Abstract
Suspension polymerization of vinyl chloride (VCM) was carried out in the presence of particle of both pristine Mg-Al layered double hydroxide (LDH) and LDH intercalated by 3-(trimethoxysilyl) propyl methacrylate (MPTMS-LDH) in a pilot-scale reactor. The experiments were conducted at different concentrations of each type of the particles (0, 1, 3, 5, 7 wt. % of VCM). The pure LDH and MPTMS-intercalated LDHs were used as co-monomer for fabrication of LDH or MPTMS-LDH/PVC nanocomposites; the particles were directly pre-dispersed in monomer phase before dispersing in the continuous aqueous phase and starting the suspension polymerization. The basal spacing obtained from the X-ray powder diffraction (XRD) showed that the reaction between VCM and MPTMS-LDH was effective, with a further intercalation of PVC chains in the interlayer space. The molecular characterization showed that the polydispersity index (PDI) and molecular weight of the poly (vinyl chloride) (PVC) resin do not change in the presence of the pristine LDH. The MPTMS-LDH, however, decrease the molecular weight and increase the PDI of PVC resin. Also, from the morphological point of view, scanning electron microscopy (SEM) showed that PVC grains produced with high particles content had a smoother surface with more regular shape. Moreover the incorporation of nanoparticles in the VCM suspension polymerization, made particles with a narrower size distribution. The LDH results in the formation of smaller primary particles with higher internal porosity whereas, the larger primary particles with lower internal porosity in the presence of modified particles was observed.
Keywords
Introduction
Poly (vinyl chloride) has been widely used as an important general plastic because of its favorable electrical properties, corrosion resistance, self-extinguishing characteristics, recoverability and inexpensiveness. However, its low heat-softening temperature and poor toughness and limit its application as a kind of engineering plastics.1–5 In recent years, the use of inorganic particles dispersed in a PVC matrix to provide high-performance novel materials has attracted intense interest lately from academic and industrial researchers. 6 Among nanofillers, the layered double hydroxides (LDHs) are one of the most specific inorganic fillers that have been used extensively in polymer nanocomposite preparation.7–13
The growth of LDH nanocomposites has increased in recent years. LDHs have been reported to significantly improve the mechanical and thermal properties of the end-use polymer products. They are anionic clays with the general formula
In situ polymerization involves initial dispersing the inorganic particles directly in the monomer phase prior to a polymerization process with the aid of mechanical stirring.
29
In this method, the LDH layers are swollen by the liquid monomer.
30
When polymerization is initiated, the intercalated monomer can convert to polymer intercalated to the interlayer of LDH and thus forms a nano-composite.
26
As the presence of the LDH particles in the reactor is expected to change the interaction patterns of the system, they can influence the drop formation mechanism, drops coalescence and break-up process and consequently, the initial droplet size distribution of the monomer dispersed phase. Subsequently, it affects the mean PVC grain size, grain size distribution, and in particular, porosity (cold plasticizer absorption) and bulk density, which eventually influences the resin processability.
31
The particles can also act as the primary particle nuclei and, in this way, affect the size, the number, and the three-dimensional network structure of the primary particles.32–34 All nanocomposites show significantly enhanced thermal stability compared with virgin PVC due to the increases of the effective activation energy (Eα) during degradation process. However, LDHs nanocomposites show much higher thermal degradation temperatures than clay nanocomposites with the same filler content because they have much higher activation energy than clay nanocomposites at the early degradation stage. Silane modification of layered double hydroxides (LDHs) plays a crucial role in controlling the surface hydrophilicity or wettability and improving the compatibility of LDHs dispersed in non-polar monomer. Moreover appropriate selection of the coupling agent which is capable of surface initiated polymerization, is of importance for optimal improvement of the thermal and mechanical properties of the final PVC nanocomposite.
8
3-(trimethoxysilyl) propyl methacrylate (MPTMS
In the present work, the intercalation of 3-(trimethoxysilyl) propyl methacrylate (MPTMS) into Mg-Al LDH hosts and application of intercalated LDH for preparation of LDH/poly (vinyl chloride) nanocomposite by in situ suspension polymerization are studied. The effect of the nature of the LDH particles surface, with and without MPTMS modification, as well as the concentration of particles on the molecular (Molecular weight distribution and k-value) and morphological (particle size distribution and internal structure) characteristics of final PVC grains are investigated.
