Abstract
The effects of four hindered phenolic antioxidants in the short-term heat-oxygen stability of High-Density Polyethylene (HDPE) was evaluated by the Melt Flow Rate (MFR) and Oxidation Induction Time (OIT) analyses. The results showed that Irganox 1330 and Irganox 1010 had more excellent processing property and oxidation resistance behavior in HDPE than Irganox 1024 and Irganox 3114. The OIT values of HDPE with Irganox 1330 and Irganox 1010 were up to 97.25 min and 89.73 min respectively. The long-term accelerated heat-oxygen stability tests of HDPE containing hindered phenolic antioxidants were carried out at 110°C in deionized water pressurized with pure oxygen at a pressure of 10 MPa. The characterization of the long-term heat-oxygen stability was performed by Fourier Transform Infrared (FTIR), Differential Scanning Calorimetry (DSC) and tensile testing. However, the results exhibited that the aging resistance of HDPE containing Irganox 1010 was inferior to that of HDPE containing Irganox 1024 and Irganox 3114, which is quite contrary to their short-term effect on HDPE stabilization. It may be speculated that the ester groups of Irganox 1010 undergo hydrolysis during the long-term heat-oxygen stability tests, leading to the formation of 3-(3,5-ditert-butyl-4-hydroxy-phenyl) propanoic acid and pentaerythritol.
Introduction
Polyethylene is a highly versatile thermoplastic used for both short-term and long-term applications. A very important one in the latter category is food packaging, textiles, lab equipment and automotive components due to the excellent mechanical properties, low cost, and superior processibility.1–6 The long-term performance of polyethylene significantly depends on their resistance against heat-oxygen aging degradation. In the presence of oxygen, degradation occurs already at ambient temperatures and the oxidation rate is considerably enhanced with increasing temperatures.7–9 Among all kinds of methods to increase the heat-oxygen stability of polyethylene, the addition of antioxidants is an effective way to prolong the life of polyethylene.10,11 Antioxidants can be classified as primary and secondary, depending on their respective mode of operation. One of the most used primary family of antioxidants in industry is the very effective hindered phenolic antioxidants(e.g., Irganox 1010, Irganox 1330, Irganox1024, Irganox3114), which have good pollution-free and non-discoloring properties compared with the toxicity and discoloration of amine antioxidants.12–15
The long-term heat-oxygen stability of polyolefins is usually evaluated by time-consuming oven aging tests.16–18 The oven aging tests are sometimes carried out at higher temperatures close to the melting point of polyethylene. This leads to changes in the polymer morphology. Moreover, the physical loss of antioxidants due to evaporation, migration or extraction is enhanced at higher temperatures.19–22 Accelerated aging tests at lower temperatures and high concentration of oxygen would be favorable.23,24
The main objective of this paper is to investigate the heat-oxygen stability of four commercial hindered phenolic antioxidants in polyethylene. Especially, accelerated heat-oxygen aging tests were carried out at 110°C in deionized water pressurized with pure oxygen at a pressure of 10 MPa. By a combination of mechanical, thermal and chemical-analytical characterization methods, the heat-oxygen aging behavior of the four commercial hindered phenolic antioxidants was systematically and precisely evaluated. These tests provided guidance for selection and application of commercial hindered phenolic antioxidants in HDPE.
Experimental
Materials and HDPE blends preparation
Benzenepropanoicacid-5-bis(1,1-dimethylethyl)-4-hydroxy-2-[3-[3,5-bis(1,1-dimethylethyl)-4-hydroxyphenyl]-1-oxopropyl] hydrazide (Irganox 1024) was purchased from Nuotai Chemical Co. Ltd (Shanghai, China). Tris(3,5-di-tert-butyl-4-hydroxybenzyl) isocyanurate (Irganox 3114) and pentaerythritol tetrakys 3-(3,5-ditert-butyl-4-hydroxyphenyl)propionate (Irganox 1010) purchased from Darui Fine Chemical Co. Ltd (Shanghai, China) were analytical reagent grade. 2,4,6-Tris(3’,5’-di-tert-butyl-4’-hydroxybenzyl)mesitylene (Irganox 1330) was purchased from Aladdin Industrial Corporation (Shanghai, China). The structures of these hindered phenolic antioxidants are shown in Figure 1. HDPE was kindly supplied by Daqing Chemical Engineering Research Center (China) and was stored in the dark.

