Abstract
Eco-friendly packing polymer materials are in the spotlight but, lack of new biodegradable polymers either natural or synthetic is yet to establish the market more competitively. So, in the present work, clay as a nano-filler is embedded and organically modified in some synthetic and natural polymers which are well established commercially to enhance their biodegradability. The impact of clay on the properties of synthetic polymers namely, poly(methyl methacrylate) (PMMA), poly(vinyl chloride) (PVC), poly(vinyl acetate) (PVAc) and natural polymer cellulose acetate butyrate (CAB) was studied. Results from differential scanning calorimetric (DSC) showed a decrease in the glass transition temperature of organically modified polymer clay nanocomposites (PCC) than pure polymers. Scanning electron microscopy (SEM) displayed a uniform surface with small-sized crystallites distributed on the polymer surface. X-ray diffraction (XRD) spectra revealed the formation of enhanced intercalated structures in PCC. Furthermore, FTIR studies showed that the interlayer bonding (Si–O bands) of pure clay is deformed in PCCs. The tensile strength of PCC increased with an increase in organo-clay loading. This unique mechanical behavior is due to the agglomeration of organo-clay particles. Finally, the biodegradation studies revealed enhanced hydrolytic degradation in PCC than pure polymers. Hence, these PCCs are environmentally friendlier than their pure synthetic polymers without significant compromise in their properties, which makes it suitable for packaging industries.
Keywords
Introduction
Organically modified polymer clay nanocomposites (PCC) possess high mechanical properties,1-3 thermal properties4-6 and liquid barrier properties7,8 than pure polymers and other conventionally modified microcomposites of clay and polymer. Conventionally, expensive clay minerals are often used in preparing microsized dispersion on polymer matrix to bring about these enhanced properties. 4 Later, sodium montmorillonite (Na-MMt) a natural smectite clay (2:1 phyllosilicate) consisting of aluminosilicate layers with a high aspect ratio and a high surface area were used to synthesize PCC. 7 Based on the mode of dispersion of clay in the polymer matrix two types of PCC are formed they are intercalated PCC and exfoliated PCC. Clay is found to be periodically dispersed in intercalated PCC while in exfoliated PCC clay nanoparticles are exfoliated from layers due to penetration of polymer chain through the layers. Hence, morphology control depends on the mode of preparation and the type of organic modifiers used.9,10 Moreover, in this present work intercalated PCC is obtained.
Organically-modified layered clay which mainly comprises of silicates is prepared by a cation-exchange reaction between a surfactant (alkyl ammonium salt) and the silicates. During the synthesis of PCC, organic modifiers replace the adsorbed water present between the layers of silicates. 11 Upon the addition of the polymer solution, the polymer slowly penetrates through the silicate layers with nanoscale thickness and suspends them in the polymer matrix.12-14 In Na.MMt, sodium cations are hydrated in the clay galleries making it hydrophilic. This reason is the main concern while mixing with organic polymers. To bring about organophilicity, interlayer inorganic cations are ion-exchanged with organic cations such as alkylammonium ions. Using this concept, the thermoplastics like polystyrene, polyamides, polypropylene were organically modified to prepare PCC.15,16 Laminated clay/polyvinyl alcohol nanocomposite food-packaging film exhibited a 99% decrease in O2 permeability. Hence, the introduction of organoclay into the polymer was found to improve its moisture barrier properties and establish in the packaging industry.
Hesham et al. 17 prepared toxicity-free expanded organoclay by modifying with stearic acid and mixed with Natureplast blend as nanocomposite and observed to have enhanced thermal, mechanical, and barrier properties. Sodium lactate loaded chitosan-poly(vinyl alcohol)/montmorillonite barrier film for water vapor, carbon dioxide, and oxygen with antibacterial activity was reported by Zhang et al. 18 Gul et al. 19 reviewed polymer-clay nanocomposites and showed that the addition of clay improved properties such as mechanical strength, thermal stability, and flame retardancy. Applications of PCC are found in automobile, aerospace, construction, biomedical fields, and barrier materials because of lightweight and low cost.
