Abstract
The present study described reduces of plastic based non-biodegradable food packaging material and sustainability toward the environmental protection. The polyvinyl alcohol (PVA) based ternary blend biodegradable films with bio-materials (corn starch and pectin) in ratio 1:5, 1:2, 1:1 and 2:1 were synthesized by solution casting method in water as a solvent to improve the tensile strength, high % elongation at break, and sustained packaging properties. Citric acid and glycerol were used for cross-linking and plasticizing purposes which are also bio-degradable and non-toxic. Blend films were characterized by FTIR and checked out the cross-linking of different components. The dispersion of contents in films was analyzed by SEM images. Thermal stability and degradation behavior of casted films were studied with thermogravimetry and thermal stability increased in blends as compared to neat PVA film. Biodegradability of cast films was also checked by Soil Burial method and bio-degradation increased with time i.e. almost 50% degrades in 30 days and 68% after 90 days. Tensile properties of blend films were tested and found that tensile strength (18.85 MPa) and elongation at break (516%) are high in blend films as compared with neat PVA film (12.04 MPa, 170%). The films are eco-friendly and biodegradable, might be a replacement of plastic material in food packaging having improved tensile properties.
Introduction
Petrochemical-based polymers are frequently used in food packaging films because of their wide availability, cheap industrial manufacturing, favorable mechanical properties, heat seal-ability and good barrier properties to water and oxygen.1-3 Most of these polymers are non-degradable in the soil and cause severe environmental problems and hence it is going to be banned for their use in straw pipes, carry bags, and packaging materials. Therefore, new eco-friendly materials have been studied to replace such non-degradable materials. 4 Research has been already shifted in this area and binary composites of natural polymer with synthetic degradable polymers like Polyvinyl alcohol (PVA), polylactic acid (PLA), polycaprolactam (PCL) have been developed by many workers.5-8 Among these, PVA is immensely used due to its easy availability, non-toxicity, water solubility, and thermal stability.9,10 Also, PVA has a good film-forming ability, better oxygen barrier property, and better tensile strength as compared to other synthetic biodegradable polymers.3,11,12 PVA has a relatively high cost, limited flexibility, low elongation at break, and low degradation rate in soil. 13 To make PVA films more reliable in commercial use, PVA is blended with natural polymeric materials, which are intended to increase mechanical performance, economic efficiency, and biodegradability.14,15 The natural polymers, especially pectin and starch are progressively used in PVA based binary blends.
Both pectin and starch are edible, inexpensive, amorphous, non-toxic and natural polysaccharides. Pectin is extracted from several types of fruits, with chemical or enzymatic methods and used in the food industry as a thickener and stabilizing agent. 16 It is also safe for the environment due to its biodegradable nature and PVA/pectin composite were fabricated in modern research due to strong interaction between hydrophilic PVA and hydroxyl groups present in the backbone of pectin. Coffin and Fishman studied PVA/Pectin blends and found a greater impact of composition and lesser impact of the degree of hydrolysis of PVA on mechanical performance and reported pectin/PVA blends. 17 Fishman and Coffin reported enhanced solubility of PVA/Pectin blends as compared to PVA. 18 Further, starch is most commonly used in blend films as it is a biodegradable polymer derived from a surplus commodity. 3 Starch is a natural polymer produced by plants in the form of granules, consisting of linear polymer amylose (20–30%) and branched polymer amylopectin (70–80%). The size of granules and ratio of amylose to amylopectin in starch depends on its source. Starch from potato and corn are commonly used in bioplastics as these are the major sources. Starch is generally derived from agricultural material that falls in the category of renewable resources, use of such materials reduces the overall cost of the product and increase the growth of farming sector but the films containing starch as a major component have some problems like comparatively weak water barrier properties and less tensile strength.19,20 Also if the starch amount has been increased more than 25% then the tensile strength and elongation at break decrease very sharply as reported in the literature by Sin et al. and Azahari et al.21,22 Blending of the PVA film with starch or pectin improved its biodegradability but reduce its barrier and mechanical properties (tensile strength and elongation at break).23-26 Therefore, it is necessary to improve the packaging favorable properties of these composites with a fixed amount of PVA, for which the concept of ternary composites is more efficient to boost mechanical and other beneficial properties for food packaging. Also, study the effect of ternary combinations on the mechanical properties, thermal degradation, and biodegradation is in objectives.
