Abstract
In this study, polypyrrole (PPy) and polypyrrole/molybdenum oxide composite (PPy/MoO3) were synthesized by the chemical oxidative method in an aqueous medium, using anhydrous ferric chloride (FeCl3) as an oxidant. The successful preparation of materials was confirmed by Fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), scanning electron microscopy (SEM) and transmittance electron microscopy (TEM). PPy and PPy/MoO3 were converted into pellets which were used as the sensor. A four-in-line probe device was used for studying DC electrical conductivity–based ammonia vapour–sensing properties at three different ammonia concentrations, that is, 1 M, 0.5 M and 0.1 M. The PPy/MoO3 sensor showed much-improved sensing performance than the PPy sensor in terms of % sensing response and reversibility. PPy/MoO3 sensor showed excellent selectivity for ammonia vapours against various VOCs. The % sensing response of PPy/MoO3 sensor towards ammonia was found to be 2.19, 2.50, 3.16, 3.87, 4.1, 5.15, 6.19, 6.55 and 7.77 times greater than ethanol, methanol, acetone, acetaldehyde, formaldehyde, toluene, benzene, chloroform and n-hexane, respectively. In the end, a sensing mechanism was also proposed, which is based on rapid adsorption–desorption of ammonia molecules on the PPy/MoO3 sensor’s surface.
Introduction
Over the years, the gas sensors based on conducting polymers (CPs) displayed high sensitivity and shorter response time when operated at room temperature.1–10 These sensing materials can be synthesized easily when employing chemical or electrochemical processes.1–3 Nowadays, widespread interest in polypyrrole (PPy) has been aroused as it can be easily deposited from aqueous and non-aqueous media, adheres to various types of substrates and exhibits high conductivity as well as stability in air and aqueous media.11–13 Several limitations restrict the usage of PPy in some applications, such as its poor mechanical properties and processibility as well as fragile nature. 14 Researchers have adopted several approaches in order to enhance these properties of PPy; among them, the most preferred one nowadays is the preparation of its conducting composites with inorganic components.11–13 This process results in the formation of stable composites, which are mechanically strong as well as have good vapour/gas sensing properties. Therefore, PPy has been widely used as an effective material for the detection of toxic, hazardous and flammable gases.11,12,15–18 Among the large assay of toxic and environmental pollutant gases/vapours, ammonia is very common, which has been released from industries and uncontrolled human activity. Continuous exposure in an ammonia environment can cause eye, throat, lung, skin and respiratory problems. At a very high concentration of ammonia, human cells may also be damaged. Therefore, prompt detection and monitoring of ammonia at ambient conditions is compulsory for protecting our environment and improving human health.16–18
Among the several intriguing transition metal oxides available nowadays, molybdenum oxide and its derivatives have attracted considerable interest as they possess unique optical as well as structural properties and are highly thermally and chemically stable.19,20 The oxidation states of molybdenum range from +2 to +6, and it exists primarily in two forms such as molybdenum (IV) and molybdenum (VI) oxide. 21 As molybdenum (IV) oxide shows electronic conductivity similar to metals, it has found application in several energy fields. MoO3 nanostructures have been utilized widely in fabricating sensors as their structure is anisotropic and their surface-to-volume ratio is high. It is highly electronically mobile as compared to its other bulk material counterparts.22,23
Over the years, conducting polymer/molybdenum oxide nanocomposites have been utilized for sensing applications. Chaluvaraju et al. carried out in-situ polymerizations of pyrrole (PPy) with molybdenum trioxide (MoO3) in the presence of the oxidizing agent ammonium persulphate to synthesize PPy/molybdenum trioxide composites (PPy/MoO3). 24 Hybrid thin films of PPy/MoO3 were synthesized and investigated for their volatile organic compounds and gas-sensing properties by Matsubara et al. 25 Sha et al. synthesized a metal-organic framework/PPy nanocomposite based on MoO3 for electrochemically enhancing the detection of the neurotransmitter dopamine. 26 Matsubara et al. synthesized layered MoO3/PPy nano-hybrids by employing the concomitant ion-exchange reaction for the detection of volatile organic compounds. 27 Zeng et al. developed a novel sensing platform based on the composite of MoO3-PPy nanowires/MWCNT and applied it for detecting luteolin electrochemically, which is used widely as an antitumoural agent 28 . Roy et al. employed the in-situ polymerization method to synthesize polyaniline/molybdenum trioxide composites and studied their gas-sensing parameters towards LPG. 29 Itoh et al. prepared layered organic–inorganic nano-hybrid thin films of molybdenum trioxide with derivatives of polyaniline for studying its sensing properties towards aldehyde. 30 Hybrid material of polyaniline with molybdenum trioxide (MoO3) aerogel was synthesized by Wei et al. and investigation for ethanol-sensing properties. 31
To the best of our knowledge, the PPy/MoO3 composite is not utilized for ammonia vapour sensing yet. So, we have synthesized PPy and PPy/MoO3 composites by the in-situ oxidative polymerization method and characterized by FT-IR, XRD and SEM techniques. For investigating ammonia vapour–sensing properties, both the materials have been converted into the pellet-shaped sensor. The sensing performance was determined in terms of % sensing response, reversibility and selectivity. Thus, PPy/MoO3 composite–based novel sensor pellet was found to be highly efficient for the rapid detection of ammonia vapours at room temperature.
