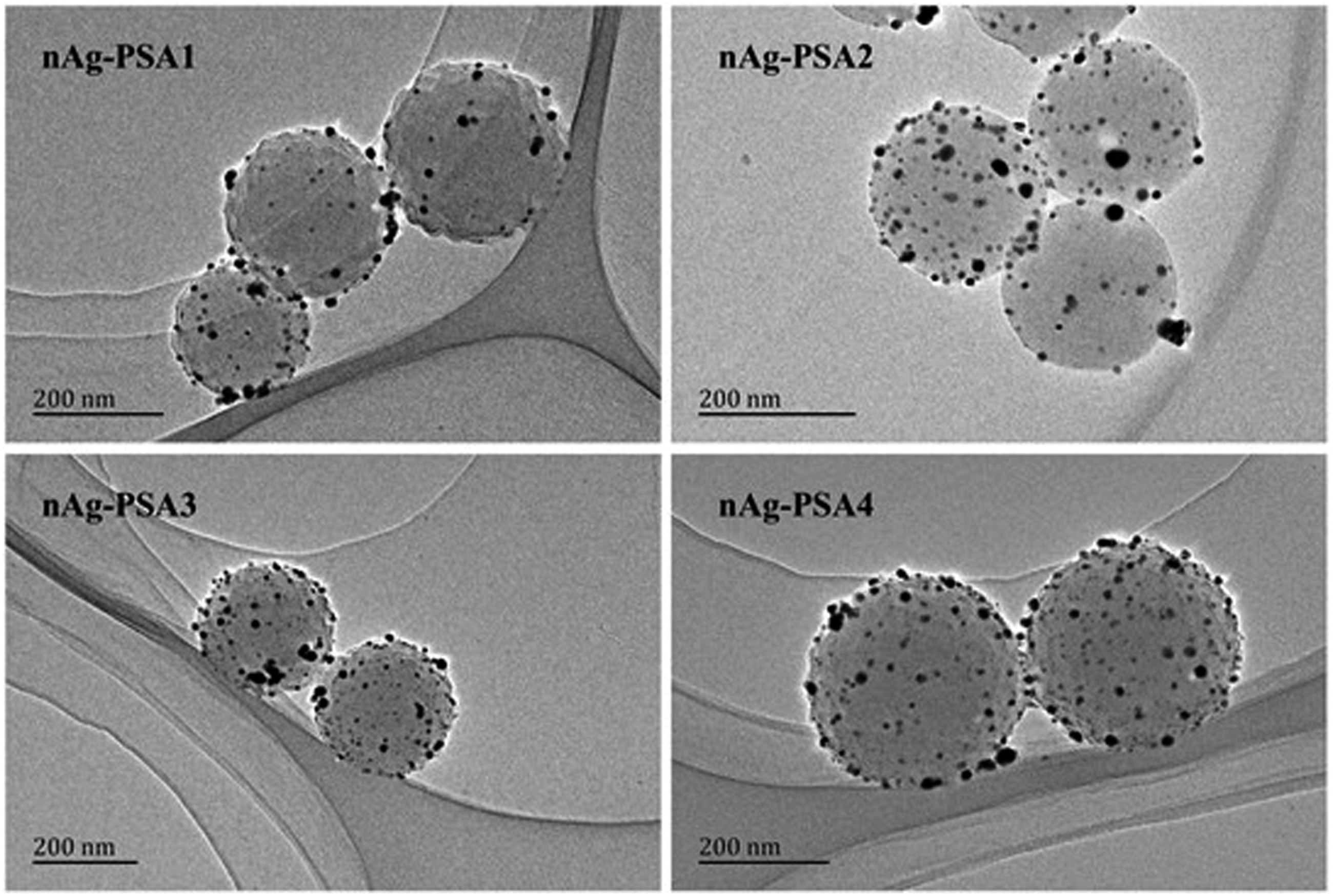
Antibacterial properties of nAg-PSA
Effect of nAg-PSA on bacterial growth
Figure 3 shows the inhibition zone of nAg-PSA1-4 against E. coli (a) and S. aureus (b), respectively, and summarizes the diameter of the inhibition zone (c). In Figure 3, it can be observed that the growth of bacterial colonies in the medium was significantly inhibited by the nAg-PSA nanocomposites. It might be because that the release of silver particles penetrated the cell wall, damaged the bacterial DNA, and affected protein synthesis of the bacteria.
