Abstract
Natural rubber (NR) has poor mechanical properties, which limits its practical application. Filler blending is a simple method that improves the inherent properties of natural rubber and expand its applicability. Using the mechanical mixing process, the effects of graphene oxide (GO) and redox graphene (rGO) on the physical properties, electrical conductivity, thermal conductivity, and air permeability of styrene-butadiene rubber (SSBR)/NR composites were studied. The results show that rGO exhibits efficient filler properties in various aspects, for example, the optimal filling amount of rGO and GO was 1.5 phr. In addition, rGO filled SSBR/NR composites showed satisfactory filler dispersibility. Notably, the better dispersibility of rGO was because of fewer hydrophilic functional groups on the surface which were difficult to agglomerate. The increase of rGO and GO content increased the maximum torque (MH) and minimum torque (ML) of the composite material, and decreases tc90 and tc10. The Payne effect of GO/SSBR/NR composites is more obvious than that of rGO/SSBR/NR composites. In addition, we found that the content of rGO (GO) reached saturation at 2phr. Notably, rGO and GO improved the properties of rGO filled SSBR/NR composites such as the tensile strength of rGO/SSBR/NR composites to 23.9 MPa. This shows the potential application of SSBR/NR composites in wearable electronic devices.
Introduction
Natural rubber (NR) exhibits electrically insulating features in nature. Static charges build upon its surface and discharges when it comes into contact with a material with the opposite charge. Therefore, to avoid electrical shock, a conductive rubber is necessary in certain places and products including conveyor belts, factories, and hospitals. 1 To form conductive rubber composites, conductive fillers, among them, carbon fiber, carbon nanotubes, graphenes, carbon black, and other conductive additives are introduced into natural rubber.2–4
For example, studies on graphene with two-dimensional planar honeycomb crystals that form a single layer of sp2 hybridized carbon atoms have largely matured in the recent past. Notably, graphene filled rubber composites harbor excellent mechanical properties, high specific surface area, high electron mobility, and high thermal conductivity.5–10 Also, graphene oxide (GO) carbon layer with oxygen-containing groups including carboxyl and hydroxyl groups at the surface has been of pivotal interest to scholars. It is a promising precursor for mass production of graphene and can be synthesized in bulk quantities by reacting graphite powder with strong oxidants. 11 GO is incompatible with most of non-polar rubber owing to the hydrophilic nature of the oxygen-containing functional groups on its surface.12–19 GO is directly or indirectly reduced on its surface to improve its compatibility with rubber and prevent agglomeration.20–23
Of note, soluble styrene-butadiene rubber (SSBR) is a synthetic rubber formed by random copolymerization of styrene and butadiene monomer. 24 SSBR has the benefits of low temperature, good wear resistance, low fatigue heat generation, as well as anti-wettability, and low rolling resistance.25,26 Further, SSBR has been utilized in making adhesive tread, particularly for high-performance car tires. Natural rubber (NR) is frequently integrated to form SSBR/NR composite material and further improve the processing, performance, and strength of the adhesive tread. 27 Zhu et al. 25 revealed that rGO could improve the mechanical properties, wet resistance and reduce the rolling resistance of SSBR/ NR composites. Similarly, Zhou et al. 5 discovered that GO formed a filling network structure in the SSBR/NR rubber blend thereby improving the tensile strength and toughness of the compounded composites. Whereas rGO and GO can improve the mechanical properties of SSBR/NR composites, their electrical, thermal, rheological, and gas barrier properties are poorly understood.
In the present study, we systematically analyzed the effects of different contents of GO and rGO on the comprehensive properties of SSBR/NR composite. Specifically, the dispersion of rGO and GO in the rubber matrix through the Payne effect and SEM was underscored to identify the effective filling quantity. Subsequently, we comprehensively evaluated the vulcanization properties, basic mechanical properties, airtightness, and electrical conductivity of the rubber composite. Moreover, this work obtained the law of the influence of different contents of rGO and GO on the properties of SSBR/NR composite. Besides, we proposed rGO and GO blocking gas transmission models based on the airtightness of SSBR/NR composite material.