Experimental
Materials
Pyrrole (99%) (Sigma-Aldrich), molybdenum oxide nanoparticles (MoO3) 99.5% pure and average particle size (APS) 90 nm (MKnano, Canada), anhydrous ferric chloride (FeCl3), ammonia solution and methanol (Fischer Scientific, India) were used as received. Double-distilled water was used in synthesis and other experiments.
Synthesis of polypyrrole (PPy) and polypyrrole/molybdenum oxide composite (PPy/MoO3)
In order to synthesize the PPy and PPy/MoO3 composites, the in-situ polymerization method in aqueous media was employed where FeCl3 was used as an oxidizing agent.11,12 The first step involved the agitation of 0.03 mol of pyrrole in 150 mL of doubly distilled water. This was followed by the preparation of an aqueous solution of FeCl3 (0.09 mol) in 150 mL of distilled water. Accordingly, there was a dropwise addition of FeCl3 into the aqueous pyrrole solution accompanied by continuous stirring at room temperature. The stirring was performed for 12 h, and it resulted in a black slurry formation. Then, the obtained slurry was simultaneously filtered and washed with doubly distilled water and methanol. Thus, the PPy obtained was kept for drying in an air oven at 50°C for 24 h and was converted to very fine powder form in order to perform further investigations.
In order to synthesize PPy/MoO3, a similar procedure was followed in which firstly, there was agitation of 0.03 mol of pyrrole in 150 mL of doubly distilled water. Then, in the aqueous solution of pyrrole, 400 mg of MoO3 was transferred. This was followed by the ultrasonication of the prepared aqueous suspension for 2 h so that the pyrrole molecules get adsorbed on the surface of MoO3. This was followed by the dropwise addition of FeCl3 solution to the prepared aqueous suspension, accompanied by continuous stirring for 12 h, which resulted in the formation of a black slurry of PPy/MoO3. This was followed by the filtration and continuous washing of the slurry with doubly distilled water and subsequently by methanol. Then, the slurry was dried in an air oven at 50°C for 24 h and was converted to very fine powder form for carrying out the further experimental work.
Characterization and instrumentation
The Fourier transform infrared (FT-IR) spectra of PPy and PPy/MoO3 were recorded in the range of 400–4000 cm−1 using a Perkin-Elmer-1725 instrument on KBr pellets. The XRD spectra of PPy and PPy/MoO3 were recorded by an X-ray beam diffractometer (Shimadzu XRD, model 6100) [Cu Kα radiation (1.540 Å)], operating at a voltage of 30 kv and 2θ value between 5 and 80°. The surface morphological studies of PPy and PPy/MoO3 were performed using a JSM-6510-LV (JEOL, Japan) scanning electron microscope. The TEM image of PPy/MoO3 was obtained by a JEM 2100 JEOL (Japan) transmittance electron microscope.
Ammonia vapour sensing experiments were done by a four-in-line probe instrument attached with a PID-controlled oven manufactured by Scientific Equipment, Roorkee, India. The equation used for the calculation of electrical conductivity is given below:
The sensor was fabricated by converting 200 mg of PPy and PPy/MoO3 into pellets by a hydraulic pressure machine. The applied pressure was about 70 kN for 100 s for making pellets of each sample.
Results and discussion
Fourier transform infrared spectroscopic studies
The FT-IR spectra of PPy and PPy/MoO3 are shown in Figure 1. In the spectrum of PPy, the peak at 3428.13 cm−1 is attributed to the N-H stretching vibrations. The peak at 1551.74 cm−1 is due to C=C stretching vibrations. The peak at 1470.20 cm−1 attributes to the C-C stretching, and the peaks that appeared 1306.91 cm−1 and 1191.13 cm−1 may be due to the C = N bending and C-N vibration, respectively. The =C-H bending vibration appeared at 1045.58 cm−1, whereas the peak at 788.72 cm−1 is due to the stretching vibrations of C-H bonds. The peak at 923.17 cm−1 is ascribed to the =C-N+-C stretching vibration. This peak at 923.17 cm−1 is the confirmation that pyrrole was effectively oxidized to PPy and doped by FeCl3 into positively charged species that act as charge carriers (i.e. polarons/bipolarons).11,12,16–18 The FT-IR spectra of PPy and PPy/MoO3.
