Abstract
A novel phosphorus-silicon containing flame-retardant DOPO-V-PA was used to wrap carbon nanotubes (CNTs). The results of FTIR, XPS, TEM and TGA measurements exhibited that DOPO-V-PA has been successfully grafted onto the surfaces of CNTs, and the CNTs-DOPO-V-PA was obtained. The CNTs-DOPO-V-PA was subsequently incorporated into epoxy resin (EP) for improving the flame retardancy and dispersion. Compared with pure EP, the addition of 2 wt% CNTs-DOPO-V-PA into the EP matrix could achieve better flame retardancy of EP nanocomposites, such as a 30.5% reduction in peak heat release rate (PHRR) and 8.1% reduction in total heat release (THR). Furthermore, DMTA results clearly indicated that the dispersion for CNTs-DOPO-V-PA in EP matrix was better than pristine CNTs.
Introduction
Epoxy resin (EP) has been widely used in various applications, such as adhesives, coating, potting, laminates and encapsulation material, due to the attractive adhesion to many substrates, low shrinkage on cure, superior electrical properties, excellent corrosion resistance and mechanical properties.1-3 However, high flammability of EP limits its high-performance applications in many fields for safety consideration.4-10 Thus, it is important to enhance the flame retardancy of EP. Carbon nanotubes (CNTs) have attracted increasing interest for researchers in recent decades and have been used to improve the flame retardancy of polymers.11-13 However, the aggregation of CNTs in the polymeric matrix always deteriorates the physical properties of polymer matrix and decreases the efficiency of flame-retardant. Therefore, the dispersion property becomes more important when CNTs are blended into polymer matrix.14-19
Up to now, much effort has been devoted to improve the dispersion of CNTs through various functionalized methods, among which, covalent functionalization can remarkable improve dispersion of CNTs in solvents and compatibility of CNTs with polymer matrix. Various polymer chains with functional groups have been employed to wrap CNTs to obtain composites with significantly performance and stable dispersions. For instance, Fang’s group 20 prepared functionalized CNTs grafted with an intumescent flame-retardant, which showed better dispersion and flame-retardant effect. Kuan et al. 21 synthesized a modified CNTs with functionalized by vinyltriethoxysilane and an increase in flame retardancy of the fabricated EP nanocomposites was obtained. Recently, cyclotriphosphazene was grafted onto the surface of CNTs, leading to simultaneous improvement of flame retardancy and mechanical properties of EP nanocomposites. 22 Although many functionalized methods have been reported, the high-performance CNTs are still necessary to be further explored at low loading.
In this work, a novel phosphorus-silicon containing flame-retardant DOPO-V-PA was synthesized and wrapped onto the surface of CNTs. The wrapped CNTs (CNTs-DOPO-V-PA) were used to prepare flame-retardant epoxy nanocomposites. The flame-retardant effects of epoxy resins containing CNTs-DOPO-V-PA were investigated and the dispersion of CNTs-DOPO-V-PA in polymer matrix were also analyzed.
Experimental
Material
Concentrated sulphuric acid (98%), nitric acid (HNO3, 65–70%), pyridine (99.5%), tetramethylammonium hydroxide (TMAOH), (3-aminopropyl)trimethoxysilane (APT), 2,2′-azobisisobutyronitrile (AIBN), N, N-dimethylformamide (DMF, 99%) and tetrahydrofuran (THF) were all purchased from Alfa Aesar Chemical Reagent Co. Ltd. 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO) was purchased from TCI Development Co., Ltd. Vinyltrimethoxysilane (VTMS), ethyl alcohol (EtOH) and benzene were reagent grade and purchased from Sigma-Aldrich Co., Ltd. Phenyltrimethoxysilane (PTMS) was reagent grade and purchased from Gelest Chemical Reagent Co. Ltd. Chloroform (CHCl3) and thionyl chloride (SOCl2) were supplied by Fisher Scientific Chemical Co. EPON 826 with an epoxy equivalent weight of 178–186 g was supplied by Hexion and used as received. The hardener, Jeffamine D230, with an amine equivalent weight of 60 g, was supplied by Huntsman Corp. and also used as received. CNTs (outer diameter 10–20 nm, inner diameter 5–10 nm, length 10–30 μm) synthesized by chemical vapor deposition was purchased from Chengdu Organic Chemistry Co. Ltd, Chinese Academy of Science.