Experimental section
Materials
Natural rubber (NR, SCR10) was purchased from Qingdao Huaxia Rubber Co., Ltd., China. Solution polymerized styrene-butadiene rubber (SSBR, T2000, mass fraction of styrene segment: 25%) was provided by Sinopec Shanghai Gaoqiao Chemical Co., Ltd. Carbon black (N 330) was purchased from Shanghai Cabot Chemical Co., Ltd., China. Reduced graphene oxide (rGO) and graphene oxide (GO) (purity: 99%) were supplied by Shandong Oxalipline New Materials Co., LTD., China. The manufacturer of industrial-grade Zinc Oxide (ZnO) was purchased from Guangzhou Jinchangsheng technology co., LTD., China. Stearic acid (SA), chemically pure, purchased from Tianjin Bodi Chemical Co., Ltd., China. N-Isopropyl-N’-phenyl-4-phenylenediamine(4010NA), n-cyclohexyl-2-benzothiazole sulfenamide (CZ), sulfur(S), microcrystalline wax, and rubber oil were purchased from Sinopharm Chemical Reagent, China.
Preparation of rGO/SSBR/NR and GO/SSBR/NR composites
First, SSBR/NR blend was plasticized using a two-roll mill (XK-160-A, Fujian Yongchun Light Industrial Machinery Factory, China) and the obtained film was then cut into thin strips. SSBR/NR was further mixed using an internal mixer at a rotational speed of 60 rpm for 1 min at 80°C before the addition of rGO or GO, carbon black (CB), stearic acid, anti-aging agent, and rubber oil. The well-mixed compound was then discharged after 5.5 min of mixing and further processed using the two-roll mill. During this processing, sulfur was added and a thin sheet was obtained as the final product. When the composites were cooled to room temperature, it was placed into a moisture-proof bag to prevent the prepared composite from getting damp. The composite was tested after 8 h. The formulations of rGO/SSBR/NR and GO/SSBR/NR composites are given in Table 1.
Formulation of graphene/rubber composites.

phr: parts per hundred of rubber.
Curing time determination and vulcanization for rGO/SSBR/NR and GO/SSBR/NR composites
The vulcanization time of all samples was measured with a rotorless rheometer (M2000FAN, High-Speed Rail Technology Co. Ltd., China) at 150°C for 60 min. About 10 g of samples from each group were cut into spherical specimens. Then, the specimens were sandwiched between two glass papers and placed on the loading table of the Rotor-Free Curing Instrument. The curing time was further determined. It equaled tc90 × 1.3 min.
A Flat-plate vulcanizer (XLB, 25t, Jiangdu Pearl Experiment Machine Factory, China) was used to press the specimens into flat sheets. This was conducted at 150°C and 10 MPa of pressure. After 12 hours of storage at room temperature, the mechanical properties of the vulcanized samples were evaluated.
Characterization and testing
Rubber processing analysis (RPA)
A rubber processing analyzer (RPA-2000, Alpha, USA) was used to measure the dynamic rheological properties of all samples. This was executed under conditions of strain scanning temperature of 120°C, a frequency of 0.1 Hz, and a strain range of between 0.7% and 70%. A sample from each group was cut into a circle of 2 cm radius and 5 mm thickness for reserve. Glass papers were placed on top and bottom of the sample and placed on the carrier of RPA2000. RPA2000 was started, then the test results were recorded.
Tensile tests
The tensile behaviors of the composites were analyzed using an Instron 5967 Universal Testing machine with a 30 kN load cell. The experiments were performed at room temperature (25°C) and normal atmospheric conditions (relative humidity of 20 ± 5%) at a crosshead speed of 500mm/min. The samples of each composite were tested and the average value of the test results was calculated.
Thermal conductivity test
The thermal conductivity of all samples was measured using the laser flash thermal conductivity analyzer (LFA447, Germany Nike Company) at 25°C. A total of samples were randomly cut from a vulcanized sheet (112 mm × 83 mm × 2 mm) prepared from each group. The sample was wiped with absolute ethanol to remove surface impurities and evenly sprayed with a thin layer of graphite to increase the laser absorption of the sample.
Electrical conductivity test
The electrical conductivity of all samples was determined using the capacitance and impedance analyzer (Hitachi, Japan) at a working frequency of 1 × 106 Hz–3 × 109 Hz and alternating current (AC) voltage of 0.1 V (rms). In total, six samples were randomly cut from a vulcanized sheet (112 mm × 83 mm × 0.5 mm) prepared from each group. The samples were scrubbed with absolute ethanol to prevent contact resistance before the measurement of their electrical conductivity.
