Abstract
Core-shell particles were synthesized employing seeded suspension polymerization. Suspension polystyrene (PS) particles were used as seeds, and poly(methyl methacrylate) (PMMA) was synthesized in the presence of polystyrene seeds. A combination of solvent extraction, Fourier transform infrared spectroscopy, scanning electron microscopy, and transmission electron microscopy techniques were employed to study the structure and morphology of synthesized particles. The results suggested a
Introduction
Core-shell particles (CSPs) are structured composite particles consisting of at least two different components, one forming the core and the other forming the shell of the particles. 1 CSPs are usually prepared by various polymerization strategies such as seeded suspension polymerization.2−4 seeded (mini)emulsion polymerization,5−7 and dispersion polymerization. 8 According to preparation methods, they can be prepared in a spherical form in nano and micron sizes. This class of materials has attracted much attention because of the combination of superior properties not possessed by individual components. CSPs have been used in many applications, including impact modifiers, surface coatings, pressure-sensitive adhesives, printing, catalysis, pollution control, sensing, and drug delivery.9−12
Seeded suspension polymerization is a multi-phase polymerization that produces unique beads with a radial composition profile. Using this process, suitable properties of one polymer can replace the weaker properties of the other. The production of particles with core-shell morphology using seeded suspension polymerization has been examined in a few studies.2−4,13,14 Gonçalves et al.,2,3 for instance, used seeded suspension polymerization with different strategies to synthesize core/shell particles with polystyrene (PS) cores. They reported that the shell thickness and amount of monomer incorporated as a polymeric shell to the seed particles were strongly influenced by the water solubility of the second monomer and swelling time; the more hydrophobic the monomer and the higher the swelling time, the thicker the shell and the higher the fraction of the second monomer formed inside the seeds as a shell.
Moreover, Heydarpoor et al. 4 used seeded suspension polymerization to synthesize expandable core/shell particles having PS cores. They observed parallel regular and onion-like particle morphologies and concluded that the morphology of beads, blowing agent content, and distribution are influenced by the type of the second monomer(s).
In addition, Besteti et al. 15 utilized combined semi-batch emulsion/suspension polymerization to synthesize core-shell particles. They concluded that the morphology of the shell depended on the glass transition temperature (Tg) of emulsified particles. Furthermore, a thin film was formed around the core when the Tg of the polymer constituting the shell was lower than the reaction temperature, while a porous shell was formed when the Tg of the polymer material was higher than the reaction temperature.
In the present study, PS-PMMA core-shell particles were synthesized by seeded suspension polymerization. Suspension PS particles were used as the seed and methyl methacrylate was employed as the second-stage monomer. The importance and the industrial interest of core-shell structures require a detailed study of the morphology and structure of these particles. Therefore, for the first time, the selective solvent extraction method was combined with Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), and transmission electron microscopy (TEM) analyses to study the morphology and structure of the synthesized core-shell beads.
Experimental section
The formulation used to synthesize PS and PS-PMMA particles via suspension polymerization.
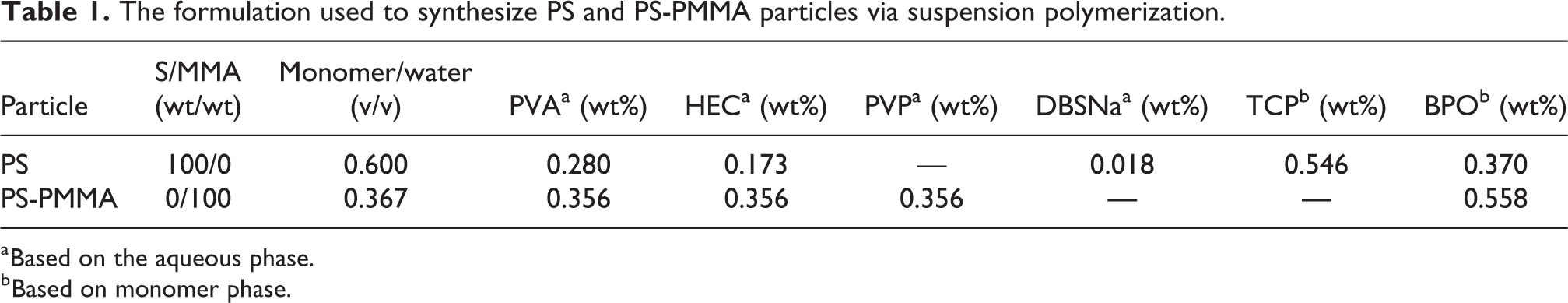
a Based on the aqueous phase.
b Based on monomer phase.