In the spectrum of PPy/MoO3, all the characteristic peaks of PPy were observed, which were weakened and shifted at smaller wavenumbers.11,12,16,17 The characteristic peaks observed at 3428.13 cm−1, 1551.74 cm−1, 1470.20 cm−1, 1306.91 cm−1, 1191.13 cm−1, 1045.58 cm−1, 923.17 cm−1 and 788.72 cm−1 in the spectrum of PPy were detected at 3425.71 cm−1, 1539.73 cm−1, 1452.55 cm−1, 1301.47 cm−1, 1167.19 cm−1, 1039.21 cm−1, 900.79 cm−1 and 783.17 cm−1, respectively, in the spectrum of PPy/MoO3. The weakening and shifting of the peaks may be due to some synergistic/electronic interaction acting at molecular levels between PPy chains and MoO3.11,12,16,17
X-Ray diffraction studies
The XRD spectra of PPy and PPy/MoO3 are shown in Figure 2. In the case of PPy, the broad, amorphous peak appeared between 2θ = 18–30°; having a maximum at 2θ = 24° revealed that pyrrole was successfully polymerized to PPy.11,12,16–18 In the case of PPy/MoO3, the characteristic peaks of MoO3 were detected at 2θ = 12.84°, 23.39°, 25.81°, 27.39°, 33.94°, 35.64°, 39.04°, 46.42°, 49.22°, 52.85° and 58.85° which are related to the (020), (110), (040), (021), (111), (041), (060), (200), (061), (002) and (081) crystal planes of orthorhombic α-MoO3 (JCPDS No. 05–0508), respectively.19,20,32,33 The XRD spectra of PPy and PPy/MoO3.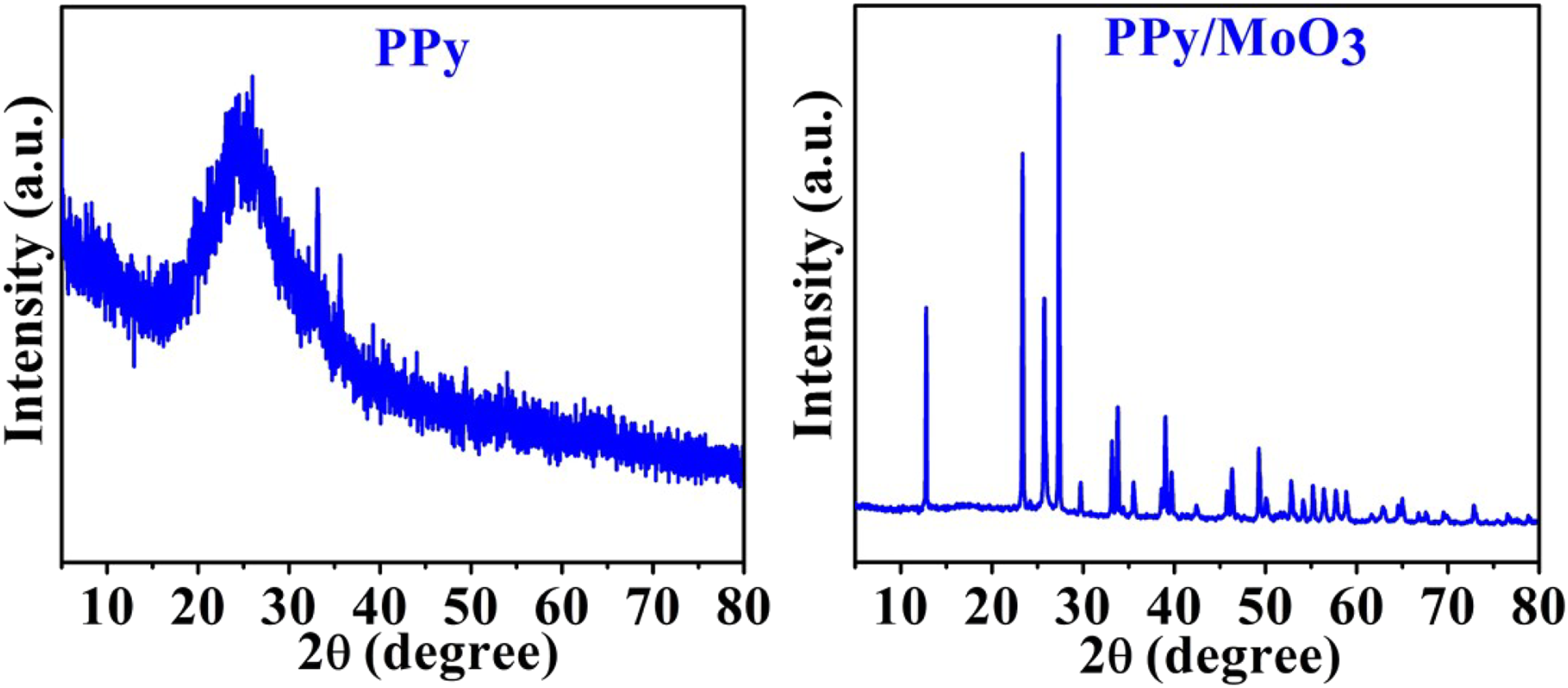
Morphological studies
The surface morphology of PPy, MoO3 and PPy/MoO3 was studied by scanning electron microscopy (SEM). The scanning electron micrographs of PPy, MoO3 and PPy/MoO3 are shown in Figure 3. The surface morphology of PPy observed by SEM images showed that there was an agglomeration of globular particles (Figure 3(a)).11,12,16–18 The SEM image of MoO3 (Figure 3(b)) showed that it majorly consists of rectangular rod-like particles along with some square-shaped particles of varying sizes. The SEM images of PPy/MoO3 (Figure 3(c) and (d)) confirmed that pyrrole was successfully polymerized on the surface of MoO3. PPy/MoO3 consists of an agglomerated globular particle of PPy on the surface of MoO3. However, some rectangular-shaped rod-like structures can be observed clearly in SEM images of PPy/MoO3, which confirms the presence of MoO3 in PPy/MoO3. The SEM images of (a) PPy, (b) MoO3 and (c & d) PPy/MoO3.