Gas permeability test
The gas permeability of the composites was determined by the differential volume–variable pressure method using a VAC-V1 Gas Permeability Tester (Labthink Co. Ltd., China) and Oxygen was used as the testing gas. The composite film was cut into a circular specimen of 97 mm diameter. Then, the specimen was placed into the testing chamber with an effective test area of 38.48 cm2. An oxygen (O2) flow of 1 atm was fed on one side of the film sample (upper chamber) while the other side of the film was evacuated (lower chamber) and connected to a differential pressure transmitter. The transmission rate (permeability) was subsequently obtained by analyzing the pressure difference for 12 hours.
Scanning electron microscope (SEM)
The microstructure of GO, rGO, and composite were examined under a field emission scanning electron microscope (FESEM) FSU8020 (Hitachi, Japan) operated under an accelerated voltage of 5 kV.
Results and discussion
Effect of different rGO amounts on vulcanization characteristics of SSBR/NR composites
Both the minimum torque (ML) and the maximum torque (MH) of the SSBR/NR composite increased with the increasing rGO content (Table 2). This was attributed to the increased binding area between rGO and rubber as a result of the grid structure formed by the well-dispersed rGO on the rubber compound. An increase in rGO content increased the viscosity of the rubber compound. This was demonstrated by the increased ML and MH. The scorch time (tc10) and cure time (tc90) of the composites were significantly decreased with an increase in rGO content (Table 2). Although increasing rGO content triggered an increase of the binding area between rGO and SSBR/ NR, it reduced the effect of the added adhesive. The concentration of sulfur and the promoter in the unincorporated glue reduced the coke burning time and positive curing time of the sizing material. Also, the high thermal conductivity of the rGO caused the filled sizing material to absorb heat much faster compared to the unfilled one, thereby reducing the vulcanization time.28–31
Effect of rGO content on rheological properties of SSBR/NR composites.

Effect of different amounts of GO on vulcanization characteristics of SSBR/NR composites
The vulcanization characteristics of GO filled SSBR/NR compound agreed with those of rGO/SSBR/NR composite. Both the ML and MH increased with increasing GO content (Table 3). Nonetheless, the reducing effect of adhesive was not observed in GO filled SSBR/NR compound. This might be due to the different polarity nature of GO and rubber32,33 which caused the GO to be poorly dispersed in the rubber matrix. This was demonstrated by the fluctuating tc10. Moreover, the lower thermal conductivity of GO compared to that of rGO led to lower heat absorption in the GO filled SSBR/NR. This caused the tc90 of GO filled SSBR/NR composite to be longer compared to that of rGO filled SSBR/NR composite.
Effect of GO content on rheological properties of rubber composite.

Effect of different amounts of rGO on dynamic rheological properties of SSBR/NR composites
Figure 1 shows the dynamic mechanical property of the SSBR/NR blends in the presence of rGO. The storage modulus G’ of the SSBR/NR blend decreased with the increase of strain. This signified the occurrence of the Payne effect in the rGO/NR/SSBR composite. The steep slope of the corresponding strain scan curve increased with the increase in the content of rGO in the composite. At the same time,
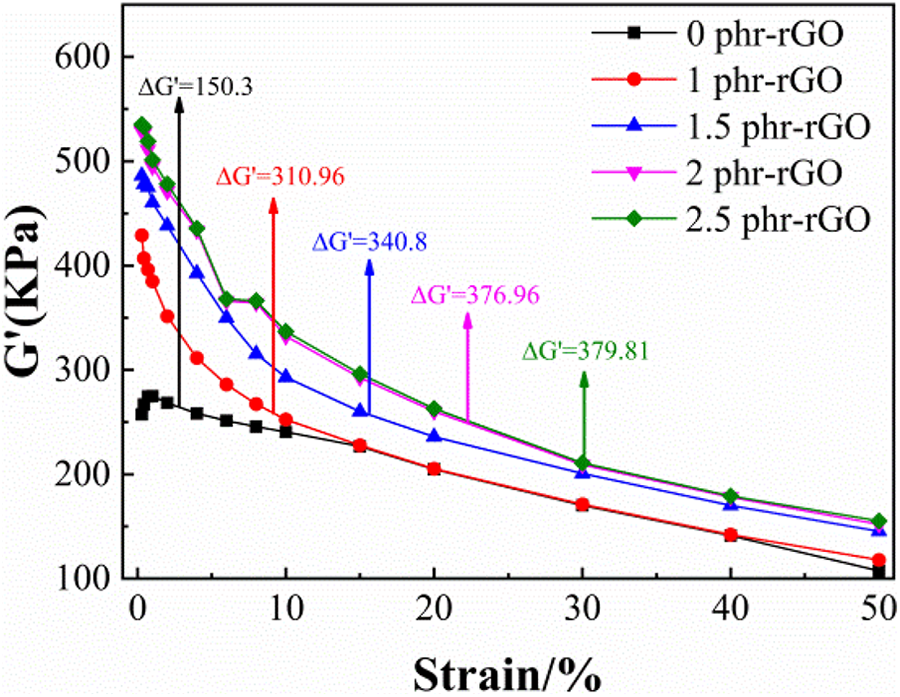
RPA strain scanning of composites prepared with different content of rGO.