Experimental
Materials
Magnesium chloride hexahydrate (MgCl2·6H2O), Aluminum chloride hexahydrate (AlCl3·6H2O), Urea (NH2CONH2) and 3-(trimethoxysilyl) propyl methacrylate (MPTMS) were purchased from Aldrich. Vinyl chloride monomer (VCM) was supplied by Bandar Imam Petrochemical Company, Mahshahr, Iran; hydroxypropyl methylcellulose (HPMC) (Methocel 65SH-50); hydroxypropyl cellulose (HPC) (KlucleJ), and sorbitan monolaurate (Span 20) as suspending agents were supplied by Shin-Etsu Chemical Co. (Tokyo, Japan), Hercules International, Ltd., and Beckmann Chemikalien KG (Bassum, Germany; Becksurf 7125), respectively. Dimyristyl peroxydicarbonate ((MYPC:
Preparation of LDH
A co-precipitation method was used for the preparation procedure of LDH-NO3; 250 ml aqueous stock solution of Mg (NO3)2·6H2O and Al (NO3)3.6H2O with Mg/Al molar ratio of 3 were mixed and 3 M Sodium Hydroxide Solution was used to control pH at 9. After reaction completion, the product was collected by centrifugation, washed thoroughly with distilled water and dried at 40°C.
Preparation of MPTMS-LDH
A full description of preparation of LDH intercalated with silan group is given elsewhere. 35 In brief, 250 ml aqueous solution of Mg and Al nitrate (Mg/Al = 3) was added with a flow rate of 5 ml min−1 into 200 ml of aqueous solution of sodium dodecylsulphate (0.6 mol l−1) by maintaining pH at about 9. The precipitation was carried out under vigorous stirring at 80°C. The resulting suspension was stirred overnight at 80°C. Then MPTMS was added to the dodecylsulfate (DS) intercalated LDHs (DS-LDH) in methylene chloride with N-cetyl-N, N,N-trimethylammonium bromide (CTAB) as a cationic surfactant. After mixing the components, the suspension was sealed in a 500 ml glass bottle under nitrogen and stirred for 6 days (AMPS and ACPA) or 2 days (AA and MAA) at room temperature. The anion exchange method of intercalation in layered double hydroxides (LDHs), based on the formation and organic phase extraction of a salt between dodecylsulfate (DS) anions and a cationic surfactant (CTA) was used to obtain a well-intercalated LDH. The product was collected by centrifugation, washed thoroughly with distilled water and dried at 40°C (see Figure 1).

Synthesis route of MPTMS-LDH and presentation of adding to VCM monomer.
Preparation of PVC nanocomposites
In situ suspension polymerization experiments were conducted by using a 15 L stainless steel jacketed reactor includes two baffles of circular cross-section and two eight-flat blade turbines. The particles were first pre-dispersed in the VCM phase for 30 min at 0°C.(As the surface and between the LDH layers are modified by MPTMS groups, the surface of LDH particles become hydrophobic to a large extent and can be well dispersed inside the monomer phase). After that, a mixture of suspending agents, initiator, and DM water was pumped into the reactor, and the mixture was stirring until forming a stable suspension. Then, the mixture was heated up to the polymerization temperatures of 52°C by utilizing hot water in the jacket. In all experiments, the amounts of water and VCM charged were 4100 and 2800 g, respectively. The polymerization reaction was quenched after a pressure drop of 5 bar of the inlet reactor pressure. Sampling is carried out by stopping the reaction via immediately quenching the temperature of the reactor to about 15°C. In all experiments, after venting the unreacted vinyl chloride to the VCM recovery unit, the reactor contents temperature was raised again up to 60°C to remove the residual monomer. Accordingly, different types of PVC nanocomposite were manufactured with a varying weight percentage of particles at X = 0, 1, 3, 5 and 7% based on the overall VCM initial charge. The samples were coded as MPTMS-LHH-PVCNCX (for PVC nanocomposites prepared in the presence of MPTMS-LDH) or LDH-PVCNCX (for PVC nanocomposites prepared in the presence of LDH) where X stands for particle content.
For the investigation of the intercalation of PVC chains into MPTMS-LDH and LDH interlayers, a series of PVC nanocomposite grains was washed thoroughly several times by using tetrahydrofuran (THF) to remove the unattached PVC chains for further analysis. The samples were coded as W-MPTMS-LDH-PVCNCX or W-LDH-PVCNCX.