PS-PMMA core-shell particles were synthesized in a semi-batch seeded suspension polymerization system. For this purpose, PS seeds (with the diameter of 300–420 μm) and the solution of PVA, PVP, and HEC in water (Table 1) were charged into the reactor and the mixture was heated to 50°C with moderate mixing (380 rpm). Then, the MMA monomer solution containing BPO was fed to the reactor at either 0.311 or 0.580 mL/min feeding rates. Afterward, the temperature was increased to 80°C and polymerization was continued for 8 h under agitation at 950 rpm. After the completion of the reaction, the reactor was cooled to ambient temperature. The reaction mixture containing PS-PMMA core-shell beads was washed with water and dried at room temperature. In this study, the PS-PMMA particles with a 420−600 µm diameter range were used for structure characterization and morphological studies. The ratio of MMA to PS seeds was 47/53 wt/wt.
Results and discussion
PS seeds characteristics
PS seeds were synthesized using both water-soluble (PVA or PVP) and dispersed (TCP) stabilizing agents. It is known that a degree of hydrolysis in the range of 70–80% for PVA provides good droplet stabilization in aqueous media. 21 Stabilizers improve the dispersion stability by not only increasing the viscosity of the aqueous phase, but also prominently preventing the coalescence of polymer particles by covering the surface of particles (steric stabilization). 22 PVA can also be grafted on the polymer, which is formed inside the droplets, forming a skin on the surface of final particles. 23 The presence of stabilizers and suspending agents can be considered as contamination in the resultant products in suspension polymerization. However, the stabilizers and suspending agents that are not grafted on the surface of particles can be removed from the final particles by washing with water or a dilute acid solution.24,25
Generally, the final particle size distribution (PSD) in suspension polymerization depends on the type and concentration of surface-active agents, quality of agitation, and physical properties (e.g. density, viscosity, and interfacial tension) of continuous and dispersed phases as well as the monomer-to-water ratio.19,26 On the other hand, the solubility of the polymer in monomer during polymerization in dispersed media leads to the softness of polymer particles. The stability of the softened particles in suspension polymerization and the acceptable appearance and shape of final products cannot be provided using only one stabilizing agent.27−29 Clearly we see that, using just one hydrophilic stabilizer or changing agitation condition such as speed or mixer displacement leads to none-spherical shape. Having spherical particles is crucial to provide meticulous information from particles morphology. Therefore, usually, a combination of various suspending agents and stabilizers with a proper agitation is used in suspension polymerization.
After synthesizing PS seeds from suspension polymerization, to have a precise PSD diagram, the particles were washed with 2 M hydrochloric acid solution for 1 h and finish washing with distilled water to remove any residual stabilizers from the surface. The PS seed particles had a wide particle size distribution with a maximum in the range of 300–420 µm (Figure 1), which was employed as seeds in core-shell polymerization. The mean particle size and particle size polydispersity index for the PS seeds were 463 µm and 1.1337, respectively.

Particle size distribution for PS seeds.
Thermal gravimetry analysis from PS particles (Figure 2) shows that nearly 4 wt% of monomer and volatiles are inside the PS seed particles. The residual monomer inside the particles would decrease the glass transition of particles and could make some unexpected matters during the core-shell polymerization.

TGA diagram for pure PS particles used as seeds in core-shell polymerization. Inset: the weight loss (∼3%) in the temperature range of 80–240°C.