Effect of different GO on the dynamic mechanical properties of SSBR/NR composites
The Payne effect of GO/NR/SSBR composite was more apparent compared to that of rGO/NR/SSBR composite because of polar functional groups including hydroxyl group and carboxyl group on the GO surface (Figure 2). The polar functional groups of GO were incompatible with the non-polar rubber matrix thus causing the GO not to be well dispersed in the rubber matrix even with the aid of mechanical shearing. Further, the agglomeration phenomena were inevitable during mechanical mixing. This caused a more significant Payne effect. Moreover, similar to rGO, the RPA curve at 2 phr was consistent with that at 2.5 phr, and the difference was not significant (Figure 2). This was because when the content of GO exceeded 2 phr, the filler network structure in the composite reached saturation. When 2.5 phr GO was added to the complex, the effect became equivalent to that of 2 phr GO.

RPA strain scanning of composites prepared by different content of GO.
Effect of rGO and GO on the mechanical properties of SSBR/NR composites
Figure 3 illustrates the effect of rGO and GO on the mechanical properties of SSBR/NR composite. As shown, the increasing content of rGO and GO in SSBR/NR composites increased the Shore hardness, strength at 300% elongation, and tear strength. Unlike conventional carbon black filled rubber fillers, rGO and GO harbors a large surface area and high surface activity.34,35 Increasing the content of rGO and GO in SSBR/NR composites increased the surface area and active surface thereby causing the rubber to bond and form the grid structure. As such, the Shore hardness and strength at 300% elongation increased the trend with the increasing rGO and GO content. On the other hand, the tear strength of the rGO and GO filled SSBR/NR was linked to the nano-size of the reinforcing fillers. Notably, the tear strength improved when smaller fillers were used. Addition of submicron rGO and GO caused the tear strength of SSBR/NR composite to improve with the increasing rGO and GO content of rGO and GO.
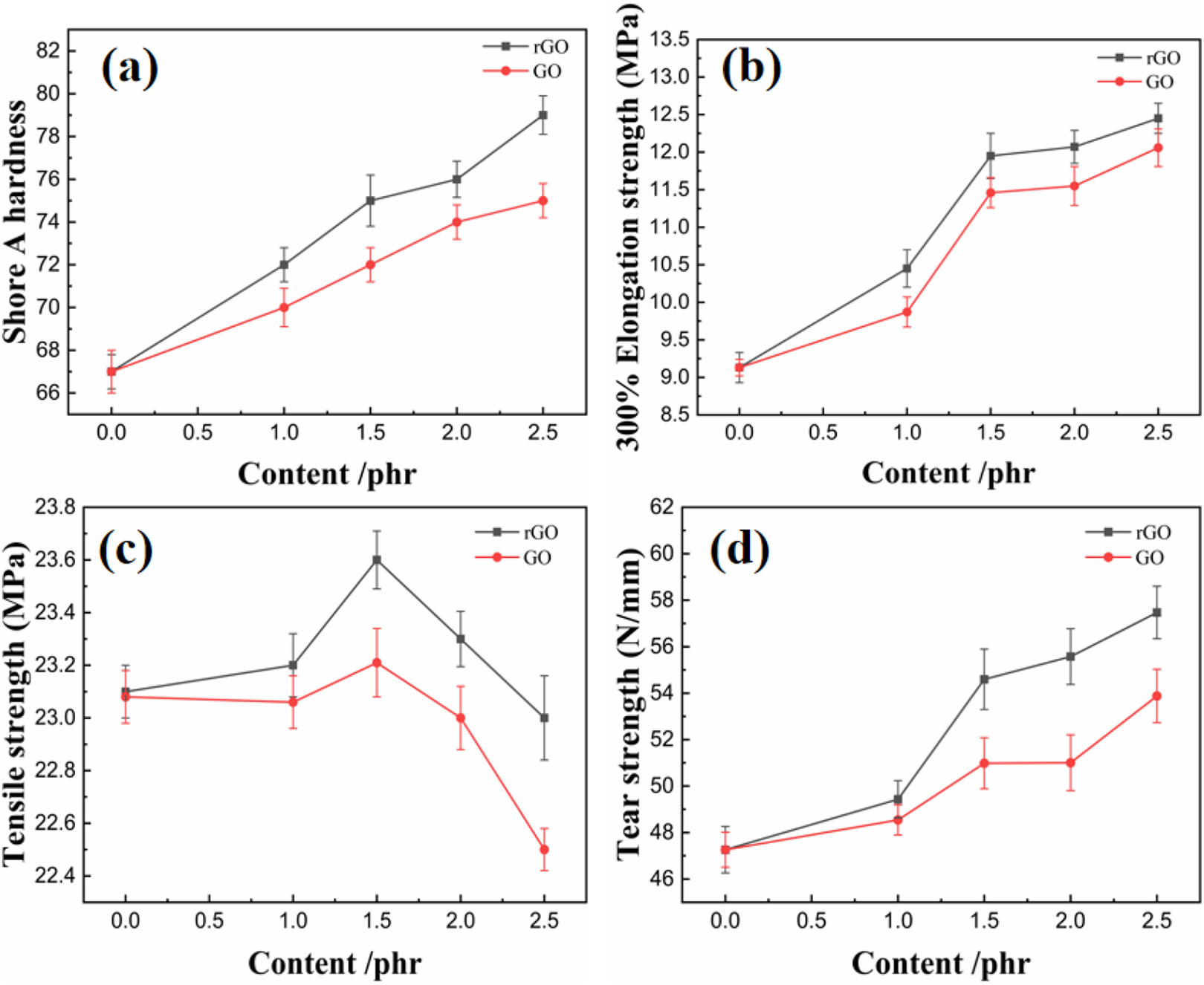