Characterization
Powder X-ray diffraction (XRD) measurements were performed using a XRD model D8 Advance, X-ray diffractometer (40 kv, 30 mA) with CuKa (λ = 1.54184 Å) source radiation at room temperature. The scanning rate was 2 °/min in the range of 5° to 70° and a count time of 0.1 s/step. Fourier transform infrared (FTIR) spectra were recorded using a Bruker Tensor 27 spectrometer equipped with a KBr beam splitter, with a wavenumber range of 4000 to 500 cm−1 at a resolution of 6 or 4 cm−1. Transmission electron microscopy (TEM) images were collected on a Philips CM30 electron microscope at 300 kV. The porosity of the PVC nanocmposites was characterized by cold plasticizer absorption (CPA) according to the standard test method (ASTM D 3367-95). A Thermo Jarell Ash ICAP 61E instrument (ICP) were used to measure the total particle content remain in the aqueous phase after polymerization.
The mean particle sizes and particle size distributions (PSD) of the PVC grains were measured with a Malvern model 2603LC particle size analyzer (Worcestershire, UK). The surface mean diameter is also known as Sauter mean diameter (SMD) was calculated by 36 :
The coefficient of variation is defined as the ratio of the standard deviation to the mean diameter according to 36 :
where Di and
Scanning electron microscopy (SEM; model XL30, Philips Co., Eindhoven, The Netherlands) was used to study the quality of produced particles as well as the skin and inner structure of the grains. Energy-Dispersive X-Ray Analyzer (SEM-EDX; model XL30, Philips Co., Eindhoven, The Netherlands) was used to identify the elemental composition of samples. The interior structure of particles was studied via the fracturing of particles. For this purpose, samples frozen in liquid nitrogen were mechanically broken down. Then, the fractured samples were coated with a thin layer of gold with a gold sputter coater (model SCD005, Bal-Tec, Hannover, Germany) in a vacuum, and then micrographs were prepared. The SEM images were analyzed by using image processing software (ImageJ) to measure the critical conversion (after which Sauter mean diameter (D32, equation 1) of primary particles remain unchanged with conversion.37–40) At least 120 primary particles for three different broken particles at each conversion were analyzed to calculate D32. The K-value was calculated from the dilute solution viscosity measurements of the PVC samples in cyclohexanone by an Ubbelohde viscometer according to ASTM D 1243-95. The polydispersity index (PDI) were measured by gel permeation chromatography (GPC; ALC, Waters) with a Styragel 6e column, a tetrahydrofuran solution of PVC at 40°C, and 1
Result and discussion
FTIR analysis
The FTIR spectra were employed to characterize the chemical composition of neat LDH, MPTMS-LDH, and pure PVC W-MPTMS-LDH-PVCNC3 (Figure 2). For LDH sample, the wideband at around 3550 cm−1 was attributed to the hydroxyl stretching vibration. The band appearing at 450–800 was recognized for Metal-Oxygen stretching vibration. FTIR spectrum for the MPTMS-LDH sample exhibited the diffraction peaks at 1060 cm−1, corresponding to the functional groups of silane compound i.e. Si–O–C. The peak at about 1620 cm−1 can be attributed to C=C group stretching vibrations in MPTMS. In the case of neat PVC, bands around at 690 and 620 cm−1 were related to the stretching vibration of C-Cl bonds. Figure 2(d) shows the spectra of the W-MPTMS-LDH-PVCNC3 indicating clearly the characteristics of bonds related to both pure PVC and MPTMS-LDH, confirming the reaction between VCM and double bond moiety in anchored MPTMS in MPTMS-LDHs.

Fourier transform infrared (FTIR) spectra of (a) LDH (b) MPTMS-LDH (c) pure PVC (d) W-MPTMS-LDHPVCNC3.
XRD analysis
X-ray diffraction analysis is one of the important techniques for the structural characterization of the crystalline material. Figure 3 shows the XRD patterns of LDH, MPTMS-LDH and W-MPTMS-LDH-PVCNC3 particles.

X-ray diffraction analysis (XRD) pattern of the (a) LDH (b) MPTMS-LDH (c) W-MPTMS-LDH-PVCNC3.
As can be seen, the pure LDH exhibit characteristic sharp peaks located at (2θ) match with the reflection of (003), (006), and (009) crystal planes.25,28
Compared with LDH, MPTMS-LDH and W-PTMS-LDH-PVCNCXs diffraction peaks are significantly shifted to a lower angle which is related to the increase in the inter-planar spacing; d003-value calculated according to Bragg’s law changes from 0.89 in LDH to 1.31 nm and 2.05 for MPTMS-LDH and MPTMS-LDH-PVC, respectively. Once again, it can be concluded that MPTMS molecules inserted between LDH interlayer, can be able to participate in free-radical polymerization reactions of VCM and consequently, further intercalation of LDH with PVC chains.
