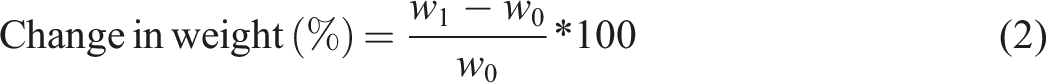Abstract
The aim of this study is to use recycled waste materials to modify the performance of composites used as dental fillings. For this purpose, depolymerized polyethylene terephthalate (DPET) polymers were prepared by recycling waste polyethylene terephthalate (PET) using the glycolysis process. DPET was then blended with polymethyl methacrylate (PMMA) at different weight percentages. Polymer blends were subjected to mechanical, physical, and biological tests. It is evident that PMMA blended with a 5 wt% of DPET exhibits the highest compressive strength which is approximately two times that obtained from neat PMMA, highlighting the suitability of these resins to replace amalgam and to be considered for dental restorations. It was also found that the water absorption of the blends investigated is relatively stable at 5 wt% DPET. The SEM images showed no distinguishable phases, highlighting the high compatibility between DPET and PMMA. The biological tests, including in vitro cytotoxicity, showed that the cell viability mean values of PMMA containing 5 wt% DPET is higher than that of control group. Interestingly, in dental practice, the reliable in vitro cytotoxic approach is considered as an important aid in clinical procedures in the use of dental filling systems. The microbiology study has shown that the inhibition zones against Streptococcus mutans of PMMA: 5 wt% DPET is higher than the control group. According to the obtained results, the blend of PMMA: 5 wt% DPET was selected as an optimum blend, which was then reinforced with various weight percentages of nano-sized titanium dioxide (TiO2). The composites were then tested under compression, microhardness, and impact tests. The results demonstrated the significant impact of TiO2 on enhancing the properties of the composites investigated. This can provide a new protocol for synthesizing nanocomposite materials for dentistry.
Keywords
Introduction
Recently, polymer-based biomaterials have found wide range of applications in dental industry. The investigations are based on innovations in compositional material design and processing to enhance the performance of polymers. Good mechanical properties and compatibility are the most requirements in choosing materials for dental applications. 1 Despite significant advances in oral hygiene education and practice, dental decay (or dental caries) treatment continues to be an integral element of routine dentistry. 2 In dentistry, as the decay proceeded, fillings are considered as the main treatment option. Filling materials used in dentistry can be polymeric composites, porcelain, and dental amalgam (i.e., combination of several materials). 3 Traditionally, amalgam has been used to treat cavities in posterior teeth; however, because of dissatisfaction with the metallic appearance and concerns about the mercury they contain, they are being used less often, especially in high-income countries. As an alternative to amalgam fillings, tooth-colored composite resin fillings have been employed. 4
Polyethylene terephthalate is considered as the most common in the polyester family. It is a semicrystalline and transparent thermoplastic polymer with good mechanical and chemical properties. Many plastic products, such as water and soft drinking bottles, video and audio tapes, synthetic fibers, photographic films and food packaging, are made of PET. Despite the aforementioned uses of PET that, its disposal in the environment poses major challenges, as it takes between 300 and 450 years to naturally degrade.5,6 Therefore, the recycling of polymers is considered as one of the most successful solutions. PET products can be reused by converting its chains to monomers or other useful compounds by depolymerization process. Based on the chemical agents used to break down the polyester chain, depolymerization can be categorized as glycolysis, methanolysis, hydrolysis, etc. Glycolysis, as chemical recycling technique, is the most common process at which PET polymer is broke down into monomers and dimers with the help of chemicals such as glycol.5,7,8 In the presence of trans-esterification catalysts, ester linkages are broken and replaced by hydroxyl terminals,
9
as presented in Figure 1. PET glycolysis.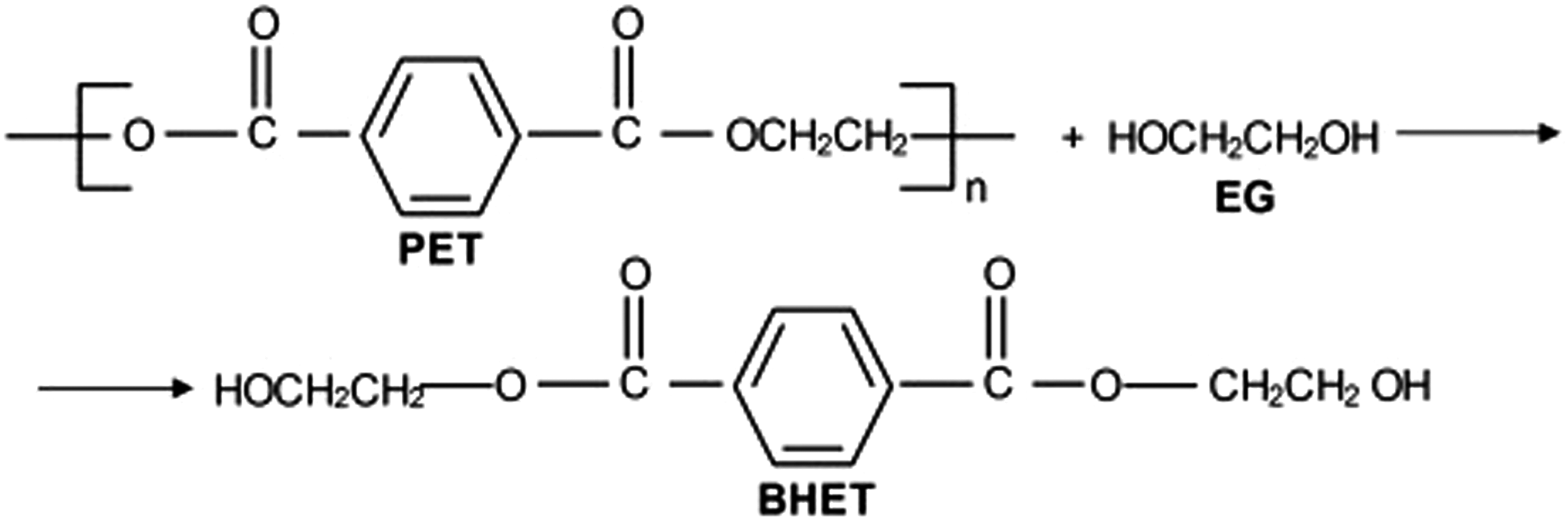
It was observed by researchers from Brunel University London that drinking bottles manufactured using recycled polyethylene terephthalate (PET) can contain higher levels of pollutants than those made of new PET, suggesting that recycling process may increase the possibility of contamination occurrence. 10 Polymer blending is one of the most important and influential methods for developing new polymeric materials. The properties of polymer blends are better than that obtained from its components. However, the properties of the blends depend on the compatibility between its parts at the molecular level. 11 Therefore, creating polymer blends based on DPET and PMMA is expected to produce high-performance polymers suited for biomaterial applications. PMMA, as the most used acrylic material, is used widely in restorative dentistry for both cosmetic and functional purposes due to its unique properties such as easy to manipulate, non-toxic, low water absorption, recyclability, solubility, and high aesthetic appeal. 12 Nonetheless, adequate survival rates for the use of PMMA as a dental material have yet to be reached. To tackle this issue, researchers are encouraged to create PMMA-based materials reinforced with various types of nanoparticles. 13
The utilization of nanoparticles as reinforcing materials has found a wide range of applications in dentistry. Efforts are directed towards the incorporation of nanofillers into acrylic resins to produce nanocomposites with superior mechanical properties compared to those containing micro-scale particles. Titanium dioxide (TiO2) nanoparticles are the best candidates as biocompatible materials due to their outstanding properties such as non-toxicity, chemically stable, resistant to corrosion, high refractive index, and efficient against a broad spectrum of pathogens, including gram-positive and gram-negative bacteria, fungi, and viruses.12,14 Faiza M. Salim 15 prepared nanocomposites based on PMMA reinforced with 40% TiO2 and 60% of calcium aluminate (CaAl2O4) in terms of different volume fractions (1%–5%). It was shown that the friction and wear coefficient of the nanocomposites improve as the volume fraction of the nanoparticles increases. It was concluded that, based on the mechanical results, a 5% volume fraction of fillers was the sufficient value for reinforcing dental filling materials.