Physical properties of GO and rGO composites: (a) shore A hardness, (b) 300% elongation strength, (c) tensile strength, (d) tear strength.
Interestingly, the optimal tensile strength of both rGO and GO filled SSBR/NR composites was observed at a concentration of 1.5 phr and the tensile strength of RGO/SSBR/NR composite reaches 23.9 MPa. This was attributed to the submicron-sized rGO and GO that readily dispersed at low concentration but agglomerated when its concentration was more than 1.5 phr. The agglomerated rGO and GO failed to bond with the rubber molecules causing a decrease in the tensile strength of the composite.
GO exhibited a lower impact on the physical properties of SSBR/NR compared to rGO. This was attributed to the polarity of GO which made it incompatible with the rubber matrix. As such, a severe Payne effect occurred in the GO filled SSBR/NR compared to the rGO filled SSBR/NR composites. For instance, both the steepest slope in RPA strain scanning and the lowest tensile strength at 2 phr loading of GO filled SSBR/NR pointed toward a higher Payne effect in GO filled SSBR/NR composite.
SEM of SSBR/NR/rGO(GO) composites
Figure 4 shows the SEM images of SSBR/NR composites filled with 1, 1.5, and 2 parts of GO (rGO). Figure 5 shows the SEM images of GO and rGO. As shown in Figure 4, with the increase of GO (rGO) content, the dispersion of GO (rGO) in the SSBR/NR composite matrix became worse. Specifically, the agglomeration increased when the content of GO (rGO) exceeded 1.5 phr. This was similar to the RPA and mechanical properties of the composite. Additionally, compared with the SEM of GO/SSBR/NR composite and rGO/SSBR/NR composite, we found that the agglomeration of GO/SSBR/NR composite was significantly severe. This is related to the oxygen-containing function of the GO surface. As described in Figure 5(b), the surface of GO was curly and the edge was somewhat convex, which was the oxygen-containing group generated in the preparation process of GO. 36 The existence of oxygen-containing groups caused the aggregation of GO in the matrix. Notably, this explains why rGO/SSBR/NR composites exhibit better properties compared to GO/SSBR/NR composites.