Chemical structure of hindered phenolic antioxidants.
Preparation of HDPE blends
The HDPE blends were prepared by melt compounding with a SJ-65 twin screw extruder, the screw diameter was 65 mm and the length-to-diameter (L/D) ratio was 130. The extruder was operated at 200°C with a roll speed of 50 rpm. At the beginning of the blending procedure HDPE was first added to the SHR-10A mixer, and then the antioxidant was added, the mixture was processed for about 15 min at high speed. All the HDPE blends were done with 1mg·g−1 of one of the hindered phenolic antioxidants. For accelerated heat-oxygen aging experiments, 1 mm thick HDPE specimens were prepared.
Hindered phenolic antioxidant performance characterization
Thermo Gravimetric Analysis (TGA) of the hindered phenolic antioxidants was determined using Thermo Gravimetric Analyzer (TGA Q5000), about 4–10 mg of the resins were heated, and the heating rate was 20°C/min from 25°C to 1200°C. Performances of hindered phenolic antioxidants in HDPE were evaluated by MFR and OIT. MFR was measured using an Italian Ceast melt flow rater 6542 at an extrusion temperature of 190°C with 21.6 kg load according to ASTM-1238. OIT was measured with JH-910 Differential Scanning Calorimeter following the standard method (ISO 11357-6:2002). The methods used for the OIT measurement proceeded were given as follows: Firstly, the samples were held at 60°C for 5 min with a nitrogen flow of 50 mL/min. The samples were heated to 210°C at a rate of 20°C/min under nitrogen flow. And then the gas was switched to oxygen with the flow rate of 50 mL/min. The oxidation of the sample was observed as a sharp increase of heat flow.
Accelerated heat-oxygen aging characterization
All accelerated heat-oxygen aging experiments were performed on dumbbell-shaped samples (thickness, 1 mm). The samples were aged in autoclaves at 110°C in deionized water pressurized with pure oxygen at a pressure of 10 MPa. With a certain time interval, the samples were taken out from the autoclaves and stored at 25°C for 24 h and the changes due to thermal oxidative degradation were monitored by FTIR, OIT and tensile strength tester (INSTRON-4467, INSTRON Inc. England). FTIR spectrum was performed by a Nicolet FT-IR750 in the range of 4000–400 cm−1. OIT was measured with JH-910 Differential Scanning Calorimeter according to the standard method ISO 11357-6:2002. The changes of the tensile strength and elongation at break the samples were monitored by using a tensile strength tester at a crosshead speed of 50 mm/min.
Results and discussion
Short-term heat-oxygen stability of hindered phenolic antioxidants in polyethylene
Thermal stability of the hindered phenolic antioxidants is an important factor during processing. Therefore, before mixing with polyolefins the thermal behavior of the hindered phenolic antioxidants was examined by TGA. The typical TGA curves for various hindered phenolic antioxidants under air atmosphere are presented in Figure 2. The initial temperature of weight loss (Ti) is defined as the temperature of the 1% weight loss. The relative thermal stability of the antioxidants was evaluated by Ti and maximum rate of weight loss (Tmax) shown in Table 1.