In this paper, an attempt is made to evaluate structural and tensile properties of synthetic and biodegradable polymer hybrids embedded with alkyl ammonium ion-exchange clays. The polymers which are in demand in packaging industries and yet to be studied such as poly(methyl methacrylate) (PMMA), poly(vinyl chloride) (PVC), poly(vinyl acetate) (PVAc), and natural polymer cellulose acetate butyrate (CAB) were selected to prepare the PCCs. Nevertheless, the PCCs synthesized using these polymers were subjected to biodegradation and found to have reduced degradation time. Hence, this work will overcome many hurdles of using expensive clay minerals, moisture sensitivity, and biodegradation without compromise in their properties.
Experimental details
Materials
The clay used is Na+-montmorillonite (NaMMt) and the organic modifier is cetyl ammonium bromide (CTAB). Sieve size of 325-mesh was used to remove impurities and the obtained clay had a cationic exchange capacity (CEC) of 119 meq/100 g for montmorillonite. PVC, PVAc, PMMA, CAB were purchased from the Merck. The number average molar mass of the corresponding polymer was 70000, 60000, 100000 and 40000, respectively. N, N,-dimethylformamide (DMF) was purified before use.
Purification of montmorillonite
In 5 L of 1 N NaCl solution about 25 g of the crude clay was dispersed and stirred for 24 h at 70°C. The mass was centrifuged to get the whitish layer accumulated as residue and a translucent gel as centrifugate. This centrifugate was washed with deionized water until no chloride was detected by testing with one drop of 0.1 N AgNO3 solutions. It was organically modified by subjecting to the cetylammonium ion exchange. The product obtained was then dried for 10 days at 75°C. Later, it was ground with a mortar and pestle and sieved using <50 µm mesh. The modified organophilic clay (organoMMT) was stored in desiccators. 20
Determination of cation exchange capacity (CEC)
The CEC of Na-MMt was determined by measuring the amount of alkyl ammonium salt being retained in organoMMT after careful washing. 21 The dried organoMMT and a blank sample of untreated clay were ignited at 1000°C in an electric furnace. From the weight loss between sample and blank, the mass of the alkylammonium salt, the milli equivalents of the organic substance held by the clay were calculated as its CEC. The maximum CEC of achieved clay was used for preparing polymer nanocomposites.
Preparation of CAB/organoMMT, PMMA/organoMMT, PVAc/organoMMT and PVC/organoMMT
Firstly, using optimized quantities of CAB, PMMA, PVAc and PVC annealing with (2, 4, 6, 8 and 10, wt%) organoMMT was performed above the softening point of the polymer, wherein the polymer chains diffuse into organoMMT. These films were dried for 24 h in a vacuum oven at 50°C. Secondly, the obtained composites were further subjected to a solution intercalation method at 25°C, using solvent DMF for PMMA/organoMMT and PVC/organoMMT. PVAc/organoMMT and CAB/organoMMT were prepared using methylene chloride (CH2Cl2) as a solvent. 22 Briefly, the procedure for solution intercalation of CAB/organoMMT is as follows: The annealed CAB/organoMMT composite was dissolved in CH2Cl2 for 24 h. It was subjected to sonication to bring about swelling to increase interlayer distance for an hour. The solution was mixed for 96 h to ensure equilibrium intercalation of polymers into layered silicates. The solution was cast on a glass dish and evaporated the solvent to obtain a dry film. The PCC was chopped into desired lengths. Similarly, other PCCs were prepared in respective solvents.
Degradation in activated sludge
BASF industrial waste treatment plant provided activated sludge for this study. The characteristic of sludge can be found in the literature. 23 The pure polymer films and PCCs were cut into 5 × 5 cm size, preweighed, and kept in jars containing fixed volumes of activated sludge solutions. To maintain aerobic conditions the jar was bubbled with air using a pump. Weights of pure polymer films and PCCs were observed for intervals of 10, 15 and 20 days by washing with double distilled water and drying in an oven at 60°C.