SEM of SSBR/NR composites with 1 phr, 1.5 phr, and 2 phr of rGO and GO: (a), (c), and (e) for rGO, and (b), (d), and (f) for GO.

SEM pictures of the rGO and GO: (a) rGO, (b) GO.
Effect of rGO and GO content on the electrical properties of SSBR/NR composites
Table 4 presents the volume resistivity as a function of rGO and GO content for SSBR/NR composite. The volume resistivity of the SSBR/NR composites decreased with increasing rGO content. The decrease was from 9.250 × 1014 Ω·cm to 2.251 × 104 Ω·cm for 0 phr and 2.5 phr rGO respectively. Of note, rGO formed a continuous electrical conducting path in the rubber only when the conductive filler reached a threshold amount of 1.5 phr. At this point, the composite had a decreased resistivity of 6.537 × 106 Ω·cm. The conducting path showed a significant “tunneling effect” with the increasing conductive filler content. As such, the charge carrier shifted from one rGO to the adjacent rGO.37,38 Similarly, GO filled SSBR/NR showed a decreased volume resistivity with increasing GO content. Also, it exhibited a similar filler threshold amount as those of rGO at 1.5 phr though it had a relatively higher volume resistivity. During the preparation of GO, the action of strong acid and strong oxidizer increased the defect area of the original two-dimensional plane, which damaged the connectivity of the π bond on the two-dimensional plane of the original graphene, thereby obstructing the movement of electrons in the plane. Thus, the conductivity of GO decreased or showed electrical insulation characteristics.39–41 Consequently, the volume resistivity of GO filled SSBR/NR composite decreased from 9.250 × 1014 Ω·cm to 4.794 × 105 Ω·cm for 0 phr and 2.5 phr of GO respectively.
Effect of rGO and GO on the volume resistivity of SSBR/NR vulcanizates.

Effect of different contents of rGO and GO on thermal conductivity of SSBR/NR composites
Figure 6 shows the thermal conductivity of the composites prepared with different rGO and GO contents. It is apparent from Figure 6 that rGO and GO effectively improved the thermal conductivity of the rubber composite and with the increase in the content of rGO and GO, the thermal conductivity also improves. This was because graphene was added to the rubber composite as thermally conductive filler, forming a network or chain structure throughout the rubber composite, and the overall thermal conductivity of the composite was improved. Nonetheless, Figure 6 demonstrates that the thermal conductivity of rGO was significantly higher compared to that of GO. This was attributed to two reasons, i.e., on one hand, the GO surface contained more oxygen-containing groups, which could not be evenly dispersed in the rubber, causing uneven coupling of filler and filler rubber, which further triggers the discontinuous heat conduction network structure. This directly influences the thermal conductivity of the rubber compound. On the other hand, due to the loss of a few functional groups in the reduction process of GO, the thermal conductivity of GO was lower compared to that of rGO, which was mutually confirmed by the influence of GO on the physical properties of rubber compound. 42
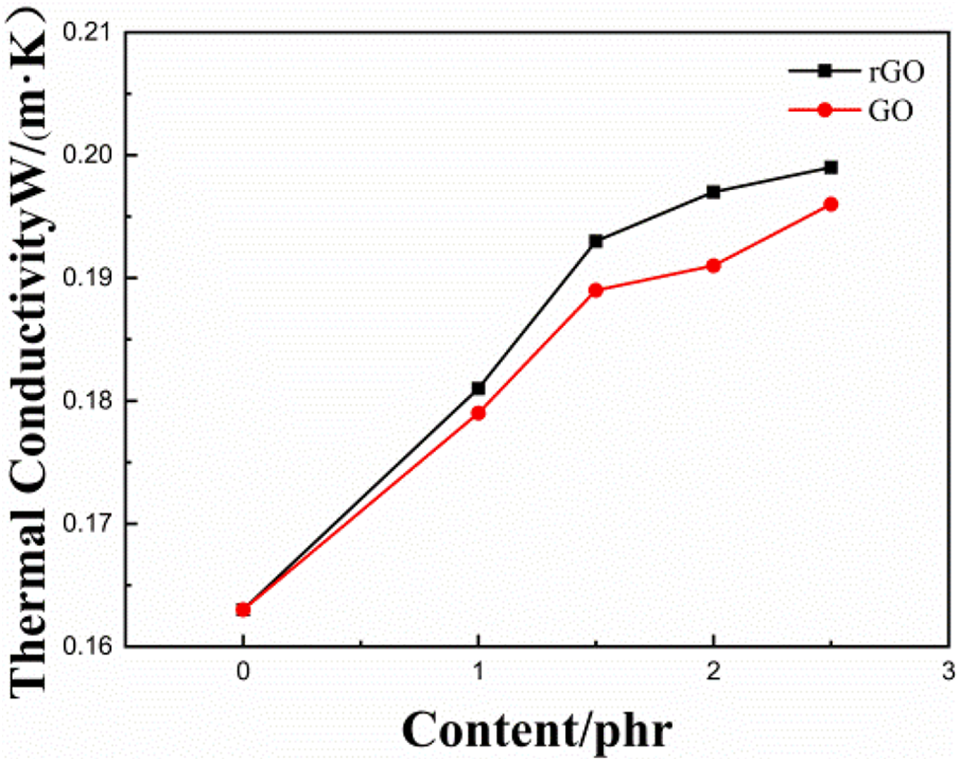
Thermal conductivity charts of composites prepared from different contents of rGO and GO.