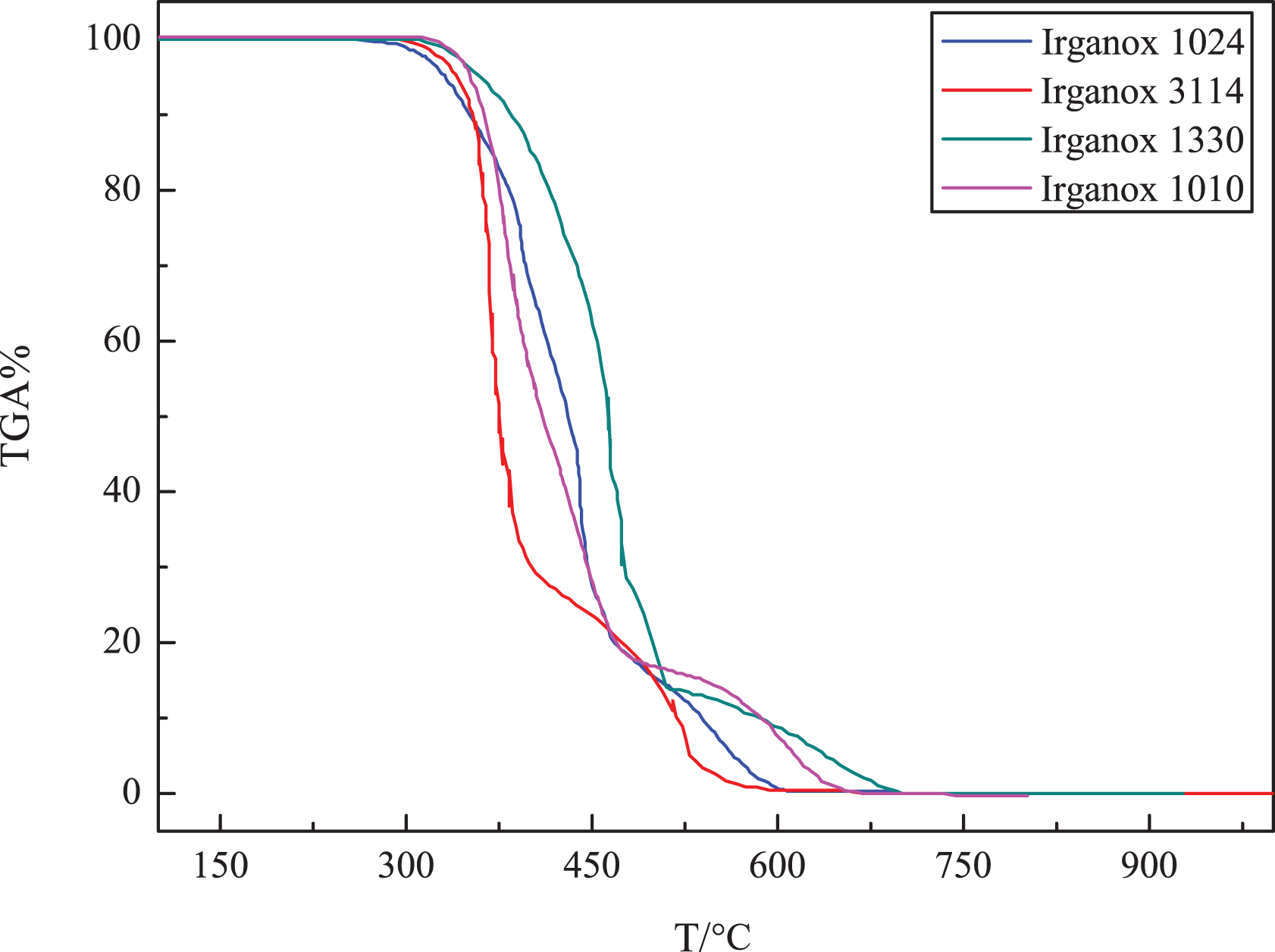
TGA curves of various hindered phenolic antioxidants in air.
Molecular weight of hindered phenolic antioxidants and their TGA data.

Clearly, for all the hindered phenolic antioxidants, the Ti loss temperature is found to be over 263°C, and these results indicate that all the hindered phenolic antioxidants are stable at temperatures used for polyethylene processing. By contrast, the Ti and Tmax for Irganox 1330 is highest, and the values are 315°C and 370°C respectively, indicating the best thermal stability of Irganox 1330 than other three hindered phenolic antioxidants.