Synthesis of DOPO-V-PA
DOPO (21.6 g, 0.1 mol), VTMS (14.8 g, 0.1 mol), and benzene (100 mL) were added into a three-necked flask with a mechanical stirrer, flux condenser, dropping funnel, and nitrogen inlet. After the mixture was saturated with nitrogen atmosphere under vigorous mechanical stirring, the temperature was warmed to 80°C. After the DOPO was dissolved completely, 0.1 g of AIBN which was predissolved in 50 mL of benzene was slowly dropped into the above reaction vessel within 2 h at 80°C and then kept at that temperature for 24 h. After that, the products were purified by filtering. Then benzene was removed by a rotary evaporator, yielding a colorless liquid product named DOPO-V. 23
As shown in Figure 1, DOPO-V-PA was synthesized by hydrolysis and condensation reactions as follows: Distilled water (25 mL), EtOH (75 mL) and TMAOH (1 mL) were mixed in a 250 mL flask under stirring. The mixture of PTMS, APS and DOPO-V at certainly molar ratios (70:10:20) was added to the above solution, maintaining 10% weight percentage. The stirring was stopped after 8 h, and the solution was aged at room temperature overnight. Precipitated condensate was collected by decantation of most clear supernatant, washed by vacuum filtration with distilled H2O/EtOH (1/3 by volume), and then washed again in pure EtOH. The rinsed powder (DOPO-V-PA) was dried thoroughly under vacuum for 20 h at room temperature.

Synthetic route of DOPO-V-PA.
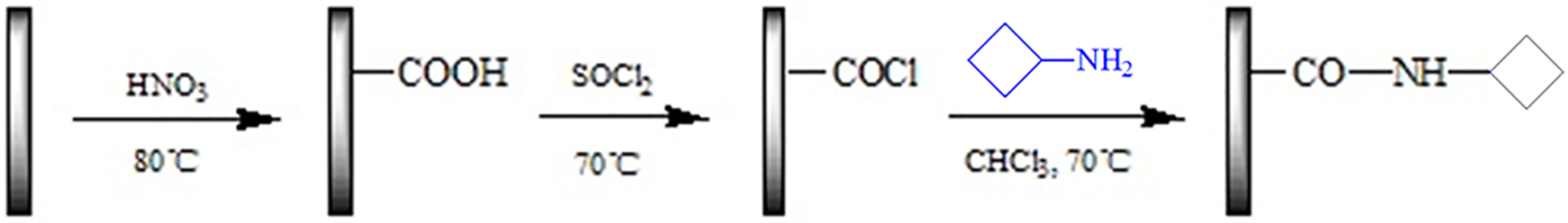
Illustration for the functionalization of the CNTs with DOPO-V-PA.
Functionalization of CNTs
The CNTs-COCl was synthesized as follows: The mixture of CNTs, HNO3 (30 mL) and H2SO4 (90 mL) was sonicated at 50°C for 2 h. After termination of reaction, it was allowed to cool down to room temperature. The mixture was diluted with a large amount of deionized water, followed by a vacuum-filtering through a nylon film (0.22 μm). The obtained solid CNTs-COOH, in which polar carboxyl groups were introduced into the convex surface of CNTs, was washed with deionize water until the aqueous layer reached neutral, and then was vacuum-dried at 80°C for 12 h. The reaction mixture of CNTs-COOH (200 mg), SOCl2 (20 mL) and DMF (1 mL) was sonicated at 50°C for 1 h, and then refluxed at 70°C for 24 h. After that, the temperature was risen to 120°C and CNTs-COCl was obtained after residual SOCl2 was removed by the reduced pressure distillation.
The CNTs-DOPO-V-PA was synthesized as follows: DOPO-V-PA (400 mg) and pyridine (1 mL, as cat.) were added into the suspension of CNTs-COCl (100 mg) and DMF (50 mL) under the protection of nitrogen, and the mixture was reacted at 70°C for 24 h. After cooling to room temperature, the dark solution was filtered and washed to remove unreacted DOPO-V-PA. The target product CNTs-DOPO-V-PA after vacuum-dryness at 80°C for 24 h was obtained.
