Abstract
It has been observed that acrylate monomers are very difficult to polymerize with the low cost nitroxide catalyst 2,2,6,6-tetramethylpiperidinyl-1-oxyl (TEMPO). Therefore, costly acyclic nitroxides such as N-tert-butyl-N-(1-diethylphosphono-2,2-dimethyl)-N-oxyl, (SG1), 2,2,5-Trimethyl-4-phenyl-3-azahexane-3-nitroxide (TIPNO) and TIPNO derivatives have to be used for the polymerization of the acrylic acid derivatives. There are very few reports on the use of TEMPO-derivatives toward the polymerization of n-butyl acrylate. Generally different reducing agents viz. glucose, ascorbic acid, hydroxyacetone etc. have been used to destroy excess TEMPO during the polymerization reaction. The acrylate polymerizations fail in the presence of TEMPO due to the strong C–O bond formed between the acrylate chain end and nitroxide. To the best of our knowledge, no literature report is available on the use of TEMPO without reducing agent or high temperature initiators, toward the polymerization of n-butyl acrylate. The present study has been carried out with a view to re-examine the application of low cost nitroxide TEMPO, so that it can be utilized towards the polymerization of acrylate monomers (e.g. n-butyl acrylate). We have been able to polymerize n-butyl acrylate using the nitroxide TEMPO as initiator (via a macroinitiator). In this synthesis, a polystyrene macroinitiator was synthesized in the first step from TEMPO, after this TEMPO end-capped styrene macroinitiator (PSt-TEMPO) is used to polymerize n-butyl acrylate monomer. The amount of macroinitiator taken was varied from 0.05% to 50% by weight of n-butyl acrylate monomer. The polymerization was carried out at 120°C by bulk polymerization method. The experimental findings showed a gradual increase in molecular weight of the polymer formed and decrease in the polydispersity index (PDI) with increase in amount of PSt-TEMPO macroinitiator taken. In all experiments conversion was more than 80%. These results indicate that the polymerization takes place through controlled polymerization process. Effect of different solvents on polymerization has also been investigated. In the following experiments TEMPO capped styrene has been used as macroinitiator leading to the successful synthesis of poly n-Butyl acrylate. It has been found that styrene macroinitiator is highly efficient for the nitroxide mediated polymerization, even in very small concentration for the synthesis of poly n-butyl acrylate. High concentration of macroinitiator results in the formation of block copolymers of polystyrene and poly (n-butyl acrylate) viz. polystyrene-block-poly-(n-butyl acrylate). The use of TEMPO toward controlled polymerization is of much importance, because it is the nitroxide commercially available at the lowest cost.
Introduction
Conventional free radical polymerization (CFRP) has been widely used for polymer synthesis at industrial level. This method has been used to synthesize a variety of polymers with a broad range of monomers for different purposes. It needs mild reaction conditions, moderate temperature during synthesis, and shows tolerance toward impurities and moisture. 1 But to overcome some important disadvantages of CFRP like poor control on the molecular weight and molecular weight distribution of synthesized polymer and the difficulty in preparation of different structured, well-defined copolymers and predetermination of functionality on polymers, scientists have invented different controlled radical polymerization methods. These controlled radical polymerization methods are called Living/Controlled free radical polymerization methods. These methods include Atom transfer radical polymerization (ATRP), Reversible addition-fragmentation polymerization (RAFT) and Nitroxide mediated polymerization (NMP). These methods have been used extensively to control molecular weight distribution and to synthesize different types of polymers and copolymers like block, comb, star and hyperbranched copolymers. 1 These types of polymers are being used as specialty polymers/smart polymers for various needs of today’s modern lifestyle.2–4 Butyl acrylate is a versatile building block for thousands of copolymers and is a very important acrylic ester monomer. It is used to improve softness, flexibility, photostability, hydrophobicity, toughness, film formation (below room temperature) and low temperature properties because of its soft nature. Butyl acrylate (Tg −45°C) is the largest volume acrylate used in the synthesis of different types of copolymers like styrene acrylic, vinyl acrylic and all acrylic copolymers. Other application areas include inks, textile, paper and leather finishes, adhesives, coatings, sealants and caulks. 5
Nitroxide mediated polymerization (NMP) is the easiest method for living radical polymerization. In this method reversible capping and de-capping of the propagating chain of polymer by nitroxide radical controls the reaction. Polymer chain grow with uniform speed. NMP shows the dynamic equilibrium between dormant alkoxyamines and propagating radicals. Polymerization of n-Butyl acrylate is difficult with TEMPO under normal conditions. 6 Different types of newer high cost nitroxides have been used for the polymerization of acrylate monomers like n-Butyl acrylate, but TEMPO could not be used successfully as an initiator for acrylate monomers.7–9,10 Acyclic nitroxides such as SG1, TIPNO and TIPNO derivatives have to be used for polymerization of acrylic acid and alkyl acrylates.11–13 There are very few reports on the use of TEMPO-derivatives toward the polymerization of n-butyl acrylate.14,15 Generally different reducing agents like glucose, hydroxyacetone, ascorbic acid etc. have been used to destroy excess TEMPO. To the best of our knowledge no literature report is available on the use of TEMPO without utilizing a reducing agent or high temperature initiators toward the polymerization of n-butyl acrylate.14,16 We have synthesized Poly n-Butyl acrylate with a low cost TEMPO by nitroxide mediated polymerization process. A series of polymers were prepared using PSt-TEMPO macroinitiator in different concentrations, from trace amount of 0.05% up to 50% by the weight of monomer. Surprisingly even 0.05 wt.% amount of macroinitiator could successfully produce controlled radical polymerized poly n-butyl acrylate. The excess amount of TEMPO nitroxide shifts the equilibrium in backward direction and leads the active polymer chains to dormant state so that no monomer addition occurs and the polymerization ceases. Difficulty associated with TEMPO mediated polymerization of n-butyl acrylate is directly related to the excess free nitroxides that build up in the reaction mixture due to termination reactions that occur throughout the course of the polymerizations. PSt-TEMPO macroinitiator is helpful for giving better results due to the presence of low concentration of TEMPO nitroxide available for capping of polystyrene backbone chains. When n-butyl acrylate monomer was added, decapping of TEMPO results in further addition of n-butyl acrylate monomer units in the polystyrene chain and form comb like structure of poly n-butyl acrylate. 17
Experiment
Materials required
All the chemicals used were purchased from SRL Pvt Ltd, India. Styrene and n-Butyl acrylate (n-BA) monomers were distilled under reduced pressure after the removal of inhibitor and stored at low temperature. TEMPO and other reagents were used as received. DMF and Toluene used were of AR grade.
Synthesis and characterization of polystyrene macroinitiator (PSt-TEMPO)
PSt-TEMPO macroinitiator was prepared by controlled living radical polymerization as per the method described below. Synthesized polymer was collected by precipitation with large excess of methanol. PSt-TEMPO was purified by reprecipitation with toluene/methanol system and then dried at 70°C for 24 h and stored in vacuo.
The molecular weight and PDI value of TEMPO capped polystyrene macroinitiator was determined by gel permeation chromatography (GPC) using polystyrene standards.
Living radical polymerization of styrene (10 ml) was performed by bulk polymerization method by BPO (0.295 mmol) as an initiator in the presence of TEMPO at 125°C in ratio of 1:1 with BPO. Experiment was run at 125°C for 12 h. under continuous stirring. The conversion was calculated by gravimetric method (Yield = 89%). The molecular weight distribution and polydispersity was obtained by gel permeation chromatography (Figure 1). PDI value of the synthesized polystyrene macroinitiator was found to be 1.46.