Basal spacing (d003) of the LDH, MPTMS-LDH and W-LDH-PVCNCXs and evaluated from powder XRD data are listed in Table 1.
d003-basal spacing of the W-LDH-PVCNCX and W-MPTMS-LDH-PVCNCX evaluated from powder XRD data.
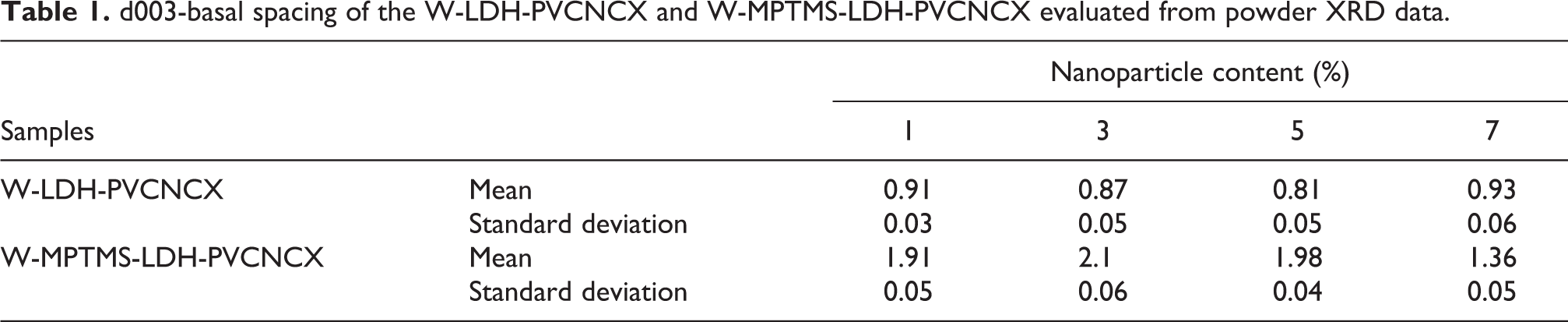
As can be seen from Table 1, Basal spacing (d003) for W-MPTMS-LDH-PVCNCX samples remains almost constant with increasing particles content up to 5%. However, a further increase in particles content up to 7 wt% causes a decline in the d003 basal spacing which can be contributed by the agglomeration of particles within monomer droplets and thus reduction of monomer diffusion into particle into the particle interlayers. For LDH-PVC samples, however, this basal spacing remains nearly constant with particles content and fluctuate around the basal spacing of the LDH.
SEM and EDX analysis
Scanning electron microscope (SEM) images were obtained to further investigate the effect of the interlayer MPTMS molecules and PVC chains on the morphology of LDH, MPTMS-LDH and W-MPTMS-LDH-PVC3 particles (as shown in Figure 4(a) to (c)). As can be seen, both MPTMS-LDH and W-MPTMS-LDH-PVCNC3 particles presented a plate like structure in which the layers have no regular edge shape. The particle size of MPTMS-LDH and W-MPTMS-LDH-PVCNC3 are estimated about 290 and 310 nm, respectively. For understanding the microstructure of the W-MPTMS-LDH-PVC, the TEM image was performed and compared with the MPTMS-LDH particles as shown in Figure 4(d) and (e). As observed, the texture of MPTMS-LDH consists generally of nanostructure regular platelets with smooth edges. The TEM image of W-MPTMS-LDH-PVCNC3 reveals the presence of increased aggregation, lateral association nanostructures layers with two types of intercalation or poor delamination (dark region) and exfoliation morphology (brighter region).

Scanning of electron microscopic (a, b and c) and Transmission electron microscopy (TEM) images (d and e) of selected samples.
Figure 5 shows the EDX quantitative test results for same three samples. The molar ratio of C/Si is 10 in MPTMS molecule (H2C=C(CH3)CO2(CH2)3Si(OCH3)3). Correspondingly, the molar ratios of C/Si would be 9, 8, or 7 if 1, 2, or 3 methoxysilane groups condense with the surface hydroxyl groups on the LDH laminates respectively.35,41 Here, the molar ratio of C/Si (Table superimposed on the Figure 5) was adopted to further confirm the successful grafting of MPTMS. As the C/Si molar ratio of 7.4 was obtained, thus, almost all methoxysilane group in each MPTMS molecule was hydrolyzed and tethered to the metal layers.

Energy-dispersive X -ray spectroscopy (EDX) of MPTMS-LDH.
The EDX test results clearly exhibit that MPTMS-LDH mainly comprised of Mg, Al, O, and Si elements, which is consistent with the above-mentioned tests FTIR and XRD.