Mana Alqahtani 12 prepared nanocomposites based on self-curing PMMA reinforced with stabilized zirconia (8Y ZrO2) and hexagonal boron nitride (hBN) nanopowders. The results demonstrated that PMMA with 5% of hBN enhanced the values of young modulus and flexural strengths by approximately 240% and 550%, respectively. The Vickers hardness (VH) increased by almost 400% when the same content of 8Y ZrO2 was added. Rihem et al. 16 developed new biocomposite by combining PMMA with Salvadora persica particles. The biocomposite were characterized using XRD, FTIR, DSC, and high-performance liquid chromatography techniques. The results showed that the biocomposites have good biological activities. The influence of different concentrations of zirconate coupling agents (ZCA) on the mechanical characteristics of hydroxyapatite (HA) reinforced PMMA is investigated by Chow et al. 17 It was indicated that the PMMA/5HA-2% ZCA composites exhibit superior flexural strength compared to untreated PMMA/HA composites. 18 Nidal et al. 19 studied the effect of silanated nanobarium titanate (Si-NBT), titanated (Ti-NBT), and pure NBT (Un-NBT) on the performance of PMMA matrix. The results indicated that the titanium-treated samples showed less ability to absorb and dissolve SBF than the silanated samples. Furthermore, the fracture toughness of titanated samples was 26% higher than that of silanated samples. Richie et al. 19 used PET as an additive to traditional fine aggregate in concrete at 5, 10, and 15 wt%. The results indicated that the specimens containing 5 wt % PET exhibited the highest compressive strength, whereas a significant drop in flexural strength was abstained comparing to the control concrete. Malik et al. 20 utilized wasted PET bottles to enhance the properties of asphalt mixtures by combining them with bitumen in proportions of 8%, 10%, and 12%. It was shown that PET-based mixtures exhibit higher deformation resistant than that offered by traditional ones. Saleem et al. 21 investigated the compatibility between PMMA and DPET using FTIR, DSC and SEM techniques. The results showed that there was no visible phase separation in blends based on 95 wt% PMMA and 5 wt % DPET.
It can be concluded that, based on the authors knowledge, there are no previous works in the opening literature focused on using recycled PET in dental filling applications. Therefore, the aim of this work is to investigate the response of a novel sustainable nano composites used as dental fillings. Here, blends of DPET and PMMA were prepared at various weight percentages. The resulted blends were then subjected to mechanical and biological tests to highlight its reliability as a good candidate to already existed dental fillings. Based on the results, an optimum blend was selected and reinforced with different weight percentages of nano TiO2. The influence of the nanoparticles on the mechanical behavior of the optimum blend was also investigated.
Materials and experimental procedure
Materials used
Commercial-grade polymethyl methacrylate acrylic (PMMA) resin was supplied from (Lama Co., Iraq) into two parts; (1) modified PMMA (powder) with a density of 1.18 g/cm3, and (2) MMA (liquid) with adensity of 0.94 g/cm3. Nano particles of TiO2 with 19–35 nm size, a specific surface area of 151–550 m2/g, and a density of 2.2 g/cm3 were used in this investigation. Ethylene Glycol (EG) with a purity of >99.0% (Thomas Baker) and Magnesium Oxide (MgO) was supplied by Sky Spring Nanomaterials, USA. Here, the wasted bottles of drinking water, made of PET, were collected from the market (Baghdad/Iraq). Initially, a PET powder was produced from post-consumer clear PET bottles from which the polyethylene caps and the polypropylene label were removed. The bottles' heads were then cut and grounded in an industrial stainless-steel grinder with a capacity of 100 g. The steps involved in the synthesis of DPET powder are: Firstly, a 100 g PET powder was mixed with ethylene glycol in a molar ratio of 4:1 (EG:PET). The mixture was then added to the batch reactor. 22 Moreover, A 0.5 wt% of MgO, based on the weight of PET, was added as a transesterification catalyst. After that, the mixture was heated to 197°C for 50 min until it reaches a consistency similar to wax. The heat treatment included complete condensation (reflux) in a closed system (no material loss) utilising a water-cooling glass condenser. In order to remove the unreacted ethylene glycol solution and any possible contaminants, the mixture was washed with distilled water, heated for 30 min, dried, and filtered using qualitative filter paper 15 cm via Buchner. Finally, the filtrate white crystalline flakes of BHET monomers were separated, air-dried, and grounded once again.
Preparation of the composites
Initially, the polymer matrix was prepared by blending different weight percentages of DPET powder with a (3:1 (P/L)) ratio of PMMA (powder) and MMA (liquid). The weight percentages of DPET were 1%, 2%, 5%, 10%, and 20%. These blends were tested according to the ASTM recommendations. Based on the testing results, an optimum blend was selected. Finally, the optimum recipe was then reinforced with various weight percentages of nano TiO2 and these being 3 wt %, 6 wt %, and 12 wt %.
Physical testing
Here, the specimens were prepared in accordance with ASTM standard D 695. The specimens, shown in Figure 2, were tested using a universal testing machine (LARYEE, China) at a cross-head speed of 0.5 mm/min at room temperature. Three samples were tested and the average value was taken. Specimens of blends (a); specimens of composites (b) Experimental before testing; samples under testing (c).
The samples for Izod impact testing were prepared, as shown in Figure 3, in accordance with ISO-180. The tests were conducted using the (XJU series pendulum Izod/Charpy impact testing equipment) with a speed of 3.5 m/s and an energy of 5.5 Again, an average value three sample tested was taken Specimens for impact testing.
The microhardness test was conducted using Vickers hardness (LARYEE), as shown in Figure 4. A diamond pyramid with a square base and an angle of 136° between the opposite faces was used as an indenter. A force of 100 kgf was applied on the surface of samples. A diamond-shaped indent was produced, and the diagonals of the indent were measured to determine the Vickers hardness (HV) using the formula: Vickers’s hardness tester.
The morphology of the specimens investigated were characterized using SEM (model Inspect F50 FE-SEM). Before SEM examination, the samples were coated with gold.
The ability of the materials investigated to absorb water was tested in accordance with ASTM D570. Here, the specimens were immersed in a distilled water at room temperature for 24 h. The samples were then removed and dried with a dry cloth. The weight values of the specimens before and after dipping in water were recorded using a precision balance (model PS 360/C/1) and the water absorption of the samples was calculated using the following equation:
Biological testing
Antibacterial activity
The antibacterial properties of the PMMA/DPET blends were evaluated using the direct contact method (agar diffusion method) against Streptococcus mutans (gram-positive) bacteria, as it is the main microorganism associated with the development and progression of carious lesions (primary and secondary caries). 23 Approximately a 20 mL of Muller-Hinton (MH) agar was put into sterile Petri dishes. After refining the organisms, 6 mm-diameter wells on agar plates were bored with a sterile tip. Different sample concentrations were employed in the bored wells. After that, plates with samples and test organisms were kept at 37°C for one night before measuring the average diameter of the inhibition zone.
Cytocompatibility
Cell culture
In a humidified incubator with a 5% of CO2, the HDFn (human dermal fibroblasts neonate) cell line was cultured and activated in a RPMI 1640 culture medium (Sigma, United States) added with 10% of foetal bovine serum (Gibco, FBS; Grand Islands, N.Y., United States) and 1% of penicillin/streptomycin (Grand Islands, Invitrogen, N.Y., United States). Confluent cells were separated utilizing 0.25% trypsin (Sigma, Saint Louis, Missouri, USA). After that, the samples were immersed in 6 mL of sterile cell culture medium in sterile vials, which were then placed onto vial holders and stored inside the incubator at 37°C. The samples of supernatants from culture media were collected after 1, 2, and 3 days.