Effect of different contents of rGO and GO on gas permeability of SSBR/NR composites
Tables 5 and 6 represent the transmission coefficient parameter tables of rGO (GO)/NR/SSBR composites prepared with different contents of rGO and GO, respectively. As presented in Tables 5 and 6, the addition of rGO and rGO significantly reduced the gas permeability of the composite. rGO and rGO are two-dimensional materials with excellent barrier properties to gas molecules, hence, adding fillers rGO and rGO improves the barrier properties of the rubber compound. Of note, graphene exhibits a large specific surface area. When graphene was dispersed in the rubber compound, two-dimensional rGO and rGO sheets formed a “tortuous” effect in the rubber, preventing direct penetration of gas molecules, thereby increasing the rubber gas barrier. Figure 7 describes a model diagram of the “brick and tile structure” of graphene in the composite material, which intuitively represents the “tortuous road” formed by the graphene sheets in the composite material. The model diagram is a cross-section of the composite material. The oval circle in the figure represents the graphene sheet, and the arrow denotes the direction of the airflow. As can be seen from the figure, when the gas passes through, the “brick and tile structure” formed by the graphene in the composite material can Barrier gas molecule penetration, improve the gas barrier properties of composite materials.34,35,43

The “brick and tile structure” formed inside rGO and GO filled SSBR/NR.
Effects of different contents of rGO on gas barrier property of SSBR/NR composites.

Effects of different contents of GO on gas barrier property of SSBR/NR composites.

Conclusion
In conclusion, this article exposes the composite material filled with styrene-butadiene rubber (SSBR) and natural rubber with different contents of reduced graphene oxide (rGO) or graphene oxide (GO). Moreover, the mechanical properties, viscoelasticity, electrical conductivity, thermal conductivity, and air permeability of composite materials were analyzed. The results reveal that reduced graphene oxide and graphene oxide exhibit different degrees of dispersion in rubber composites with different contents. On one hand, when the content of reduced graphene oxide and graphene oxide was 1.5 phr, the dispersion effect was better. On the other hand, when the content of reduced graphene oxide and graphene oxide exceeded 1.5 phr, a large amount of agglomeration occurred, which affected the performance of the composite material. As the content of reduced graphene oxide and graphene oxide increased, the composite material exhibited the Payne effect; The tensile strength of SSBR/NR composites increases with the increase of rGO and GO content. The electrical conductivity, thermal conductivity, and airtightness of the composite material increased with the increase of GO (rGO) content, while the electrical conductivity threshold of the composite material was 1.5 phr. In summary, the application of rGO filled SSBR/NR composites in wearable electronic devices is largely promising.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NSFC-Shandong Joint Fund for Marine Science Research Centers [grant no. U1606401], the Qingdao Science and Technology Development Plan [grant no. 17-6-3-16-gx] and National Natural Science Foundation of China [grant no. 51803010].