Thus, successful preparation of PPy and PPy/MoO3 can be confirmed by the results obtained from FT-IR, XRD and SEM studies.
The TEM image of PPy/MoO3 is shown in Figure 4, which confirmed the presence of MoO3 nanoparticles of similar morphology as explained by the SEM image (Figure 3(b)). The MoO3 nanoparticles were covered by PPy matrix which can also be observed by SEM images of PPy/MoO3 (Figure 3(c) and (d)). The TEM image of PPy/MoO3.
Ammonia vapour sensing
Over the years, highly efficient ammonia sensors have been fabricated by utilizing PPy and its composite materials. A completely selective and reversible ammonia sensor based on PPy/SnO2/MWCNT was synthesized by Husain et al. 16 The response of this sensor was evaluated at a concentration range of 10–1500 ppm and was calculated to be 70.4%, 66.1%, 62.2%, 55.4%, 50.8% and 39.7% at 1500 ppm, 1000 ppm, 500 ppm, 200 ppm, 100 ppm and 10 ppm concentration of ammonia, respectively. In another study, Husain et al. 17 synthesized a completely reversible and selective ammonia sensor based on a PPy/ZnO/SWCNT nanocomposite. The sensing response of the sensor was calculated within the concentration range of 50–2000 ppm and was found out to be 76.3%, 60.5%, 54.8%, 52.6%, 50.2%, 48.5%, 40.5% and 36.6%, at 2000 ppm, 1000 ppm, 500 ppm, 400 ppm, 300 ppm, 200 ppm, 100 ppm and 50 ppm concentration of ammonia, respectively.
Khan et al. 34 synthesized composite of PPy with V2O5 via a modified in-situ chemical oxidative polymerization method. This composite was employed as a sensor for detecting ammonia vapours at room temperature in the concentration range of 5–500 ppm. The sensing response was calculated to be 8%, 19.5%, 125% at 5 ppm, 10 ppm and 500 ppm, respectively.
Mahajan et al. 35 used the in-situ polymerization method at room temperature to synthesize PPy nanocomposites, in which graphene oxide multi-walled carbon nanotubes RGO–MWCNT and zinc oxide (ZnO) were employed as fillers. This material was found suitable for sensing vapours of ammonia at ambient temperatures, and the sensing response was calculated to be 325%, 50% and 36% at 2000 ppm, 100 ppm and 50 ppm, respectively. A nanocomposite of PPy and carboxylated multi-walled carbon nanotubes (MWCNT) was synthesized by Bachhav et al. 36 by employing the in-situ chemical oxidative polymerization method. The sensing response of the fabricated pelletized sensor was calculated at temperatures ranging from 28°C to 65°C within the concentration range 200–2000 ppm of ammonia. It showed a sensitivity of 3.07% at 200 ppm and 17.11% at 2000 ppm, respectively.
Albaris et al. 37 synthesized a hybrid nanocomposite PPy-GO-WO3 by polymerizing PPy in the presence of the GO-WO3 composite. This nanocomposite was used for detecting ammonia vapours in the concentration range of 5–15 ppm. The maximum sensing response was calculated to be 58% at 10 ppm. The fabricated sensor was found to be stable over a period of 50 days. Malook et al. 38 synthesized an ammonia sensor operating at ambient temperature based on the PPy/MnO2 composite by employing the chemical oxidative polymerization method. The composite was used to detect ammonia vapours in the concentration range of 1–30 ppm, and the maximum response was calculated to be 46.44% at 5 ppm. A PPy/MWCNTs nanocomposite was fabricated by Jang et al. 39 using the in-situ polymerization method. They used this composite for sensing ammonia vapours at temperatures ranging from room temperature to 100°C and calculated the sensitivity to be 65%. Tang et al. 40 fabricated an ammonia sensor based on the PPy/rGO composite by the electropolymerization method. This sensor was employed for detecting ammonia vapours in the concentration range of 1–4 ppm and showed maximum sensing response of 6.1% at 1 ppm. Sun et al. 41 synthesized a PPy-reduced graphene oxide (rGO) hybrid sensor by utilizing the in-situ polymerization method. The fabricated sensor was used for detecting ammonia vapours at room temperature in the concentration range of 1–10 ppm and showed maximum response of 50% at 10 ppm.
Comparison of the present study with the other ammonia sensors based on PPy composites.

