Abstract
Guar gum hydrogels may be dried to form polymer films which have the potential for use as biodegradable alternatives to polymers such as low-density polyethylene. In this study, the tensile strength and tensile modulus of guar gel films having moisture contents ranging between 15% and 18% (wet basis) were measured at a strain rate of 1 mm min−1. Mean tensile strengths of the films ranged between 25 MPa and 40 MPa (dependent on composition) which is of similar magnitude to the tensile strength data for polyethylene and cellophane that are reported in the literature. The mean tensile modulus of the films (1.5–2.5 GPa) was higher than the tensile modulus values reported for low-density polyethylene but comparable to those for cellophane (3 GPa).
Introduction
The significant environmental impact of plastic material refuse has triggered research into the development of plastics that degrade rapidly, leading to, in some cases, complete mineralization or bio-assimilation of the material. 1 Despite recent advances in this area, however, many large urban areas still suffer extensively from packaging litter since much of the material does not biodegrade rapidly.
Guar gum is obtained from the ground endosperm of the guar legume (Cyamopsis tetragonolobus), 2 and has a range of uses in the food, cosmetics and pharmaceuticals industries. 2 –8 The galactomannan chains of guar gum are also amenable to copolymerization, for example, by grafting with vinyl acetate-ethylacrylate 9 or methyl-methacrylate. 10 While it is most commonly used as a binder or thickener, the addition of a cross-linker can produce stable hydrogels under suitable conditions. 4,6,11 –16 The resulting gel may then be dried to produce clear ‘plastic’ films (Figure 1), which biodegrade rapidly upon rehydration. 6,17 As such, guar hydrogel films offer a sustainable alternative to petroleum-derived polymers (e.g. polyethylene) for products such as shopping bags and packaging films. Alternatively, the films could be used as food coatings.

Guar-gel film prepared as described in the second section, and used in tensile testing.
A number of studies have been performed investigating the mechanical properties of guar gum (or a derivative) mixed with other polymeric materials. Xiao et al. 18 measured the tensile strength of films produced from blends of chitosan and hydroxypropyl guar gum. They found that the maximum tensile strength (59 MPa) occurred with a 60% guar gum blend. Rao et al. 19 also measured tensile properties of guar gel/chitosan mixtures, although it appears there may have been some error in reporting their data, since the tensile strength values in Figure 2 varied between 4 kPa and 12 kPa (when it would be expected that the data would be in the region of MPa). Huang et al. 20 measured the tensile strength of films prepared from cationic guar gel mixed with poly acrylic acid and measured a maximum tensile strength of 41 MPa, at a composition of 50% guar gel. Shenoy and D’Melo 21 measured the tensile strength of films prepared from blends of acrylated guar gum and acrylic emulsion and reported tensile strengths varying between 0.5 MPa and 3 MPa. Mikkonen et al. 22 measured the tensile strength of films formed from guar gum plasticized with glycerol or sorbitol, and found that the maximum tensile strength (13 MPa) was obtained with 20% glycerol. Das et al. 23 modified the chemical structure of guar to make the film water-resistant and biocidal. They reported tensile breaking points of 21.95 MPa. Saberi et al. 24 prepared edible biofilms based on guar gum and pea starch with a variety of plasticizers and reported a maximum tensile strength of 34.1 MPa. They observed that the addition of plasticizers tended to decrease tensile strength. 25 Strand et al. 26 investigated the use of spray-on cationic guar gum biofilms made with 50% sorbitol for use as paper coatings. They reported a relatively low tensile strength of 18 MPa, probably due to the presence of the sorbitol plasticizer.

Chemical structure of guaran (guar gum repeat unit).
Polymer blends are considered advantageous since their physical properties may be tailored (within practical limits) to a given application. 18 However, there is no reason why guar gum could not be used on its own which would remove the need for the blending stage of an industrial scale production. There does not appear to be any data in the literature for the tensile properties of films based solely on guar gum as the polymeric material. The aim of this work was to measure the tensile properties of guar gel films in order to assess whether they would be sufficient for use as packaging material.