To improve the performance of starch-based blend films citric acid, sorbitol, glycerol etc. are used as additives and citric acid is a better cross-linker for starch/PVA blend films.27,28 However, the combination of citric acid and glycerol within optimum ratio gives better results in terms of tensile strength also glycerol acts as a plasticizing agent. 27 It has been reported that citric acid can form strong hydrogen bond interactions with starch and improves its thermal stability. 29
To examine the suitability of blend films for food packaging, thermal stability and tensile properties are important parameters. Thermogravimetric analysis (TGA) and derivative thermogravimetry (DTG) are widely used techniques to investigate the thermal stability and thermal decomposition behavior of polymers in different conditions. Moreover, it is always desirable and beneficial to study the thermal profiles of the blending materials to optimize the processing and check any change in the thermal stability after cross-linking or not. Further, the thermal degradation kinetic energy was evaluated by Horowitz-Metzger 30 and Coats-Redfern 31 method. The tensile strength and elongation at break of blended films should be high to use them in food packaging industries. The main motive of this work is to evaluate the effect of corn starch (CS) and pectin on the tensile strength and biodegradation of PVA/CS/pectin blends.
Materials and methods
Pectin (molecular weight 75000, degree of esterification of 65–70%) and citric acid were supplied from HiMedia Laboratories Mumbai, India. Corn Starch, PVA (average molecular weight 60000–125000, degree of hydrolysis of 98–100%) and glycerol were purchased from sd fine-chem limited Mumbai, India. De-ionized water was used to prepare the solutions.
Sample preparation
The blend films of PVA/starch/pectin were prepared by using solution casting technique.32,33 First PVA was solubilized in 150 ml hot water at 80°C with constant stirring on a magnetic stirrer for 20 min. Pectin and starch were also individually dissolved in 100 ml water at 95°C with constant stirring for 45 min. resulting homogenous dispersion in solutions and also in this process, starch was completely gelatinized.34-36 All three solutions were mixed in specific ratios (Table 1) and further stirred for 90 min at 70°C. Then citric acid 30% by weight of total polymer contents, was added to this mixture and further stirred for 15 min. At last, 1 ml of glycerol was mixed to the whole solution with stirring and the sample was poured in a glass casting tray. The sample was then dried in a vacuum oven (under reduced pressure) at 60°C for 48 hours to remove moisture content and blend films were obtained. A neat PVA film was also cast with the same method without any additive.
Composition of blends.

Characterization techniques
Fourier-transform infrared spectroscopy (FTIR)
FTIR spectra were used to characterize the presence of specific groups and the interaction of different chemical species in blend films showing the effectiveness of the developed. Two FTIR scans of each sample were recorded on the MB-3000 ABB spectrophotometer at a resolution of 8 cm−1 in transmission mode over the wavenumber range of 4000–600 cm−1with Attenuated Total Reflectance (ATR) accessory having ZnSe crystal was used. To avoid absorption of moisture, the samples were kept in a desiccator for 24 hours before scanning.
Scanning electron microscopy (SEM)
JEOL JSM-6390LV scanning electron microscope (SEM) was used to study the surfaces of casted films. Highly magnified images of 2500× were taken at accelerating voltage of 10 kV and before scanning, the samples were kept in a desiccator for 1 day to avoid absorption of moisture and coated with gold particles to make them conducting.