GPC result of polystyrene macroinitiator.
Synthesis of poly n-butyl acrylate (PBA)
n-Butyl acrylate has been polymerized with TEMPO capped polystyrene macroinitiator at 120°C for the time duration of 6 h. Amount of macroinitiator taken for polymerization was varied from 0.05% to 50% by weight of n-Butyl acrylate monomer. In all the experiments 4 g. of n-butyl acrylate and required amount of macroinitiator were placed in a three-necked flask. The flask was then degassed with nitrogen for 15 min, sealed and heated at 120°C for 6 h. For solvent study 0.8 g. of macroinitiator (20%) and 4 g. n-butyl acrylate was taken in a three neck flask. Now 20 ml of DMF was added and the flask was degassed with nitrogen for 10 min, sealed and heated at 120°C for 6 h. Same procedure was used with toluene solvent. The synthetic scheme for the preparation of poly(n-butyl acrylate) has been represented in Figure 2.

Schematic representation of synthesis of poly(n-butyl acrylate) by macroinitiator.
Polymer characterization
Gravimetric method is used for monomer conversion determination. The molecular weight and molecular weight distribution of polymers were determined with gel permeation chromatography (GPC) using Turbo Matrix-40 Perkin Elmer (USA) instrument, molecular weight ranging from 400 to 400,000 calibrated with the polystyrene standard sample. Calibration of the column was done with polystyrene sample. For mobile phase, tetrahydrofuran was used at a flow rate of 1.0 mL/min at column temperature 30°C.
FTIR Spectra were recorded on a Perkin Elmer Spectrum GX spectrometer (USA) from KBr pellets in 400–4000 cm−1 of wavelength range. Impurities as like atmospheric vapor and carbon dioxide in the air were made neglected and the spectrum gives only the results of characteristics of samples.
1H NMR Spectra were recorded on a 400 MHz FT NMR, Avance III, Bruker using CDCl3 as the solvent and tetramethylsilane (TMS) as the initial standard.
Result and discussion
Characterization of n-Butyl acrylate copolymer