Polyethylene materials are commonly exposed to heat, shearing forces, oxygen, radiation, water, and metal catalyst residues during processing and their service life. These external factors cause oxidative degradation of the polyethylene, resulting in deterioration of their chemical, physical and mechanical properties. MFR of multiple-extruded polyethylene can be used to evaluate the oxidative degradation of polyethylene during melt processing. The more the oxidative degradation of polyethylene, the better the fluidity of melt polyethylene became, and the higher the MFR values.10,25,26 The MFR curves for the four hindered phenolic antioxidants at the antioxidant concentration of 1mg·g−1 are presented Figure 3. The MFR values of pure HDPE remarkably increase with the increase of extrusions. Compared with the pure HDPE, the change of the MFR values of HDPE stabilized with hindered phenolic antioxidants is small. The results show that hindered phenolic antioxidants can prevent HDPE from thermal oxidative degradation in melting process by donating H-atoms to free radicals.10,27,28 When Irganox 1330 is used to stabilize HDPE, the MFR value is only 5.4 g/min after three extrusions, and the MFR values present a lowest increase than other three hindered phenolic antioxidants, indicating that Irganox1330 has the best process stabilization. The possible reason is rigidity chemical structure of Irganox 1330. Three 3,5-ditert-butyl-4-hydroxy-phenyl are attached to the benzene ring, which can enhance molecular rigidity, limit the free rotation of the molecular chain. Thus, the molecular structure is hard to break down into small molecules, the heat resistance is improved and the loss of evaporation is slow.29,30

MFR of multiple-extruded HDPE stabilized with hindered phenolic antioxidants.
OIT is one of the most commonly used parameter to evaluate the thermal oxidative stability of polyolefins in academic and industrial environment. The OIT is the time it takes before the sample starts to be oxidized. The more resistant the sample is to oxidation, the longer the OIT. To get further insight on the heat-oxygen stability of HDPE with hindered phenolic antioxidants, the OIT values of pure HDPE and HDPE in combination with antioxidants were measured, as shown in Figure 4. The OIT value of pure HDPE is 54.12 min, during hindered phenolic antioxidants are introduced, and the OIT values increase in various degrees. The results show that the hindered phenolic antioxidants have good effectiveness of stabilization in the HDPE. When Irganox1024, Irganox1010 and Irganox 3114 are used to stabilize HDPE, the OIT values are 75.84 min, 89.73 min and 86.48 min respectively, and the OIT value presents a further increase to 97.25 min when stabilizing HDPE with Irganox1330, indicating that Irganox1330 can endow HDPE with good protection and give better protection than other three hindered phenolic antioxidants.13,31

OIT of the HDPE stabilized with hindered phenolic antioxidants.
Long-term accelerated heat-oxygen stability of hindered phenolic antioxidants in HDPE
Although the short-term oxidation induction time measurement represents a fast and reliable tool to evaluate the antioxidant effectiveness in academic and industrial environment, the differences between short-term oxidation induction time and long-term heat-oxygen stability in polyethylene has been questioned by several authors.13,32 Therefore, the changes in chemical composition, long-term oxidation induction time and mechanical properties were measured to evaluate the antioxidant effect of the hindered phenolic antioxidants on the long-term heat-oxygen stability of prepared samples.
Infrared spectrometry is a powerful tool for studying the degradation of polyethylene. The long-term accelerated heat-oxygen stability of HDPE containing hindered phenolic antioxidants was tested by FTIR spectroscopy. As shown in Figure 5, FTIR of the pure HDPE subjected to thermal oxidation at 110°C in deionized water pressurized with pure oxygen at a pressure of 10 MPa shows obvious increases of absorption intensity in the regions of around 1800 cm−1–1650 cm−1 and 1300 cm−1–1000 cm−1 over time, which can be attributed to characteristic signals of carbonyl and C-O bond groups respectively. However, the evolution of absorption intensity of the carbonyl with a more dramatic change is chosen to study the degradation of HDPE in the thermal oxidation aging test. The carbonyl index (CI) serving as an indicator was derived by integrating the spectral absorbance of the carbonyl region from 1650 cm−1 to 1800 cm−1.The integral from 2750 cm−1 to 2900 cm−1 was used for normalization.9,13,33–35 The CI was expressed as follows
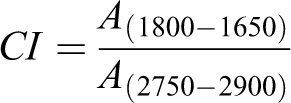
A(1800–1650) was the spectral absorbance of the carbonyl region from 1650 cm−1 to 1800 cm−1, and A(2750–2900) was the spectral absorbance of the standard peak from 2750 cm−1 to 2900 cm−1.
