Abstract
In this study, camphorsulphonic acid-doped conducting polyaniline/silk fibroin (Pani/SF) composites were prepared by in-situ polymerisation. The Pani/SF composite fibres were characterised by using Attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spectroscopy, thermogravimetric analysis, scanning electron microscopy and digital photographs. Results indicated that the Pani was successfully coated on silk fibroin. The electrical properties of Pani/SF composite fibres were influenced by the extent of loading of aniline monomers. The as-prepared material was studied as a sensing material by observing the change in their electrical conductivity on exposure to ammonia and acetaldehyde, followed by ambient air at room temperature. It was found that the composite fibres showed good reversibility and conductivity decreased on exposure to higher concentration of ammonia and acetaldehyde vapours at room temperature.
Keywords
Introduction
Conducting polymers are very versatile materials with potential applications in various evolving technologies such as electrodes in primary and secondary batteries,1,2 energy storage devices,3,4 microelectronics, 5 photocatalysts, 6 optoelectronic devices,7,8 sensors and actuators 9 . Among all conducting polymers, polyaniline (Pani) drew widespread attention because it possesses unique properties such as low cost, high environmental stability, reversible and tailorable electrical properties by controlled charge-transfer processes.10–12 Polyaniline can experience changes on adsorption and desorption of chemical species onto its surface, making it a potential sensing material for humidity, toxic solvents,13–16 and organic vapours such as methanol, ethanol, chloroform, dichloromethane and hexane.17,18
Silk fibroin (SF) is an attractive scaffold biomaterial because of its excellent mechanical properties, biocompatibility, biodegradability, tissue regeneration and various other biomedical applications. 19 Natural silk fibroin fibres (a filament core protein) is obtained by removing the outer sericin (glue-like coating of a nonfilamentous protein) from silk fibres with anhydrous sodium carbonate solution at appropriate temperature. 20 A number of composites of silk fibroin have been reported for various potential applications. Ismail et al. 21 reported the use of a fibroin/polyaniline composite for electrochemical characterisation as a reactive sensor. Xia et al. 22 reported work on a silk fibroin/polyaniline (core/shell) coaxial fibre for application in cell proliferation. The development of gas sensors by conducting composite materials has drawn more attention over the last few years. 23
Among various toxic compounds, acetaldehyde and ammonia are classified as potentially dangerous to the environment, even at low concentration. Thus, the development of new materials for the detection and determination of these toxic volatile compounds in the atmosphere are of immediate concern to environmental researchers. Herein we have tried to explore the ammonia and acetaldehyde sensing capabilities of Pani by modifying the non-conducting silk fibroin into Pani-coated conducting composite fibres (Pani/SF). To the best of our knowledge, a sensing application of Pani-modified SF fibres (Pani/SF) has not yet been reported elsewhere.
Experimental
Materials
In the preparation of Pani and Pani/SF composite fibres, the chemicals used were: aniline 99% (E. Merck, India), CSA (camphorsulphonic acid) ≥98% (TCI, Tokyo), cocoons for silk (Banaras, India), potassium persulphate (E. Merck, India) and methanol. Double-distilled water was used throughout the experiments.
Preparation of Silk Fibroin
Cocoons were degummed twice for 1.5 h in aqueous solution of 0.02 M Na2CO3 and then rinsed thoroughly with double-distilled water to extract the silk fibroin (SF). The extracted SF was then dried at 40°C for 24 h.
Preparation of Pani and Pani/SF Composites
Aniline (2.0 mL) was mixed with 100 mL of 0.1 M camphorsulphonic acid (CSA) under constant stirring for 1 h. A solution of potassium persulphate (PPS) was added dropwise and the mixture was allowed to react for 24 h. The final dark green coloured reaction mixture was then filtered, washed several times with double distilled water and methanol and then dried in an air oven at 70°C for 10 h.
The Pani/SF composite fibres were prepared by in-situ oxidative polymerisation of aniline in the presence of CSA modified SF fibres using potassium persulphate as an oxidising agent. The extracted silk fibroin fibres were dipped in 100 mL of 0.1 M CSA aqueous solution and stirred for 1 h. The resultant SF fibres were filtered and dried at 40°C. Thus the CSA modified SF fibres (300 mg) each were added to different amounts of aniline solutions prepared in 0.1 M CSA, and stirred for 1 h. A solution of K2S2O8 (6 g) in 100 mL of 0.1 M CSA was then added dropwise into the mixture at room temperature with constant stirring. The colour of the SF fibres changed from light yellow to greenish black, indicating the polymerisation of aniline on the silk fibroin fibres. The fibrous reaction mixture was then stirred for a further 24 h. The final Pani/SF composite fibres was then filtered, washed thoroughly with double-distilled water and methanol to remove excess acid, potassium persulphate and Pani oligomers. Thus-prepared Pani/SF composite fibres were dried at 50°C for 20 h in an air oven and were stored in desiccators for further experiments. For electrical conductivity measurements, 250 mg of material from each sample was pelletised at room temperature with the help of a hydraulic pressure machine at 80 kN load for 15 min. The preparation details are given in
Proportions of the constituents in the preparation of Pani-SF composites
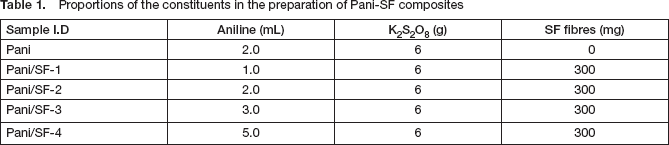
Proportions of the constituents in the preparation of Pani-SF composites