In this study, ammonia vapour–sensing studies of PPy and PPy/MoO3 sensors were carried out at three different ammonia concentrations, namely, 1 M, 0.5 M and 0.1 M. The sensing performance of the sensor was evaluated on the basis of its % sensing response, reversibility and selectivity. The sensing experiments were performed at normal laboratory conditions. The temperature was 32°C, and relative humidity was about 48%. First of all, the sensor (in the form of a pellet) was connected with the four-probe properly. Then, it was kept in a compartment which comprised ammonia solution of known concentration for producing ammonia vapours (Figure 5). Then, the sensor was exposed to the ammonia vapours for a continuous 60 s. After that, the pellet was separated from the compartment and was again exposed to the air for another 60 s. Upon exposure to the vapours of ammonia, the electrical conductivity of the sensor began to decline with time. The electrical conductivity of the sensor started to increase with time immediately after keeping the pellet in the open air for 60 s. Both the sensors showed a dynamic response of conductivity change in the ammonia vapours and air (Figure 6). The instrumental set-up used in sensing experiments. The variation in conductivity of sensors in ammonia vapours and air with respect to time.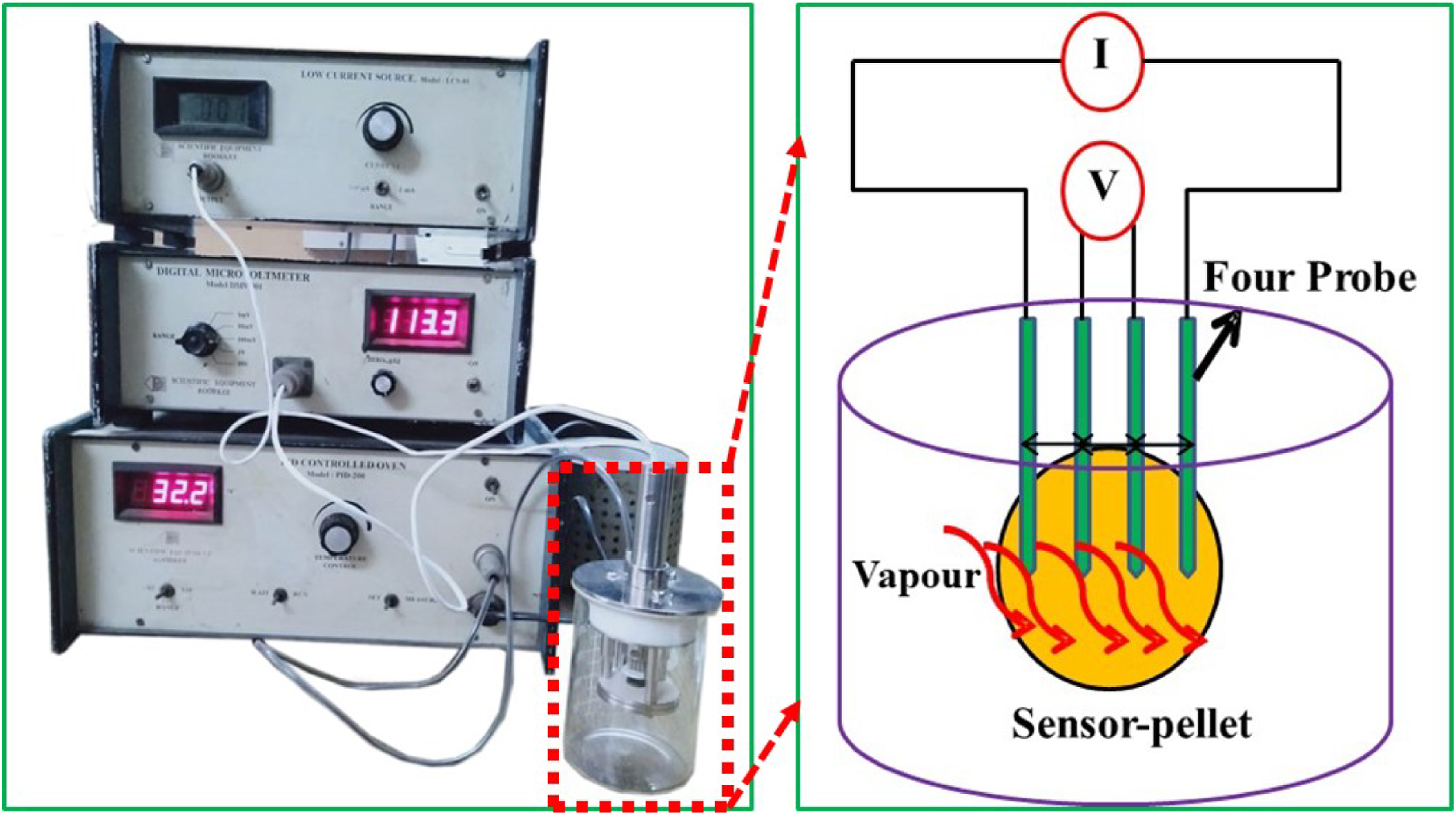

The % sensing response (S) of the sensors was evaluated by the following formula:
Sensing response
The % sensing response of the PPy sensor was found to be 53.7%, 29.5% and 19.0% at 1 M, 0.5 M and 0.1 M ammonia concentration, respectively. On the other hand, the % sensing response of the PPy/MoO3 sensor was found to be 79.3%, 38.3% and 28.9% at 1 M, 0.5 M and 0.1 M ammonia concentration, respectively. Both the sensors showed a greater response at higher concentration because of a greater number of ammonia molecules available which neutralize a large number of charge carriers. Therefore, a greater change in conductivity of the sensor was observed at a higher concentration than in lower concentrations. Also, at all three concentrations, the PPy/MoO3 sensor showed a much greater response than the PPy sensor that may be due to the greater surface area provided by MoO3 nanoparticles. The greater surface area consists of a greater number of active sites for the adsorption of ammonia molecules on its surface, which interact with charge carriers and neutralize them, causing a decrease in the conductivity of the sensor. Thus, the PPy/MoO3 sensor performed better than the PPy sensor as far as % sensing response is concerned.
