Abstract
In recent decades, rational management of agricultural residues presented a new approach for extraction, characterization, and utilization of cellulose nanofibers (CNF). In this context, the valorization of flaxseed fibers, providing an annual yield of millions of metric tons, as an abundant sustainable fiber source, was carried out. The cleaned and ground raw material was delignified and bleached, followed by 2,2,6,6-tetramethylpiperidine-1-oxyl radical (TEMPO)/NaBr/NaClO oxidization along with mechanical homogenization. The resulting extracted cellulose and cellulose nanofibers were characterized by various analytical methods. The overall yield of CNF based on the raw material was 31.2% ± 1.5%. This study explored a simple method for converting flaxseed fibers to fluorescent, high quality, nano-sized cellulosic precursors for novel applications in pharmaceutical and bio-composite applications.
Introduction
Nanotechnology has countless potential applications in industry. Nanomaterials isolated from cellulose sources are cellulose nanocrystals (CNC) or cellulose nanofibers (CNF), with the latter showing the most commercial importance. Nanocellulose has a proven potential in nanocomposite material applications, such as energy storage, production, and biomedical;1-3 membranes, aerogels, and hydrogels;4,5 and antibacterial, electrical, and industrial applications.6-8 Various studies derive CNFs from underutilized plants, such as piassava 9 and sansevieria.10,11 Industrially, nanocellulose of <100 nm in diameter is of prime interest, which further encourages the derivation of crystals or fibers from biomass sources. 12 CNFs contain both amorphous and crystalline cellulose areas, possess micrometer-scale lengths, and are 50-60 nm in diameter. 13 The natural ben-efts of cellulose, such as biodegradability, sustainability, and abundance, also include enhanced crystallinity, aspect ratio, optical, thermo-mechanical properties, and low density of CNFs. 14 Hence, CNF derivation from all underutilized cellulose sources, especially from agricultural waste or bio-mass, is of prime interest. 15
Flax is grown in two varieties. Fiber fax is known for its fibers producing fine yarn and linens. The agricultural crop variety, called flaxseed or linseed, is grown only for its seeds and oil derivative applications. Fiber fax has already established its use in conventional as well as composite industry applications for a considerable time.16-18 However, the flaxseed variety, having a higher production and GDP value than common fax, is widely produced worldwide for human, animal, and oil industry consumption. 19 However, it has been underutilized due to the annual burning of millions of metric tons of wasted byproduct straws lacking alternative uses. 18 Hence, disposal of agriculture biomass,20,21 particularly flaxseed straws, poses a serious challenge of increased pollution, domestically 22 and eventually worldwide. The top two producers globally are Canada and China, as per the United Nation's FAOSTAT (1994-2016). 23 A broad study to individualize the flaxseed fibers resulted in very short, non-spinnable fibers and environmental pollution. 24 Hence, flaxseed fibers that are unusable for spinning for use in conventional textiles can be an abundant and valuable cellulose extraction resource for nanocellulose applications.
In our previous study, an extensive attempt was carried out to investigate and compare the properties, characterization, and morphology of untreated and alkali treated flaxseed fiber bundles and their resultant composites. 25 Reusing valuable lignocellulosic resources can achieve remarkable benefits as an abundant, economical, and reliable supply of cellulose and cellulose nanofibers (CNF) for developing new biomaterials having low cost, low density, abundance, and biodegradability. 26-28 The use of discarded flaxseed biomass as a potential commercial source for the extraction of cellulose is of industrial and scientific importance. Scientific literature lacks studies reporting cellulose extraction from flaxseed plants, probably due to its enormous volume and difficult handling. Hence, methods for using this low-value material for a high-value applications could help to conserve energy and the environment, and promote economic development.
This study reviews processes of cellulose extraction from flaxseed fiber bundles and its characterization. X-ray diffraction (XRD), scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FTIR), thermo-gravimetric analysis (TGA), differential scanning calorimetry (DSC), and transmission electron microscopy (TEM) were used for characterization of the extracted cellulose. XRD was applied to examine the long-range order produced due to very short-range interactions, and crystallinity percentage estimation. SEM images revealed the cellulose morphology. FTIR investigation classified functional groups in the molecular structure of cellulose. TEM micrographs revealed the nanocellulose morphology. Finally, correlation with thermal measurements was evaluated. This study provides the essential fundamental data about the potential of using this valuable and abundant, but currently wasted, cellulosic resource as CNFs for multifunctional applications.