PSD analysis
The particle size distribution of LDH, MPTMS-LDH and W-MPTMS-LDH-PVCNC3 are shown in Figure 6(a). We observed that the all types of particles presented a normal distribution; this demonstrated that the as-prepared sample was very uniform. The particle size of MPTMS-LDH-PVCNC3 is larger than MPTMS-LDH and that is larger than LDH; this could be explained by the effect of intercalated MPTMS and PVC chains. The intercalation enlarged the interlayer spacing of MPTMS-LDH and W-MPTMS-LDH-PVCNC led to a corresponding increase in the particle size.

(a) Particle size distribution (b) the content of particles loaded inside the final PVC grains versus the total particles dispersed in VCM phase (c) K-value (d) polydispersity index of PVC nanocomposite grains.
PVC grains
Determination of the particles embedded inside the PVC grains
As particles dispersed initially in the monomer phase were likely to exit from monomer droplets and may tend to move into aqueous phase, it is essential to study the content of particles loaded inside the final PVC grains. Figure 6(b) shows the trends of nanoparticle content of nanocomposites versus the filler loading.
As can be seen from the figure, in all particle contents, the nanoparticle content of nanocomposites of MPTMS-LDH is higher than that of LDH. By increasing the number of dispersed particles, a small decrease (especially in particles percentage more than 5%) can be seen in the case of MPTMS-LDH, while there is a significant increase in the nanoparticle content of nanocomposites of LDH. The reason for this can be explained by the fact that particles aggregate earlier by increasing their concentration in the reaction leads to the formation of more stable dispersions in the media with the opposite nature. When the particles are placed in environments with the opposite character (hydrophobic/hydrophilic), they tend to aggregate into bigger clusters to minimize the surface energy. With enlarging the size of aggregates of organically modified particles, the surface energy decreases, and as a result, the chance of dispersing into aqueous media increases. A similar observation can be seen in the case of unmodified particles dispersion in the organic phase. This phenomenon becomes more pronounced for a concentration of higher than 5%wt. Due to the hydrophobic character of MPTMS-LDH, they are wetted well by organic VC monomer and then become dispersed easily within droplets. For this reason, the final PVC grains prepared in the presence of modified particles exhibited higher residual particles compared with those produced with unmodified particles. The aim of this work to study the influence of the type of treatment and the concentration of particles on molecular and morphological characteristics of the final PVC grains, so each effect will be explained in detail below.
Effect on molecular characteristics
Figure 6(c) shows the variation of k-values with increasing particles for the PVC nanocomposites.
As shown in Figure 6(c), the k-values of PVC samples produced in the presence of MPTMS-LDH particles (MPTMS-LDH-PVCNCX) decreased with increasing particles content, while in the case of the LDH-PVCNCX grains, it remained relatively constant. This can be explained in this way that the organic functionality of the surface of MPTMS modified particles can act as a chain transfer agent and decrease the molecular weights of neighboring polymer chains. On the other hand, chain transfer to particles surfaces becomes more important with increasing inorganic particles content. But at high concentration of particles (more than 5%wt) the k-value slightly increases. The reason for the slight increase of k-values is enhancing the probability of the aggregation of particles at 7 wt% particles. When particles aggregate, the contact surface area between the particles surface and growing polymer chains becomes smaller, thereby decreasing the chance of encountering and chain transfer reactions. It can be concluded that the aggregation of the modified particles occurs at contents higher than about 5%wt. The polydispersity indexes of PVC samples prepared in the presence of both MPTMS-LDH and LDH are shown in Figure 6(d).
It can be clearly seen that MWD is broadened dramatically, using MPTMS-LDH particles. After complete wetting of the particles with the vinyl chloride monomer, a two-region polymerization system is formed; regions near (1) and far away (2) from the surface of the particles (The regions of polymerization formed around a LDH particles within VCM droplet is depicted in Figure 9(d)).
Due to the proximity of free radicals to the organic surface of the particles, chain transfer reactions became more prominent in region 1 relative to region 2.5,38 As the chain transfer to the surface functional groups of particles plays an important role in the termination of the growing chains, the VCM polymerization in region 1 is very likely to produce PVC chains of lower molecular weight than that occurs in region 2. The mechanism of having two different modes of termination is expected to broaden MWD. By increasing particles content, the surface area of region 2 increases, as a consequence, PDI increases. The agglomeration of MPTMS-LDH at higher filler loadings than 5%wt decreases PDI by decreasing the surface area of region 2.
Effect on final particle size
The SEM micrographs of the pure PVC grains and the PVC grains produced with MPTMS-LDH and LDH at 5% concentration are shown in Figure 7(a) to (c).