Cell viability (MTT assay)
Mosmann’s Tetrazolium Toxicity Assay (MTT) is the most common, quick, and simple method to evaluate the vitro cytotoxicity of the DPET/PMMA blends using a yellow dye called 3-[4,5-dimethylthiazol-2-yl]-2, 5-diphenyl tetrazolium bromide that changes into purple formazan crystals. This reaction happens when the enzyme succinate dehydrogenase in the mitochondria of living cells breaks the tetrazolium ring. This enzyme is only found in the healthy and living cells. 24
Human dermal fibroblasts were cultivated (1 × 105 cells mL−1) on a 96-well plate to a final volume of 200 µl of full culture media per well, i.e., eight wells cultured with cells were employed for each subgroup of the groups. The plates were covered with a sterile parafilm, stirred gently, and put in an incubator at 37°C for 24 h with 5% CO2. The culture media was discarded, and a 200 µl of culture media, including specimens from each immersion period, was added. To assess the responsiveness of the cells, a negative control (8 wells; cells and medium only) and a positive control (8 wells; cells treated with 10% ethanol) were utilized.
25
A triplicate (i.e., three repeats) was done for each group. The plates were then incubated at 37°C with 5% CO2 for 24 h. Each well received a 10 µl of MTT solution and then incubated at 37°C with 5% CO2 for another 4 h. After that, the media was carefully removed, and 100 µl of a dimethyl sulfoxide solubilization solution was added to each well, followed by a 5-min incubation. Finally, the absorbance (or optical density) was measured using an ELISA reader at 575 nm. The measurement of the formazan product is done according to the International Standard (ISO-10993). The effect was calculated according to the following formula:
Statistical analysis
The antimicrobial activity was evaluated descriptively. Graph Pad Prism was used to statistically analyze the data. The results of three experiments presented as mean standard deviation (SD) indicating that the difference is statistically significant when p value is less than 0.05. A one-way analysis of variance (ANOVA) was performed to analyze the consistency between the data obtained from the MTT test to determine the influence of DPET blend on HDFn cell viability using Graph Pad Prism (software version 8). Tukey’s post hoc test was used to determine statistically the significant differences between the experimental and control groups.
Results and discussion
Results of PMMA/DPET blends
Physical tests
Compressive strength plays an important role in the mastication process. The values of strain-stress obtained from compression tests for DPET/PMMA blends are shown in Figure 5. Clearly, the blends showed a relatively similar stiffness (i.e., the initial slope of the strain-stress curve). It is evident that all samples investigated exhibited elastic, plastic deformation, and fracture regions, and the addition of DPET up to 5 wt% to the PMMA enhanced the plastic deformation region before fracture. Interestingly, pure PMMA exhibited a sharp fracture point, whereas all other blends showed gradual failure points. Stress-strain curves of PMMA/DPET blends.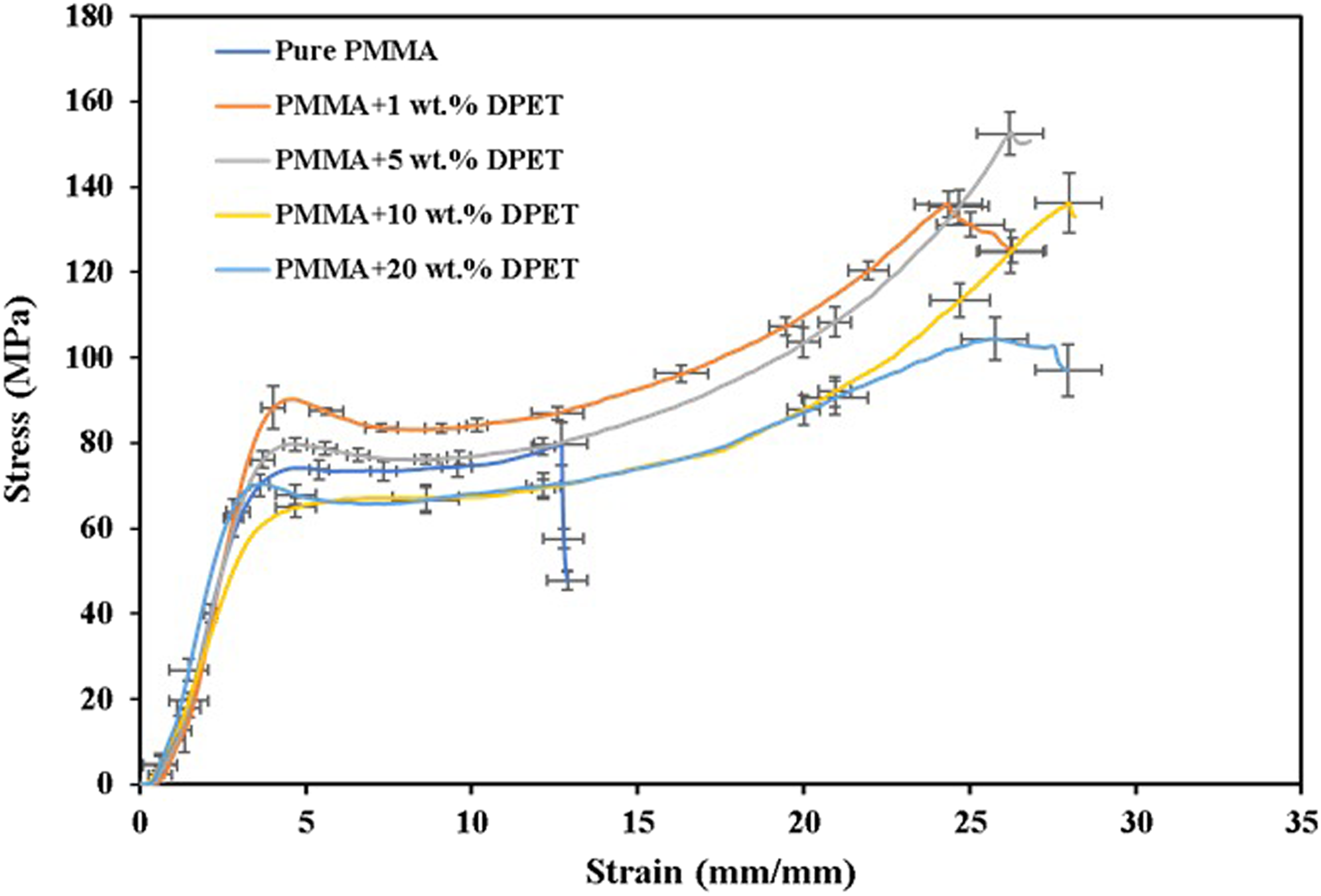
It can also be noted that the compressive strengths of the blends increase as the weight percentage of DPET increases, and this response reflects the nature of the strengthening mechanism that results in a good bonding strength between the blends used (up to 5 wt%). It is also worth mentioning that PMMA: 5 wt% DPET blends exhibit a compressive strength of 153 MPa compared to only 44 MPa for commercial filling-type composite resins (Charm Fill Plus, Korea) and it was very close to the value of enamel parameter (95–140 MPa). 26 However, blends with a percentage of DPET greater than 5% showed a noticeable reduction in compressive strength. This falling beyond 5% DPET can be attributed to the random molecular structure of blended materials as a result of unsymmetry in the structures of side groups. Therefore, it produces a completely amorphous structure with a high free volume, resulting in samples of polymer blends with low compressive strength values.
