Abstract
This article discusses observations on thermal stability, decomposition properties and degradation of organic components of friction composite materials fabricated by powder metallurgy techniques. N-Methylaniline modified phenolic resin used as a binder material in the preparation of composite materials. Thermogravimetry method was used to study the thermal properties of the samples. The experiments were performed on a TGA Q50 (TA Instrument) in an oxygen atmosphere. In order to better assess the thermal characteristics of the composites, the analyses were carried out by separating thermographs into three parts according to the degradation mechanism. The obtained results helped to assess the thermal stability of the friction materials. The degradation of phenolic resin was observed in the temperature range of 312–362°C. It was found that barite and copper-graphite particles improve the thermal characteristics of the samples.
Introduction
Friction materials are considered the main working element in the brake system. 1 Thus, the quality of the brake material affects the whole performance of the equipment and applications. Contacting pair materials must be able to provide a high friction coefficient during a wide range of sliding speed variations and various normal load regimes. 2 Products made with friction materials are widely used in various technical equipment which is covers important areas of industry. However, the heat generated during friction leads to the wear and destruction of contact materials. 3 The tribological behavior of friction materials depends on the surface characteristics of the brake pads, discs and the so called third body between them. 4 High temperatures cause rapid changes in the mechanical properties of brake materials. The structural changes in the surface layers, the formation of oxide and other chemical films, diffusion processes and etc. are one of the main processes observed between rubbing materials.5,6 As a result, mentioned effects, frictional properties and the nature of the interaction of materials are changes. Tribotechnical materials must not only provide a high friction coefficient and wear resistance and also the high stability of these parameters, which depends on the processes occurring on the surface of the contact materials.7,8
Friction materials are multi-component composites consisting of metallic and non-metallic elements. Commonly, these elements are categorized as binders, fillers, abrasives and fibers.9,10 The main advantages of non-metallic composite materials over metal-based materials are that such materials are technologically lightweight, low density and in many cases have higher strength and stiffness. Besides that, non-metallic matrix composite materials have important properties such as high corrosion resistance, good thermal insulation, antifriction and friction properties. Another important advantage is that they are economically efficient. 8
The growing polymer industry has also had a significant impact on the production of friction materials. Nowadays, asbestos substitutes have begun to be used more intensively.11,12 The progress of nanotechnology has accelerated research into the application of polymer-based composite materials at various friction junctions. Whether they are fibers in terms of new material or new nanoparticles in terms of size, all this has given a new perspective to the production of composite material. 13 Polymer-based compositions can improve properties such as thermal conductivity, higher modulus, hardness and tribological durability, depending on the number of particles at the level of a nanoscale.14,15 Epoxy, phenol-formaldehyde (phenolic resin) and polyamide are the most common polymer matrix materials among non-metallic composite materials. 16 However, the mechanisms of friction and wear of friction composite materials are very complex due to differences its ingredients. Especially, determining the tribological characteristics of polymer-based materials with organic components has its own complexities.
Degradation of organic components from frictional heat effects is one of the main reasons for the reduction of tribological properties in friction brake materials. 17 Thermal degradation causing the effect of “fade” which is decreasing coefficient friction in the early stages of sliding. 18 In addition, the fade phenomena are one of the responsible factors for friction layer formation. 19 The friction layer has significant effects on the tribological performance of friction pair materials. 20
For these reasons, the study of the thermal stability of materials is very important. The main purpose of thermal analysis is to study the changes in the properties of materials during heat transfer and evaluate thermal stability of composites. 21 The basic principle of the thermogravimetry analysis is to record the amount of weight change depending on temperature and time in a controlled atmosphere. The study of the thermal characteristics of friction materials at elevated temperatures as function time can be helpful to predict the tribological behaviors. In the present study, thermogravimetry method was used to determine the relation between the temperature change and the physical properties of friction composites.
Recently, many articles have been devoted to the study of the effect of modified phenolic resin on the mechanical, physical, and tribological properties of composite materials.22,23 However, very few of them have studied the effect of modified phenolic resin content on the thermal stability of brake friction materials. This paper explains N-Methylaniline modified phenolic resin effect on thermal properties of new eco-friendly brake friction composites.
Experimental details
Development of the composites
The friction composite materials were produced by powder metallurgy techniques. Technological sequences such as ball milling, dry mechanical mixing, pressing (pre-forming at 10 MPa and hot-pressing at 25.5 MPa) and post-curing (at 140°C for 5 h) were used in the preparation of the friction specimens (Table 1).
Details of fabricating process of friction samples.