Preparation of epoxy nanocomposites
Briefly, the EP/CNTs-DOPO-V-PA nanocomposites were prepared as follows: The CNTs-DOPO-V-PA (2 g) was dispersed in acetone (100 mL) and sonicated for 1 h to form a uniform black suspension. Then, EPON 826 (73.5 g) was added into mixture and dispersed by a mechanical stirrer for 30 min.The mixture was heated in a vacuum oven at 50°C for 10 h to remove the solvent. Subsequently, D230 (24.5 g) was added into the mixture and stirring for 30 min. After degassed in vacuum for 10 min to remove any trapped air, the samples were cured at 80°C for 2 h and post cured at 135°C for 2 h. For comparison, pure epoxy (EP) and 2 wt% CNTs/epoxy (EP/CNTs) nanocomposites were also prepared at same processing condition.
Characterization and measurement
FTIR spectra of the dried samples were recorded using a Broker Equinox-55 IR spectrometer at a resolution of 2 cm−1 with 20 scans. The samples were mixed with potassium bromide and pressed to a disc, which was used to measure. X-ray photoelectron spectroscopy (XPS) was carried out in a Thermo Scientific ESCALAB 250Xi X-ray photoelectron spectrometer equipped with a mono-chromatic Al Kα X-ray source (1486.6 eV). The surface morphology of the carbon nanotubes was observed with a JEOL JEM-2100F transmission electron microscope (TEM). Thermogravimetric analysis (TGA) measurement was carried on a TA instrument Q5000 thermogravimetric analyzer. The sample (about 10 mg) was heated from 50°C to 600°C (or 700°C) at a 10°C/min heating ramp rate in nitrogen atmosphere. Cone calorimeter measurement was performed on an FTT cone calorimeter (Britain) according to ASTM E1354 with heat flux of 50 kW/m2. The dimensions of each specimen was 100 × 100 × 3 mm3. The limiting oxygen index (LOI) was measured on an oxygen index instrument JF-3 produced by Jiangning Analysis Instrument Factory and performed according to GB2406-93. Dynamic mechanical thermal analysis (DMTA) was determined using a Rheometric Scientific SR-5000 dynamic mechanical analyzer and the data were collected from 60°C to 130°C at a scanning rate of 5°C/min.
Results and discussion
Structural characterization
In order to compare the suspension stability of CNTs and CNTs-DOPO-V-PA, tetrahydrofuran (THF) was used as solvent to prepare CNTs solutions with concentration of 0.5 mg·mL−1. Figure 3 showed the results of solubility of CNTs and CNTs-DOPO-V-PA in THF after 5 min sonication. For CNTs, THF was a poor solvent and the dispersion of CNTs was totally unstable with a solid precipitate. In contrast, the solubility of CNTs-DOPO-V-PA was siginificantly improved after covalent functionalization of the DOPO-V-PA molecules onto a CNTs surface, and the CNTs-DOPO-V-PA forms a stable dispersion in THF, which makes the solution deeply black. It could be attributed to that the THF was a good solvent for DOPO-V-PA and the group of -NH- in DOPO-V-PA can enhance hydrogen-bonding abilities, which lead to the improved suspension stability compared with the pristine CNTs.

Comparison of solubility of CNTs (a) and CNTs-DOPO-V-PA (b) in THF.
The FTIR spectra of pristine CNTs, CNTs-DOPO-V-PA and DOPO-V-PA were shown in Figure 4. After the covalent functionalization, a sharp peak at 1655 cm−1 corresponding to the stretching vibration of the C=O group of the amide functionality was observed in the CNTs-DOPO-V-PA. The spectra of CNTs-DOPO-V-PA also exhibited the expected –CH2– stretching at 2940 cm−1, which was attributed to the grafter DOPO-V-PA. Moreover, Comparing with the FTIR spectra of CNTs, another new peak appeared at around 1105 cm−1, which was due to the symmetric stretching vibration of Si–O–Si bond.24-25 These results revealed that DOPO-V-PA were grafted to the surface of CNTs.

FTIR spectra of CNTs (a), CNTs-DOPO-V-PA (b) and DOPO-V-PA (c).
The XPS spectra of CNTs and CNTs-DOPO-V-PA were displayed in Figure 5, and the results of element composition were summarized in Table 1. Pristine CNTs only showed a strong C1 s peak at the BE of 285 eV, and a very weak peak at 532 eV assigned to O1s. The existence of oxygen might originate either from air contamination or thermal oxidation during the purification process. In comparison with CNTs, more other photoemission peaks appeared in the XPS spectra of CNTs-DOPO-V-PA, namely at BEs of 102 eV (Si2p), 133 eV (P2p), 153 eV (Si2s) and 400 eV (N1s).26-27 Moreover, the higher surface oxygen content (as high as 10.64 atm%) was obtained in CNTs-DOPO-V-PA. The results indicated from XPS analysis provided powerful proof of the covalent functionalization of CNTs by DOPO-V-PA, which was consistent with the FTIR spectra.

