Abstract
In the present study, chitosan oligomer was modified to sulfated chitosan oligomer (ShCsO) to mimic heparin. Its chemical structure was determined by infrared spectroscopy (FT-IR), X-ray diffractometry (XRD), differential scanning calorimetry (DSC), and thermogravimetric analysis. The results showed that the FT-IR spectrum band at 799 cm−1 corresponds to C–O–S and that at 1212 cm−1 corresponds to S=O bond stretching, which prove that the sulfate groups are incorporated into chitosan oligomer successfully. The antimicrobial activity of ShCsO against to Bacillus subtilis in 1% concentration was 89.1 ± 1.7%. The IC50 (μg/ml) of ShCsO was 67.75, 56.07, 85.47, and 84.68 for A2780, MCF-7, DU-145, and HepG2, respectively. The results show that this newly synthesized material is a potential candidate to heparin-like chitosan derivatives. According to the literature, it was the first time that chitosan oligomer was modified to mimic heparin.
Introduction
According to the GLOBOCAN database, cancer is a major threat to health life around the world and cancer cases will increase in the next 15 years. 1 On the basis of this data, scientists need to develop new anticancer agents with fewer (or no) side effects. 2 Cancer is a complex disease, and metastasis plays an important role for cancer mortality rate. Cell–cell interactions including cell adhesion, proliferation, migration, and invasion lead to separation of cancer cells from initial tumor tissue to other part of body. There are two important cell attachment material families, selectins (vascular cell adhesion molecules) and lectins (contribute the cell adhesion) and their ligands have major roles in to attachment and metastasis of tumor cell in blood vessel. Heparin (inhibits both selectins and lectins) is also identified as an important molecule in metastasis. 3 Heparin is a member of glucosaminoglycan (GAG) family and is highly negatively charged because of its carboxyl and sulfo groups. 4 In the natural state of heparin, it has heterogeneous polysaccharide chains with different length and sulfonation degree. 5 The linear structure of the heparin consists of 1–4 linked repeating disaccharide units including (D-glucuronic acid (GlcA) or L-iduronic acid (IdoA)) and D-glucosamine (GlcN). The main repeating disaccharide unit of heparin is L-iduronic acid, while the heparan sulfate that is also similar to heparin is D-glucuronic acid. 6 According to in vivo studies, mucin, sulfated glycolipids, and glycosaminoglycans bind to selectins as functional selectin ligands.7,8
Chitosan is a polysaccharide, consists of glucosamine units, and obtained via deacetylation of chitin found in organisms such as crab, crustacean, and mushroom. 9 One of the most important polysaccharides, chitosan, the deacetylated form of chitin, 10 has different applications such as medicine,11,12 food industry,13,14 and environment . 15 Due to its biocompatible and biodegradable properties, it is an interesting polymer. Chitosan oligomer (CsO) has more advantages compared to high molecular weight chitosan. Higher MW chitosan limits the biological application in different area because of low solubility and higher viscosity. CsO found a lot of application areas due to its low MW and water solubility characteristics. 16 The literature shows that CsO prevents tumor growth and malaria, improves bone strength, and treats asthma .17, 18 Biological activities of CsO consist of antibacterial, antifungal, anti-tumor, antioxidant, blood pressure control, hypocholesteromic effects, immune alert agent, decreasing fatty acid,19,20 and also fight bacteria. 21 CsO evolved products are used in medicine such as gene transferring, 22 anti-adipogenic effect and decreasing the nitric oxide releasing,23-25 and heparin mimicking.26,27 Another application of the sulfated chitosan is to prevent metastasis. Sulfonation of chitosan has been one of the most interested modifications to obtain molecule-like heparin. Sulfonation of chitosan was made using various reagents by scientists. 28 Sulfonated derivatives of the chitosan are used as blood anticoagulant activity.29,30 The anticoagulant activity of sulfonated chitosan arises due to the sulfur concentration in the chitosan. It has been reported that the modified sulfated chitosan demonstrated anticoagulant activity, and this result is similar to the anticoagulant activity of heparin. 31
The aim of the submitted study depends on the synthesis, chemical characterization, and determination of biological activities of heparin mimicking sulfated chitosan oligomer. For this purpose, it is modified from chitosan oligomer and after characterization studies, its antimicrobial activities and cytotoxicity studies were performed.
Experimental progress
Materials
Chitosan oligomer was purchased from Carbosynth (product code: OC28900). Its molecular weight was 3 kDa, and its deacetylation degree was at least 85%. The other mainly used chemicals were acetic acid (AA), ether, ammonia, acetone, sulfuric acid (65%), chlorosulfonic acid (HClSO3), formamide, and dimethylformamide (DMF) which were bought from Sigma. The other chemicals in the article were of reagent grade and they were purchased from Merck AG (Germany).