Reversibility
The reversibility of a sensor may be correlated with its ability to perform consistently for several continuous cycles. In this facet, the reversibility of both the PPy and PPy/MoO3 sensors was evaluated for five consecutive cycles of 40 s each. The sensors were exposed to ammonia vapours for 20 s and then to air for another 20 s to complete one cycle. Similarly, the sensors were tested for five consecutive cycles comprising a total of 200 s (Figure 7). The conductivity of the PPy sensor kept decreasing after each cycle at all three ammonia concentrations. However, for the first and second cycles, the conductivity of the PPy sensor returned close to the initial value, showing good reversibility. After completion of the fifth cycle, there was a considerable loss of conductivity. Herein, the reversibility of the sensor was calculated as a function of conductivity retention after completion of the fifth cycle. The reversibility of the PPy sensor was found to be 70.5%, 73.1% and 66.3% at 1 M, 0.5 M and 0.1 M ammonia concentration. At the same time, the PPy/MoO3 sensor showed excellent reversibility as compared to the PPy sensor. After completion of the fifth cycle, the conductivity of the PPy/MoO3 sensor returned very close to the initial value. There was a very minor loss of conductivity after the fifth cycle. The reversibility of the PPy/MoO3 sensor was found to be 98.2%, 95.1% and 90.6% at 1 M, 0.5 M and 0.1 M ammonia concentration. Thus, the PPy/MoO3 sensor was also found to be more efficient than the PPy sensor in terms of reversibility. The reversible response of sensors in ammonia vapours and air with respect to time.
Selectivity
Due to the better performance of the PPy/MoO3 sensor than the PPy sensor in terms of % sensing response and reversibility, its selectivity towards ammonia was also tested in the exposure of various volatile organic compounds (VOCs) at 1 M concentration (Figure 8). The selected 1 M aqueous solution was composed of ethanol, methanol, acetone, acetaldehyde and formaldehyde. Toluene, benzene, chloroform and n-hexane were used in pure form as received from the company. The PPy/MoO3 sensor was exposed to all the selected VOCs for 60 s, and the % sensing response was calculated. The % sensing of the PPy/MoO3 sensor was found to be 36.2%, 31.6%, 25.1%, 20.5%, 19.3%, 15.4%, 12.8%, 12.1% and 10.2% for ethanol, methanol, acetone, acetaldehyde, formaldehyde, toluene, benzene, chloroform and n-hexane, respectively. PPy/MoO3 showed the highest response for ammonia (i.e. excellent selectivity) than other VOCs tested, as ammonia has the maximum tendency to donate electrons among the other selected VOCs. There is an interaction