XPS scan spectra of CNTs and CNTs-DOPO-V-PA.
Element composition of CNTs and CNTs-DOPO-V-PA.

Changes in the morphology can further indicate successful grafting of DOPO-V-PA to CNTs, and a comparison of the microstructures of CNTs and CNT-DOPO-V-PA is shown in Figure 6. Figure 6(a) shows a typical TEM image of CNTs with a very smooth and clear surface without any adhering material. However, the CNT-DOPO-V-PA shown in Figure 6(b) appears stained with an additional phase that may potentially be attributed to grafted DOPO-V-PA molecules. Furthermore, an increase in the nanotube diameters can be observed from the image of CNT-DOPO-V-PA in Figure 6(b). This increase indicates that DOPO-V-PA was successfully grafted to the surface of CNTs.

TEM images of CNTs (a) and CNT-DOPO-V-PA (b).
Figure 7 shows the TGA curves of pristine CNTs, DOPO-V-PA and CNTs-DOPO-V-PA from 50°C to 600°C under N2. Obviously, the pristine CNTs did not exhibit significant weight loss and about 97.08 wt% residues are remained at 600°C. From the TGA curve, DOPO-V-PA starts to decompose at 192.1°C, with 52.32 wt% of residue char at 600°C. For CNTs-DOPO-V-PA, the weight of final chars of 89.97 wt% was observed at the end of decomposition in this test. In addition, DOPO-V-PA grafting amount can be roughly estimated by TGA data. As a result, the content of DOPO-V-PA in CNTs-DOPO-V-PA was calculated to be about 15.88 wt%.
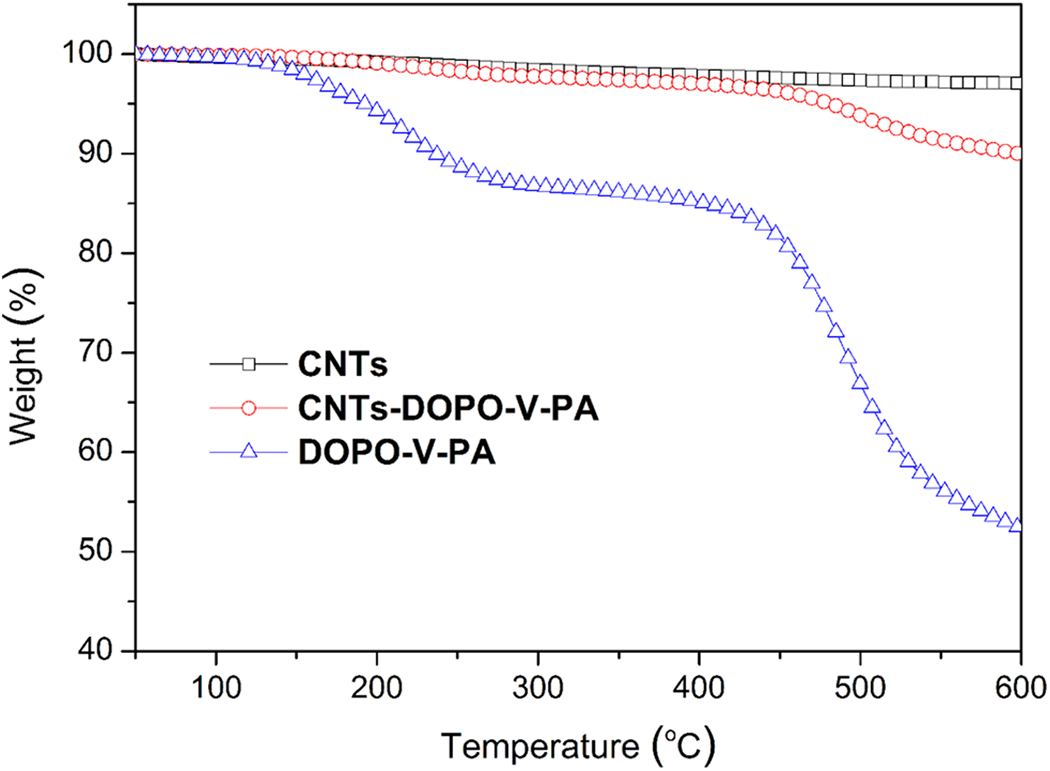
TGA curves of CNTs, DOPO-V-PA and CNTs-DOPO-V-PA.