Synthesis of sulfated chitosan oligomer (ShCsO)
The sulfated chitosan oligomer (ShCsO) (Figure 1) was synthesized according to the literature.
23
First of all, chitosan oligomer (1 g) was dissolved in the 100 mL N, N- dimethyl formamide via stirring at 60°C.
32
8 mL HCISO3 was added into the solution and continued stirring for 14 h. Then acetone (5 fold) was put in solution. To precipitate the modified chitosan oligomer, the mixture was spun at 5000 g for 30 min at 4°C. The precipitated material was cleaned once with acetone. The product was dialyzed versus distilled water during 48 h. The dialyzed sample was lyophilized under the – 60°C. Structure of chitosan oligomer (a), heparin mimicking sulfated chitosan oligomer (b), and heparin structure from PubChem databank Compound ID: 71312100 (https://pubchem.ncbi.nlm.nih.gov/) (c).
Chemical Characterization
Fourier transform infrared spectrophotometer (FT-IR) analysis
The FT-IR analysis of chitosan oligomer and sulfated chitosan oligomer was made with Fourier transform spectrophotometer (Perkin Elmer, UK). ShCsO and CsO samples were mixed with potassium bromide (KBr) separately and pressed into a disc. KBr was used as a blank (reference molecule). Then the FT-IR spectrum was recorded.
Differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA)
These analyses were used to determine the effect of sulfate group incorporation into the chitosan oligomer structure. DSC and TGA data were obtained using a Shimadzu TA60 thermal analysis system. Chitosan oligomer and sulfated chitosan oligomer (3–5 mg) were heated up to 500°C and 700°C with a heating rate of 10°C/min in a dynamic nitrogen atmosphere for DSC and TGA analysis, respectively. A sealed empty aluminum pan was used as a reference.
X-ray diffraction (XRD) analysis
XRD patterns were recorded by a (D2, Bruker, Germany) over a 2-theta range from 5 to 50° using a scan rate of 0.04 min−1. During the analysis, the operating voltage and current were 40 kV and 30 mA. The X-ray source was Cu-Kα radiation with a wavelength of 1.54 Å.
Biochemical properties
Antimicrobial activity
Antimicrobial activity was tested against Gram-negative bacteria (E. coli) and Gram-positive bacteria (Bacillus subtilis).
33
Serial dilution method was used to dilute the bacterial suspension with sterile physiological saline (0.85%) to 102–103 CFU/ml.
34
The inhibition rates of chitosan oligomer and sulfated chitosan oligomer against E. coli and B. subtilis, were studied by the agar plate method. The different concentrations of chitosan oligomer and sulfated chitosan oligomer (0% (blank), 0.01%, 0.05%, 0.1%, 0.20%, 0.50%, and 1.0% (w/v)) solutions were prepared using distilled water. After sterilization of the LB (Luria Bertani), agar culture medium was poured to obtain solid petri dishes. After solidifying, 50 μl of bacterial suspension and 100 μl of each sample (chitosan oligomer and sulfated chitosan oligomer) with different concentrations were mixed. Then the bacterial mixture was spread uniformly onto the petri plates. The sterile water was also used to compare as a reference. The experiments were performed in triplicate, and the colonies were counted after incubation at 37°C for 24 h. The following equation (equation (1)) was used to calculate the percent inhibition rate (IR%)
Cytotoxicity
Cell viability assay of the chitosan oligomer and sulfated chitosan oligomer was evaluated by using 36 a colorimetric method to produce a colored formazan compound using the 3-(4, 5-dimethylthiazol-2-yl)-2,5 diphenyl tetra sodium bromide or MTT by live cells. Colored formazan compounds were then dissolved using a solubilizing solution, and absorbance was measured at 550 nm using a plate-reader. Human ovarian cancer (A2780), breast cancer (MCF-7), prostate canser (DU-145), and liver cancer (HepG2) cell lines were used. The cell cultures were enhanced at 37°C in media with 5% CO2 and atmosphere of 95% air. When the cells reached at 70–80% growth rate, they were passaged. Before treatment, the cancer cells containing 1.5 × 10 4 cells were seeded into each well of 96-well plate and permitted to attach for 24 h. Then cells were treated with various doses (1, 5, 25, 50, and 100 μg/ml) of chitosan oligomer and sulfated chitosan oligomer for 24 h. 50 μl of MTT solution (5 mg/mL) was added to each well incubated at 37°C in media with 5% CO2 and atmosphere of 95% air for 3 h. Then 100 μl of DMSO was mixed and stirred for 15 min. At room temperature, the absorbance values were recorded at a wavelength of 550 nm using ELISA (enzyme-linked immunosorbent assay) (Synergy HTX, ABD). The obtained average absorbance value for control wells was accepted as 100% viable cells. The absorbance values obtained from wells treated with chitosan oligomer or sulfated chitosan oligomer were proportional to the control absorbance value and were accepted as percent viability. These MTT studies were performed 10 times on different days.