Guar gel film preparation method
Guar gum
Since guar gum is a natural product, its molecular structure is highly dependent on the climate, the planting season and other environmental parameters.
4
It is therefore impossible to characterize guar gum as a specifically defined molecule or polymer, but only as a mixture of carbohydrates which share a general structure. Eighty per cent by mass of food-grade guar gum powder is comprised of the galactomannan guaran which consists of a (1→4)-beta-mannopyranosyl backbone with (1→6) linked alpha-
Mixing
The solution and gelling characteristics of guar gum are sensitive to a variety of factors including time (both mixing and setting), temperature, concentration, pH, powder particle size and the type of cross-linker used. 4,5,27 The guar powder is extremely hygroscopic which requires either a very slow rate of addition to the solvent (impractical other than at small scale) or high-shear mixing, which has a significant influence on the temperature and viscosity of the solution; factors which in turn influence the gel-forming characteristics. 28 High-shear mixing can break polymer chains, which is not desirable for gelation 29 and produces a significant quantity of heat. Centrifugation is a potential alternative to high-shear mixing; however, it may be prohibitively expensive on an industrial scale.
The dissolved guar gum forms a viscous, non-Newtonian fluid with thixotropic behaviour, due to the entanglement of the galactomannan chains
4
. The viscosity is dependent on the concentration of guar gum in the solvent, mixing time and the solvent pH.
28
Jussen
4
measured the viscosities of guar gel solutions as functions of time (at 40°C) in acidic, neutral and basic solutions in order to determine the optimum mixing time, shear rate and solvent pH and concluded the following: To maintain polymer chain length, guar gum gels should be mixed for as short a time as possible. For 10 g L−1 solutions the minimum mixing time was approximately 10 min (at 8000 r min−1). In order to increase the guar powder loading (and hence reduce the drying load), acidic solvents should be used. Of the solvents considered, 1M HCl was the most effective solvent at reducing the solution viscosity. Acidic solutions produced more ductile films than alkaline or neutral solutions.
In this study, all guar gel film samples used for tensile testing were prepared by mixing at 8000 r min−1 for 10 min in 1M HCl solution. Three concentrations of guar powder were used: 10, 12 and 15 g L−1.
Setting
It is important that the mixing process does not proceed too long since air bubbles become entrained in the solution as the gel starts to set (the greater the guar gum content of the solution the greater the susceptibility to air entrainment). The gels were spread out on a smooth surface to dry as soon as possible after the guar gum powder had fully dissolved. A Perspex grader was drawn over the gel once it had been poured onto the drying surface to ensure uniform thickness of the gel and resultant film.
Drying
To prevent brittleness and cracking, the gels were dried at relatively low temperature (<60°C). The drying needed to occur at a sufficient rate to prevent the growth of microbial organisms typically present within bulk feedstocks of the guar gum powder. In these experiments, the gels were dried by small (10 mm diameter) fans at near ambient temperature.
Moisture content of guar gel films
Since mechanical properties depend on the composition of the films, the moisture content of the films was determined by drying the films at 100°C until the mass of the sample had reached an equilibrium value. The wet-basis moisture content was determined from equation (1)
where W is the wet-basis moisture content, m i is the initial mass of the guar gel film and m f is the final mass of the film. Figure 3 shows the moisture content of the films as a function of the guar concentration in the initial gel. Each point represents the average of five film moisture content measurements. As expected the moisture content of the film decreases as the concentration of guar gum in the initial gel increases.

Moisture content of films versus guar concentration of gels.
The guar gels have a wet-basis moisture content of approximately 99%, while the films have wet-basis moisture contents of less than 20%. This means that 90–100 g of water must be evaporated per gram of film produced, which represents a substantial drying load. However, because the gels were spread out in a thin layer (5 mm or less) drying rates were high even at ambient temperature, provided forced convection was employed.