Experimental
Materials
Flaxseed fiber bundles were supplied by the Agriculture Research Academy, Inner Mongolia, China. Dimethyl sulfoxide (DMSO), 12 wt.% sodium hypochlorite (NaClO) solution, 2,2,6,6-tetramethylpiperidine-1-oxyl radical (TEMPO, 98%), sodium bromide (NaBr), and ethanol were of laboratory grade (Shanghai Aladdin Chemical Regent Inc. and Sinopharm Chemical Reagent Co. Ltd. China) and used without further purification. To avoid any chemical reactions, only deionized water was used for cellulose nanofibers extraction in this study.
Method
Pretreatment of Flaxseed Fibers—Waterlogging
Flaxseed stems were obtained from the plants by cutting the mid-section to the length of 30-40 cm (Fig. 1a), followed by waterlogging in boiling water for 24 h inside an ambient temperature room. The bast was manually peeled of and left to dry naturally. The resulting material is hereafter referred to as untreated fiber bundles (Fig. 1b).

(a) Flaxseed fiber stems, (b) conversion into small pieces (bundles), (c) flaxseed fibers powder (LC-RAW), (d) chemically-treated samples, and (e) cellulose fibers (LC-NF).
Non-Cellulosic Material Removal
For removing non-cellulosic constituents, flaxseed fiber bundles were grounded to a 0.3-mm particle size powder labeled as LC-RAW (Fig. 1c), soaked in a 15 wt.% NaOH solution, and heated at 60 °C for ∼4 h according to published work. 29 Samples were thoroughly dried and treated with DMSO solution at a liquor ratio (LR) of 1:15 at 70 °C (Fig. 1d). Similarly, again after washing several times with distilled water, it was dried in an oven at 70 °C for 24 h. The acquired cellulose fibers were labeled as LC-NF (Fig. 1e).
Preparation of Cellulose Nanofibers
Preparation of cellulose nanofibers suspension was performed according to the literature procedure. 30 Flaxseed cellulose fibers LC-NF (15 g) were fully dispersed in a solution with water (285 mL), and calculated amounts of NaBr (0.30 g) and TEMPO (0.03 g) were added. The oxidation reaction was initiated with the addition of 13% NaClO solution (27 g) under constant stirring. The solution's pH was kept between 10 and 10.5 for 2 h using a 2 wt.% NaOH solution with continuous monitoring using a pH meter. Finally, 5 mL of anhydrous ethanol was added. The final solution was washed five times with deionized water by subsequent centrifugations at 10,000 rpm for 10 min and ultra-sonication for 10 min each. The resulting 1 g of cellulose nanofiber jelly was dried in a vacuum oven at 90 °C for 24 h and named LC-CNF. The remaining aqueous suspension was stored at 4 °C in the refrigerator for later use.
Characterization
SEM
Surface morphology was examined by micrographs using a JSM-6360LA (JEOL, Japan) SEM using an accelerating voltage of 15 kV. Samples were gold-coated by sputter-coating with evaporated gold.
TEM
The microstructural study of CNF morphology imaging was performed by a Philips CM120 TEM at an accelerating voltage of 120 kV by depositing a drop of each LC-CNF sample in an ethanol dispersion solution with a concentration of 0.5 wt.% on a glow-discharged carbon-coated Cu grid and examined by TEM.
FTIR
An S-100 spectrometer (Perkin Elmer) was used for FTIR analysis. An attenuated total reflectance (ATR) accessory was used, and the samples were scanned from 4000 to 400 cm−1 in transmission mode (T%).
XRD
The Diffract-AC software program on a Siemens D500 XRD was used for the study of dried CNFs at 15 mA and 30 kV operating in transmission mode, using a CuKα lamp (λ = 1.54056 A), in the range of 10° to 40°, with a step size of 0.015°, and a count time of 100 s for each 1.05°. The degree of crystallinity index
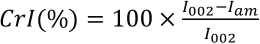
TGA
A Perkin Elmer TGA-7 was used to investigate the thermal stability of the samples, with a gas flow of 19.8 mL/min under a nitrogen atmosphere. Each sample was kept within a 0.3−0.5 mg weight range and heated from 25 °C to 600 °C at a rate of 10 °C/min.