between the polarons of the PPy chain and the lone pairs of electrons present on the nitrogen atom of ammonia which neutralize the positive charge of polarons. As soon as the polarons of PPy are neutralized, a decrease in the DC electrical conductivity is observed. In the case of ethanol, methanol, acetaldehyde, acetone and formaldehyde, the PPy/MoO3 sensor showed less response as the electron donation capacity of oxygen is lower than that of nitrogen of ammonia, leading to a lesser number of polarons of PPy being neutralized. Thus, there is a lower change in electrical conductivity. For the other hydrocarbons such as toluene, chloroform, n-hexane and benzene, the variation in electrical conductivity was found to be the lowest among all the VOCs tested as there was no atom having lone pairs of electrons for donation. Therefore, the % sensing response of the PPy/MoO3 sensor towards ammonia (79.3%) was found to be 2.19, 2.50, 3.16, 3.87, 4.1, 5.15, 6.19, 6.55 and 7.77 times greater than ethanol, methanol, acetone, acetaldehyde, formaldehyde, toluene, benzene, chloroform and n-hexane, respectively, which shows its excellent selectivity towards ammonia vapours. The selectivity of the PPy/MoO3 sensor towards ammonia at 1M concentration against various VOCs tested.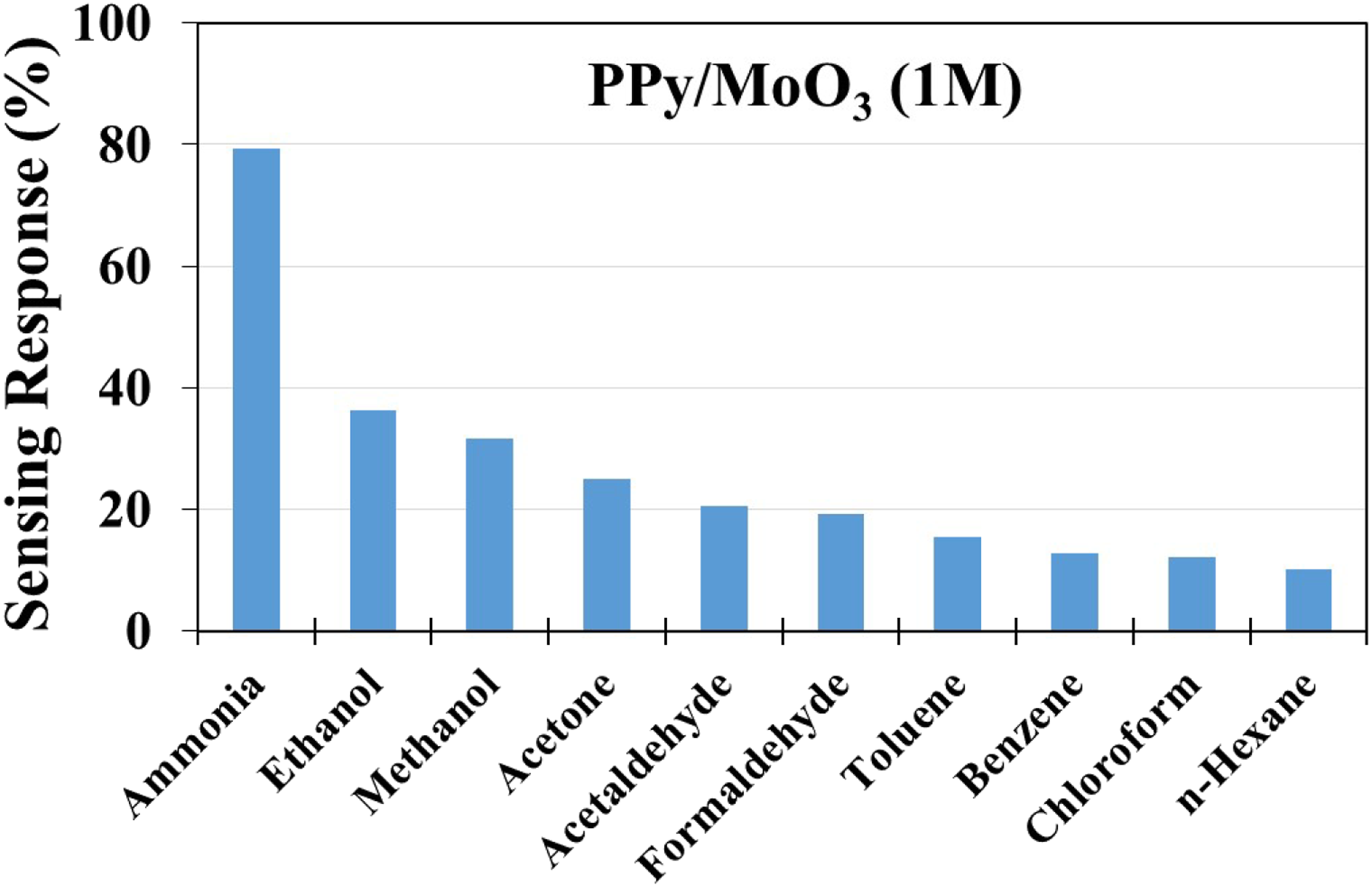
Sensing mechanism
The sensing response of the PPy/MoO3 sensor with respect to changes in electrical conductivity towards ammonia vapour is based on the fact that upon exposure of the sensor to the gas’/vapours’ environment, its electrical conductivity reduces with time and returns towards its initial value when exposed to air.11,12,16–18,43,44 First of all, ammonia molecules adsorb on the sensor’s surface and then interact with polarons/bipolarons and neutralize them. When the sensor is exposed to the air, the ammonia molecules are replaced by air. The significantly improved ammonia vapour–sensing performance of the PPy/MoO3 sensor might be associated with its higher electrical conductivity along with the greater surface area provided by MoO3, which facilitated a greater number of active sites for the adsorption of ammonia molecules. The superior % sensing response and reversibility of the PPy/MoO3 sensor over the PPy sensor have also revealed the prospect of more rapid adsorption and desorption processes of ammonia molecules on the PPy/MoO3 sensor pellet as compared to that of PPy. SEM and TEM results showed that MoO3 nanoparticles were thoroughly covered by PPy. Therefore, only the interaction between lone pairs of ammonia molecules and PPy chains has been considered for suggesting the mechanism involved in the ammonia-sensing properties of PPy/MoO3.