Core-shell particles characteristics
Particle size distribution
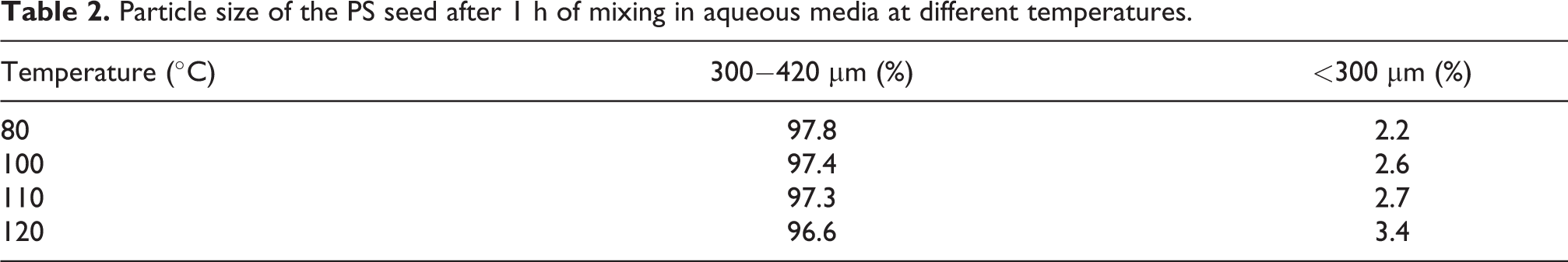
Figure 3 shows the size distribution of PS-PMMA core-shell particles obtained from seeded suspension polymerization. The mean particle size of the synthesized core-shell particles using both 0.580 and 0.311 mL/min MMA feeding rates was about 450 µm. Moreover, the PSD of the core-shell particles showed a low fraction of small particles (about 0.11% for 0.58 mL/min and 0.53% for 0.311 mL/min MMA feeding rates) (Figure 3). These small particles were probably the result of the residual monomer in PS seeds (about 4 wt% determined by TGA measurement). To understand the origin of these small particles, the PS seeds which were used to synthesize core-shell particles were mixed with aqueous phase in a nonreactive medium (without initiator) in the reactor for 1 h at different temperatures (80−110°C). Table 2 shows the PSD results. It can be seen that, in all experiments, the fraction of small particles was below 3 wt%. Therefore, the formation of small particles in core-shell products is most likely due to residual monomers in the particles, which led to the formation of flabby particles. 19 Moreover, the diffusion of monomer into polymer seeds in the second step of polymerization reduced the glass transition temperature of polymers, increasing the probability of particle breakage by the shear force of agitation. The formation of secondary particles (during MMA feeding step) may be another reason for these small particles as previously reported by Gonçalves et al. 3
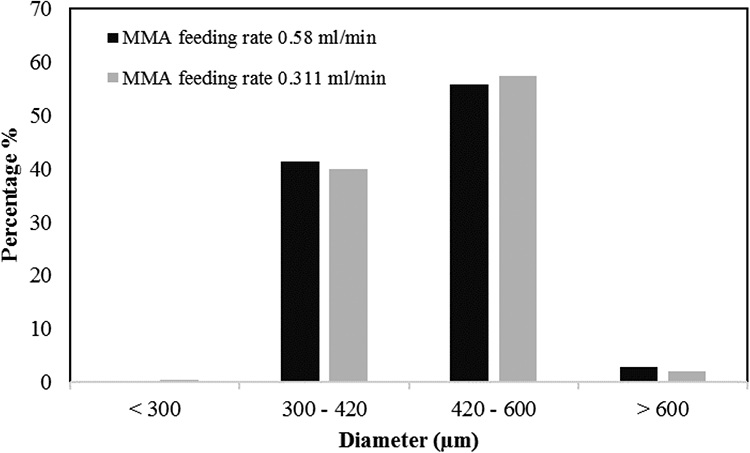
Particle size distribution of PS-PMMA core-shell particles obtained with different monomer feeding rates.
Particle size of the PS seed after 1 h of mixing in aqueous media at different temperatures.
Structure and morphology of the synthesized PS-PMMA particles
The morphology and structure of the core-shell particles were studied by a combination of dissolving particles in a selective solvent, SEM, and TEM techniques. The selection of a good selective solvent is important in using FTIR analysis. Figure 4 illustrates the FTIR spectrum of the liquid phase obtained from mixing PS seeds in acetic acid for 1 h at 99°C. The absence of absorption bands in 700−750 cm−1 confirms that acetic acid is a non-solvent for PS.