Water absorption testing for polymer blends is mostly used to figure out how much water a material absorbs and, in cases where the relationship between moisture and mechanical properties, dimensions, or appearance has been established, to figure out how these properties change when exposed to water or humid conditions. Figure 6 illustrates the relationship between the values of water absorption for the polymer blends and DPET content. It was found that the addition of DPET to PMMA leads to increased water uptake, and this behavior is expected due to the hydrogen bonds with the hydroxyl groups of DPET resulting water molecules. It was also noted that blends based on PMMA and 20 wt% of DPET exhibit the highest value of water absorption. Interestingly, the addition of DPET up to 5 wt % causes no significant change in water absorbed by the resulted blends. Therefore, no serious deterioration of the mechanical properties is expected. Water absorption values versus DPET weight percentages.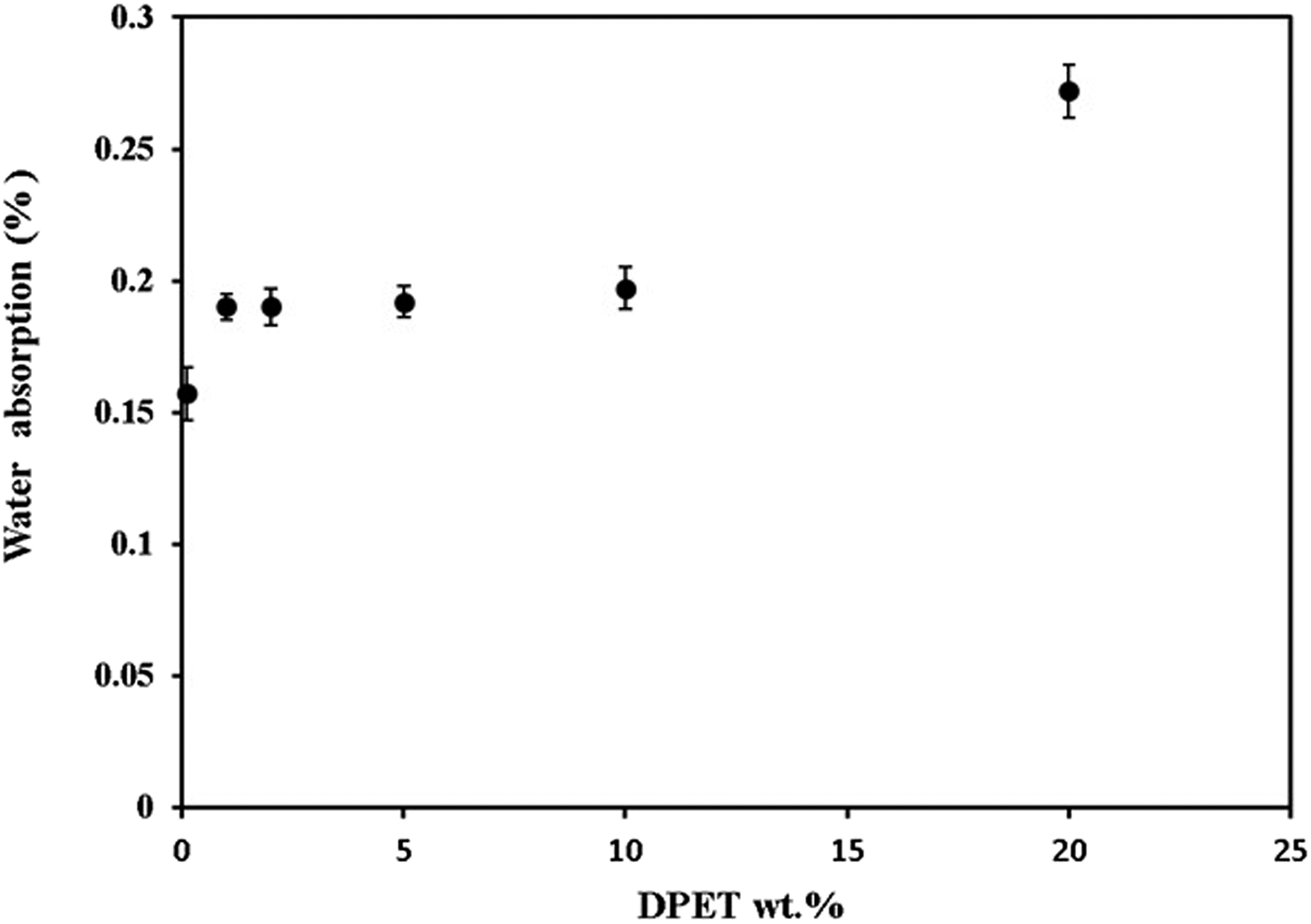
Biological tests
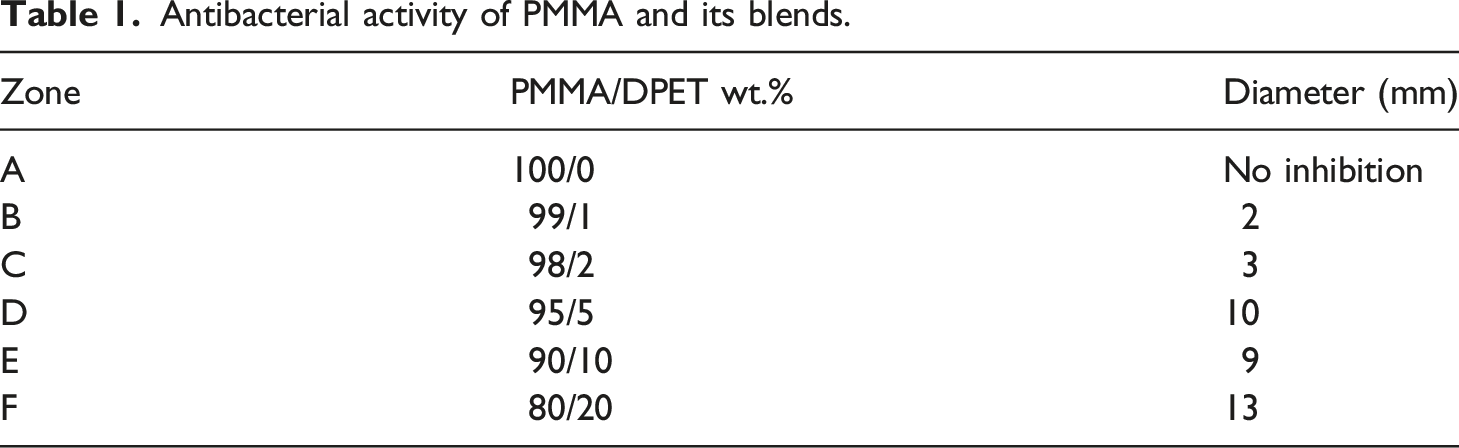