Statistical analysis
The KOLMOGOROV SMİRNOV test was used to evaluate group normality. LEVENE’s method was used to measure the homogeneity of variances. Group comparison was made by one-way analysis of variance. Test of TAMHANE T2 was used for multiple comparisons due to nonhomogeneity of variances. If the P was lower than 0.05, it was considered as significant. The results were expressed as mean ±SD
Result and discussion
Chemical characterization
FT-IR
FT-IR analysis was used to evaluate incorporation of the functional group (sulfate) into chitosan oligomer. The FT-IR spectrum of chitosan oligomer showed bands at 1616 cm−1 (Amide-I), 1516 (Amide-II) cm−1, and 1411 cm−1(-CH2-bending) like in the literature,
37
and Figure 2 shows FT-IR spectrum bands at 799 and 1212 cm−1 due to C–O–S and S=O bond stretching, respectively, which provide that the sulfate groups are incorporated into chitosan oligomer successfully; similar changes or results were also observed.38-45 The main functional groups in the disaccharide unit of heparin are COO−, SO3−, OH, NH, and C-O-C, while the main functional groups of sulfated chitosan oligomer are COO−, SO3−, OH, NH, and C-O-C (Figure 1). When compared with the FT-IR spectrum of standard heparin in the literature, the similarities between the FT-IR peaks of heparin and sulfated chitosan oligomer could be seen. The broad peak in the range of 3100 cm−1 to 3700 cm−1 suggesting the stretching of the hydroxyl group. The peaks recorded in the range of 1600 cm−1 to 1700 cm−1 in both of heparin and sulfated chitosan oligomer proved the presence of N–H bend and C=O stretching, the peak at the range of 1200 cm−1 to 1250 cm−1 mainly pointed toward the strong S=O stretching of sulfate.46,47,48 Thus, the FT-IR spectra of heparin and sulfated chitosan oligomer show characteristic peaks of their main functional groups. FT-IR of chitosan oligomer (CsO) and sulfated chitosan oligomer (ShCsO). A1) 1616 cm−1 (Amide-I), a2) 1516 (Amide-II) cm−1, a3) 1411 cm−1(-CH2-bending), a4)1212 cm−1, and a5)799 cm−1.
DSC and TGA
The DSC and TGA thermograms of CsO and ShCsO were compared as shown in Figure 3. Two peaks were observed both of CsO and ShCsO in the DSC curve. The first one is related with weight loss due to water molecule. The second one is also related with weight loss which might be due to decomposition of pyranose ring of CsO and ShCsO.
49
The second peak of ShCsO shifts to higher temperature (approximately 240°C). This shifting might be evidence of the sulfate group incorporation in the structure. DSC and TGA results of chitosan oligomer (CsO) and sulfated synthesized chitosan (ShCsO). Abbreviations: DSC = differential scanning calorimetry; TGA = thermogravimetric analysis.
The TGA results of the CsO show one water loss (25–100°C) including two decomposition events may have occurred for CsO. The second decomposition appears between 250 and 500°C with a maximum weight loss of the CsO. The TGA results of the ShCsO show one more peak which corresponds to the sulfate group decomposition. This event of the ShCsO extends from around 160°C up to around 300°C. This difference of decomposition between the two samples at 100–250°C is because of sulfonation. 50 The further decomposition of ShCsO is because of the bulk decomposition. 51
XRD
The XRD result of CsO and ShCsO molecules is given in Figure 4. Chitosan oligomer have two reflections at 2θ = 14° and 2θ = 24°. The reflection at 2θ = 14° was associated with crystal form I. The strongest reflection is at 2θ = 24° and corresponds to crystal form II. Sulfated chitosan has more intensive peak at 2θ = 14°; the peak was increased with degree of sulfonation, suggesting that chemical modification increased its capability to form a hydrogen bond.
52
XRD results of chitosan oligomer (CsO) and sulfated synthesized chitosan (ShCsO).