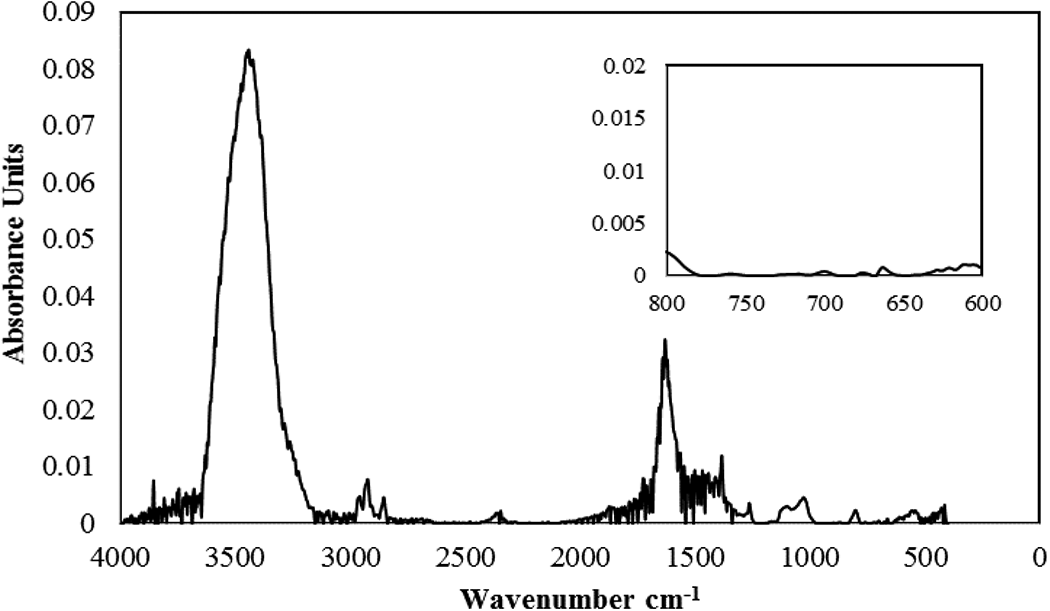
FTIR spectrum of the liquid phase obtained from exposing PS seeds to acetic acid for 1 h at 99°C. Inset is a magnified spectrum in the range of 600−800 cm−1.
As mentioned before, two types of experiments were carried out to study the structures of particles using the polymer extraction method. In the first experiment, core-shell particles were mixed in acetic acid for 80 min while samples were taken every 10 min. In the second experiment, particles were mixed in acetic acid for 10 min and then filtered and dried in a vacuum oven for 4 h at a high temperature to eliminate the solvent residue. Afterward, the dried particles were mixed with acetic acid for another 10 min and then filtered and dried in a vacuum oven for 4 h at a high temperature. This process was repeated 8 times. In the first experiment, the clear acetic acid changed to an opaque solution due to the presence of PS and PMMA chains in the solution. The FTIR spectra of the taken sample confirmed the presence of both PMMA and PS in the acetic acid solution (Figure S1). Therefore, some PS chains, together with PMMA chains, immigrated from the particles into the solution. In the second experiment, due to the elimination of acetic acid in each step (mixing every 10 min), the diffusion of the solvent to core-shell particles and therefore polymer extraction were expected to decrease substantially. The particles extracted by this method showed a bumpy and rough surface (SEM and optical microscopy images presented in Figures 5 and S2, respectively). Therefore, the structure and morphology of core-shell particles were studied by the first extraction method.

SEM images of PS-PMMA core-shell particles after (a) 3, (b) 4, and (c) 5 steps of extraction by the second method.
The mean particle size of the core-shell particles before and after extraction using the first method was calculated by measuring the diameters of more than 100 particles using the Image Analyzer software and summarized in Table 3. The SEM images of core-shell particle surfaces being held in acetic acid for 10, 20, 30, and 40 min are depicted in Figure 6, and the SEM images of the cross-section of the corresponding extracted particles are given in Figure 7 (The particles were cut in half under liquid nitrogen). Figure 6(a) shows that the diameter of particles decreased after 10 min of extraction in solvent due to polymer dissolution, whereas the diameter of particles after 20 min of extraction increased due to the diffusion of the solvent inside polymer particles and particle swelling. Moreover, it seems that solvent diffusion and particle swelling increased with extraction time (Figures 6(c) and (d)). Besides, the shape of particles was deformed with increasing the extraction time, most probably because of the easier movement and diffusion of PMMA chains inside the particles, which led to particle shape deformation.

SEM images of PS-PMMA core-shell particles extracted by the first method for (a) 10, (b) 20, (c) 30, and (d) 40 min.

SEM images from cross-sections of PS-PMMA core-shell particles extracted by the first method for (a) 10, (b) 20, (c) 30, and (d) 50 min.