Seven composite materials with the same composition but different weight percentages were used for the study (Table 2). In addition to the components listed in Table 2, 1 wt.% of molybdenum disulfide, magnesium oxide and brass particles (as reinforcing element) were also added to the composition. Glycerin was used as a plasticizer to improve the powder processing properties.
Formulations of friction samples (wt.%).

Phenolic resin modified by polycondensation in an alkaline environment with N-Methylaniline. By the modification process, the amount of free phenol was reduced by five to sic times, and free formaldehyde was reduced by three to four times (Figure 1). 24 Copper-graphite particles obtained by electrochemical method (80%Cu 20%C). 25

Molecular structure of N-Methylaniline modified phenol-formaldehyde.
Thermogravimetric analysis
Thermal analyses were performed to study the mechanisms of decomposition caused by heat effects in composite materials. The experiments were carried out on a thermogravimetric analyzer (TGA Q50, TA Instrument). For comparison, all experiments were performed under the same conditions. The thermogravimetric furnace is connected to a computer-controlled heating and data entry system, which allows recording the variable weight loss in materials at increasing temperatures under dynamic conditions. The specimens were analyzed in an oxygen atmosphere (flow rate: 60 ml/min). A platinum pan was used to heat the powdered samples (25 mg) in the furnace with a heating rate of 10°C/min. Materials were kept in a furnace from 25°C to 1000°C and examined.
Results and discussion
Depending on phenolic resin content some differences were observed in the surface structure of specimens. It is reported in a previous study the strong interfacial adhesion between the components and polymer matrix is very important for obtaining improved mechanical properties. 26 Mechanical properties of polymer composites depend on factors such as the reinforcement or filler relationship with matrix and interface bonding. Optical microscope examinations (AmScope) showed that composites containing 20–25 wt.% phenolic resin have a smooth surface than 15 wt.% (Figure 2). It shows that using 20–25 wt.% phenolic resin provides more effective bonding between components and ensuring good physical and mechanical properties. But depending on formulation phenolic resin may have a different effect on the thermal properties of composites which are discussed below.

Surface images of (a) C-3 (b) C-5 (c) C-1 composites obtained by optical microscope.
Weight loss
The thermogravimetric (TGA) curves showing the changes in the weight of the samples according to the elevated temperatures in the oxygen environment are given in Figure 3. In order to better assess the thermal stability of the composites, criteria were selected according to the temperature-weight loss relationship, and the values corresponding to these criteria are given in Table 3.

TGA curves of composites.
Critical temperature points determined by weight loss.

These criteria include T0 (starting temperature of decomposition), T10 and T20 (temperature for weight loss of 10 and 20%, respectively) and Tmax (maximum weight loss temperature). The higher values of T0, T10, T20 and Tmax, suggest thermally stable properties.
As shown in Table 3 the highest values for T0, T10, T20 and Tmax were mainly in the C-3 and C-7 samples. Although the T20 and Tmax values for C-7 composite were lower than C-3, the weight loss at 1000°C was less in C-7 composite (Table 4). The low T20 and Tmax values can be explained by the low Wt.% of barite and copper-graphite, and the low weight loss at 1000°C can be explained by the high Wt.% of silicon dioxide. If we take a look at the graph in Figure 2, the thermal stability of the composites can be characterized as C-3 > C-5 > C-7 > C-6 > C-1 > C-2 > C-4. This characterization helps to determine how the thermal stability changes depending on components. Low content of copper-graphite can be cited as one of the reasons why weight loss at 1000°C is in C-4 specimen. As main components which are can influence the heat conductivity and thermal behavior of composites, barite and copper-graphite particles, a graph of the dependence between Wt.% and weight loss was established in order to determine the effects of these components (Figure 4).
Percentage of weight loss in specimens investigated at various temperature stages.