Biochemical Characterization
Antimicrobial activity
Antimicrobial activity of CsO and ShCsO against E. coli and B. subtilis is shown in Figure 5 and Figure 6, respectively. Chitosan oligomer (3 kDa) inhibited bacterial growth of both E. coli and B. subtilis, at a 1.0% concentration. Water-soluble chitosans such as chitosan oligomers show bactericidal activity against both E.coli and B. subtilis.
19
The main antimicrobial activity of chitosan depends on the protonated amine groups which interact electrostatically with the phosphoryl groups of the phospholipids, found the cell membrane structure. The antimicrobial activity is also affected by the physical conditions such as temperature, pH, and salinity.
53
In the literature, chitosan oligomers showed different antimicrobial activity against Gram-negative and Gram-positive bacteria according to their molecular weights differences and chitosan concentrations.
54
However, this behavior was not observed with Gram-negative bacteria for sulfated chitosan.
55
The sulfated chitosan showed inhibition rate as 21 ± 17% to E. coli at 1% sample concentration (Figure 5). Distribution of the cell surface negative charge of Gram-positive bacteria is lower than that of Gram-negative,
19
and it causes more charge–charge repulsion between cell surface and sulfated chitosan oligomer. Moreover, the sulfated groups increase the negatively charged ShCsO and interact with biological macromolecules such as proteins, phospholipids, and fatty acids which are found at the surface of bacteria cell. After the reaction, it prevents their biosynthesis, blocking the mass (foods) transport across the cell wall, and leads to the death of the bacteria terminatively. In the current study, inhibition rate of sulfated chitosan on B. subtilis is 89.1 ± 1.7% at 1% sample concentration. Inhibition rate of chitosan (CsO) and sulfated chitosan oligomer on E. coli. Inhibition rate of chitosan (CsO) and sulfated chitosan oligomer on B. subtilis.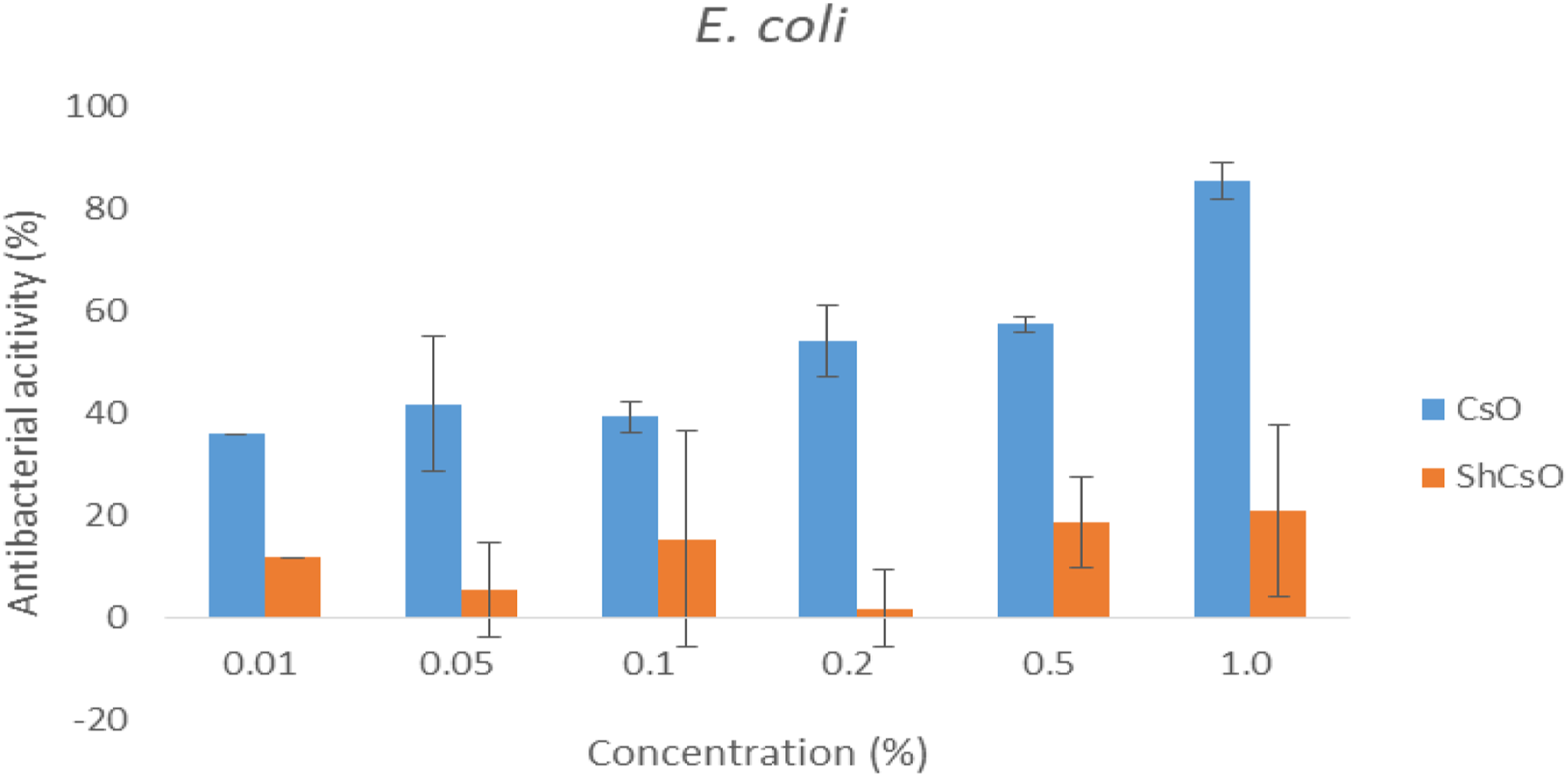

Cell viability
In the current study, cytotoxicity of the chitosan oligomer (CsO) and sulfated chitosan oligomer (ShCsO) was determined by MTT assay which was performed by using different concentrations between 1 and 100 μg/mL at 24 h. Figures 7–10 show cytotoxicity on A2780, MCF-7, DU-145, and HepG2 cancer cells, respectively. Table 1 shows the IC50 and Log IC50 values calculated from the graph of Figures 7–10. In the current reported data, sulfated chitosan oligomer had an IC50 value of 67.75, 56.07, 85.47, and 84.68 μg/ml (Table 1) for A2780, MCF-7, DU-145, and HepG2 cells, respectively. The cell viability test of chitosan and its degraded product or derivatives on some cancer cells has been reported in the literature.56,57 In the literature, sulfated chitosan was studied with breast cancer cells (MCF-7 and MCF-7ras),