Mean particle sizes of PS seeds and PS-PMMA core-shell particles before and after solvent extraction through the first extraction.

Solvent diffusion and polymer extraction can be observed more obviously in the SEM images of cross-sections (Figure 7). Based on Figure 7(a), 10 min of extraction was not enough for solvent diffusion inside the particles, while the solvent diffusion and swelling of polymer particles were completely clear after 20 and 30 min of extraction times. On the other hand, it can be observed that extraction for 50 min was enough for extracting some parts of PMMA (the presence of holes in Figure 7(d)), but the main part of the shell and PMMA chains presented in PS-rich domains were not yet extracted. It was reported that the average molecular weight of PMMA in core-shell particles is almost 5 times higher than the PMMA obtained in unseeded batch suspension polymerization due to high polymer concentration and diffusional limitations (gel effect) that take place from the beginning of the second step of polymerization, decreasing the termination rate. 4 Therefore, the high-molecular-weight PMMA synthesized in the second step of polymerization needed more time to be completely extracted.
To shed light on the structure of core-shell particles, they were extracted for 2 h in acetic acid. Due to the adequate extraction time, it was expected that the shell, which was supposed to be formed with pure PMMA, would be completely dissolved into acetic acid. Surprisingly, however, the SEM images of these extracted particles (Figure 8) show that the shell remained unchanged even after 2 h of solvent extraction and the particles exhibited a core-shell structure (Figures 8(a)−(c)). Since acetic acid has a high selectivity to PMMA (PS is not soluble in acetic acid, Figure 4), the outer shell layer was expected to be PS. The ATR-FTIR analysis of the shell (Figure 9) shows that the intensity of the PS characteristic absorption peak was substantially higher than that of PMMA. Besides, Figures 8(a) and (b) demonstrate the presence of hollow particles. The PS central core in some particles may have jumped out of the particles. Figure 8(c) presents the PS core and outer shell of the particles after 2 h of solvent extraction. This figure clearly shows that the particles were swollen and their size increased during solvent extraction process.
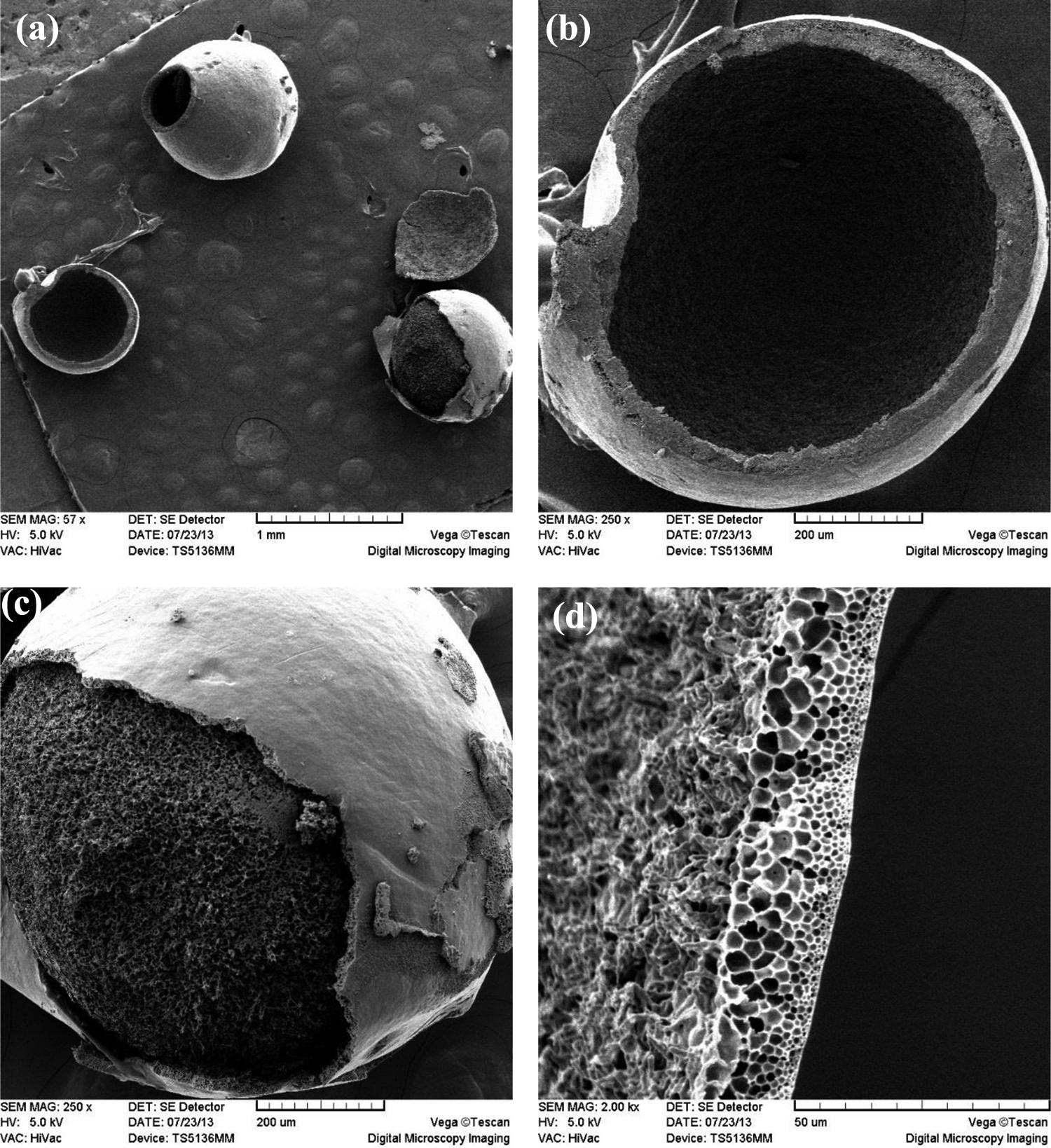
SEM images of (a), (b), (c) PS-PMMA particles, and (d) cross-section of the outer shell of PS-PMMA particles after 2 h of extraction in acetic acid using the first extraction.
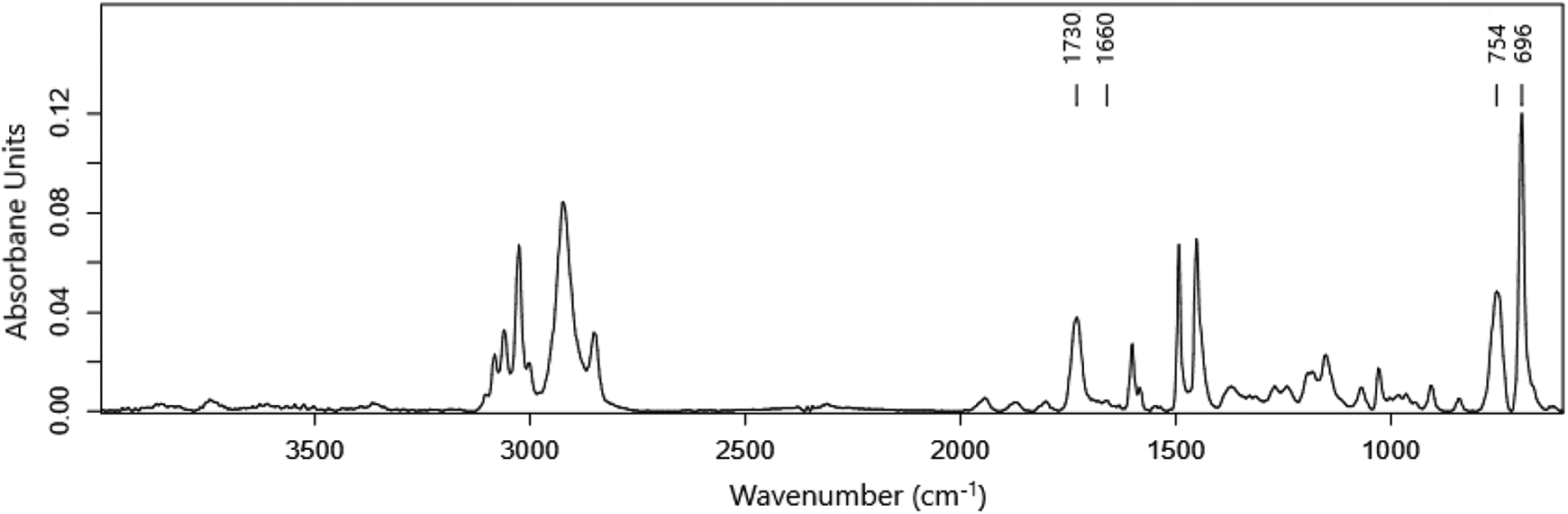
ATR-FTIR spectrum of the outer shell of PS-PMMA particles after 2 h of extraction using the first extraction. The peaks appearing at 700–750 and 1720−1740 cm−1 belong to PS and PMMA, respectively.