The SEM photographs showing the appearance of (a, d) pure PVC (b, e) LDH-PVCNC5 (c, f) MPTMSLDH-PVCNC5.
By comparing the micrographs, it can be seen that the MPTMS-LDH-PVCNC5 grains are more uniform in size and spherical in shape than LDH-PVCNC5 and that than pure PVC. As will be discussed in more detail in the next section, primary particles were exposed to an earlier aggregation in polymerization with particles, resulting in the formation of an inner porous, strong network at the initial stages of polymerization, preventing the droplet from collapsing with increasing monomer conversion and from coalescence in sticky stage.5,36–40,42,43 As can be seen from the images, the morphologies of the irregular-shaped particles have started to change into a spherical shape, and the mean size of the particles reduced. The corresponding particle skins are visualized in the micrographs shown in Figure 7(d) to (f).
According to Figure 7, the skin of the MPTMS-LDH-PVCNCXs and LDH-PVCNCXs grains is found to be porous, and the pore size of the latter is larger. To explain the above observations, the formation mechanism of the PVC grains in the presence of particles must be understood. The porous grain skin of PVC grains can be interpreted by the early aggregation of particles preventing the newly formed PVC nano-domains from precipitating at the skin.36,37 As the aggregation of LDH particles inside VCM droplet occurs earlier than MPTMS-LDHs. Early aggregation of LDHs at the early stage of polymerization increases the interstitial pores type of the primary particles where cannot be totally filled by polymer chains and thereby resulting in pores and defects in grain skin. 38 Adsorption of a fraction of suspending agents from the aqueous phase on the particles surface decreases the number of the suspending agent molecules tending to adsorb at the VCM/water interface and thus decreases the tendency to “skin-healing”.5,36,38 Due to higher surface energy and more hydrophilic nature of the LDH particles, larger amounts of suspending agent molecules adsorb onto their surface. Figure 8(a) exhibits the line graph of the mean Sauter mean diameter (D32) of final PVC grains prepared with LDH and MPTMS-LDH.
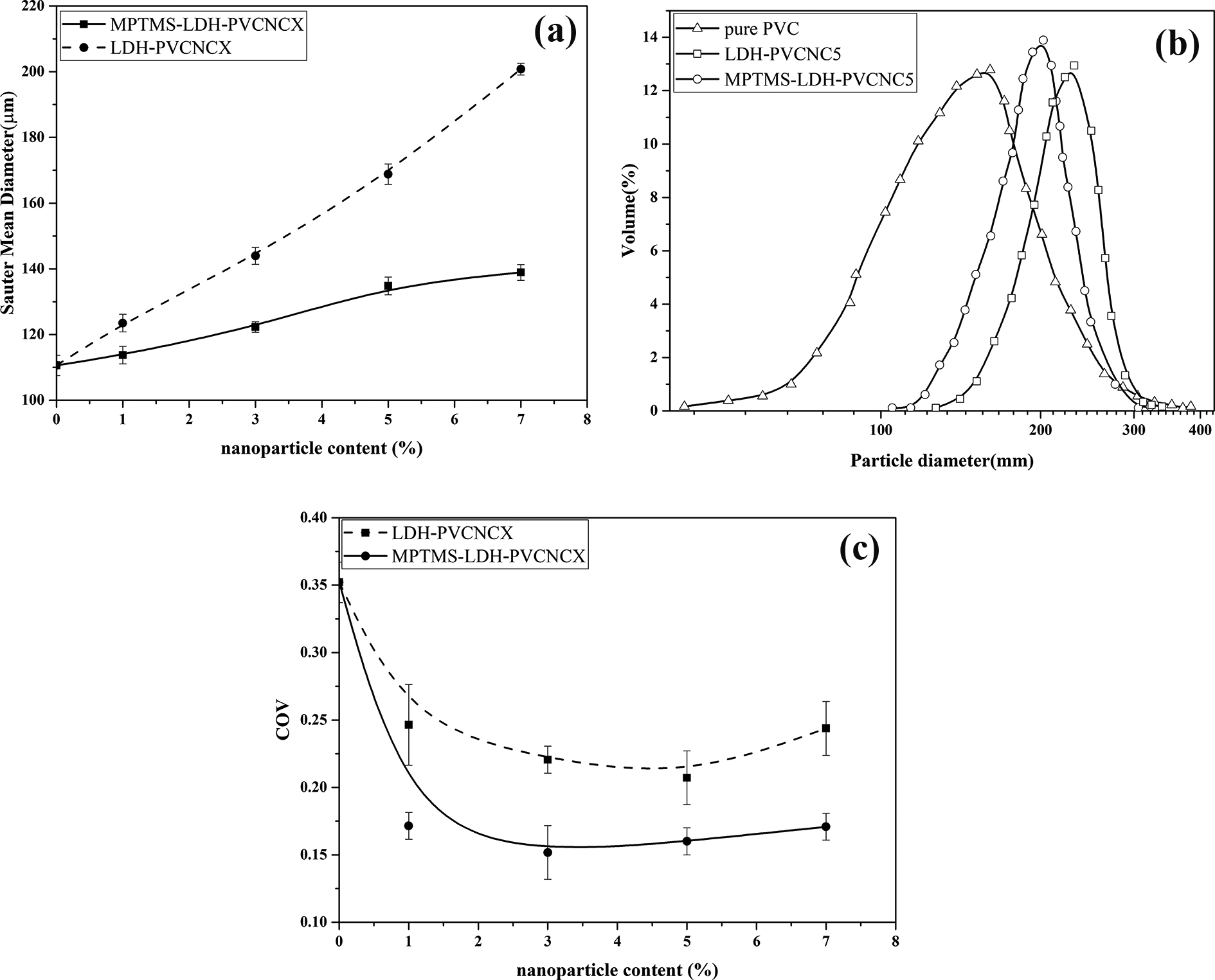
(a) Sauter mean diameter (b) particle size distributions (c) the coefficient of variation of grain size distribution of the samples.
It is found that the final achieved average grain size is influenced by the type and the concentration of particles.2,24,44 It can be observed that, Sauter mean diameter of the final PVC grains shows an upward trend versus the percentage of dispersed particles. This is explicable by the fact that the pre-dispersion of the particles into the monomer causes an increase in the viscosity of the organic phase, thereby facilitating the coalescence and preventing the breakup. Therefore, a higher percentage of both types of particles leads to larger grain sizes, which becomes even more pronounced for the cases produced in the presence of LDH particles.