Results and Discussion
Chemical Composition Analysis
The chemical composition of the flaxseed fibers in powder form was determined (Table I) using a standard gravimetric method,
31
and α-cellulose content was determined using a calorimetric method with anthrone reagent.
32
The content percentages of different constituents, determined by the chemical composition analysis of flaxseed fiber bundles in our previous study,
25
were compared with those from various underutilized cellulosic sources reported in the literature such as wheat straw,
33
rye husk,
34
sugarcane bagasse,
31
grass/ reed
Comparison of the Chemical Composition of Flaxseed Fiber Bundles with Various Underutilized Cellulosic Sources
Morphological Evaluation
Fig. 1 shows the physical appearance of the original flaxseed fibers in bundle form (Fig. 1a), in cut pieces form (Fig. 1b), and in powder form after passing through 60 mesh sieves (Fig. 1c). The pronounced effectiveness of chemical treatments (e.g., alkaline solution, bleaching, and DMSO) was observed since white-colored flaxseed fibers were observed (Fig. 1d). After the chemical treatments, the white color of the residual material indicated that a considerable part of the initial non-cellulosic material was removed, resulting in pure cellulose fibers after filtration and washing (Fig. 1e), which were suitable for CNF extraction.
SEM revealed information on the geometry of fibers in the powder form LC-RAW (Fig. 2a) and the cellulose fibers form LC-NF (Fig. 2b). The LC-RAW sample fibers were roughly messy and exhibited an uneven structure significantly different than those fibers after chemical treatments LC-NF, which were more isolated and aligned due to the removal of non-cellulosic impurities. The SEM images (Fig. 2) showed LC-NF fibers as bundles opened up with significant fibrous form, indicating the removal of non-cellulosic impurities, and the distribution of micro-fibrils by alkali treatment, with improved surface modification. 40 Treatment with DMSO broke down the intermolecular hydrogen bonding, resulting in cleaner, aligned, and individualized microfibers. 41

(a) Flaxseed fibers powder (LC-RAW) and (b) cellulose fibers (LC-NF).
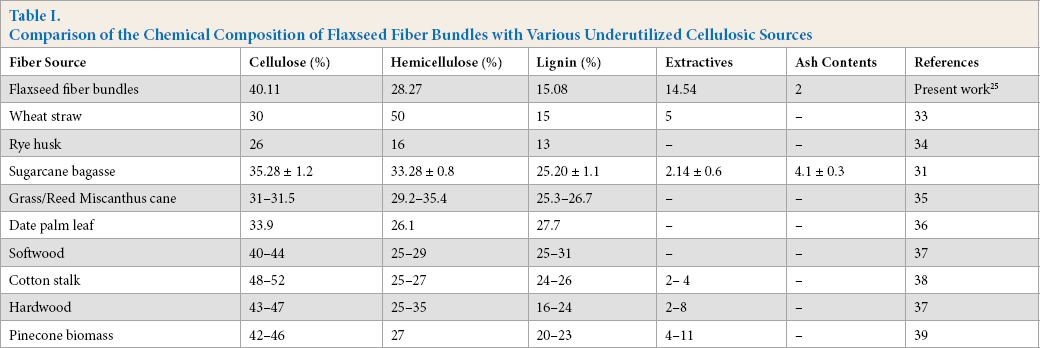
The LC-CN (Fig. 3) showed short and thin fibers of CNF as use of TEMPO reduced the amorphous cellulose regions. Fig. 3 provides the TEM images of LC-CNF, which showed that the obtained structures were relatively isolated with defined shapes. The dimensions of CNFs depended on the operating conditions of the extraction process. CNF samples were examined by evaluating the TEM images using Image J. software for size dimension; 100 samples were taken for that test. Most nanoparticles displayed a diameter in the range of 16 to 35 nm (Fig. 4). As observed in the morphology of CNF obtained after TEMPO oxidation, LC-CNF demonstrated an average diameter of 23 nm and a length of several hundred nanometers. Similar studies showed that the size dimensions of CNFs extracted from curaua fiber were 6-10 nm in width and 80-170 nm in length. 42 Also, CNFs of 5-60 nm in diameter and several micrometers in length were extracted from fax, sugar beet, potato tuber, wood, and hemp. 43

TEM of cellulose nanofibers extracted from flaxseed fibers.