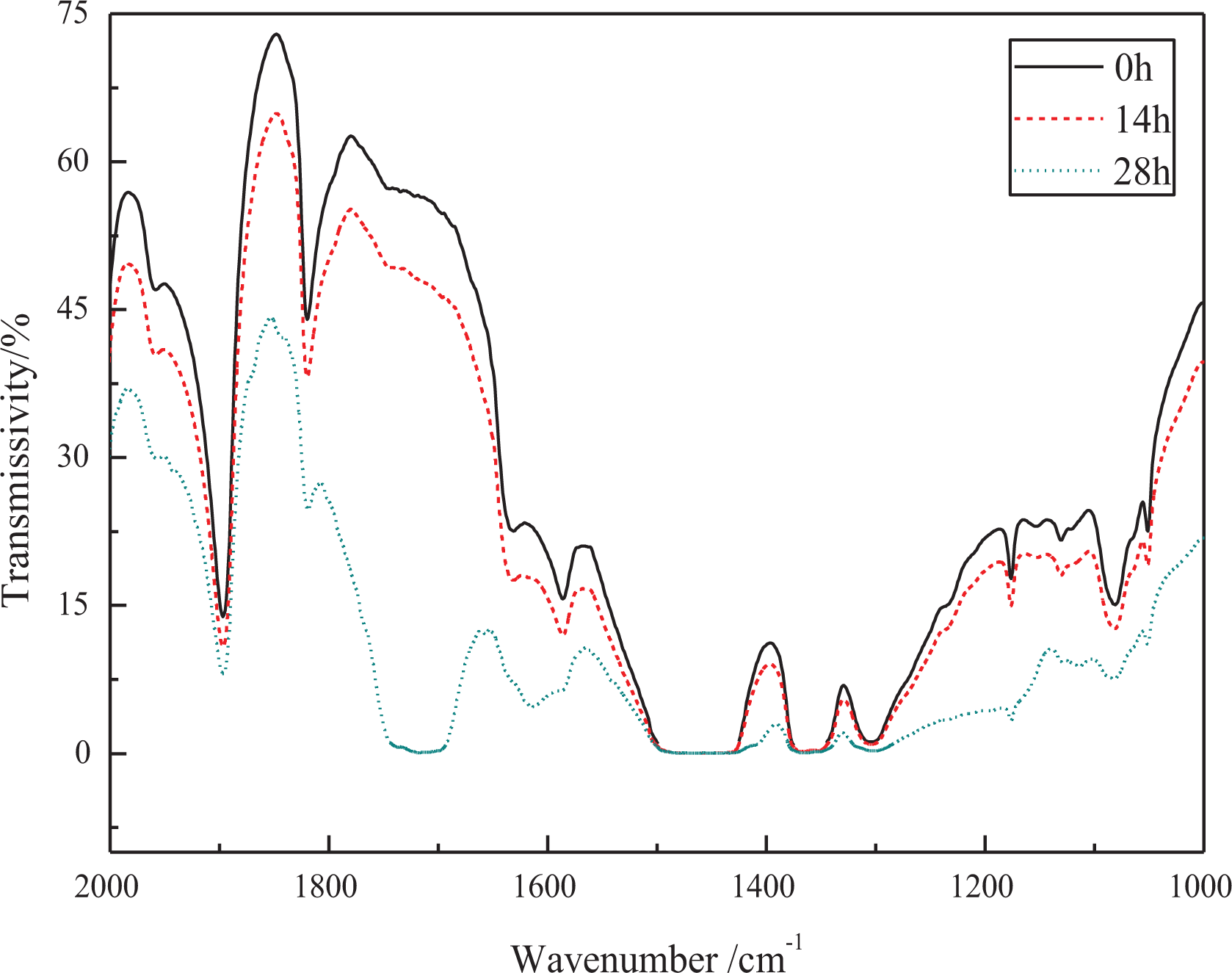
FTIR spectra of pure HDPE at increasing aging times.