Graphs of weight loss depending on (a) barite and (b) copper-graphite content.
Despite the minor differences, it can be seen from the presented graphs that barite and copper-graphite content generally improve the thermal characteristics of the compositions. It was reported that the high thermal conductivity of graphite content helps in dissipating frictional heat from the contact surface of the materials, thus leading to better thermal stability. However, at a lower concentration of graphite, the dissipation process can be poor due to the low physical contact of graphite particles. 27 Small differences can be understood as a combination of different components, the most important of which are phenolic combinations. In order to better analyze the thermal behavior of the composites, the degradation process, and especially the phenol-formaldehyde effect, the thermogram is divided into three main phases, according to the onset and end of significant temperature changes (Figure 3). Table 3 shows the weight loss percentages for all three ranges. First range represents weight loss up to 300°C. In this section, weight loss in the temperature range of 25–300°C was generally up to ∼3% for all samples. At this stage, weight loss can be attributed mainly to the separation of water and gas from organic ingredients. 28 In the second range, weight loss in the temperature range of 300–650°C was in the ∼21–30%. Weight loss was specifically high in the 2nd range for all compositions. The main reason for this is the release of gaseous components such as CO2, H2 due to the degradation of the organic components in the composites as a result of chemical reactions from the effects of temperature. The presence of phenol-formaldehyde has a significant effect on the thermal stability of materials. With the thermal degradation of phenolic resin, the release of a large number of volatile elements causes a change in the structure of the material while binder softening.29,30 In the third range, the weight loss in the temperature range of 650–1000°C was ∼30–36%. Although this is very low compared to the 2nd range, the decomposition process continued at a gradually increasing rate. Weight loss in this range is mainly due to structural changes in the phenolic resin content due to the disruption of the methylene bond. Also, chemical reactions at high temperatures and disruption of molecular bonds result in the decomposition of the components. One of them is molybdenum disulfide. Decomposition of molybdenum disulfide occurs around 540°C to form MoO3 with the release of sulfur dioxide, and sublimation of MoO3 oxide occurs at about 700°C, which has had an effect on weight percentages. 31 At 700°C, graphite is also decomposing with turning into gas. 32 However, depending on the composition of the materials, the decomposition temperature of any component may be different.
If we summarize the data given in Table 3, the average growth rates for each of the three stages were as follows: (1) 1.3%, (2) 23.5% and (3) 7.9%.
Although no significant decrease in the weight of the composites was observed after approximately 700°C, the reduction process continued gradually. However, the low rate of weight loss at this stage can be explained by two reasons: The delaying decomposition process by increasing the thermal conductivity of the composites due to elements such as copper-graphite and barite particles. Glycerin prevents early degradation by preventing thermal oxidation of phenolic resin, which in general serves to increase the thermal stability of composites.
At the same time, it should be noted that no significant weight loss is observed up to 1000°C, because of the high decomposition temperature of other ingredients, like barite, silicon dioxide and metal components. It was reported that the maximum weight loss rate of barite was only 0.15% at 1000°C. 33 Considering this, due to thermally resistant characteristics, the effect of barite on weight loss can be considered very small. However, as discussed above, barite content can improve thermal stability with increasing properties such as heat resistance and thermal conductivity. 34
In addition to the use of modified phenolic resin in experiments, glycerin also had a significant effect on the onset of the degradation process. Thus, if the degradation of phenol-formaldehyde normally occurs at a temperature of ∼300°C, in the samples prepared with modified phenolic resin, this process took place mainly in the range of 312–362°C.
Oxidation
Oxidation of metals during heating in TGA is one of the factors affecting the thermal characteristics. Although weight loss is mainly due to decomposed components, sometimes oxidation can also lead to an increase in weight loss. As the metal oxidizes, the weight of the sample increases as a result of the decomposition or oxidation of the organic components. 35 Since the oxidation of metal elements such as Pb, Sn and Cu in the proposed composition is theoretically a maximum of ∼3–4%, the difference is almost imperceptible due to the increase in weight loss during decomposition of other components. Since this fact is already known in advance, the number of metal elements in the composition is the same for all specimens and wt.% of polymer components were used in different proportions and compared.
In short, the thermal stability of a material with a high oxidation index will also be high. 30 The oxidation index (OI) was determined on the basis of the empirical equation by the number of carbonaceous char (CC) in the residual mass of thermogravimetrically analyzed samples in an oxygen environment at a temperature of 1000°C. 30 The char content is mainly due to phenolic resin. At temperatures above 550°C, the carbonization process of phenolic resin is completed. The coefficient was taken as 0.005875 according to the approximate average value of CC and the OI was calculated as follows:
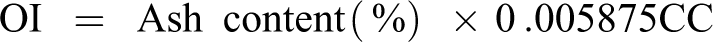
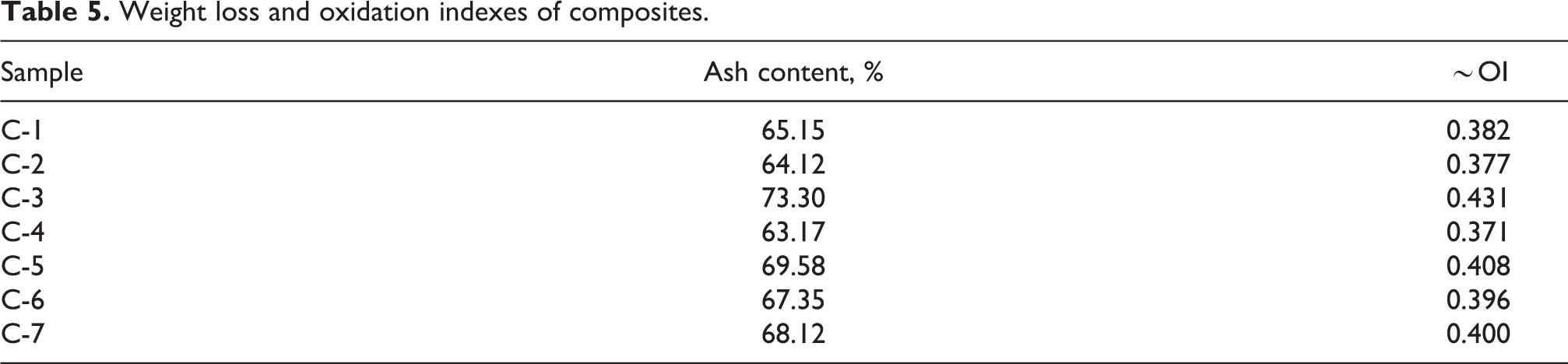
At the end of the experiments, the approximate value of CC was calculated based on the amount of graphite, phenol-formaldehyde and glycerin properly to 25 mg of mass, as it is difficult to determine the carbonized components from the residual materials. The percentage of CC amount that can be carbonized in phenolic resin was determined based on molar mass. The results of the OI are given in Table 5 and Figure 5. As can be seen from the figure, the highest OI were in the samples C-3, C-5 and C-7, which confirms the discussion of the above results.
Weight loss and oxidation indexes of composites.