Tensile strength tests
The tensile strength (failure stress) and tensile modulus (Young’s modulus) of the guar gum films were measured using an Instron 33R4204 machine (Melbourne, Australia) at a strain rate of 1 mm min−1. For each of the three gel concentrations five film samples were prepared and five strips from each film were cut into samples 160 mm length and 15 mm width (25 test specimens for each gel concentration). The gauge length of the samples was 124 mm. The thickness of the specimens was measured using a micrometer at three different positions along the film length, and typical values were of the order of 0.05 mm.
A typical tensile test plot (stress vs. elongation) is shown in Figure 4. Stress was consistently observed to initially increase slowly up to a plateau after which it increased more rapidly with respect to strain, although the gradient decreased slightly before final fracture. The consistency of this trend suggests that it represents structural mechanisms and implies some type of yielding occurring at lower stresses, possibly due to the failure of a particular type of intermolecular bond. Overall, behaviour was generally brittle; there was no obvious macroscopic yielding and failure occurred at strains between 0.015 and 0.025 (Figure 5).

Typical stress versus extension plot for guar gel films.

Mean failure strains for guar gel films.
The mean tensile strengths for each gel concentration are shown in Figure 6 (the error bars correspond to the standard deviation in the results). The tensile strength increased as the concentration of guar gum in the hydrogel increased from approximately 25 MPa at 10 g L−1 to 40 MPa at 12 g L−1; however, the tensile strength decreased from 12 g L−1 to 30 MPa at 15 g L−1. The increase in strength between 10 g L−1 and 12 g L−1 may be attributed to the fact that an