Abstract
Polyaniline (PANI) is one of the most important conducting polymer and has been widely used in various fields for its tunable electrical conductivity. In this manuscript, conductive polyaniline/ramie fabrics were developed by in situ polymerization and characterized. The effect of aniline content, polyetherimide (PEI) pretreatment, padding and the color depth (K/S value) on electrical resistivity was analyzed. The result showed that electrical conductivity was increased with the increase of aniline (ANI) contents. PEI pretreatment and padding method improved the conductivity of the fabric significantly. The result of K/S value was reversely consistent with the conductivity. Both scanning electron microscope (SEM) and Fourier transform infrared spectroscopy (FTIR) analysis confirmed that polyaniline particles were successfully deposited on the surface of the ramie fabrics. Thermal degradation (TG) measurement showed that polyaniline (PANI) polymerization decreased the onset decomposition temperature. Mechanical measurement showed that pretreatment process reduced the tensile strength of conductive ramie fabrics by 30%. K/S value in full range showed that the color of PANI/ramie fabric was sensible to pH value. Contact angle (CA) measurement disclosed the conductive PANI/ramie fabric was hydrophobic and this hydrophobicity was stable to the change of pH value. The obtained PANI/ramie fabrics can be used as potential conductive multifunctional textiles.
Introduction
Since the first intrinsically conducting polymer was discovered, numerous studies have been published considering the production methods and possible applications of conducting polymers.1-3 Polyacetylene, polypyrrole, polyaniline and polythiophene are the most common intrinsically conductive polymers applied in wearable textiles due to their biocompatibility, easier surface modification, and large surface area. 4 Among them, polyaniline has drawn enormous attention from researchers and industry people because of its low cost, simple fabrication, high conductivity and better chemical stability compared to other conductive polymers.5,6 Polyaniline has been widely used in solar cells, 7 chemical sensors, 8 electromechanical devices, 9 electromagnetic shielding devices, 10 filtration 11 and antistatic materials 12 due to its electrochemical activity and electrochromic properties. 13 Polyaniline (in its emeraldine base with the same number of oxidation units and reduction units) becomes electrically conductive after doping with protonic acid. Generally, this unique doping mechanism makes the doping and de-doping of polyaniline reversible.14,15
In 1992, polyaniline conductive fibers were fabricated by melt blending for the first time. 3 However, the polyaniline composite materials prepared by melt/solution blending methods have many problems, including high stiffness, difficulty in formation, poor strength, brittleness and high requirements for solvents and spinning equipment.16,17 On the other hand, conductive polyaniline has been deposited on fibers and fabrics to develop functional textiles, by simple in situ polymerization process. This in situ polymerization method can maximize polyaniline deposition on the surface of the matrix and this method has been successfully applied on different textile substrates, such as cotton, 18 polyester, 19 nylon, 20 silk, 21 wool, 22 and acrylics, 23 to develop multifunctional textiles based on electrical conductivity. However, few papers report improving electrical conductivity of ramie fabric by depositing PANI. Actually, ramie, also named China grass, is an environmentally friendly natural fiber, is also promising materials as a template for the preparation of multifunctional textiles due to its outstanding mechanical strength, good thermal stability and relative chemical stability.24,25
Conducting polymer layers can be deposited on fabric surfaces chemically or electrochemically.26-28 Chemical deposition is one of the most commonly used methods because of its bulk production, and negligible influence on the strength and comfort of the fabric. 29 In chemical synthesis method, aniline monomer is oxidized to dope polyaniline by strong oxidant in acidic medium. Oxidant type, pH value of the medium and reaction temperature have great influence on the structure and properties of the obtained polyaniline. 30
In the present work, ramie fabric was used as base substrate due to its high mechanical strength and chemical stability. Conductive PANI/ramie fabrics was developed by in situ polymerization. The effects of aniline (ANI) content, PEI pretreatment and mechanical padding on surface resistivity and color depth (K/S value) were discussed. The structure and properties of PANI/ramie were also characterized.
Experimental
Materials
Scoured and bleached plain-weave 100% ramie fabrics (236 warp yarns per 5 cm, 188 weft yarns per 5 cm; fabric surface density 70g/m2) were obtained commercially. Aniline monomer and ammonium persulphate (APS)((NH4)2S2O8) were purchased from SCR chemical Co., Ltd., sodium hydroxide (NaOH), hydrochloric acid (HCl) and polyetherimide ((CH2CH2NH)n) were purchased from Aladdin Chemistry.