Figure 6 shows the change of the CI as a function of the time for the HDPE until the 70 h. Clearly, the CI of the pure HDPE increases sharply just in 28 h, implying very easy oxidation of the pure HDPE. The CI values of HDPE containing Irganox 1024 and Irganox 3114 are 0.86 and 0.74 respectively when aging for 70 h, indicating that Irganox 1024 and Irganox 3114 can protect HDPE from oxidation to some extent. However, for HDPE with Irganox 1330 as a stabilizer, the CI value is only 0.32 when aging for 70 h, and Irganox 1330 endows PE with better aging resistance than Irganox1024 and Irganox 3114. When it comes to HDPE with Irganox 1010, it is a surprise that Irganox 1010 endows HDPE with much worse aging resistance than other three hindered phenolic antioxidants. The HDPE containing Irganox 1010 get embrittlement and pulverization when aging for 55 h. In contradiction to the above results, the result of short-term heat-oxygen stability show that antioxidant performance of Irganox 1010 is better than antioxidant 1024 and antioxidant 3114. The reason probably attributes the difference in their chemical structures. Hydrolysis is a known degradation mechanism (Figure 7) of Irganox 1010 where the reaction proceeds through successive nucleophilic attack of a water molecule at the carbonyl carbon atom of the ester groups ultimately leading to the formation of 3-(3,5-ditert-butyl-4-hydroxy-phenyl) propanoic acid and pentaerythritol.30,36,37

CI of various HDPE versus aging time.
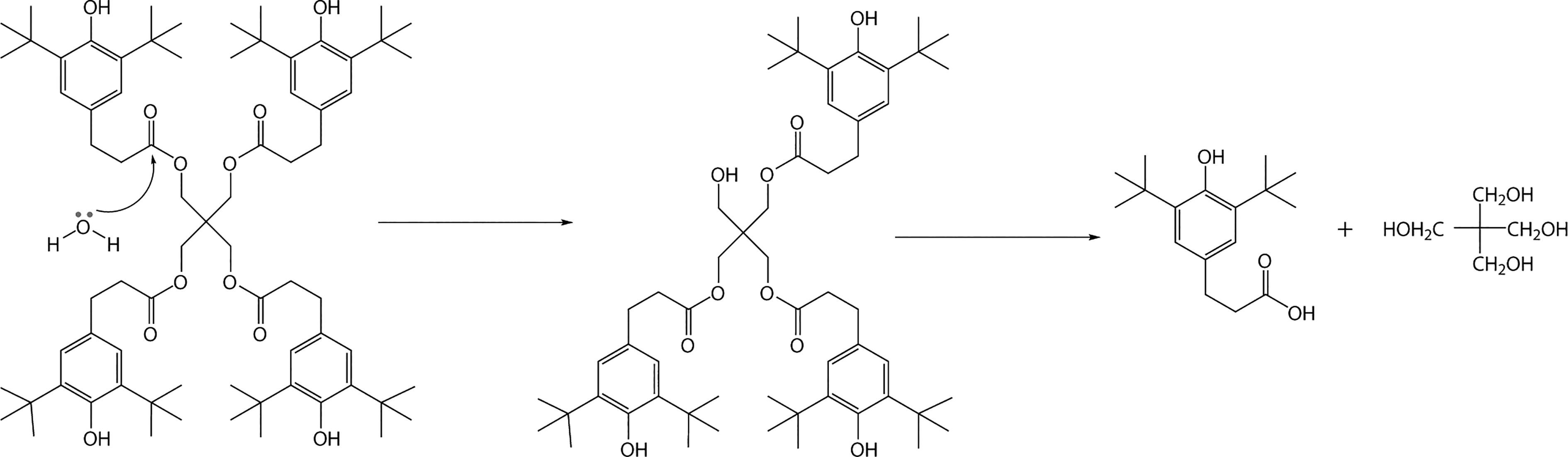
Hydrolysis of Irganox 1010.
The OIT values of the HDPE containing various antioxidants aged at 110°C in deionized water pressurized with pure oxygen at a pressure of 10 MPa for different aging time was shown in Figure 8.

OIT of various HDPE versus aging time.