The presence of harmful bacteria in the mouth may cause far-reaching consequences for the general health of people. Therefore, minimizing the burden of bacteria in the oral cavity is one of the most important biological objectives for preventing oral diseases. The inhibition zones for the material tested against Streptococcus mutans bacteria are shown in Figure 7, and the results of the tests are presented in Table 1 and Figure 8. According to the results, the addition of DPET leads to get very effective antibacterial blends against S. mutans bacteria. It is also clear that materials with a high antibacterial activity have significantly large inhibitory zones. Even though blends with 20 wt% of DPET showed the highest antimicrobial activity, its response against water and compression loading was not promising. Inhibition zones for PMMA/DPET blends against Streptococcus mutans bacteria. Antibacterial activity of PMMA and its blends. Graph showing the diameter of the zone of inhibition (in mm) for pure PMMA and its blend against S. mutans.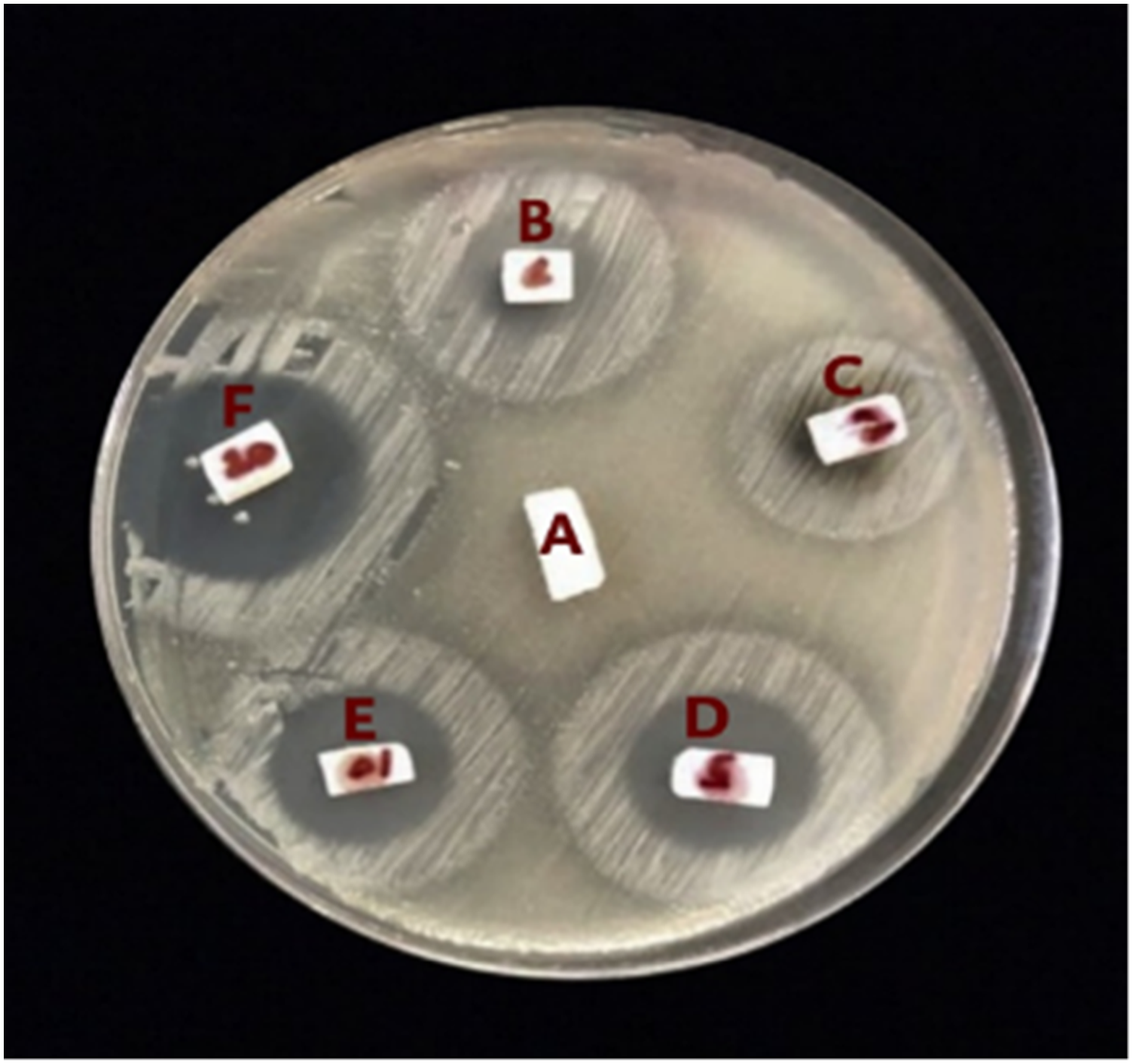
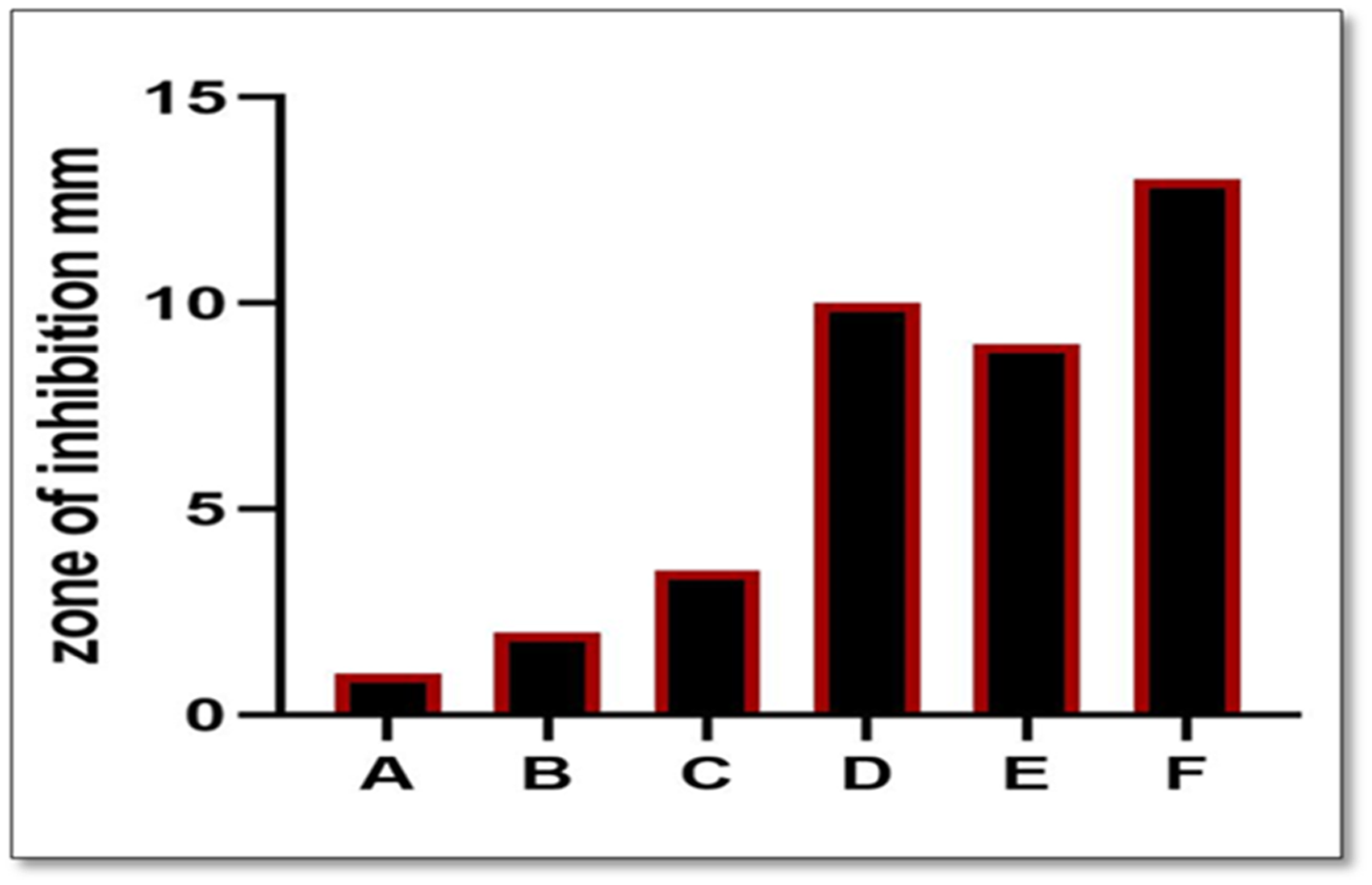
MTT cell viability (%) responses for all groups at the three immersion time periods.