Preparation of ramie/polyaniline fabric
PANI/ramie fabrics were prepared using in situ chemical polymerization. 12.5 ml aqueous solution containing 0.23 g HCl and 0.28 g aniline was prepared. 0.46 g ramie fabric was immersed in each aniline/hydrochloric acid solution. 12.5 ml aqueous solution containing 0.23 g HCl and 0.58 g APS was slowly and uniformly dripped into each aniline/hydrochloric acid solution, and the solution was shaken evenly. This fabric was immersed in this solution for 48 h, and the ramie fabric was taken out. Then this coated ramie fabric was rinsed with cold water for 10 min and dried in oven at 65°C 30 min. The produced PANI coated ramie fabric was labeled as “PANI/ramie.” In addition, to investigate the effect of aniline contents on electrical conductivity and K/S value, various aniline content (0.14 g, 0.18 g, 0.23 g) was selected.
In second method, firstly each 0.46 g ramie fabric was immersed in a PEI solution (3 wt%) with bath ratio 1:50 at 65°C for 1.5 h. Then the fabric was taken out and dried in the air. After PEI pretreatment, the process of PANI polymerization was same to “PANI/ramie” fabric. To evaluate the effect of mechanical padding, some ramie fabrics were pressed by padding roller keeping about 80% liquid pick-up before final drying. Finally, samples were rinsed with cold water for 10 minutes and dried in oven at 65°C for 30 min. The resultant PANI coated two ramie fabric were labeled as “PANI/ramie/PEI” and “PANI/ramie/PEI/padding” respectively.
Characterization
The morphology of ramie and PANI/ramie fabrics was characterized using scanning electron microscope (JSM5600LV, JEOL Ltd., Japan) with an acceleration voltage of 30 kV. Before SEM observation, samples were first glued into a conducting resin and then coated with Pt layers with thickness of 10 nm under vacuum to prevent charging during imaging.
Fourier transform infrared (FT-IR) spectra of ramie and PANI/ramie fabrics were obtained on Avtar360 (TENSOR 27X, Bruker, German). FTIR spectra were recorded in the range of 600–4000 cm−1 by 32 scans with 4 cm−1 resolution.
Thermal degradation behavior of ramie and PANI/ramie fabrics was evaluated by thermal gravimetric (TG, NETZSCH STA409PC, German). 5 mg sample was heated from room temperature to 600°C at a rate of 10°C/min under nitrogen atmosphere to yield the onset temperature of decomposition, mass loss and maximum decomposition peak.
The electrical conductivity of PANI/ramie fabrics was measured at room temperature by using a MF30 universal resistance tester (China). Five different locations were selected for measuring the electric resistance and the value was taken as the conductivity of this fabric.
Apparent color depth (K/S) of PANI/ramie fabrics was determined by a color i7 spectrophotometer (X-Rite, USA), using the CIElab color space at D65/10°.
The contact angle (CA) of ramie and PANI/ramie fabric was measured by contact angle meter (SL200C, Shanghai Shenrui Instrument Co., Ltd., China) with stationary droplet method. The CA of PANI/ramie fabric was investigated by dipping the fabric in baths having pH value 1 and 11 alternatively for five cycles. Firstly, PANI/ramie fabric was dipped inside the bath having pH 1 for 2 min. After drying and contact angle measurement, the fabric was dipped inside the bath having pH 11 for 2 min. After contact angle measurement, the fabric sample was again dipped inside the bath having pH 1 for the second cycle.
The breaking strength of untreated and the treated ramie fabrics was determined by a dynamometer (Instron 5566, USA) at ambient temperature. The size of the samples was 50 mm × 20 mm. At least five samples for each fabric were tested and the average value was recorded for analysis.
Results and discussion
SEM observation
Scanning electron micrographs of control ramie, PANI/ramie, PANI/PEI treated ramie and PANI/PEI treated ramie with padding are shown in Figure 1 respectively. Figure 1(a-1) shows the micrograph of the control ramie fabric. There are some parallel ridges and grooves in fiber surface and the unevenness of these ridges and grooves can be seen in the magnified image of the fiber morphology (Figure 1(a-1) and (a-2)). It can be clearly seen from the image of the “PANI/ramie” fabric (Figure 1(b-1)) that PANI is successfully deposited on the ramie fabric surface, and the magnified image (Figure 1(b-2)) indicates most PANI is in a particular shape. It can be seen from the images of “PANI/ramie/PEI” fabric (Figure 1(c-1)) that PANI is densely and unevenly deposited on the fiber surface and the magnified image (Figure 1(c-2)) shows some PANI is in cluster form, which indicates PEI pretreatment is benefit to enhance the polymerization of aniline on ramie. PANI deposition on ramie surface can also be obviously seen from the image of “PANI/ramie/PEI/padding” fabric (Figure 1(d-1) and (d-2)), and the magnified morphology of fiber surface reveals that coating density and continuity of PANI along ramie fiber length are increased further. It may be considered that padding treatment can change the state of aniline on ramie surface and thus the PANI deposition.