Characterizations
Miniflex-Rigaku, X-ray diffractometer with CuKα characteristic radiation (scan speed of 1°/min in a range of 2θ = 10–30°) was used to measure XRD. Glass transition temperature (Tg) was determined under a nitrogen atmosphere using SHIMADZU DSC-60 model from 35–250°C at 10°C/min. Surface morphology was studied using a JEOL JSM-630LA scanning electron microscope. FTIR spectra were obtained from Nicolet Avatar 5700 FTIR spectrometer. All the above tests were conducted on PCC having 6 wt% of organoMMT in polymers. Tensile strength was tested using INSTRON with 5 pts/sec of chart speed and 1.52 mm/min of crosshead speed for all the composition of organoMMT in the polymer. The tests were carried out at ambient temperature and 75% relative humidity.
Results and discussion
The modification of clay using the alkyl ammonium molecule generates an organophilic silicate. The interaction happens due to silicate layers having partial negative charges. Alkyl ammonium molecule having a cationic head group will reside on the surface due to these negative charges on the silicate layers and long aliphatic tail protrudes from the surface. 14 Schematically as shown in Figure 1 the polymer intercalates in organoMMT to form a hybrid composite.
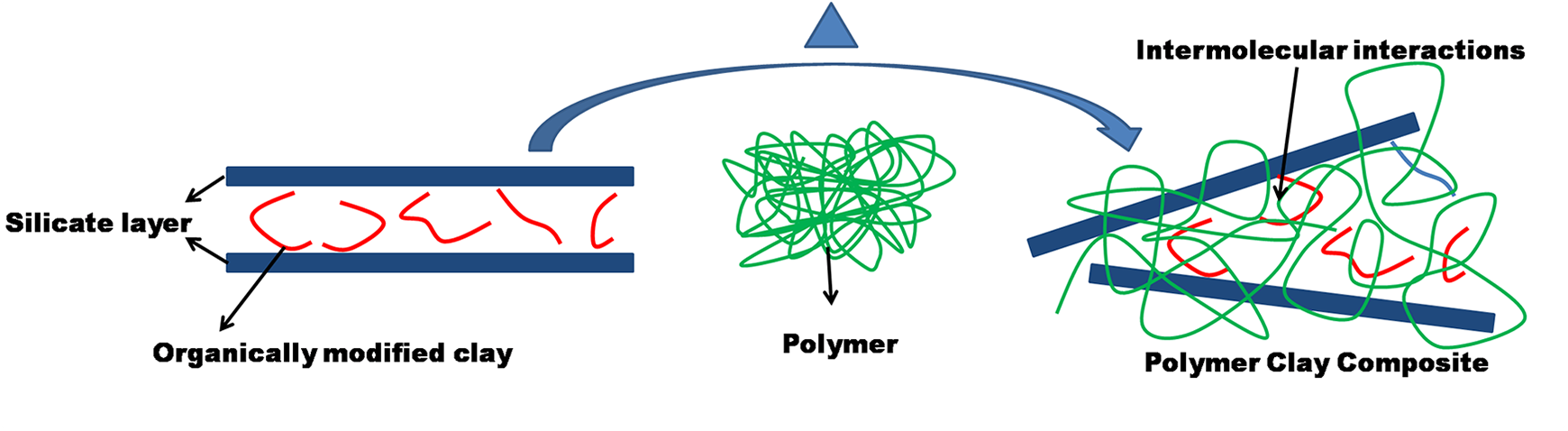
Schematic representation of intercalation between polymer solution and organoMMT.