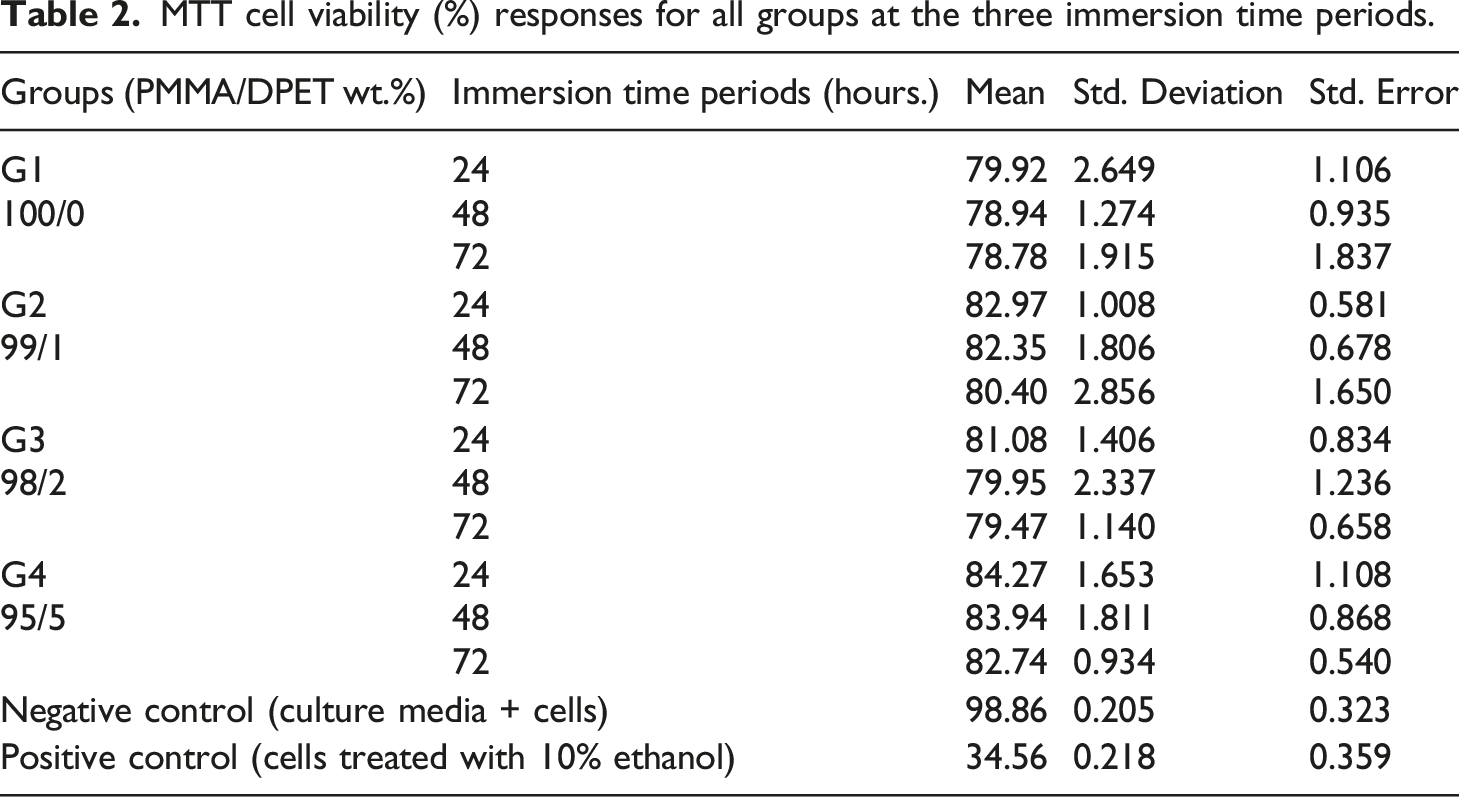
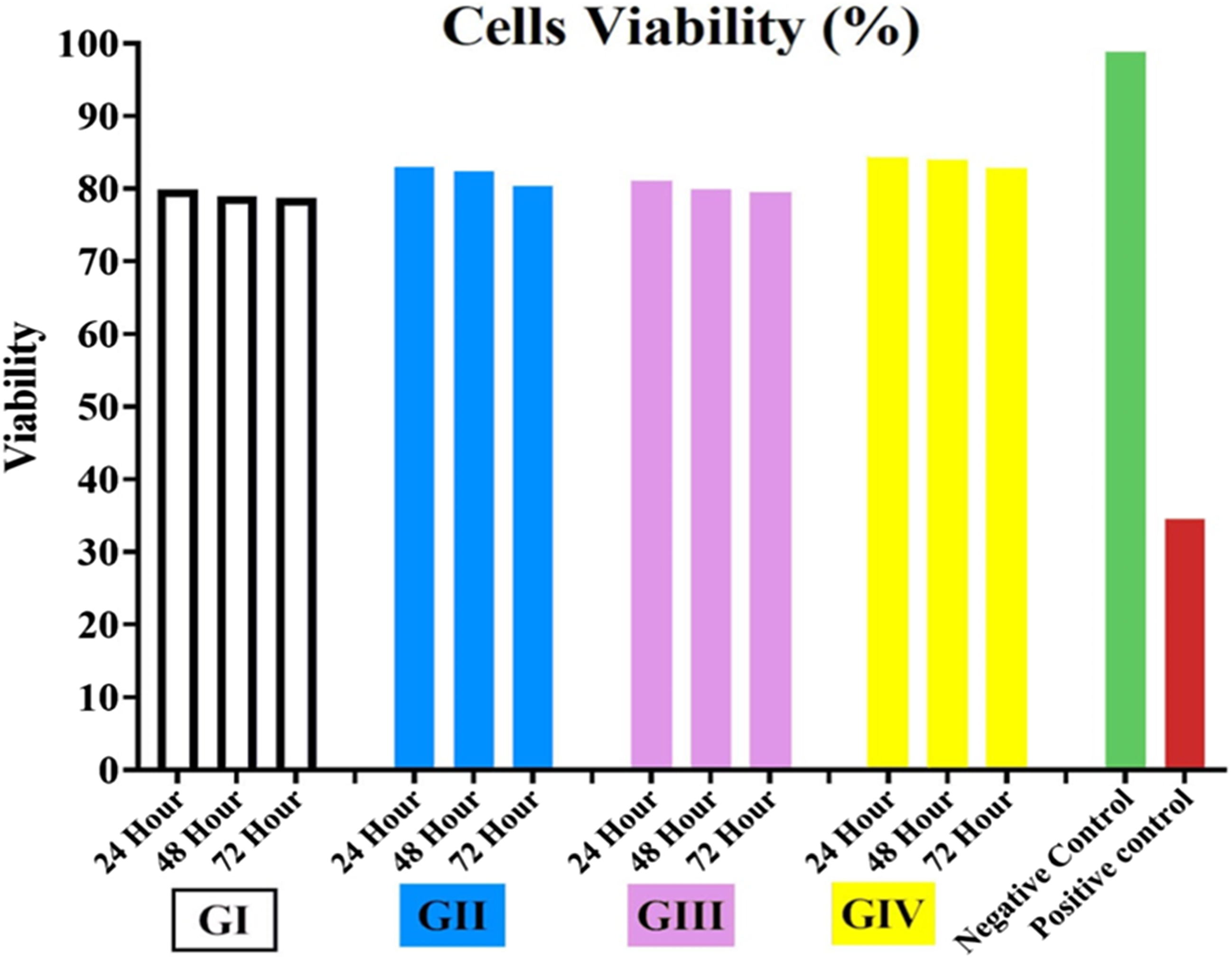
Graph of (%) cell viability over time for PMMA and its groups.
One-way ANOVA (Tukey’s test) for determining the significance of differences in the mean cell viability across the three immersion times for each group.

NS = not significant at p > .05.