Figure 8(b) shows the comparison of the particle size distribution of the pure PVC with the LDH- and MPTMS-LDH-PVCNC5 samples.
It is evident from the figure that the PSD of MPTMS-LDH-PVCNCXs is the narrowest, arising from the most uniform distribution of these particles in VCM droplets and consequent grains. The trend of the size distribution of PVC samples with a different concentration of particles is evaluated through its coefficient of variation (COV) and reported in Figure 8(c).
The Coefficient of Variation (COV) is the mean-normalize standard deviation, which is used to describe distribution width. For the VCM suspension polymerization experiments with the addition of both LDH and MPTMS-LDH, no significant change in COV values is observed by increasing the particles. Larger grain sizes, which were seen by the addition of LDH particles, can be attributed to the hydrophilic feature of these particles that hinder their distribution in the hydrophobic monomer phase. One important reason is the adsorption of some of the suspending agents on the surfaces of particles. 5 As the final size of the PVC particles is strongly influenced by the amount of suspending agents used, it is expected that the addition of particles can certainly increase grain size by adsorption of some of the suspending agents on to their surfaces. 38 This phenomenon is more pronounced for LDH particles owing to their higher surface energy. This reason also can justify the observations such as delay in the occurrence of the skin-healing point, and the early motionless point, a small agglomeration of primary particles, and thus a high porosity of grains PVC in the presence of unmodified LDH particles. In general, VCM suspension polymerization in the presence of particles causes a larger grain size as the monomer droplets tend to coalesce more. 36
The effect on the internal structure
Figure 9 compares the SEM micrographs of the fractured pure PVC grains and the LDH- and MPTMS-LDH-PVCNC5 grains.

PVC primary particles at final conversion a) pure PVC b) LDH-PVCNC5 c) MPTMS-LDH-PVCNC5.
The final primary particles size in the MPTMS-LDH
Effect on critical conversion (Xc)
In VCM suspension polymerization, there is a conversion at which the primary particle size remained almost constant. At the critical conversion (Xc), a three-dimensional network of the primary particles is formed within the monomer droplets which influences on skin porosity, the size of primary particles, grain porosity and so on.36,38 Figure 10(a) exhibits the critical conversion for pure PVC grains and the LDH- and MPTMS-LDH-PVCNCX grains.

(a) The critical conversion (b) motionless conversion and (c) CPA of the pure PVC grains and the LDHand MPTMS-LDH-PVCNC grains.