To better understand the morphology of the particles and see whether or not the outer shell of the particles was PS, the particles were extracted for 48 h at a high temperature in cyclohexane, which is a good selective solvent for PS (PMMA is not soluble in cyclohexane and the characteristic peak of PMMA, around 1720−1740 cm−1, is absent in Figure S3). The SEM micrographs of the extracted particles are shown in Figure S4. The polymer shell was not dissolved in cyclohexane, while the PS core completely disappeared, confirming that the polymer shell(s) were less likely PS.
The SEM image of the cross-section of the outer shell of a particle extracted with acetic acid is illustrated in Figure 8(d). It is seen that solvent diffusion and PMMA extraction formed a cellular structure across the shell with increasing the pore size in the path of going into the seed part. Based on the solvent extraction and SEM analysis results, the particles most probably included a PS core, an inner shell containing PMMA-rich domains, and an outer shell containing PS-rich domains, as schematically shown in Figure 10. The solvent extraction of PMMA-rich domains in the inner shell together with the high temperature and shear forces are the most likely reasons for the hollow particles formed during solvent extraction (Figures 8 (a) and (b)).

Schematic representation of the synthesized PS-PMMA particles.
TEM analysis is a useful technique to study precisely the distribution of PS and PMMA domains in the outer shell layer. Staining by OsO4 provides enough contrast to distinguish PS and PMMA domains in a PS-PMMA composite polymer shell. The TEM micrographs of the ∼15 µm thick outer shell of the particles synthesized at 0.58 mL/min monomer feeding rate are displayed in Figure 11. It can be observed that the outer shell was covered with PS, and PMMA brighter domains were dispersed in the PS matrix. Also, the size of the PMMA domains decreased by going toward the inner shell. The same trend was observed for the particles synthesized at a lower monomer feeding rate (0.311 mL/min, Figure 11). Moreover, the higher monomer diffusion time to synthesize core-shell particles at 0.311 mL/min led to a deeper diffusion path. The same behavior was reported by Gonçalves et al. 2 It is worth noting that, in both cases, the well-defined core-shell morphology was not obtained (Figures 11 and 12) as the surface of the particles was composed of PS with PMMA domains below the surface. This kind of morphology was achieved because the system was not under thermodynamic equilibrium as the polymerization rate was much faster than the diffusion of PMMA/MMA in the swollen seed particles. This resulted in the formation of PMMA domains close to the surface of the particle instead of a continuous PMMA shell.

TEM micrographs of outer shell cross-section of a PS-PMMA particle with 420 µm diameter. The feeding rate of MMA was 0.58 mL/min. The TEM micrographs toward the seed part are shown from (a) to (d).

TEM micrographs of outer shell cross-section for a PS-PMMA particle with 420 µm diameter. The feeding rate of MMA was 0.311 mL/min. The micrograph (a) corresponds to the area close to the particle surface. The TEM micrographs toward the seed part are shown from (a) to (d).
Conclusions
Core-shell particles were synthesized through seeded suspension polymerization. Suspension polystyrene particles and methyl methacrylate were used as seeds and second-stage monomer, respectively. The synthesized core-shell particles showed a narrow particle size distribution, and the feeding rate of the second monomer did not affect the mean particle size of the particles. The particles presented a complex morphology with a suggested core-inner shell-outer shell structure. Therefore, the PS core was covered by a PMMA-rich inner shell and a very thin outer shell composed of PS-rich domains. The shell thickness and PMMA domain distribution in the polymer shell(s) were influenced by the strategy adopted for the monomer feeding into the system; the higher the swelling time, the higher the MMA diffusion toward the PS core and the higher the shell thickness.
Supplemental material
Supplemental Material, supporting_information - Insight into the structure of polystyrene-poly(methyl methacrylate) core-shell particles synthesized by seeded suspension polymerization
Supplemental Material, supporting_information for Insight into the structure of polystyrene-poly(methyl methacrylate) core-shell particles synthesized by seeded suspension polymerization by H Rostami, F Abbasi, K Jalili, E Mehravar and M Najafpour in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