24
The diameters of the inhibition zone for nAg-PSA1-4 against S. aureus were bigger than that for nAg-PSA1-4 against E. coli, implying that the antibacterial activity of nAg-PSA against Gram-positive bacteria was stronger than that against Gram-negative bacteria. The size of the inhibition zone of nAg-PSA was as follows: nAg-PSA4> nAg-PSA3> nAg-PSA1> nAg-PSA2, consistent with the mass density of silver nanoparticles (Figure 1(b)). Image of the inhibition zone of nAg-PSA1-4 against E. coli (a) and S. aureus (b). The medial position corresponds to the control of 0.9% normal saline. PSA: polystyrene-co-acrylic acid.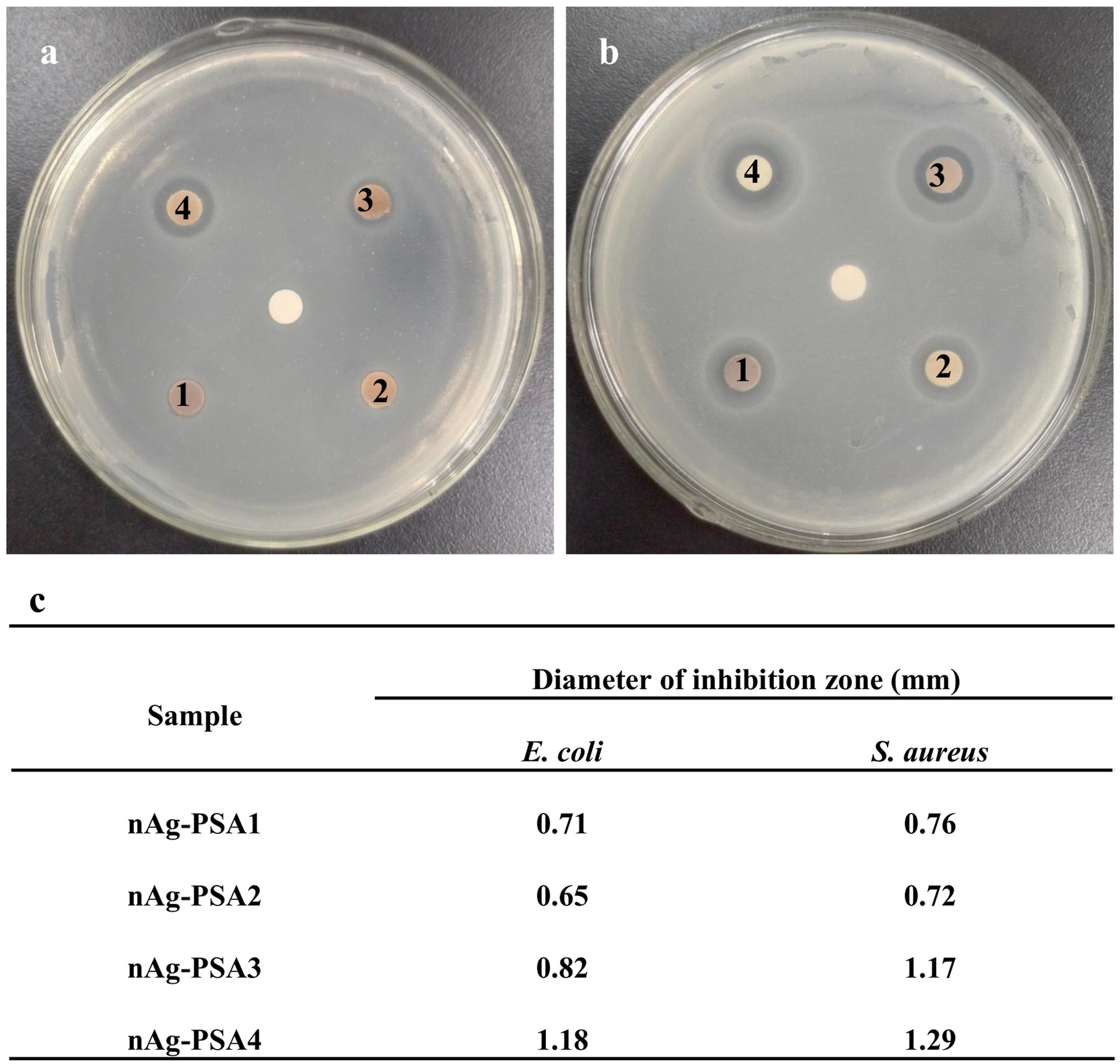
Figure 4 is the growth curves of E. coli and S. aureus treated with and without nAg-PSA1-4. It shows that the bacterial growth was significantly inhibited under the influence of nAg-PSA, especially after 6 h of culture. The inhibiting effect of nAg-PSA was as follows: nAg-PSA4> nAg-PSA3> nAg-PSA2> nAg-PSA1, consistent with the number density of the silver nanoparticles on nAg-PSA (Figure 1(b)). It indicated that except for the mass density of silver nanoparticles, the size of the silver nanoparticles on nAg-PSA also played a crucial role in the antimicrobial effects of nAg-PSA. The silver ions on the surface of nAg-PSA might adhere to the bacterial cell membrane and cause the separation of the plasmolysis, thereby inhibited the synthesis of bacterial cell membranes.
25
Growth curve of E. coli (a) and S. aureus (b) with nAg-PSA nanocomposites. PSA: polystyrene-co-acrylic acid.
Effect of nAg-PSA on bacterial morphology