OI results of specimens.
The formed CC as a result of TGA, is carbon or carbonaceous materials. These materials remain in the chamber at high temperatures because they can no longer break down into volatile components. Char residue is usually formed from polymers with aromatic ring compounds. The greater the number of aromatic rings in the composition, the higher the amount of char will be formed. This process takes place in a stable form, usually up to 600°C, in a nitrogen environment. However, in an oxygen environment, organic molecules burn at lower temperatures, leaving only ash of organic compounds instead. And in a nitrogen environment, all the carbon is removed and a residue remains.
Conlusions
In this study, the thermal stability of N-Methylaniline modified phenolic resin friction composites was investigated using the TGA method. The following conclusion can be drawn from the presented study. The TGA studies have indicates that the thermal stability of composites is in the following order C-3 > C-5 > C-7 > C-6 > C-1 > C-2 > C-4, according to the criteria determined on the basis of temperature - weight loss relationship. The highest weight loss at 1000°C was found in the C-4 sample (63.17%) and the lowest in the C-3 sample (73.3%). Due to the degradation of phenolic resin used in the composition at 312–362°C, the weight loss of the samples was high in this temperature range. The results suggest that barite (30–35 wt.%) and copper-graphite (8–10 wt.%) particles improve the thermal characteristics of the compositions depending on the content percentage. The composite samples with 15 Wt.% N-Methylaniline modified phenolic resin exhibited better thermal stability. The highest oxidation indexes were observed in C-3, C-5 and C-7 specimens.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