X-ray diffraction
XRD traces, reveals that the nanocomposites formed with the clay are expected to be exfoliated or intercalated. 24 The XRD pattern of pure organoMMT, PVC/organoMMT, PMMA/organoMMT, CAB/organoMMT, and PVAc/organoMMT are shown in Figure 2. If exfoliation occurs in the composite, it means that there is a disorder introduced during the preparation process and more amorphous peaks are observed in XRD. This is not reflected in the XRD pattern; hence it is not an exfoliation method. In the present work, intercalated polymer clay nano-composites occur when the periodic layered structure of clay is maintained in nano-composites. This is reflected as crystalline peaks in XRD. In Figure 2, all the PCCs contain characteristic XRD diffraction peaks of the pure organoMMT at the region of 2θ = 20.71° and 26.52°, 25 suggesting crystallinity although the incorporation of polymers. However, these peaks in PCCs were of less intensity than that of organoMMT. Moreover, in respective PCC the d001 values at these regions increased indicating an increase in interlayer distance between silicates layers of organoMMT due to penetration of polymer. 9 These variations of d001 value were tabulated in Table 1. In respective PCC the full-width at half-maximum (FWHM) calculated value at regions 2θ = 20.71° and 26.52° was less than that of organoMMT. Reduced FWHM values in PCCs indicate that there is an absence of ordered stacks of silicates which leads to a slight widening of the peaks. 26 Furthermore, the relaxation of tensile stress decreases the FWHM 27 indicating that the PCCs structure is having less stress between molecules. This observation is also confirmed in a study of the tensile strength of these PCCs. This confirms the intercalation effect observed in the composites. Although the polymer chains are intercalated between the silicate layers, they possess restricted segmental motion. Hence, the preparation of PCCs using a solvent casting and melt intercalation can lead to the manufacture of an effective nanocomposite for packaging application without using any kind of coupling agents or modifications of guest monomer.

XRD pattern for different polymer modified clay at (a) organoMMT, (b) PVAc/organoMMT, (c) CAB/organoMMT, (d) PMMA/organoMMT and (e) PVC/organoMMT.
XRD data for the polymer clay composites.

FTIR spectrum
Figures 3(a) and (b) show a comparison of FTIR spectrum pure clay and organoMMT, respectively. The pure clay showed characteristic bands of OH, H–O–H, Si–O plane stretching, Al–Mg–OH bending, Si–O stretching of silica, Si–O bending parallel to the optical axis and Si–O–Si vibration at 3632, 3186–3520, 1652, 1046, 850, 800, 696 and 480 cm−1, respectively. As observed in Figure 3(b), the peaks at 3668 to 3517 cm−1 shifted to higher wavenumbers and 1658–480 cm−1 shifted to lower wavenumbers. Additionally, new peaks of ionic bonded NH stretching, CH asymmetric stretching, CH symmetric stretching, combination of OH and NH bending, and Si–O bending perpendicular to the optical axis at 3152–3517, 2923, 2852, 1658, and 723 cm−1, respectively were observed in modified organoMMT. 28 The presence of CH stretching peaks, shifts in characteristic peaks and formation of new bending bands in the organoclay indicates that the pure clay has been successfully modified using the organo-exchange method. 29
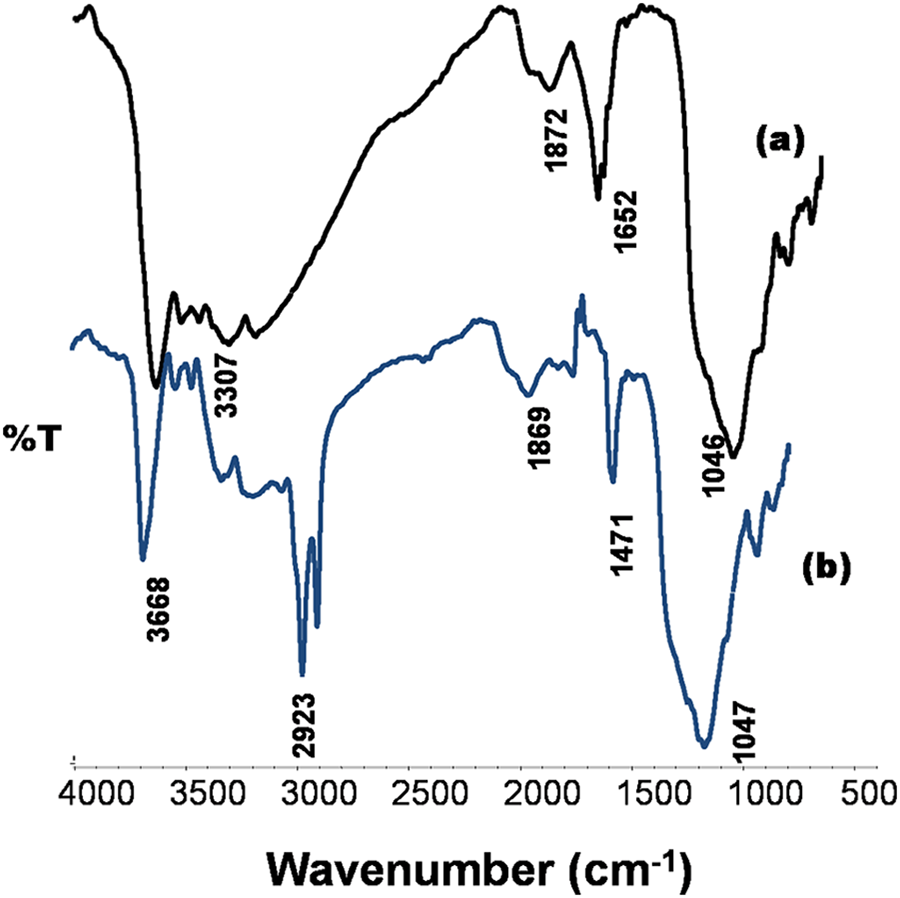
FTIR spectra of (a) pure clay, (b) organoMMT.