Figure 3 shows the 1H NMR spectrum of synthesized poly(n-butyl acrylate). The chemical shift δ = 0.9 ppm was attributed to the protons of CH3, the chemical shift δ = 1.4 and δ = 1.6 ppm was assigned to the protons of –CH2–, δ = 1.8 ppm attributed to –CH– backbone chain, δ = 4 ppm corresponds to O–CH2– protons and very small peak at δ = 7.3 ppm for the aromatic ring of polystyrene (from macroinitiator present in very small concentration i.e. 0.05%).

NMR spectrum of poly(n-butyl acrylate) with 0.05% of PSt macroinitiator.
Figures 4 and 5 shows FTIR spectrum of PBA polymer with polystyrene macroinitiator. The characteristic absorption bands of the C=O group stretching vibration appeared at 1725 cm−1 (and the overtone of C=O at 3444 cm−1), and the characteristic absorption band of the C–H stretching vibration at 2852, 2924 cm−1. The bands at 1452, 1395 and 1378 cm−1 represent CH2 bending modes. The C–O–C (asymmetric str.) can be seen at 1257 cm−1 and the C–O–C (symmetric str.) can be seen at 1163 cm−1. 18

FTIR spectrum of poly-(n-butyl acrylate) with 0.05% of PSt macroinitiator.
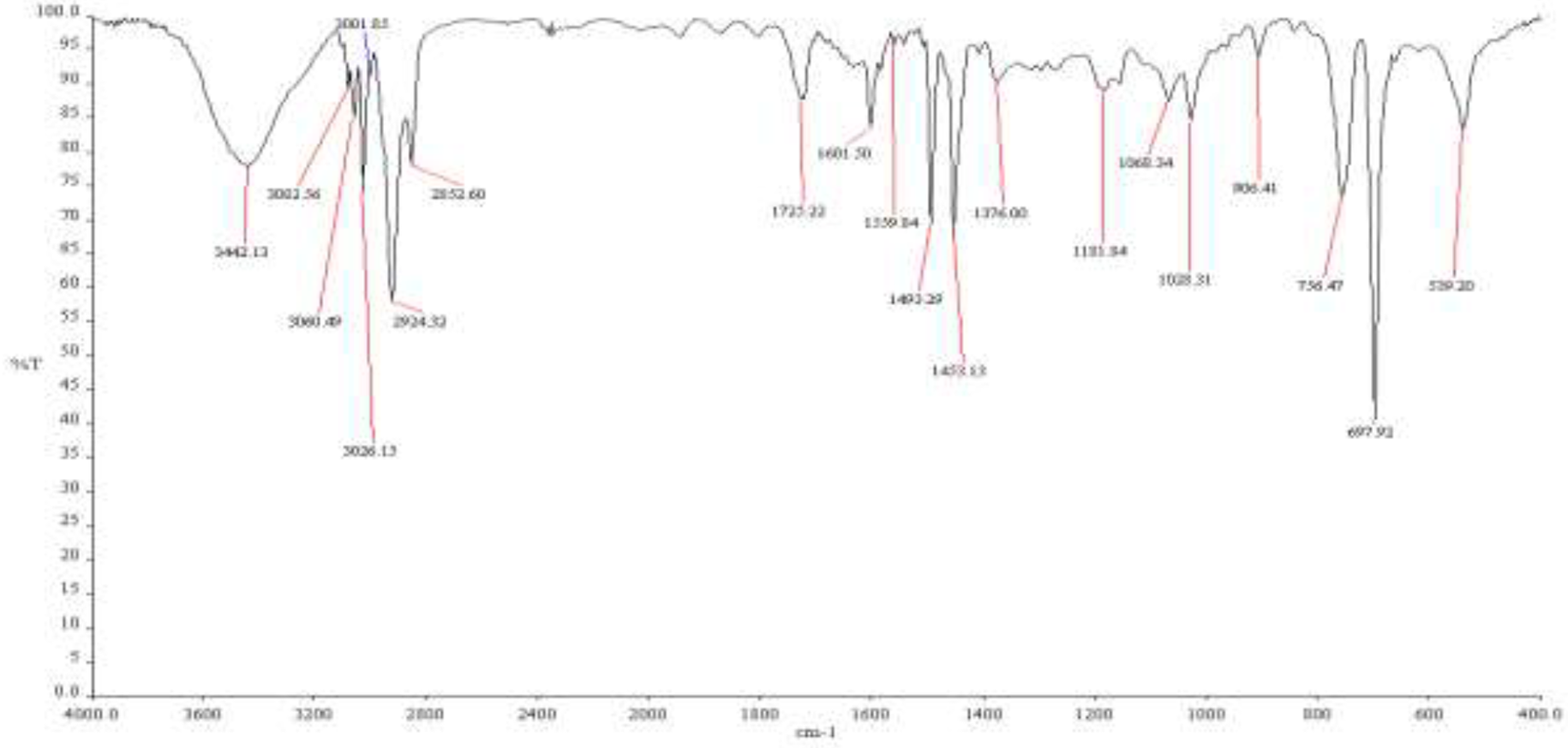
FTIR spectrum of polystyrene-block-poly(n-butyl acrylate) with 50% of PSt macroinitiator.
In Figure 5 characteristic absorption bands at 3001, 3082.56 are seen for the aromatic ring of polystyrene because the concentration of macroinitiator was 50%, but these bands are not visible in Figure 4 because the concentration of macroinitiator is very small (0.05%).
As shown in Figure 6, the PDI is low when the amount of macroinitiator is above 5% and it reaches 1.46 at 50% macroinitiator concentration. This shows that the very small amount of macroinitiator is also effective for controlled polymerization of n-butyl acrylate. PDI results are better with increase in the amount of macroinitiator, however in that case a block copolymer is formed. In Figure 7, we can see conversion percentage is uniform after 4 h. It shows that long time duration is not required for polymerization process. It can be concluded that TEMPO can be utilized easily for the controlled polymerization of acrylate esters via macroinitiator. Almost pure homopolymer poly(n-butyl acrylate) can be prepared by using very small quantity viz. 0.05% of macroinitiator. Block copolymers of polystyrene and poly(n-butyl acrylate) were formed by using large concentration of macroinitiator.