Diameter distribution of cellulose nanofibers LC-CNF.
XRD
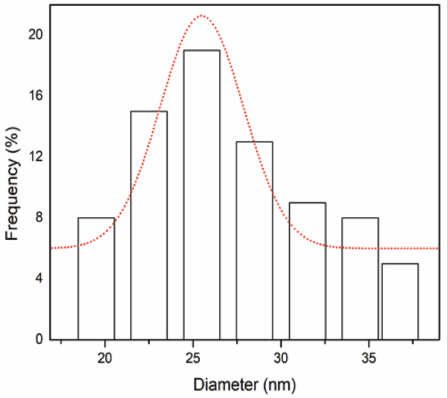
XRD analysis was performed to evaluate the crystallinity of flaxseed fiber powder, cellulose fibers, and CNFs obtained after chemical treatments. Cleaning, alkali treatment, bleaching, DMSO treatment, and TEMPO performed on the flaxseed fibers affected the crystallinity of the samples. It's evident that, due to hydrogen bonding interactions and Van der Waals forces between the molecules, cellulose fibers exhibit crystalline structure, but lignin and hemicellulose show amorphous structure. 44 When treated with an alkali solution, natural plant fibers become increasingly stiff due to the removal of non-cellulosic materials. 45 By investigating the crystallinity of the treated samples (LC-NF and LC-CNF) and comparing them with the untreated sample (LC-RAW), chemical treatment effectiveness can be assessed.
The diffraction patterns obtained from XRD techniques for untreated, treated (alkalization, bleaching, DMSO treatment), and TEMPO-oxidized CNFs are shown in Fig. 5. All the samples are typical of cellulose-I, with diffraction peaks at 2Θ angles of 16°, 22°, and 35° corresponding to 110, 002, and 004, 12 respectively. Chemical effectiveness was evaluated by determining the crystallinity index of every sample. Alkali treated samples LC-NF showed a sharp peak compared to the flaxseed fiber powder LC-RAW due to higher crystallinity (54%), which is ascribed to the progressive removal of amorphous non-cellulosic materials. The crystallinity of flaxseed was 40% (Table II). Similarly, DMSO tended to open the amorphous region of the cellulose fibers and helped the TEMPO oxidation materials to penetrate rapidly. As the hydroxyl groups of cellulose were oxidized to carboxyl groups, LC-CNF showed higher crystallinity (63.40%) corresponding to the CNFs, and displayed the sharpest peak at 22° (Fig. 5). Similar results were also noted in the case of CNFs extracted from various sources of biomass, such as 64%, 69%, and 73% crystallinity when treated under various experimental conditions from curaua fiber. 42
Crystallinity of LC-RAW, LC-NF, and LC-CNF
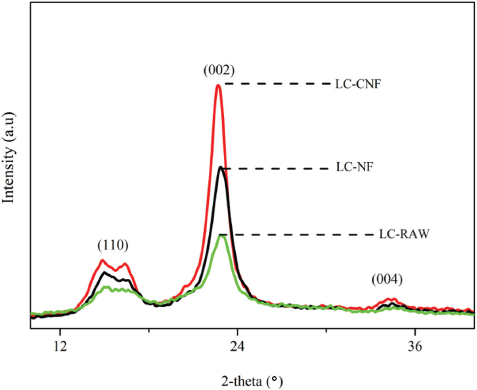
XRD patterns of flaxseed fibers (LC-RAW), cellulose fibers (LC-NF), and cellulose nanofibers (LC-CNF).
FTIR
The study of changes in the chemical structure of flaxseed fibers after chemical treatments through the final product as CNFs, was carried out through FTIR spectroscopy. Fig. 6 shows the FTIR spectra recorded on CNFs obtained after TEMPO oxidation. The different chemical treatments were expected to induce morphological and chemical changes, as shown in the previous sections.