Figure 5(a) shows the smooth surface of the original E. coli, while Figure 5(b) exhibits short, wrinkled surface of E. coli, indicating that the nAg-PSA caused significant damage to the bacterial cells of E. coli. Figure 5(c) shows S. aureus with spherical and smooth surface, while Figure 5(d) reveals the depressed surface of S. aureus treated with nAg-PSA, suggesting that nAg-PSA broke the cell of S. aureus as well. It might be because that the appropriately sized silver nanoparticles on the surface of nAg-PSA were easily bound to the bacterial cell membrane and cause pores. This made the intracellular material flow out of the cell, eventually leading to cell death.
26
SEM of E. coli (a), E. coli treated with nAg-PSA (b), S. aureus (c), S. aureus treated with nAg-PSA (d). SEM: scanning electron microscope.
Therefore, nAg-PSA is a nano-scale material with excellent bacteriostatic properties due to the presence of silver nanoparticles on the surface. Different preparation conditions of nAg-PSA1-4 resulted in discrepant size and density of silver nanoparticles, playing an important role in the antibacterial ability of nAg-PSA.
Conclusions
In this study, a new antibacterial material, the nAg-PSA coated with silver nanoparticles, was successfully synthesized. The antibacterial properties of nAg-PSA against E. coli and S. aureus were confirmed by the paper disk diffusion method, bacterial growth curve mapping, and bacterial morphology observation. It demonstrated that the prepared nAg-PSA had excellent antibacterial properties against both Gram-negative bacteria and Gram-positive bacteria. The antibacterial property of nAg-PSA is closely related to the size and quantity of silver nanoparticles covered on the surface. The new nAg-PSA nanospheres with high antibacterial activity would have wide application prospect as an antibacterial material to prevent food contamination and spoilage.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Scientific Research Project of Tianjin Municipal Education Commission (2018KJ091).