58
and their results showed that IC50 value was 35.65±1.44 μM; however, in the present study, the sulfated chitosan showed that IC50 is 56.07 μg/ml against MCF-7 cancer cells. The literature also showed that sulfonated chitosan efficiently inhibited the MCF-7 cell growing as dose-time dependent manner via apoptosis.
59
In addition, the literature showed that sulfated chitosan did not inhibit the (BL6) melanoma cells,
60
while the 6-O-sulfonation of chitosan inhibited P selectin binding to the human melanoma cells (A375).
61
Sandhya et al. declared that 9 μg/ml and less sulfated chitosan did not show any cytotoxic effect on RAW 264.7 cells.
62
In vitro cytotoxicity of chitosan oligomer and sulfated chitosan oligomer on A2780 cells. Cell viability is expressed as mean ± S.D. All experiments were performed 10 times. In vitro cytotoxicity of chitosan oligomer and sulfated chitosan oligomer on MCF-7 cells. Cell viability is expressed as mean ± S.D. All experiments were performed 10 times. In vitro cytotoxicity of chitosan oligomer and sulfated chitosan oligomer on DU-145 cells. Cell viability is expressed as mean ± S.D. All experiments were performed 10 times. In vitro cytotoxicity of chitosan oligomer and sulfated chitosan oligomer on HepG2 cells. Cell viability is expressed as mean ± S.D. All experiments were performed 10 times. After 24 h incubation, A2780, MCF-7, DU-145, and HepG2 cells IC50 values.




Conclusion
In conclusion, the findings of this research showed the chemical and biological potential of sulfated chitosan oligomer with characteristics comparable to standard heparin. Prepared heparin mimicking chitosan oligomer derivative is a potential anti-metastatic drug candidate in the future. The chitosan oligomer was modified (ShCsO) by sulfonation of chitosan oligomer, and its characterization was made by FT-IR, XRD, TGA, and DSC analyses. The ShCsO demonstrated good antimicrobial activity against only Gram-positive, and the comparable IC50 values occurred when evaluated statistically against the A2780, MCF-7, DU-145, and HepG2 cells. Further studies will be focused on molecular docking research on sulfated chitosan oligomer and its receptors (heparanase). In the future, the interested experiments will be carried out on microfluidic cell adhesion experiments and anti-metastatic effects both in vitro and in vivo.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Cumhuriyet University Scientific Research Project (CUBAP) financed this project (Project number:F-575).