Thermal analysis
Thermal behavior was studied using EXSTAR SII 6300 instrument at 10°C/min heating rate in the nitrogen atmosphere with a flow rate of 40 ml/min. The samples were stored in a desiccator for 5 days before analysis and run of each sample was conducted under the same set of experimental conditions. Samples were heated from 30–800°C. Before each run nitrogen was flushed through the furnace for 20 minutes to build an inert atmosphere to avoid unwanted oxidation. The TG/DTA analyzer was calibrated before recording the thermograms. Dried alumina powder was used as reference material and ceramic crucible as a sample holder. To ensure uniform heating, the samples were taken in small amounts (3–7 mg). 37 Thermal data obtained was used to calculate the activation energy of the blends. The activation energy was calculated by the model-based methods of Horowitz-Metzger and Coats-Redfern, assuming the degradation process to be a first-order reaction and their final equations are given as following

where,
Method of Biodegradation studies
Soil Burial test was carried out for biodegradation according to work reported by Riyajan et al. 38 Three samples of each blend film were analyzed and their average value has been reported. To study the biodegradation of the cast films, 3 × 3 cm squares pieces of each blend were cut and placed in agricultural soil in a 12 × 18 cm square pot. Pieces of films were kept at 6 cm depth from the surface of the soil. Samples were packed in nylon net and connected through a thread while another end is tagged outside the soil so that the sample can be easily identified after a time period. The pot was placed in contact with the free environment for 90 days. The moisture of soil was maintained by adding water regularly with time. A hole was made at the bottom of the pot and plugged with cotton to drain the excess water. The measurements were performed after 1, 2 and 3 months. In this duration, temperature of the environment was in the range of 30–37°C and relative humidity was 50–60%. After the fixed time interval of 30 days, the samples were dry cleaned with a brush, quickly washed in cold water and dried in the oven at 60°C for an hour and finally weighed.
Mechanical properties
The mechanical analysis of the films was studied by tensile testing on Universal Testing Machine (UTM) with the load capacity of 5 kN. Before testing the samples were stored for 24 hours at 25°C and 50% relative humidity. The samples were tested according to ASTM D882 standards. The tensile properties of the sample were recorded at a cross-head speed of 50 mm/min. Five samples were tested in each category and the average value of tensile strength, tensile modulus and % elongation is reported.
The thickness of films is an important parameter while calculating tensile strength. Screw gauge was used to measure the thickness of the conditioned films. The thickness of the film at 10 random positions was measured and the average of obtained results is reported.
Results and discussion
FTIR spectroscopy
There is a slight difference in the intensity of peaks in the spectra of blends as a function of starch/pectin ratio as shown in Figure 1. The FTIR spectra of PVA, starch, pectin, citric acid, and ternary blend film with cross-linker is depicted in Figure 2. A broad band at 3340 cm−1 corresponds to O–H stretching vibrations due to hydroxyl groups present in pectin, starch and glycerol. Further –CH stretching peaks observed at 2923 cm−1 and peaks at 1234 cm−1 and 1087 cm−1 corresponds to –C–O– from ether linkage and C–O–H groups, respectively. The band at ∼1740 cm−1 in pure citric acid can be attributed to vibrations of –C=O– in carboxylic acid groups while the band at ∼1712 cm−1 in all the blend films can be related to the vibration of carbonyl in ester groups39,40 formation between the carboxylic group of citric acid and the hydroxyl group of other added components (PVA, starch, pectin, and glycerol) and such type of cross-linking is also reported in the literature by Seligra et al. 25 Although there is the absence of a peak in blended films at 1740 cm−1 which is present in free citric acid, indicated that citric acid has been cross-linked with added components and FTIR results inferred the formation of blend films.