Thus, the interaction between ammonia molecules and polarons/bipolarons, which cause a decrease in conductivity, finishes and conductivity returns towards the initial value with time (Figure 9). Thus, the adsorption–desorption of ammonia molecules on the PPy/MoO3 sensor’s surface is the reason for showing the excellent reversible nature of the sensor with a high sensing response. The proposed reversible ammonia vapour–sensing mechanism of the PPy/MoO3 sensor showing electronic interaction between lone pairs of ammonia molecules and polarons.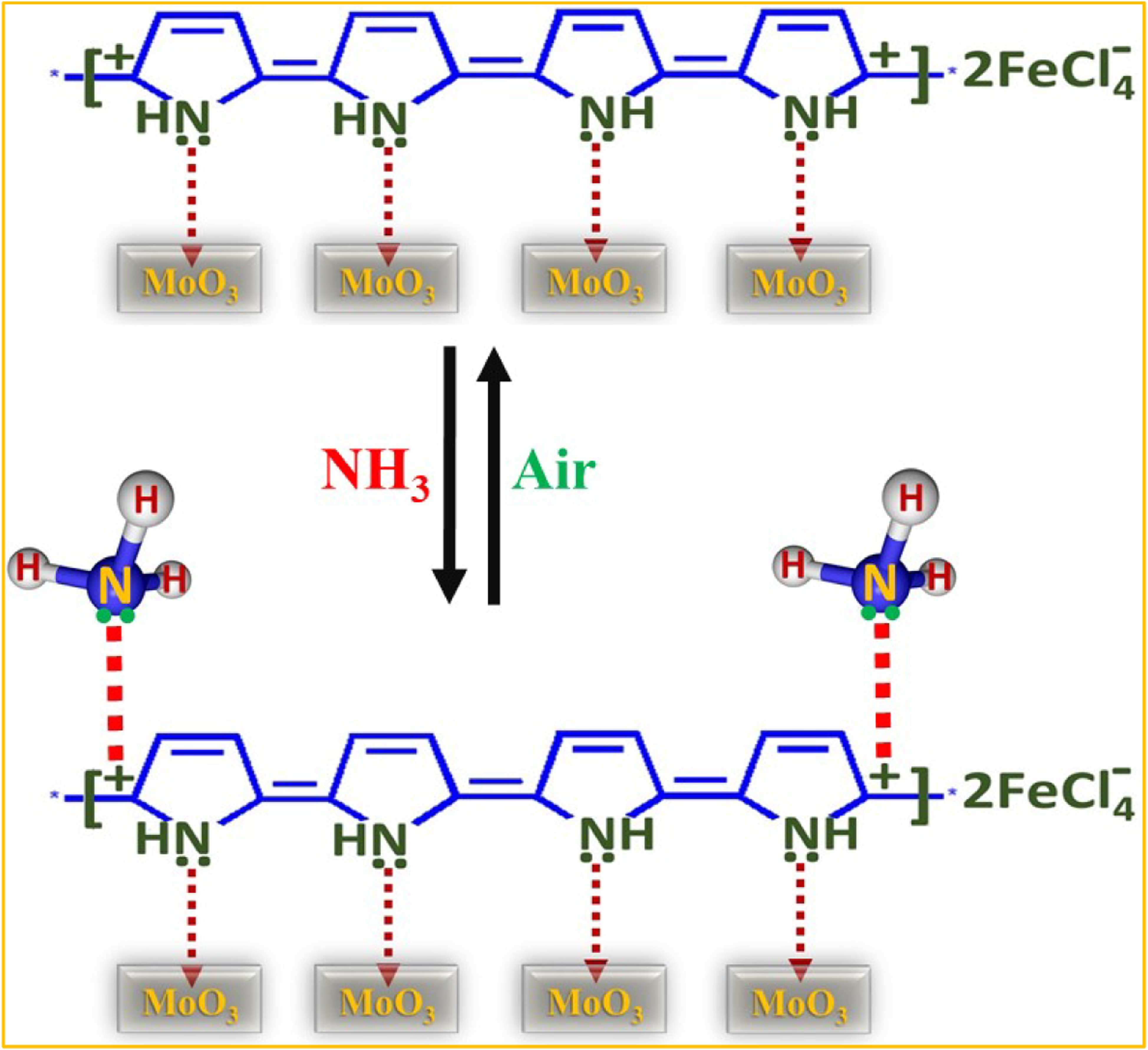
Conclusions
This study presents the successful preparation of PPy and polypyrrole/molybdenum oxide composite (PPy/MoO3) in an aqueous medium using anhydrous ferric chloride (FeCl3) as an oxidant which also acts as the dopant. The structure and surface morphology of materials were studied by various techniques like FT-IR, XRD, SEM and TEM. Both the PPy and PPy/MoO3 were converted into pellets for testing their ammonia vapour–sensing ability at room temperature towards 1 M, 0.5 M and 0.1 M ammonia concentrations. The PPy/MoO3 sensor was found to be more efficient than the PPy sensor in terms of % sensing response and reversibility. The % sensing response of the PPy sensor at 1 M, 0.5 M and 0.1 M ammonia concentrations were found to be 53.7%, 29.5% and 19.0%, respectively, while its reversibility was found to be 70.5%, 73.1% and 66.3%, respectively. On the other hand, the PPy/MoO3 sensor’s response was 79.3%, 38.3% and 28.9% at 1 M, 0.5 M and 0.1 M ammonia concentration, respectively, and was found to be 98.2%, 95.1% and 90.6% reversible, respectively. The % sensing response of the PPy/MoO3 sensor towards ammonia (79.3%) was found to be 2.19, 2.50, 3.16, 3.87, 4.1, 5.15, 6.19, 6.55 and 7.77 times greater than ethanol, methanol, acetone, acetaldehyde, formaldehyde, toluene, benzene, chloroform and n-hexane, respectively, which shows its excellent selectivity towards ammonia vapours. Thus, PPy/MoO3 can be a promising material for the fabrication of a very sensitive, extremely reversible and excellently selective ammonia vapour sensor at room temperature.
Footnotes
Acknowledgments
We are very much thankful to Dr Faiz Mohammad for his indispensable guidance and support during the preparation of this manuscript.
Author contributions
Mohd Urooj Shariq and Ahmad Husain contributed equally.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