As can be seen, the incorporation of the particles into reaction media, accelerates networking between the primary particles inside the polymerizing monomer droplets. Furthermore, primary particles produced by LDHs creates earlier three-dimensional aggregations with smaller size, thereby leading more rapidly to the generation of the final size and shape of the PVC grains. 36 It should be pointed out that the critical monomer conversion decrease with increasing in particles content leading to an increase in the porosity of the final PVC grains, as discussed in CPA measurements section.
Effect on motionless conversion (Xm)
The motionless conversion (Xm) is the conversion at which the three-dimensional networks of primary particles are formed inside droplets. An experimental methods for Xm determination of the PVC grains was discussed in detail in our previous work. 38 The conversion Xm is an important factor in the morphological development and determining the structure and strength of the primary particle network, skin healing, and the subsequent final porosity of PVC grains.43,45,46 The inner skeleton of the droplets would have been formed at this conversion, and the droplets become stiff enough and cannot be collapsed anymore. 5 The estimated Xm for PVC samples is given in Figure 10(b).
It can be seen that the motionless conversion (Xm) is the most delayed when MPTMS-LDH are loaded in VCM polymerization. The Xm is strongly affected by both the interstitial pores and discrete zones between irregular shaped three-dimensional networks of primary particles.37,38 The presence of particles as the nucleating site for primary particles decreases the interstitial pores types, and increases the discrete zones by early aggregation. As a consequence, the MPTMS-LDH-PVCNC5 samples will have the highest motionless conversion. The Xm is a morphological characteristic of PVC depending strongly on the particles content inside the monomer droplet.5,38 MPTMS-LDH-PVCNCX grains contain a greater content of particles compared with LDH-PVCNCX, and as a consequence, their internal structures are expected to be affected to a greater extent, resulting in a more delay in the occurrence of the motionless point. This means that loading the LDH accelerates networking between the primary particles inside the polymerizing monomer droplets. Under this circumstance, the primary particles undergo an earlier aggregation and form a kind of strong inner skeleton in initial stages of polymerization, preventing the droplet from collapsing too much with increasing monomer conversion. While the aggregation of LDHs inside the PVC droplets creates a solid 3D porous network that acts as nucleation sites for primary particle formation, the well-dispersed MPTMS-LDH particles are rather stable against considerable aggregation and prefer to grow individually with precipitated PVC chains.
Effect on the porosity
The porosity of the PVC grains was measured by the determination of cold plasticizer absorption (CPA) of the resin at room temperature. Figure 10(c) shows CPA versus particles contents for different PVC resin samples. As can be seen, the cold plasticizer adsorption in the MPTMS-LDH-PVCNCXs grains displays a smaller value compared with those formed in the presence of LDHs. The cold plasticizer adsorption (CPA) in the LDH- and MPTMS-LDH-PVCNC7 grains have the highest and the lowest CPA value, respectively.
The CPA of MPTMS-LDH-PVCNCXs shows a decreasing trend while LDH-PVCNCX samples increases with increasing particles content. This can be due to the fact that the existence of the larger mean diameter of the primary particles caused by the earlier aggregation, at low conversions in the LDH-PVCNCX grains, increases interstitial pores and discrete zones between the three-dimensional network of primary particles and thus CPA. The second reason is that it can be noted that the greater number of pores on the skins in the LDH-PVCNCX grains can directly contribute to better accessibility of the internal grain pores and, thus, to the greater plasticizer uptake by the PVC grains. The third reason for increasing porosity is that the LDH-PVCNCX and the corresponding primary particles are less stabilized and, therefore, agglomerate faster, and a porous three-dimensional inner skeleton of smaller particles with larger voids is formed.
Conclusion
A series in situ VCM suspension polymerization was separately performed in the presence of MPTMS-intercalated and pristine LDH in a pilot-scale reactor. The results showed that the reaction between VCM and MPTMS-LDH was effective, with a further intercalation of PVC chains in the interlayer domain and increasing the basal spacing. While the MWD and MW of produced resin in the presence of LDH remained unchanged, MPTMS-LDH broadened MWD and decreased MW. The particle size and particle size distribution of PVC nanocomposite also affected by three factors of the type and the concentration of particles. The loading of particles, especially in the case of LDHs, provided larger particles with a more regular shape and a smoother surface. The skin of the PVC grains prepared with particles is found to be porous, and the pore size of the PVC skin in the prepared by LDH is larger than that in PVC grain prepared by the MPTMS-LDH. The SEM micrographs showed that the adding LDH, results in the formation of an early three-dimensional skeleton with smaller primary particles and subsequently, higher internal porosity whereas, the larger, more uniform, and distinct primary particles with lower internal porosity in the presence of MPTMS-LDH is observed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