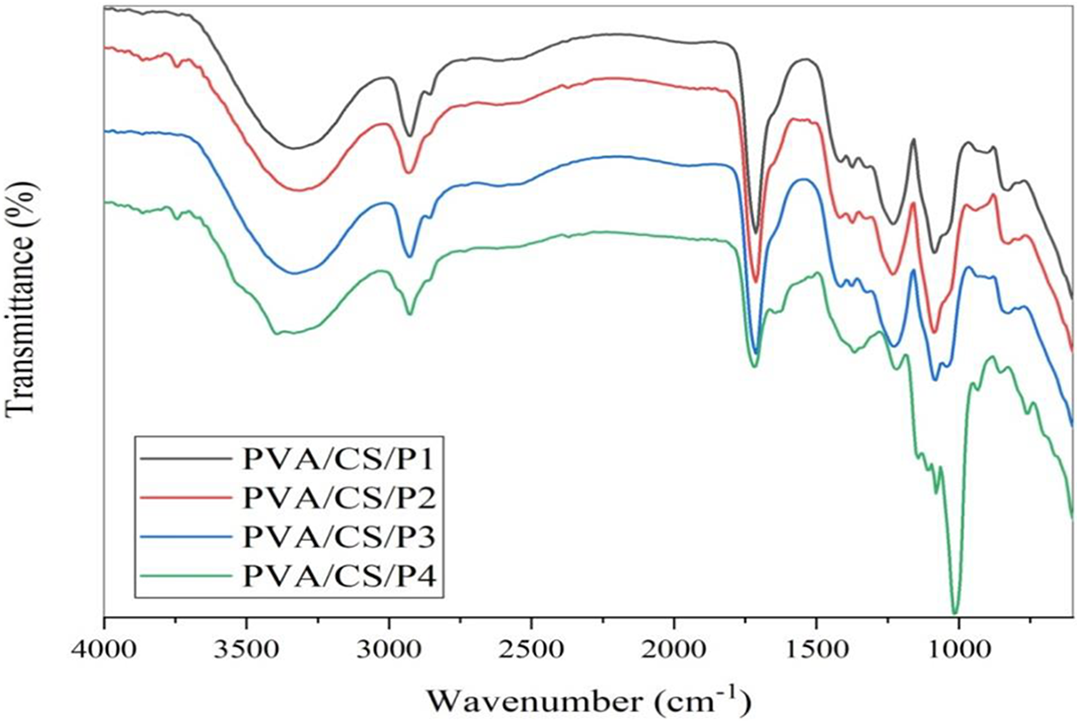
FTIR spectra of blend film PVA/CS/P1, PVA/CS/P2, PVA/CS/P3, and PVA/CS/P4.

FTIR spectrum of PVA, pectin, starch, citric acid, and blend film PVA/CS/P1.
Thermogravimetric analysis
Thermograms of blend films obtained by heating the samples from 30–800°C at a heating rate of 10°C/min in an inert atmosphere of Nitrogen are presented in Figure 3. From ambient temperature to 200°C, thermograms showed a small loss of weight (1–2%) because of moisture evaporation and the blend films are stable up to 200°C, which is slightly less than neat PVA film. 41 The onset decomposition temperature of blends with different compositions slightly varies in the range 208–218°C. The overall thermal decomposition of blend films occurred in four stages, as also shown in Derivative Thermogravimetry (Figure 4), the first decomposition stage from 200 to 260°C is accompanied by the removal of glycerol and citric acid contents from blends. 29 Further in this temperature range, the glycosidic linkages in pectin are broken down to form smaller chains of pectin molecules. 42 In the second decomposition stage 280–370°C,29,41 starch decompose completely and also side chain of PVA removed in this range of temperature. Further PVA decomposes to give organic volatiles by cyclization reaction in the third decomposition stage in the temperature range 400–470°C. 41 In the last decomposition stage depolymerization of pectin occurs leading to the formation of volatile compounds up to 570°C temperature.

TGA curves of PVA/S/P1, PVA/S/P2, PVA/S/P3, PVA/S/P4, and PVA.