SEM images of (a) control ramie fabric, (b) PANI/ramie fabric, (c) PANI/ramie /PEI fabric, (d) PANI/ramie/PEI/padding fabric.
FTIR analysis
Figure 2 shows the FTIR spectra of ramie fabric and PANI/ramie fabrics.
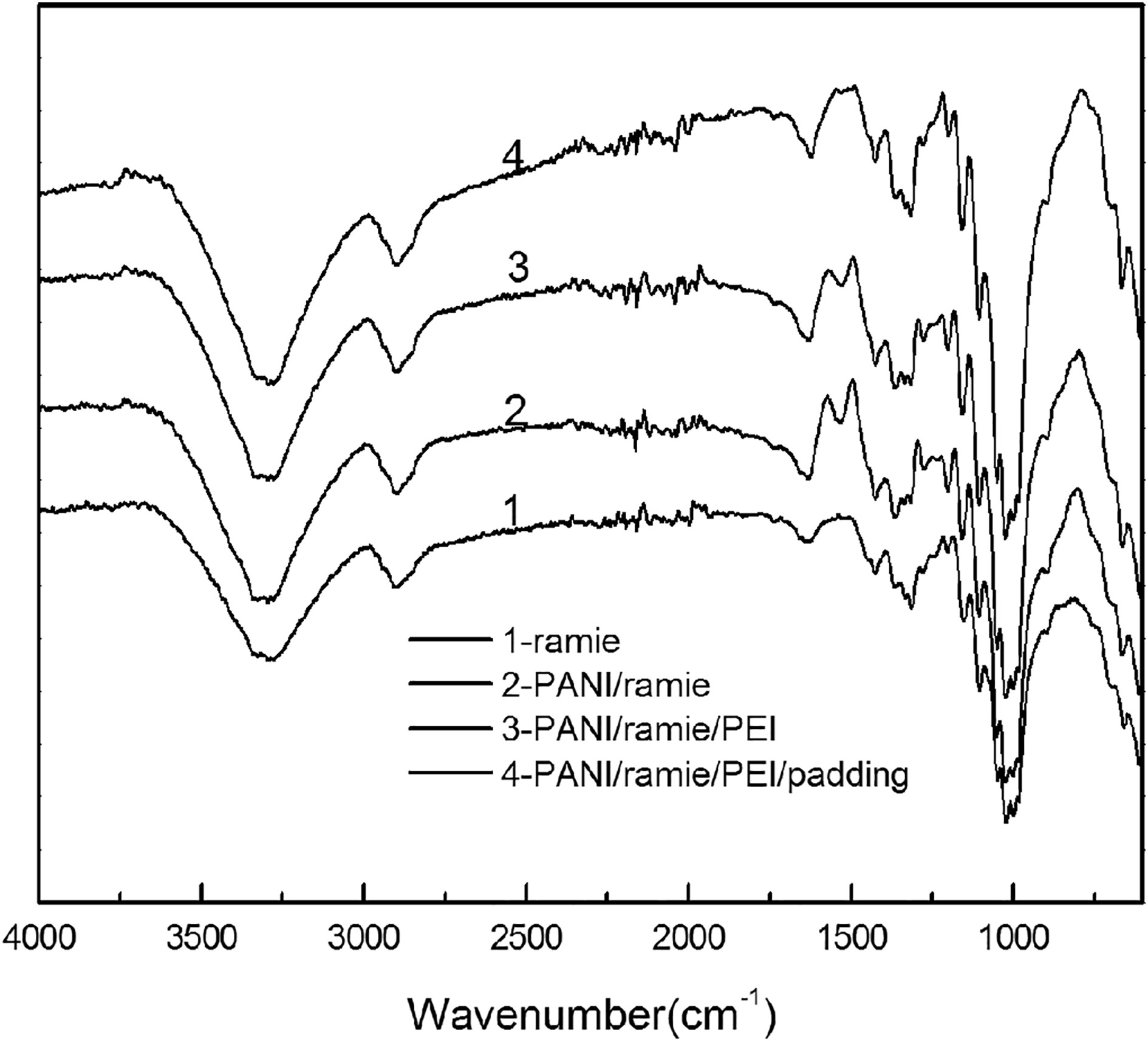
FTIR spectra of ramie fabric and PANI/ramie fabrics: (1) ramie fabric (2) PANI/ramie fabric (3) PANI/ramie/PEI fabric (4) PANI/ramie/PEI/padding fabric.
The bands centered at 3300, 1634, 1428, 1336, 1155 and 1051 cm−1 in Figure 2(1) correspond with the cellulose structure of ramie fabric, attributing to the stretching vibration of O–H in cellulose, H–O–H bending of the absorbed water, the bending vibration of C–H2 in cellulose, the O–H bending of CH2–OH group in cellulose and the skeletal vibration of C–O–C pyranose ring respectively. 18
The FTIR spectra of PANI/ramie (Figure 2(2) to (4)) showed a hump at 1540 cm−1 and 1456 cm−1, which is attributed to the stretching of quinoid ring N=Q=N and the stretching of benzene ring respectively. 31 The band centered at 1278 cm−1 is characteristic of the stretching vibration of C–N bonds of PANI in ramie fabric. The peak at 1234 cm−1 is attributing to the stretching vibration of the polaron structure of the PANI, which is the conducting protonated form of C–N. 32 A clear band centered at 1155 cm−1 in ramie fabric (Figure 2(1)) is attributed to the antisymmetric bridge stretching of C–O presence in the cellulose structure. 33 In Figure 2(2) to (4), the peaks at 1159 cm−1 can be assigned to the vibration mode of the –NH+= structure which is related to the protonation of PANI on the ramie fabric and considered to be a measurement of the degree of delocalization of electrons.34,35 The peaks at 810 cm−1 related to the vibration mode of 1,4-substituted benzene rings also can be observed in the FTIR spectra of PANI/ramie(Figure 2(2) to (4)). However, there are no clear difference between Figure 2(2) and (3), which may be due to the fact that the spectral characteristics of PEI are obscured by PANI.
For PANI/ramie composite fabrics, FTIR spectra combine the characteristics of PANI and ramie, and most of their characteristic peaks can be clearly observed. However, there are a few red or blue shifts in some peaks, and the shape of the peaks is widened to some extent, which indicates that there is a bond between PANI and ramie fiber. The spectral characteristics of PANI/ramie composite fabrics mentioned above indicate that during in situ polymerization, molecular bonds was formed between PANI and ramie in addition to the polymerization of aniline monomers. Hydroxyl and glycoside bonds in cellulose can interact with imino-nitrogen in polyaniline molecule by doping or hydrogen bonding, which makes the bond between polyaniline and ramie fiber stronger.
Electrical conductivity
Figure 3 shows the surface electrical resistance of ramie fabric treated with different methods. From the figure, it is seen that, the trend of electrical resistance of three fabrics is monotonically decreasing with the increase of aniline monomer content in the solution. Aniline monomers in fabrics are initiated in the oxidation solution where sufficient ammonium persulphate is available, which is conducive to the formation of higher molecular weight polyaniline. The growth of polyaniline is an autocatalytic reaction. 36 In other words, aniline radicals can become new cationic radicals based on the highest oxidized state of polyaniline through electron transfer, and the new cationic radicals are oxidized on the surface of PANI polymer to reach the highest oxidized state and finally PANI polymer deposition is achieved. Therefore, high aniline content is conducive to the formation of polymers. The formation of conductive PANI is guaranteed due to the presence of doped acids in the oxidation solution.