Dependence of PDI value on different amount of macroinitiator for NMP of Nba.

Conversion % of poly(n-butyl acrylate) with increase in time.
The corresponding PDI values are shown Table 1. The traces shift cleanly confirming the decrease in PDI value of polymers with the increase in the amount of macroinitiator taken. However, very small amount of macroinitiator (0.05 wt.%) has also given the good PDI value of 1.67.
Effect of different amount of PSt macroinitiator for n-butyl acrylate polymerization.

Effect of the polar and nonpolar solvents on NMP of n-butyl acrylate has also been studied. Results in Table 2 shows that PDI values are found to be lower in DMF than that in Toluene.
Effect of solvent on NMP of n-butyl acrylate with 20 wt.% PSt macroinitiator.

Chain extension of PBA
In order to confirm the active nature of the chain end of the synthesized poly(n-butyl acrylate), chain extensions of the synthesized polymer were performed in DMF with styrene monomer. The solution mixture of 1 g. poly(n-butyl acrylate) (PDI = 1.48) and 2 g. styrene monomer was taken in a two neck round-bottom flask was heated at 110°C under nitrogen atmosphere for 4 h. The chain extensions were successful. The chain-extended copolymer has PDI value 1.56. It is confirmed from this result that the synthesized polymer of n-BA was end-capped with TEMPO and active for further chain extension with living radical polymerization method. Thus, the amount of the chain-extended PBA with styrene increased to 2.608 g. It is clear from this result that end groups of polymers are active in chain extension reaction.
Conclusions
The nitroxide mediated polymerization of n-butyl acrylate was successfully performed with styrene macroinitiator by bulk polymerization method at 120°C. Different resultant PDI values (<1.7) of the synthesized polymers indicate that the polymerization proceeds via living/controlled radical polymerization process. Livingness of PBA or Polystyrene-graft-PBA were proved by chain extension reaction. Results shows that even very small amount of macroinitiator (0.05 wt.%) is effective for polymerization of n-butyl acrylate with TEMPO nitroxide. A wide range of initiator concentration from 0.05 wt.% to 50 wt.% has been studied. The method can be utilized for the preparation of either homo or co-polymers of acrylate esters. For the preparation of poly(n-butyl acrylate) low concentrations of initiator viz. 0.05 wt.% are ideal, as the percentage of polystyrene component present in the polymer is very small. At high concentrations of macroinitiator, block copolymers [polystyrene-block-poly-(n-butyl acrylate)] can be prepared. The PDI values of synthesized polymers decreases with the increase in macroinitiator content. The PDI is found to be lower in DMF than that in Toluene. This shows polar solvents are more suitable for polymerization than non-polar solvents. Hence it has been possible to utilize TEMPO as the low cost nitroxide catalyst for the controlled polymerization of acrylate esters like n-butyl acrylate.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