FTIR spectrum of PMMA/organoMMT, CAB/organoMMT, PVC/organoMMT, PVAc/organoMMT are compared in Figures 4(a), (b), (c) and (d), respectively. The sharp peak at 3632–3000 cm−1 for OH and NH stretching observed in organoMMT is broadened in all the PCCs indicating specific interaction of organoMMT with polymers. Furthermore, Si–O in-plane stretching in range of 1003–1087 cm−1; Si–O bending parallel to the optical axis in the range of 696–750 cm−1 observed in all PCCs are sharper and shifted to lower energy by 10–12 cm−1 when compared to the spectrum of organoMMT. The shift is probably due to the formation of hydrogen bond interaction of the polymer with silica tetrahedral oxygen in the intercalated layers of organoMMT. 30 The shift to low energy by Al–Mg–OH bending peaks observed at region 924 to 854 cm−1 and presence of weak Si–O–Si at 514 to 490 cm−1 in PCCs compared to organoMMT indicates that there is a formation of hydrogen bonding with polymers and breakage of interlayer bonds in the clay. 31 The characteristic peaks of polymer in PMMA/organoMMT, CAB/organoMMT, PVC/organoMMT and PVAc/organoMMT are observed as follows: C=O stretching at 1722, 1735 cm−1, absent, and 1718 cm−1; CO–H bending at 1437, 1466, 1425 and 1494 cm−1; and symmetric R–COO− stretching at 1386, 1398, absent and 1372 cm−1, respectively. These peaks in PCCs are vibrating in lower energy than the pure polymers 32 indicating polymers have interaction with organoMMT and it is embedded with exfoliated organoMMT layers.