increase in the solids composition of the gels (and hence the films) allowed for a greater number of cross-links between polymer chains. The decrease in strength between the 12 g L−1 gels and 15 g L−1 gels may be attributed to the greater number of bubbles that were unavoidably entrained (Figure 7) during the preparation of these gels, as a result of the increase in viscosity that accompanied the increase in guar gum concentration. 27 The bubbles would have caused stress concentrations and reduced the cross-sectional area of the test samples, which would explain the reduction in tensile strength.
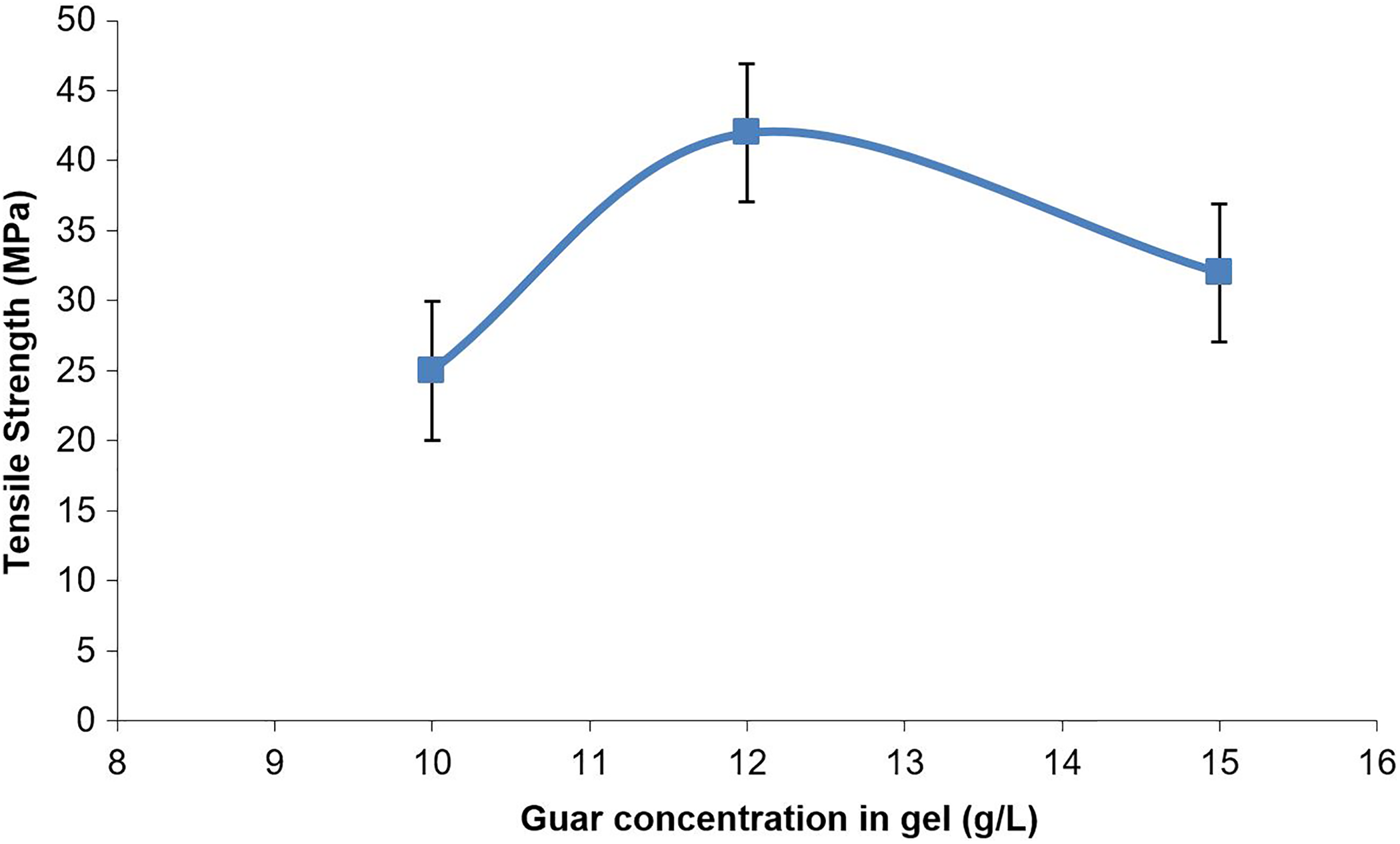
Mean tensile strength at yield values for guar gel films.

Bubble entrainment in guar gel films (a) 10 g L−1, (b) 12 g L−1 and (c) 15 g L−1.
The tensile modulus was calculated from the second linear portion of the stress–strain curve (Figure 4), rather than the initial linear portion, which often was not as well defined, perhaps due do extension or ‘straightening’ of the samples rather than due to strain. Generally, the calculation was based on the change in strain between stresses of 10 and 20 MPa, although in some cases the sample failed below 20 MPa, in which case other criteria were used. The tensile modulus (Figure 8) appeared to decrease as the gel composition was increased from 10 g L−1 to 12 g L−1, although there was no apparent decrease in elastic modulus from 12 g L−1 to 15 g L−1. It is difficult to draw conclusive inferences about trends in the tensile modulus data since the standard deviation in the results (which was used to plot the error bars in Figure 7) was high; however, the lack of change between gel composition of 12 g L−1 and 15 g L−1 could again probably be attributed to the influence of the large number of bubbles in the films made from the 15 g L−1 gels (Figure 7(c)).