In order to investigate the morphology, structure and chemical composition of Pani/SF fibres, a variety of methods were used including attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR). The spectra were recorded using a Perkin-Elmer 1725 instrument. Scanning electron microscopy (SEM) studies were carried out on a JEOL, JSM, 6510-LV (Japan). Thermogravimetric analysis (TGA) was done by using a Perkin Elmer instrument in the temperature range from 35 to 700°C. The thermal stability in terms of DC electrical conductivity of Pani and Pani/SF composite fibres under isothermal and cyclic ageing conditions was also studied. For this study, a four-in-line probe with a temperature controller, PID-200 (Scientific Equipment, Roorkee, India) was used to measure the DC electrical conductivity and its temperature dependence. The equation used in the calculation of DC electrical conductivity was:

Mechanism of Preparation of Pani/SF Composite Fibres
The mechanistic view of the polymerisation process seems to involve the anilinium cations (phenyl-NH3+) getting trapped by Coulombic attraction between anilinium cations and the sulphonate group of camphorsulphonic acid adsorbed on the surface of the silk fibroin. Thus the SF fibres get uniformly surrounded by anilinium cations. This arrangement comes into contact with K2S2O8, the anilinium cations become polymerised on the surface of the SF fibres, forming a uniform smooth layer of Pani (emeraldine salt). The uniform deposition of Pani on the CSA modified surface of silk fibroin may be supported by the interaction between negatively-charged sulphonate groups and polarons of Pani. The similar mechanism is mentioned by Youyi Xia et. al. in which SF fibres were modified by methyl orange (MO) and then aniline was polymerised on the surface of MO modified SF fibres.
22
The schematic presentation of coulombic attraction is given in

Schematic representation of possible interaction between Pani and CSA modified SF fibres in Pani/SF composite fibres
ATR-FTIR spectra of the SF and Pani/SF-4 composite fibres are shown in

ATR-FTIR spectra of: (a) SF fibres and (b) Pani/SF-4 fibres
The thermal stability of the Pani/SF-4 composite fibres was investigated and shown in
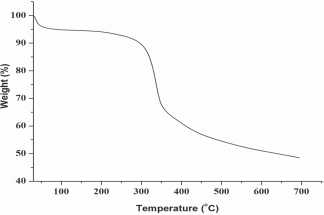
TGA thermogram of Pani/SF-4 composite fibres
The morphology of SF fibres and Pani/SF-4 composite fibres were studied by SEM at different magnifications as presented in

SEM micrographs of: (a and b) SF fibres, (c and d) Pani/SF-4 composite fibres at different magnifications

Digital photographs of: (a) SF fibres and (b) Pani/SF-4 composite fibres
For the electrical conductivity measurements and sensing experiments, 250 mg material from each sample was pelletised at room temperature with the help of a hydraulic pressure machine at 80 kN force for 15 min. The initial DC electrical conductivity Pani and Pani/SF composite fibres were measured by the standard four-in-line probe method. The variations in DC electrical conductivity in Pani/SF composites with loading of different amounts of aniline monomer are shown in
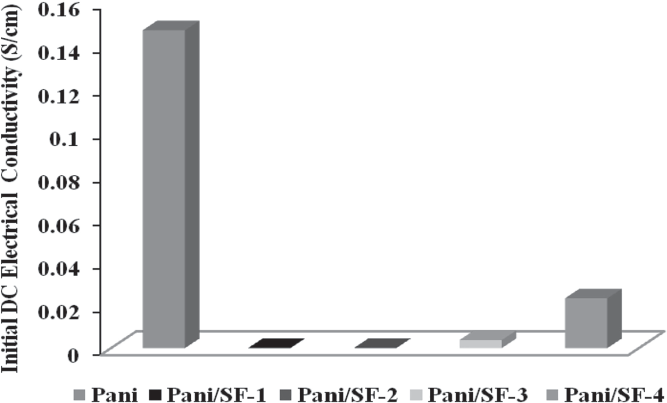
Initial DC electrical conductivities of Pani and Pani/SF composite fibres
Stability under Isothermal Ageing
The stability of Pani and Pani/SF-4 composite fibres in terms of DC electrical conductivity was studied under isothermal ageing conditions as shown in 