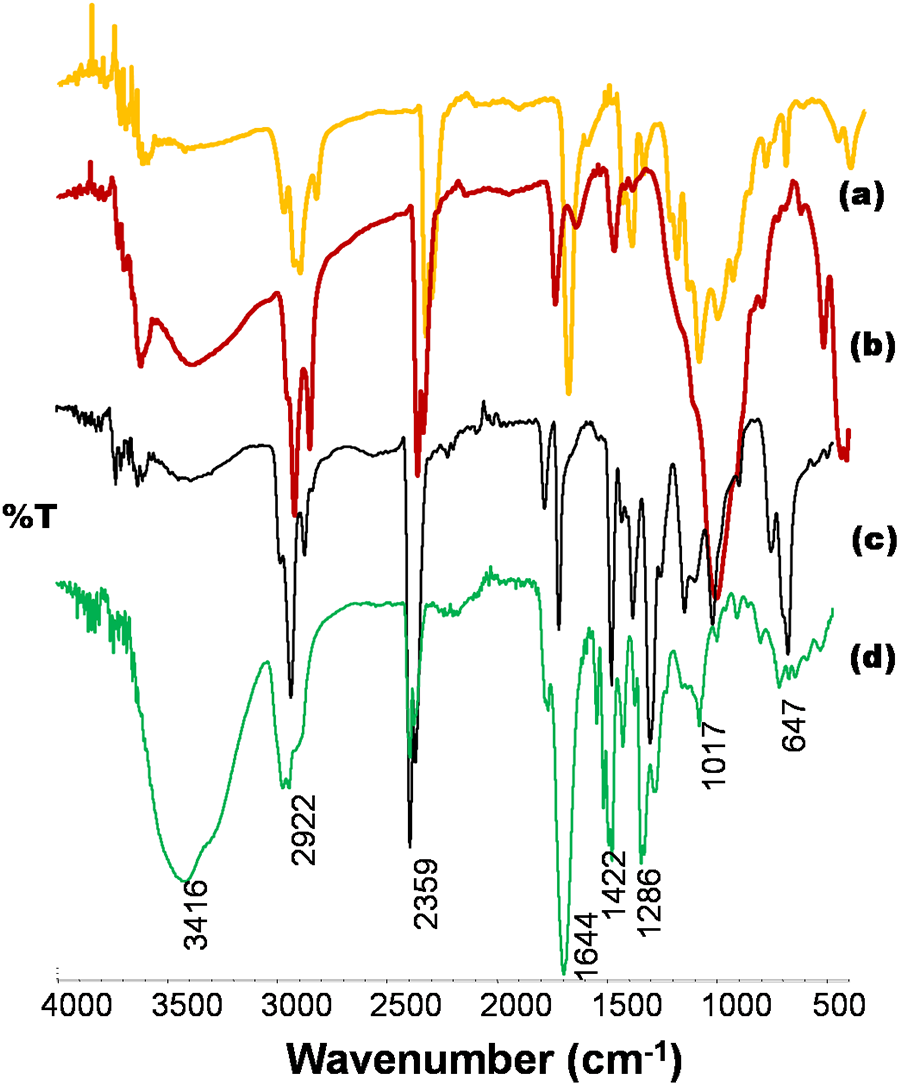
FTIR spectra of (a) PMMA/organoMMT, (b) CAB/organoMMT, (c) PVC/organoMMT, (d) PVAc/organoMMT.
Surface morphology
Figure 5(a) to (d) show the SEM images of PMMA/organoMMT, CAB/organoMMT, PVC/organoMMT, PVAc/organoMMT composite films, respectively. In PMMA/organoMMT (Figure 5(a)) the flakes of exfoliated organoMMT are dispersed over the surface of the polymer. In CAB/organoMMT (Figure 5(b)) tiny crystallites of organoMMT are observed scattered over the surface. In PVC/organoMMT (Figure 3(c)) the fractured surface of organoMMT intercalated on polymer surface is observed. In PVAc/organoMMT (Figure 3(d)) more fractured and voids due to the presence of organoMMT are observed. The fractured surfaces and voids are due to the finely dispersed clay particles in the polymer matrix. Deformed parts may be resulted due to the agglomeration of the fractured surface and increase of basal interlayer spacing in organoMMT. Therefore, the uniform dispersion of clay in the polymer matrix possesses good mechanical properties. 27

SEM images of (a) PMMA/organoMMT, (b) CAB/organoMMT, (c) PVC/organoMMT, (d) PVAc/organoMMT.