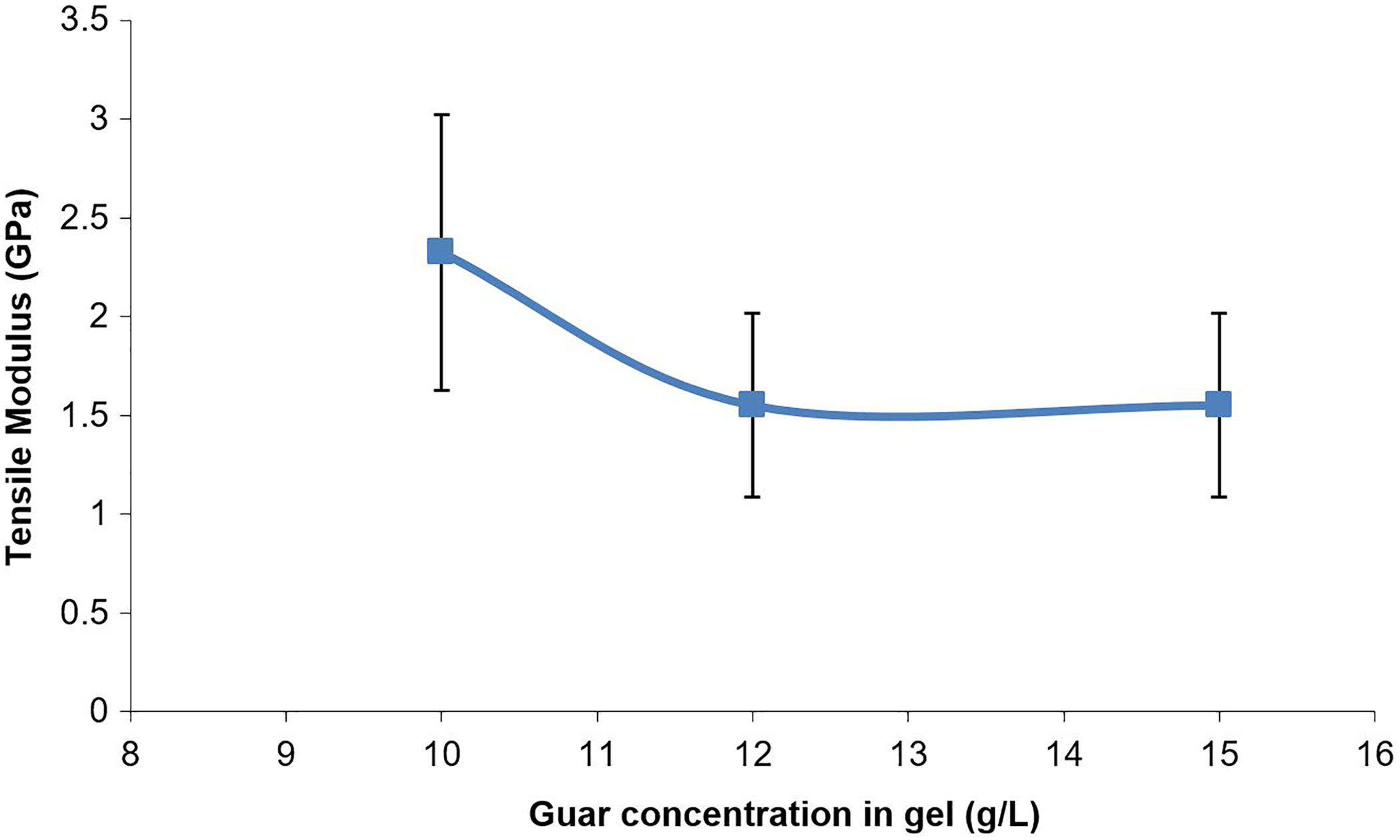
Mean tensile modulus values for guar gel films.
The tensile strength of the guar gel films was of similar magnitude to the tensile strength of polyethylene (e.g. 20 MPa 30 ) and cellophane (35–75 MPa 31 ). It is also similar in magnitude to tensile strengths of the blended polymer film data referred to above, 18 –22 and demonstrates that guar gum itself is capable of providing sufficient tensile strength for packaging purposes. The guar gel films were three to five times stiffer than low-density polyethylene (tensile modulus 590 MPa 30 ), but similar to cellophane (3 GPa 31 ).
More work is required in order to optimize the preparation of gels with 15 g L−1 guar gum concentration without the entrainment of so many bubbles, and subsequently to determine whether tensile strengths higher than 40 MPa might be achieved.
Conclusion
Tensile testing was performed on films prepared by drying guar gum hydrogels. The moisture content of the films varied between 15% and 18% (wet basis). The tensile strength of the films initially increased with increasing guar content, but subsequently decreased as the guar content continued to increase (this decrease was attributed to weakening of the films by air bubbles entrained in the films). The scatter in the tensile modulus was high and it was therefore difficult to draw conclusions about the dependence of tensile modulus on composition. The tensile strength was of similar magnitude to the tensile strength values for low-density polyethylene and cellophane that are reported in the literature, while the tensile modulus was higher than polyethylene and slightly lower than cellophane.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.