The effect of aniline monomer on conductivity.
Treatment methods also have obvious influences on conductivity. Figure 3 shows PEI pretreatment significantly reduces electrical resistance. PEI, a water-soluble polymer, is alkaline due to its repeating unit CH2 CH2 NH and has the ability to absorb protons in a wide range of pH, namely “proton sponge.” Because of the multiple functions of PEI, more polyaniline can deposit on the surface of PEI treated ramie fabric, thus reduce the surface resistance of ramie fabric. In addition, it can be seen that padding can significantly improve the conductivity, due to mechanical extrusion that can make more aniline filling the gap between warp and weft yarns. Therefore the polyaniline deposits on the fabric surface densely and more uniformly.
K/S value is an important index to characterize the surface color of traditional dyed products. With the increase of oxidation degree, the color of PANI polymer will become darker. The K/S values measured at the maximum absorption wavelength are also shown in the Figure 4. The results of K/S value correspond to the variation of resistance value. The fabric with lower electrical resistance has higher K/S value, thus sometimes the conductivity of PANI/ramie fabrics can be judged by their shade of color.

The effect of aniline monomer on K/S value.
The electrical conductivity of PANI/ramie fabric shows good time durability as shown in Figure 5. After placed in condition room(20°C, 60%RH) for 14d, the electrical resistivity of PANI/ramie fabric without PEI treatment only increases from 16 to 21 MΩ, and that of PANI/ramie fabric with PEI treatment and padding slightly increases from 0.35 to 3.5 MΩ. Thus the results indicate that the time produces slight effect on the stability of electrical conductivity property for PANI coated ramie fabric.