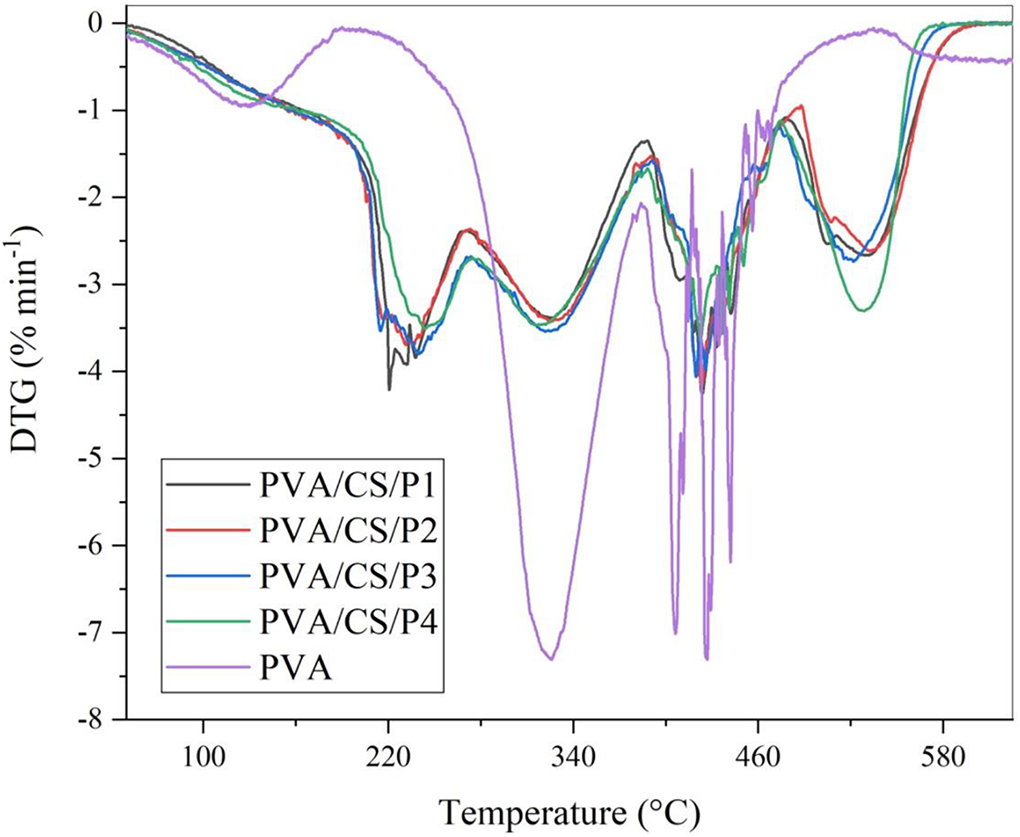
DTG curves of PVA/S/P1, PVA/S/P2, PVA/S/P3, PVA/S/P4, and PVA.
The decomposition of pectin starts from 280°C to 570°C. In this temperature range, decarboxylation of pectin and depolymerization occurs leading to the formation of volatile organic compounds. 42
There is no major shift in the thermogram of blends as the starch/pectin ratio increases from 1:5 to 2:1 indicating the retention of thermal stability with the increasing starch content. The maximum decomposition temperature (Tm) for first decomposition stage has been increased from 220.93°C to 243.65°C with the increase in starch/pectin ratio and maximum weight loss rate (MWLR) for this decomposition also decrease from 4.21°C/min to 3.50°C/min (Table 2) and it may be inferred that citric acid and glycerol have a stronger interaction with starch that leads to shifting in DTG peak maxima
The onset degradation temperature (To), maximum degradation temperature (Tm) and endset temperature (Te), activation energies (Ea) of all degradation stages.

The TGA data obtained at a heating rate of 10°C/min was used to calculate the activation energy corresponding to four stages of decomposition of blends by Horowitz-Metzger (HM) and Coats-Redfern (CR) methods. The decomposition was assumed to be first-order and the values of activation energies (Ea) and regression coefficient (R2) are given in Table 2. The regressions are good for the first three decomposition stages and the activation energies are almost similar for each decompostion stage of all blends as calculated by the corresponding method. The value of regression for the fourth stage is lower and the activation energies calculated by the methods are different which indicates that the decomposition behavior does not follow first-order kinetics. Further, the activation energy for the blend films is almost similar and supports the fact that the blends are homogeneous as well as equally thermally stable.
Biodegradation study
The degradation percentage of blended films as calculated by soil burial test are reported in Table 3. In the first 30 days, blends were 47–50% degraded and 61–63% degradation occured in 60 days. After a time period of 90 days, 67–68% of blends were degraded which is comparable to reported binary blends of starch/PVA.43,44 PVA neat film degraded slowly over approximately 21 days and then entered in a “steady-state period” despite the presence of glycerol and starch. According to Abdullah and Dong, 14 these components substantially increases degradation rate. The degradation process in soil depends upon the amount of water absorbed by the blends and also on the susceptibility of components toward the micro-organism/enzymatic attack. Since starch and pectin are the naturally derived polymers, their susceptibility toward attack by micro-organisms is high and thus the degradation percentage of blend films is higher than neat PVA film. 14 The reported degradation percentage is the sum of completely/partially degraded products and the polymer chains that are dissolved/disrupted from the sample. The percentage degradation in the blends is higher than expected, from the composition and degradation % of neat PVA film, that might be because of presence of porous which could be formed as biopolymers molecules were degraded by microorganisms, leading to an increase in water diffusion inside the film and consequently, an increase in cleavage of PVA molecules by hydrolysis.