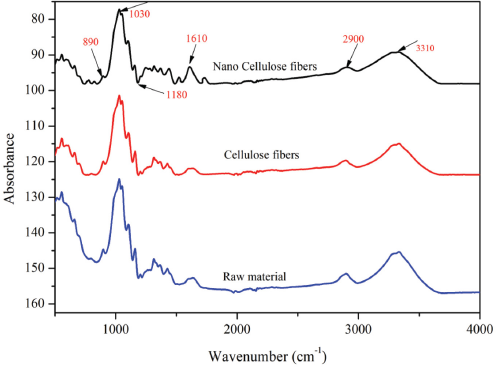
FTIR spectra for flaxseed raw material (LC-RAW), cellulose fibers (LC-NF), and cellulose nanofibers (LC-CNF).
Absorptions around 3310, 2900, and 890 cm−1 exhibited in all spectra were associated with the native cellulose-I characteristics. 46 The broad absorption band located between 3000 and 3500 cm−1 was due to the -OH group stretch, and absorption at 2900 cm−1 was related to the CH2 groups of cellulose. 47 The peak at 890 cm−1 was associated with C-H due to the vibration of cellulose present in all samples. Similarly, the peak at 1610 cm−1 for cellulose samples was associated with TEMPO oxidation resulting in sodium carboxylate groups. The band at around 1028 cm−1 was due to pyranose ring stretching due to C-O-C. The FTIR revealed that the chemical swelling by DMSO and TEMPO oxidation reactions did not affect the chemical structure of the cellulosic material, which indicated that the chemical groups of the resulting materials were stable and no strong chemical reaction occurred.
Thermal Stability
The thermal stability of CNFs after chemical processing was studied by TGA. The TGA curve is graphically represented in Fig. 7 for the flaxseed fibers in powder form and extracted CNFs. For each sample, during thermal degradation, two main mass losses were observed. The first mass loss, under 100 °C, attributed to the evaporation of water, 48 was similar for all samples, which indicated that moisture was present inside the raw material and even in the CNFs. The second mass loss was due to the decomposition of the chemical structure of lignin, hemicellulose, and cellulose present in LC-RAW and also in LC-CNF. 26
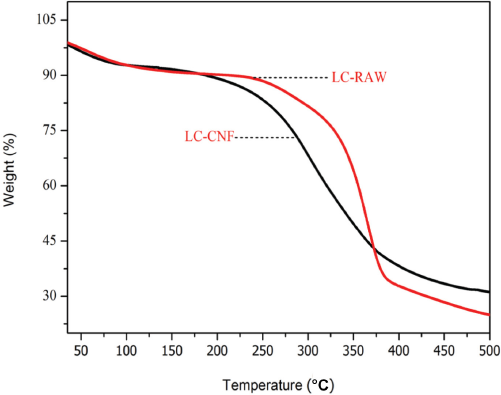
Thermal properties of flaxseed fiber powder (LC-RAW) and cellulose nanofibers (LC-CNF).
The onset degradation temperature of LC-RAW was about 300 °C, which is possibly due to the presence of hemicellulose, lignin, and the other non-cellulosic constituents, and the degradation ended at a final temperature of 362 °C. 49 In the case of LC-CNF, the onset degradation and final degradation temperatures were less than those of raw flaxseed fibers due to the chemical treatments. TEMPO oxidation caused the destruction and conversion of macro-cellulose into micro-cellulose chains, which might have caused the lower onset degradation temperature of 248 °C, and final degradation temperature was about 270 °C.
Conclusion
A suitable and practicable chemical process was used to extract cellulose nanofibers (CNFs) from an abundant yet underutilized material (i.e., flaxseed fiber bundles) by TEMPO oxidation followed by homogenization. Flaxseed CNFs were analyzed to explore their chemical composition, morphology, structural features, and thermal behavior. Chemical analysis results confirmed a greater percentage of cellulose and less of non-cellulosic components in the extracted CNFs than in the pristine flaxseed fibers. Chemical purification changed the morphology of the flaxseed fibers as the agglomeration was broken down into individual cellulose fibers. Through different techniques, the results revealed that the produced CNFs exhibited lower aspect ratios, better thermal properties, and dimensions at nano-scale, hence, making these suitable as reinforcing agents in bio-renewable composite preparation. This study provided vital fundamental data, which further established their significance and potential use in green composites or bio-materials.
Footnotes
Acknowledgment
This work was financially supported by the DHU Distinguished Young Professor Program, open fund of Shanghai Center for High Performance Fibers and Composites, Shanghai Natural Science Foundation (Grant No. 17ZR1400800).