Surface morphology
The morphology of the fracture surface can give more details about the compatibility of the blended polymers. Here, the fracture surface of the blends based on PMMA with 5 wt% of DPET was investigated using the SEM imaging technique. The SEM images of the fracture surface at two magnification values are shown in Figure 10. The SEM images showed homogeneous morphological features for the blend investigated without phase separation, highlighting the high compatibility between the PMMA and DPET polymers. SEM images of the 95/5 blend at 7000× and 10,000× magnifications.
Mechanical properties of nanocomposites
Based on the results for the blends investigated, the PMMA: 5 wt% DPET blend was selected as an optimum blend. This blend was then reinforced with various volume fractions of nanoTiO2, and these being 3, 6, and 12 vol%. In dentistry, treating tooth decay requires using conservative methods and materials that provide enough support for the teeth's chewing function. The compressive strength, as a performance criterion, is used to assess the resistance of the materials against chewing forces. The influence of nano TiO2, on the compressive strength of PMMA: 5 wt% DPET blend is illustrated in Figure 11. It is clear that blends reinforced with 12 wt% of TiO2 showed the highest compressive strength. Compression strength values of 95/5 blends with different percentages of nano-TiO2.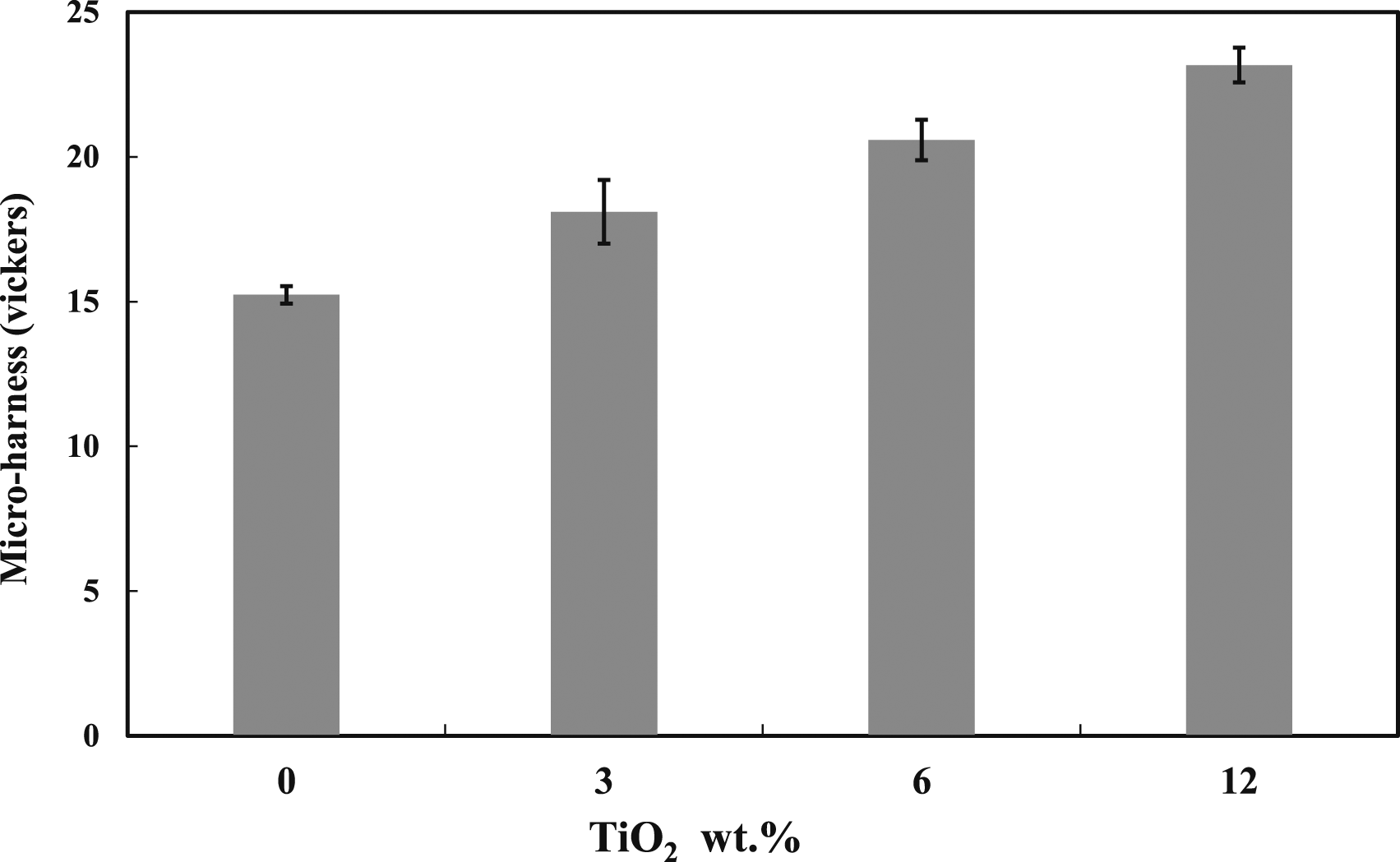
Here, the Vickers micro-hardness tests were used to assess the influence of TiO2 addition on the ability of the polymer blend investigated to withstand load penetration, and the results are shown in Figure 12. It is clear from the figure that, for the range of reinforcement used, the addition of TiO2 nanoparticles to the PMMA: 5 wt% DPET blend has a significant effect on its hardness values as it increases as the TiO2 content increases. For example, the hardness values were increased by 19%, 35%, and 52% with the addition of TiO2 at 3 vol%, 6 vol%, and 12 vol%, respectively. This can be attributed to TiO2 particles acting as rigid bodies in the matrix, resulting in an increase in the stiffness of the composites, a reduction in its mobility, and its free volume, without affecting the composite’s elemental or structural properties. Similar observations were mentioned by other authors.