Thermal stability
The thermal stability of EP and its nanocomposites under nitrogen conditions was analyzed in Figure 8, with detailed data including the onset decomposition temperature (T5wt%) and the maximum decomposition temperature (Tmax) listed in Table 2. Pure EP starts to decomposite from 346.0°C and the maximum weight loss takes place at 381.4°C, with the residual char of 7.02 wt%. For EP/CNTs, both the T5wt% and Tmax decrease, occurring at 341.7°C and 375.1°C, respectively. This is attributed to the addition of nanotubes into the resin, which may inhibit the curing of EP and therefore decreases the cross-link density. 28 However, slight increases in the T5wt% and Tmax of EP/CNTs-DOPO-V-PA were obtained from Table 2, which increases by 0.6°C and 4.0°C compared with EP/CNTs. It is apparent that the CNTs-DOPO-V-PA offers more advantages over pristine CNTs in the thermal stability of EP nanocomposite. Moreover, the amount of residual char of EP/CNTs and EP/CNTs-DOPO-V-PA both increases dramatically compared with the pure EP, indicating the char formation of EP is to some extent enhanced.

Thermal stability of EP nanocomposites.
TGA data of of EP nanocomposites.

Flame retardancy
Cone calorimeter has been widely used to evaluate the combustion properties of polymer materials. Figure 9 illustrates the heat release rate (HRR) and total heat release (THR) curves of EP nanocomposites. The parameters of samples investigated by the cone calorimeter test are presented in Table 3. It is readily seen that the pure EP exhibits the highest peak heat release rate (PHRR) of 1983.1 kW/m2 and THR of 93.9 MJ/m2. The incorporation of pristine CNTs into EP (EP/CNTs) changes the combustion process of nanocomposite and leads to a 26.6% reduction in PHRR when compared with pure EP system. This effect is also evidenced by the decrease of the THR from 93.9 MJ/m2 for EP to 86.6 MJ/m2 for the EP/CNTs nanocomposite. Adding CNTs-DOPO-V-PA into the EP matrix at the same loading further decreases the flammability values of the composites, such as a 30.5% reduction in PHRR and 8.1% reduction in THR, reflecting the improvement of flame retardancy for the polymer materials.

HRR and THR curves of EP nanocomposites.
The parameters of samples investigated by the cone calorimeter test.

It could be obviously seen from Table 3 that the average effective heat of combustion (EHC), the average CO yield (Av-COY) and the average CO2 yield (Av-CO2Y) were apparently decreased after adding CNTs and CNTs-DOPO-V-PA. It was attributed to the physical barrier effect of CNTs or CNTs-DOPO-V-PA and the strong interfacial interactions between CNTs or CNTs-DOPO-V-PA and EP. As compared to the pure EP, the maximum reduction was occurred in EP/CNTs-DOPO-V-PA. The above results confirmed that the EP/CNTs and EP/CNTs-DOPO-V-PA can suppress the formation of smoke and toxic gases. Moreover, the flame retardancy of EP nanocomposites was also investigated by the LOI measurement. It was obtained that the LOI value of pure EP was only 19.2. In contrast, the LOI of EP/CNTs and EP/CNTs-DOPO-V-PA were 23.8 and 25.4, respectively.
Dispersion
The mechanical properties of the EP nanocomposites were investigated using DMTA, and the graphical form of the storage modulus and tangent delta as a function of temperature was shown in Figure 10. The position of tanδ peak in tan d-temperature curve was used to identify the Tg of the material. Figure 10 clearly showed the Tg of EP/CNTs-DOPO-V-PA was slightly decreased in comparison to pure EP. The Tg for pure EP was 92.7°C, which decreased to 89.2°C upon incorporation of CNT-DOPO-V-PA. When a filler was homogeneously distributed in a polymeric matrix, the glass transition temperature (Tg) of the composites should increase with filler content. Thus, it could be obtained that some aggregation of CNTs-DOPO-V-PA in EP matrix still existed. On the other hand, the tangent delta curve of EP/CNTs exhibited double-peak phenomenon, reflecting that the microphase seperation between CNTs and EP matrix took place. Therefore, the DMTA results showed that the aggragation and microphase seperation of CNTs in EP matrix was remarkly improved after grafting CNTs with DOPO-V-PA.