The effect of time on surface resistance at condition room.
TG & DTG analysis
Thermal properties of ramie fabric/PANI were analyzed using TG and DTG. The TG and DTG curves are shown in Figure 6. According to TG curves (Figure 6(a)) all the samples show a minor endothermic peak in the temperature range of 51–60°C (Figure 6(a)), which is owing to the loss of the moisture present in the samples. 80–90% weight loss occurs during the second stage at 200°C–400°C (Figure 6(a)), which is due to the decomposition of the cellulose structure of ramie. Pristine ramie fabric shows an endothermic peak at 373°C, while PANI/ramie fabric without PEI treatment shows an endothermic peak at 363°C (Figure 6(b)), indicating a slight decrease of the thermal stability, due to the damage of the cellulose structure of ramie in the presence of acid. For PEI treated fabric the endothermic peaks have shifted to much lower temperature 332°C. This may be due to the degradation aggravation of the cellulose fiber in the presence of PEI and acid. For all samples a residual weight is observed after 400°C. Certainly PANI/ramie fabric has more residual weight than pristine ramie fabric since the presence of nitrogen containing carbon of PANI.

TG and DTG analysis of ramie and PANI/ramie fabric: (a) TG, (b) DTG.
The effects of pH value on water contact angle and K/S value
PANI/ramie fabric shows a relative stable surface behavior when immersed in solutions having different pH values. As Figure 7 shows, the contact angle of PANI/ramie fabric varies from 100° to 112° and remains almost constant when dipped in a solution between pH 1 and pH 13. It indicates that PANI/ramie fabric has some hydrophobicity and the infiltration property is stable to acid-base environment.

Effect pH value on contact angle of PANI/ramie fabric.
The contact angle results are presented in Figure 8. It is observed that although there are some fluctuations, the change of contact angle is not significant. The fluctuations of contact angle may be due to the unevenness of PANI decomposition and the degree of protonation of PANI changes. Ramie fibers have special structural cracks. After in situ polymerization, PANI can be compactly coated in the cracks of the fibers, which also increases the difficulty of complete de-protonation of the PANI structures on the fabric. Therefore, PANI/ramie fabric presents a certain alkali resistance and can adapt to different acid-base environment.

Cyclic wettability behavior of PANI/ramie fabric after immersing in baths having pH 1 and pH11.
When the PANI/ramie fabrics are immersed in solutions having different pH values, the fabrics present different perceptible colors, which indicate PANI/ramie fabrics have pH responsiveness with color recognition. The effect of pH value on K/S value in full range is presented in Figure 9. It can be seen that with the increase of pH value, the maximum absorption wavelength of fabrics shows obvious blue shift. When pH value is 3∼7, the fabric is blue-green, while when pH value is 9∼11, the fabric is blue-black. The color of PANI/ramie fabric is pH-responsive, which is caused mainly by the change of resistivity of doped PANI with the change of pH value. This property can be used to detect the change of acidity in the environment.

Effect of pH value on K/S value of PANI/ramie fabric.
Mechanical strength analysis
The breaking strength test results are shown in Figure 10. It is seen that breaking strength is decreased by 10% after PEI treatment and 30% after further PANI coating. The reduction in breaking strength might be due to the reduction of flexibility caused by coating. 37 Since ramie is cellulose fiber, the contact with oxidant and HCl during PANI polymerization also led to the loss of breaking strength. However, the retained strength may meet the requirements of conductive fabrics.

Breaking strength of ramie and PANI/ramie fabric.
Conclusion
In this work PANI/ramie fabrics were fabricated using simple in situ chemical polymerization of aniline on ramie. Combing PEI pretreatment and padding process with in situ polymerization, the electrical conductivity of PANI/ramie fabric was improved. SEM images showed PANI was deposited on ramie fiber surface in particular or cluster form. FTIR analysis showed chemical interaction of PANI with ramie fabric and this interaction was formed in the cellulose structure. According to the TG and DTG results, the thermal stability of ramie fabric decreased slightly after PANI polymerization. Strength retention of PANI/ramie fabric was 70%. K/S value in full wave-band showed the color of the PANI/ramie fabric was sensible to pH value changing. While contact angle results showed surface hydrophobicity of PANI/ramie fabrics was stable to acid-base environment. In situ oxidant polymerization would be a promising method to prepare PANI/ramie fabrics and these fabrics can be used as potential conductive multifunctional textiles.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