As shown in Figure 8, the OIT value of pristine HDPE decreases to 2.47 min after only 28 h aging, indicating that the pristine HDPE is easily suffered from thermal degradation. Obviously, HDPE containing Irganox 1010 shows much quicker decrease than other three hindered phenolic antioxidants as a stabilizer, indicating that Irganox 1010 is sacrificed much faster. Moreover, the OIT values of the HDPE with Irganox 1330 are the biggest among the HDPE with other three hindered phenolic antioxidants at the same aging time. These results are in good accordance with the above results obtained by FTIR. This may be attributed that Irganox 1330 donates phenolic hydrogen to a peroxy radical formed as a result of the attack of oxygen on the polymer chain, or disproportionation of two molecules, there by forming the mono quinone (Figure 9b). Subsequently the remaining hydroxyl groups of Irganox 1330 are oxidized in the same manner to form the fully conjugated tri-quinone(Figure 9c, d). Moreover, Irganox 1330 with high molecular weight are insensitive to physical loss through migration.29,37,38
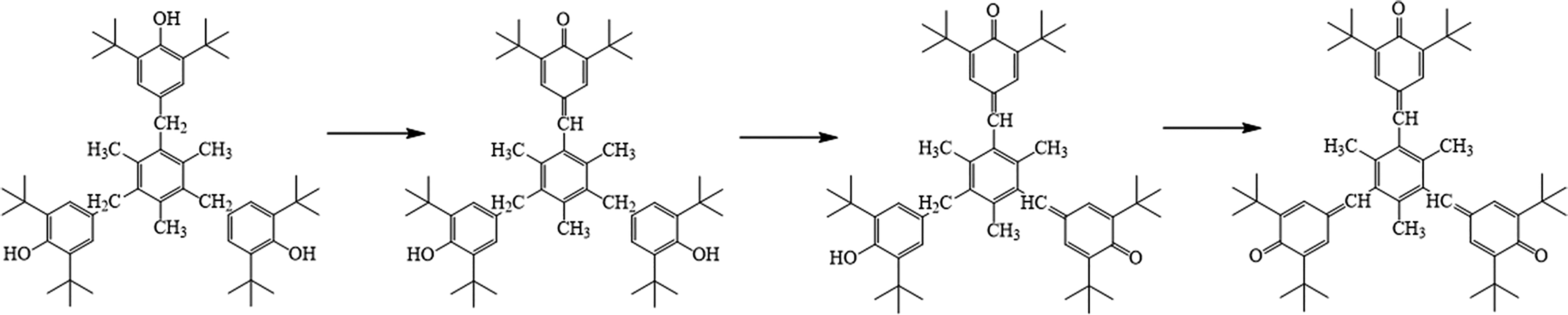
Formation of quinones during the oxidation of Irganox 1330.
Figure 10 and Figure 11 display the mechanical properties of HDPE without and with hindered phenolic antioxidants for the different aging time. The tensile strength and elongation at break of HDPE with hindered phenolic antioxidants are higher than that without antioxidant. Moreover, The tensile strength and elongation at break values of pure HDPE decrease to 5.2 MPa and 160% after only 28 h aging respectively. For the HDPE with hindered phenolic antioxidants, HDPE with Irganox 1010 fully cracked aging for 56 h, and HDPE containing Irganox 1024 and Irganox 3114 completely cracked after 70 h. While Irganox 1330 with higher molecular weight and excellent hydrolytic stability is best in stabilizing mechanical property of HDPE. These results are in good accordance with the above results obtained by FTIR and DSC.

Tensile strength of various. HDPE versus aging time.

Elongation at break of various. HDPE versus aging time.
Conclusion
The heat-oxygen stabilities of hindered phenolic antioxidants in HDPE were evaluated. The result of short-term heat-oxygen stability showed that the MFR and the OIT values of the HDPE with hindered phenolic antioxidants were higher than those of the pure HDPE. Moreover, Irganox 1330 and Irganox 1010 had better processing properties and oxidation resistance behavior in compound than Irganox 1024 and Irganox 3114. Long-term accelerated heat-oxygen aging tests of HDPE were carried out at 110°C in deionized water pressurized with pure oxygen at a pressure of 10 MPa and the results confirmed that Irganox 1010-stabilizing HDPE presented much worse antioxidant efficiency than other three hindered phenolic antioxidants, which is quite contrary to their short-term effect on HDPE stabilization. It may be speculated that the ester groups of Irganox 1010 undergo hydrolysis leading to the formation of 3-(3,5-ditert-butyl-4-hydroxy-phenyl) propanoic acid and pentaerythritol.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