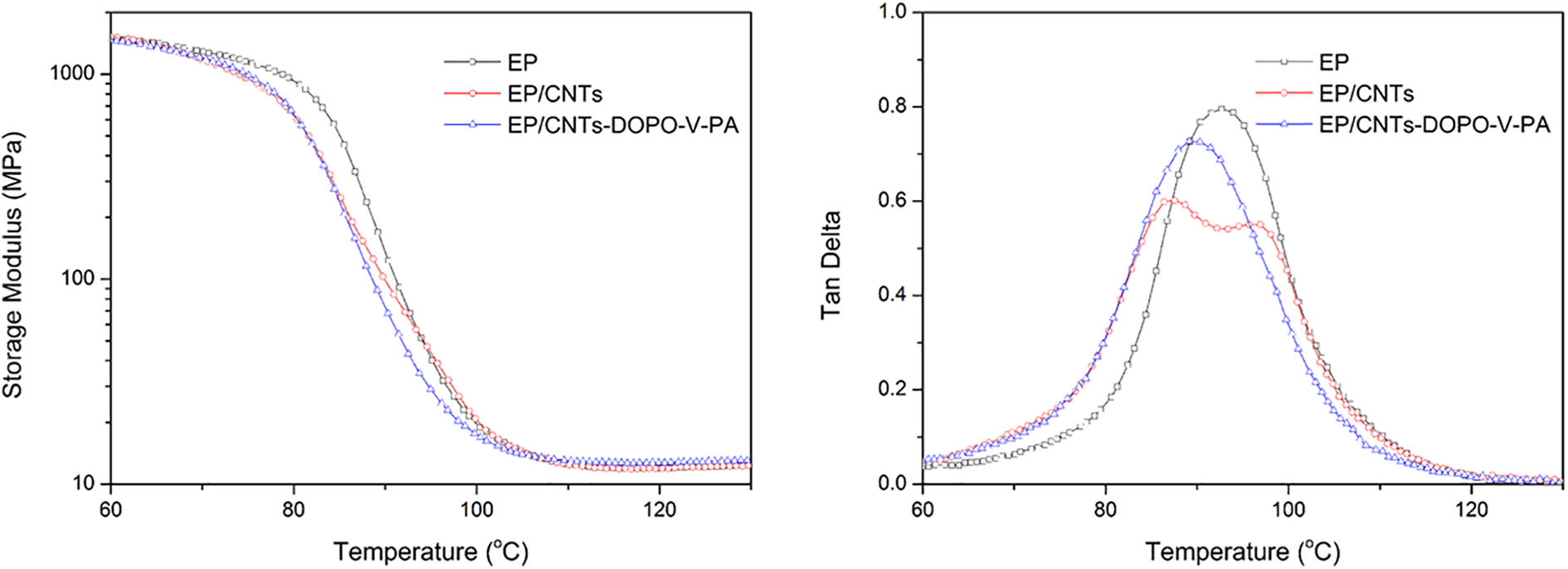
DMTA curves of EP nanocomposites.
Conclusions
In summary, an efficient and simple method was successfully developed to synthesize a novel flame-retardant-wrapped carbon nanotubes (CNTs-DOPO-V-PA) via covalent functionalization of carbon nanobutes (CNTs) with a phosphorus-silicon containing flame-retardant DOPO-V-PA. The results of FTIR, XPS, TEM and TGA measurements showed that DOPO-V-PA has been successfully grafted onto the surfaces of CNTs. Furthermore, CNTs-DOPO-V-PA was incorporated into epoxy resin (EP). Compared with primary CNTs, the addition of 2 wt% CNTs-DOPO-V-PA into the EP matrix could achieve better flame retardancy, such as a 30.5% reduction in PHRR and 8.1% reduction in THR, which was mainly attributed to the combined action of the DOPO-V-PA and CNTs on the flame retardancy of EP nanocomposites. In addition, DMTA results revealed that better dispersion was obtained for CNTs-DOPO-V-PA in EP matrix than pristine CNTs.
Footnotes
Acknowledgments
The author wishes to thank the National Natural Science Foundation of China (51703099), Natural Science Foundation of Zhejiang Province (LQ13E030002) and Ningbo Natural Science Foundation (2019A610032) and Sanmen Natural Science Foundation for financial support. This work was also supported by the Shanghai Key Laboratory of Multiphase Materials Chemical Engineering.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