Relative electrical conductivity of: (a) Pani, (b) Pani/SF-4 composite fibres under isothermal ageing conditions
The stability in the term DC electrical conductivity retention of Pani and Pani/SF-4 composite fibres was also studied by a cyclic ageing method within the temperature range of 50 to 130°C, as shown in 
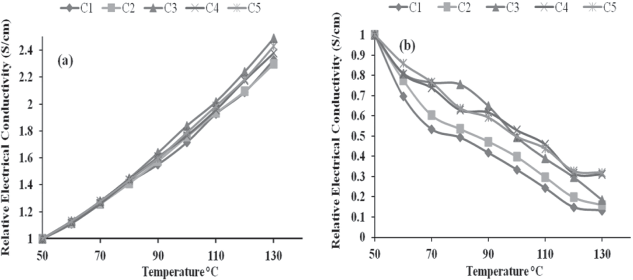
Relative electrical conductivities of: (a) Pani, (b) Pani/SF-4 composite fibres under cyclic ageing conditions
As each cycle ran from temperature 50 to 130°C the relative electrical conductivity of Pani seemed to increase, maybe due to the increment of number of charge carriers which may be attributed to the formation of greater number of polarons/bipolarons. From
Acetaldehyde is a highly reactive volatile organic compound which may be responsible for dizziness, headache and even cancer if someone is exposed to its vapour for prolonged periods. Although ammonia solution does not usually cause problems for humans and other mammals, it is highly toxic to aquatic animals even at very low concentration. Thus both the compounds are classified as dangerous for the environment and the detection of acetaldehyde and ammonia vapours are important in environmental monitoring and chemical control. The acetaldehyde and ammonia vapours sensitivities of Pani/SF-4 fibres were monitored by measuring the changes in the electrical conductivity at room temperature, using a four-in-line probe electrical conductivity device as shown in

Schematic representation of acetaldehyde and ammonia vapours sensor unit by four-in-line probe technique
The electrical conductivity of 250 mg pelletised Pani/SF-4 composite fibres showed significant response on exposure to different concentrations (0.1, 0.2, 0.3, and 0.5 M) of aqueous acetaldehyde at room temperature, as shown in

Effect on the DC electrical conductivity of Pani/SF-4 on exposure to different concentrations (0.1 M, 0.2 M, 0.3 M, 0.5 M) of acetaldehyde with respect to time
The reversibility response of Pani/SF-4 composite fibres towards 0.5 M aqueous acetaldehyde was investigated as shown in

Reversible electrical conductivity response of Pani/SF-4 composite fibres in the vapour of 0.5 M acetaldehyde solution
The similar experiment was done in presence of different concentrations (0.1, 0.2, 0.3, and 0.5 M) of ammonia solution at room temperature, as shown in

Effect on the DC electrical conductivity of Pani/SF-4 composite fibres on exposure to the vapour of different concentrations (0.1 M, 0.2 M, 0.3 M, 0.5 M) of ammonia with respect to time
The reversibility of the response of Pani/SF-4 composite fibres towards 0.5 M ammonia solution was also investigated, as shown in

Reversible electrical conductivity response of Pani/SF-4 composite fibres for 0.5 M ammonia solution
The sensing mechanism of ammonia and acetaldehyde vapours through the DC electrical conductivity response of the composite fibres may be explained by simple adsorption and desorption affecting the conduction mechanism at room temperature. In Pani/ SF-4 composite fibres, the DC electrical conductivity decreases after exposure to acetaldehyde and ammonia vapours, which may be attributed to a decrease in mobility of the charge carriers in Pani. The decrease in mobility of charge carriers may be due to interaction of lone pairs of oxygen and nitrogen of acetaldehyde and ammonia respectively with positive charged polarons concentrated at the nitrogens of polyaniline.24,29 Thus the mobility of the charge-carrying polarons decreases, resulting in a decrease in the electrical conductivity. When the adsorbed vapours of ammonia and acetaldehyde on the surface of polyaniline get released from the surface under ambient conditions, it leads to an increase in the electrical conductivity.
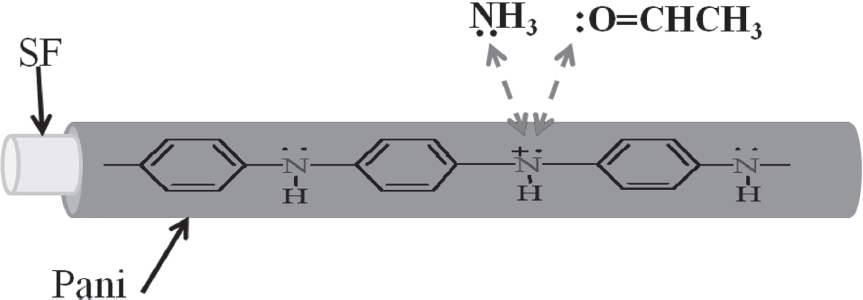
Proposed interactions of Pani/SF-4 composite fibres with acetaldehyde and ammonia
Pani/SF composite fibres were successfully prepared by an in-situ polymerisation method and confirmed by FTIR and TGA analysis. SEM analysis confirmed the uniform deposition of Pani on CSA-modified SF fibres. Pani/SF composite fibres showed lower conductivity but greater thermal stability in terms of their DC electrical conductivity under isothermal and cyclic ageing conditions than pristine Pani. The results highlighted excellent reversible acetaldehyde and ammonia sensing, indicating that the Pani/SF-4 composite fibres could be utilised as a sensor material.