The degradation percentage of blend films at different intervals.

Scanning electron microscopy (SEM)
SEM micrographs of the PVA/CS/P1 (with lowest starch/pectin ratio) and PVA/CS/P4 (with highest starch/pectin ratio) ternary films are shown in Figure 5 and the result indicates that the surfaces of PVA/CS/P ternary films are heterogeneous. The smaller size of corn starch as shown in SEM images might be due to gelatinization of starch. 20 As the starch content increases, more starch particles are visible in the micrographs. The visibility of more number of starch particles is not good for tensile properties as observed but it is good for starch based film formation which leads to homogeneity in blend films. The size of the starch particles is almost the same (5–6 µm) in all images indicating the homogeneous dispersion of these particles in blend films. This homogeneous dispersion is facilitated by the binding agent that ensures lesser agglomeration of starch. 45

SEM images (×2500) of blends (a) PVA/CS/P1 (b) PVA/CS/P4.
Mechanical properties
The observed and calculated results of thickness, tensile strength, Young’s modulus, and elongation at break of blend films are tabulated in Table 4. The neat PVA film has less tensile strength and it is increased to 19.03 MPa in blends of PVA with pectin and starch. Decrease in the S/P ratio from 2:1 to 1:2, the tensile strength increases from 12.39 MPa to 19.03 MPa and further remained almost same upto S/P ratio of 1:5. This increase in strength might be a result of better cross-linking between starch and citric acid. 46 Elongation at break in neat PVA is 170% which is increased in blend films in the range 362–516% due to consequences of both cross-linking and plasticizing effect. The Young’s modulus of blend films is decreased up to 80% as the S/P ratio increased from 1:5 to 2:1. This sharp decrease in modulus may be attributed to the plasticizing effect of glycerol. 15 In blend with starch/pectin ratio of 1:2 cross-linking predominate which results in its highest tensile strength (19.03 MPa) and lowest % elongation (362%). With the further increase in starch content, the plasticizing effect predominates as indicated by the increase in % elongation and a sharp decrease in Young’s modulus. blend with starch/pectin ratio of 1:5 has highest % elongation (516%) with comparable tensile strength (18.85 MPa). In nutshell, it may be inferred that tensile strength is mainly directed by the cross-linking effect, Young’s modulus by plasticizing effect and % elongation by both cross-linking and plasticizing effects in the present study.
Tensile properties of blend films.

The result of mechanical studies shows that the ternary blends have high tensile strength and high elongation at break which are better than that of binary PVA/starch and PVA/pectin blends24,26,47 and might be used in food packaging and other applications in which high mechanical properties are required.
Conclusion
This research report represents the eco-friendly synthesis of ternary blend films of PVA/starch/pectin in water using glycerol as a plasticizer. FTIR spectrum indicated that the cross-linking in blends through citric acid. Blend films were 50% degraded in soil within 30 days in naturalistic conditions and almost 68% degraded over 90 days. Blend films have improved tensile properties and blends with the Starch/pectin ratio of 1:5 have high tensile strength (18.85 MPa) and highest elongation at break (516%). All the films were thermally stable up to 210°C and their degradation behavior was almost similar. The overall results of the thermal study, biodegradation and mechanical properties inferred that the blend films might be used in sustainable packaging and blend with S/P ratio 1:5 is optimized for better packaging properties as well as cost effective.
Footnotes
Acknowledgment
Senior Research Fellowship to Mr Vinod Kumar is gratefully acknowledged. Acknowledgment is also due to Chairman, Chemistry Department, Kurukshetra University, Kurukshetra, for providing necessary laboratory facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from the Council of Scientific and Industrial Research (CSIR), New Delhi.