28
Microhardness results of 95/5 blends with different percentages of TiO2.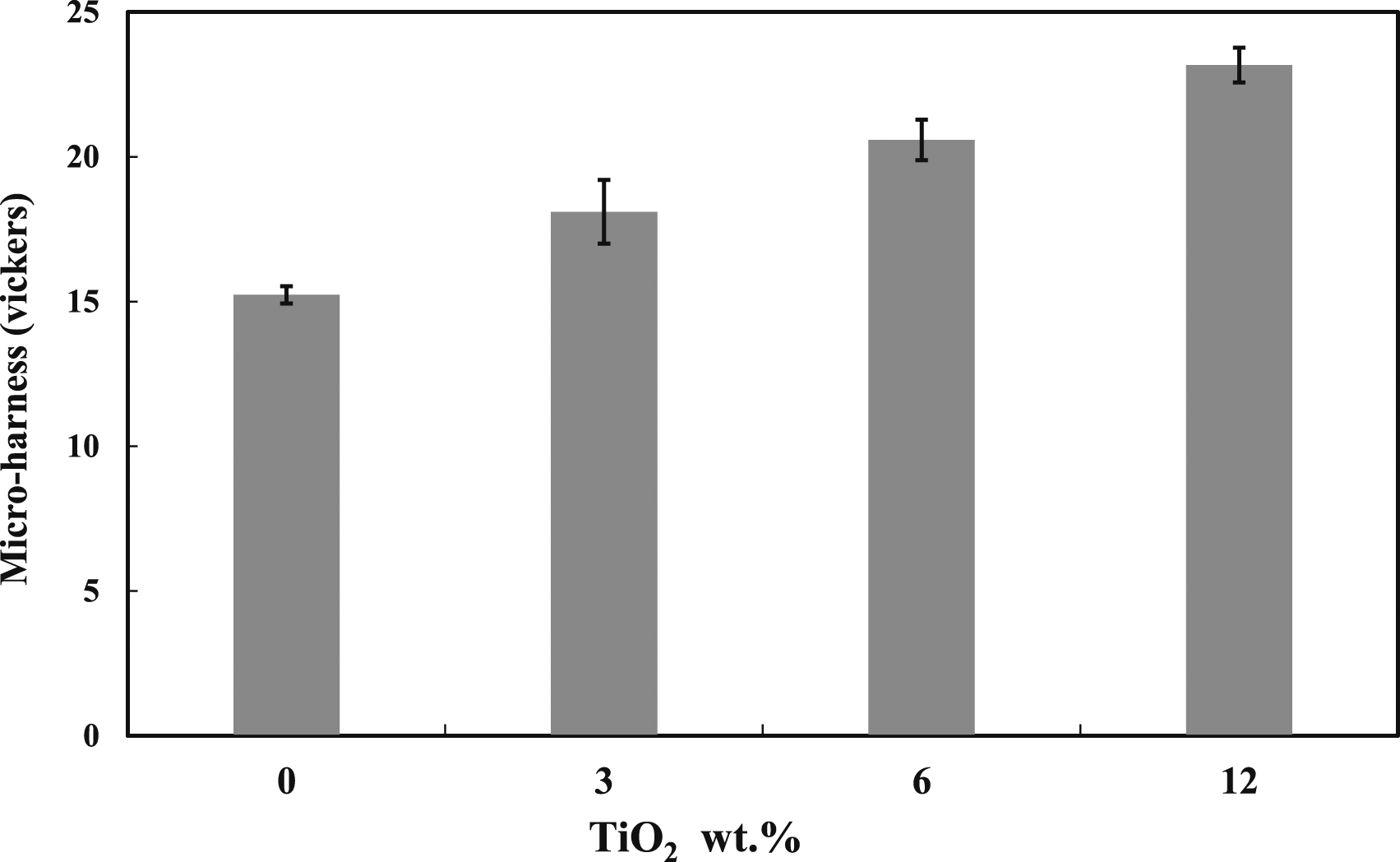
Impact properties are important when designing new dental fillings and biomaterials. Therefore, Izod impact testing was used to figure out how adding TiO2 affects the toughness of the blend investigated, and the results are shown in Figure 13. The results indicated that the energy absorbed during impact before fracture (i.e., toughness) increases as the TiO2 content increases, and the blends reinforced with 6 vol% of TiO2 exhibited the highest impact resistance at a value of 0.6 kJ/m2. However, there was a significant drop in the fracture toughness beyond this value (6 vol%). This can be attributed to the disability of the nanofillers at high concentrations to support transferring the applied stress to the polymer matrix, resulting a weak filler-polymer interfacial adhesion and creating potential sites (i.e., voids), which serve as a crack initiation.
29
The significant properties of the blends and their composites are shown in Table 4. Impact strength of PMMA: 5 wt% DPET blends with various percentages of TiO2. Characteristics of the dental composite.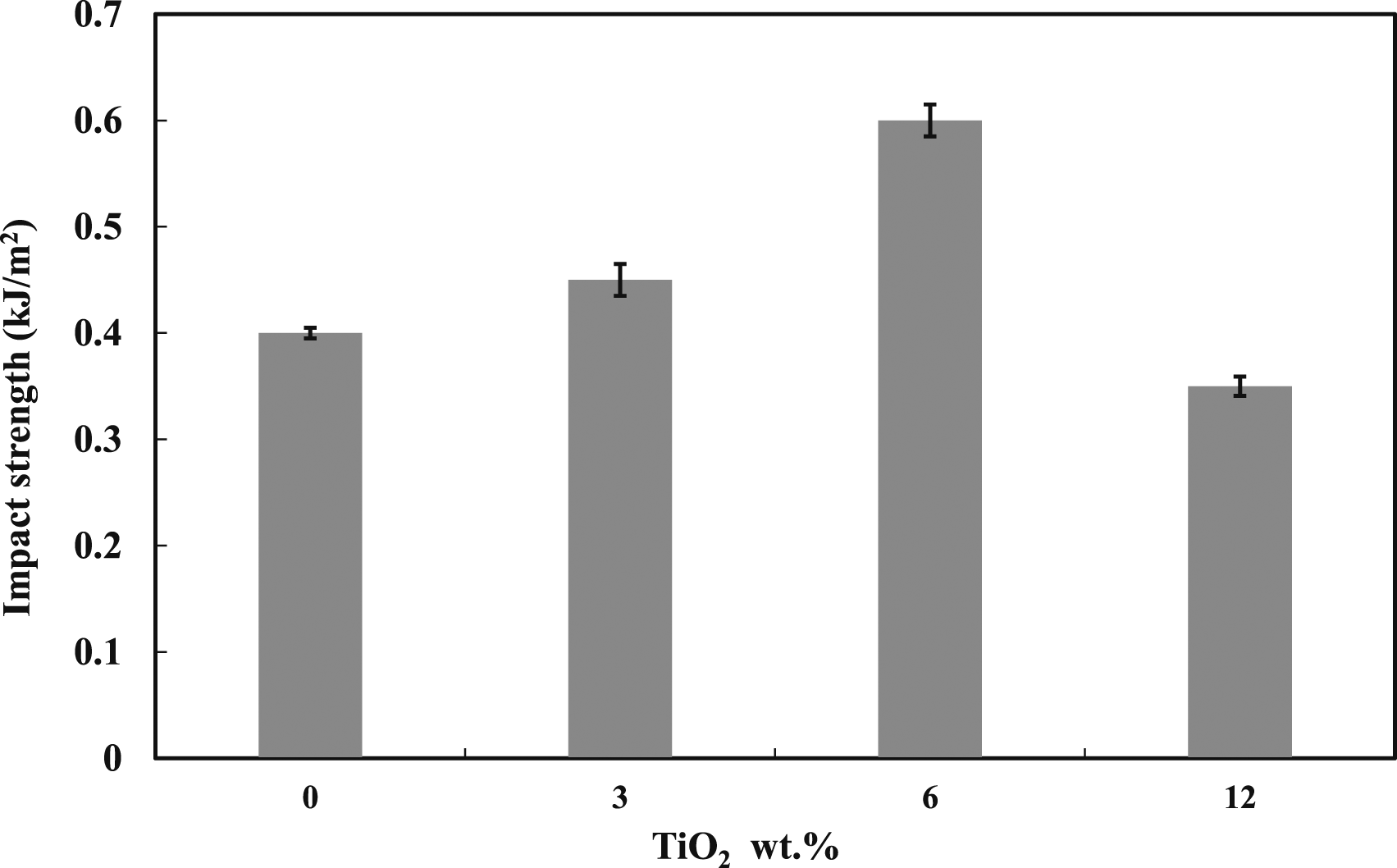

Conclusions
Initially, DPET powder was prepared by recycling the wasted drinking water bottles to preserve the environment and provide a great solution to waste plastic issues. The resulting powder was then blended with PMMA polymer at various weigh percentages. The mechanical and biological tests for the PMMA/DPET blends have been investigated. Based on the obtained results, the optimum blends were then reinforced with different weight percentages of nano-TiO2. It was shown DPET has a significant effect on the mechanical properties of PMMA. For example, PMMA blended with 5 wt% of DPET has a compressive strength value of 153 MPa which is far higher than the standard observed in resin-based dental composites. Furthermore, it is substantially closer to the compressive strength of enamel, which is unquestionably a significant achievement. Such positive findings demonstrate the potential utility of DPET as a binder in dental applications. The obtained results also showed DPET-based blends have the potential to effectively defend against microorganisms. The in vitro MTT assay results highlighted the good compatibility of blends used with cells as well as it helps cell growth and activity without toxicity. It was also shown that the addition of titanium dioxide nanoparticles to PMMA/DPET blends improve the mechanical properties of the resulted composites comparing to the neat blends. In conclusion, using DPET with PMMA gives a promising material as a high-performance dental filling that compete the commercial fillings. In conclusion, further studies on the shrinkage of dental filling over time and its resistance to wear and breakage should be considered to establish the potential of these composites in dental fillings.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.