Thermal studies
The effect of the organoMMT on glass transition temperature (Tg) of pure polymers was investigated using the DSC technique. DSC thermograms of PMMA/organoMMT, CAB/organoMMT, PVC/organoMMT, PVAc/organoMMT are shown in Figure 6(a), (b), (c) and (d), respectively. As observed in the Figure the Tg of PMMA (120°C), CAB (70°C), PVC (60°C), PVAc (45°C) had decreased upon addition of organoMMT.9,12 This indicates that the intramolecular interactions in polymer have reduced due to the presence of entrapped exfoliated organoMMT layers. Although there is an interaction between organoMMT and polymers as observed in FTIR and XRD, the rise in temperature increases the segmental motion of the polymer thereby decreasing the Tg. 27
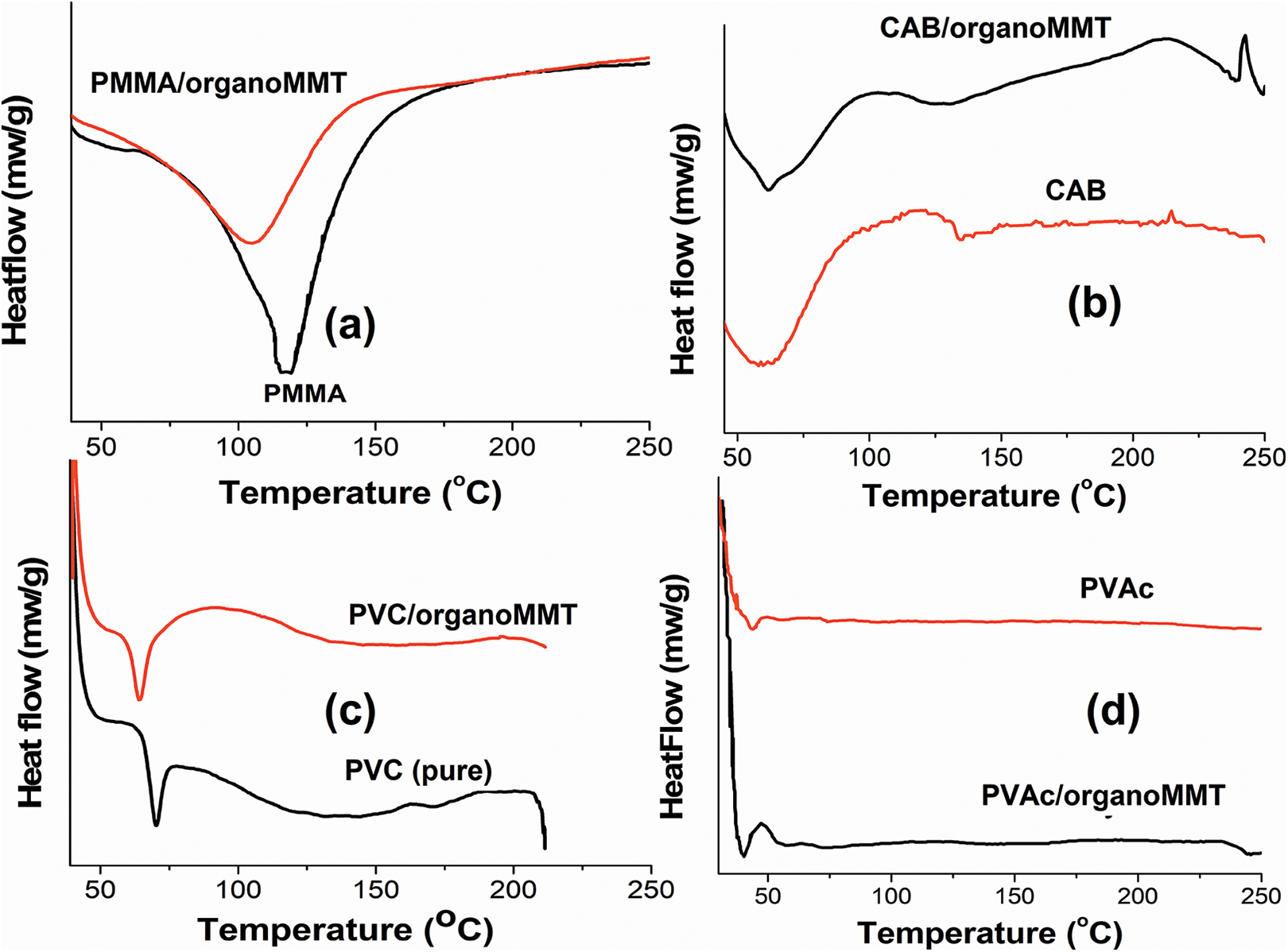
Thermal behavior of (a) PMMA/organoMMT, (b) CAB/organoMMT, (c) PVC/organoMMT, (d) PVAc/organoMMT.
Tensile properties
Figure 7 shows the plots of (%) organically modified clay vs. tensile properties of PMMA/organoMMT, CAB/organoMMT, PVC/organoMMT, and PVAc/organoMMT composites. The tensile strength of the PCCs increased as the clay content was increased from 0 to 6 wt %, but increase beyond 6 wt % the tensile property of the composite decreased. Nonetheless, it implies that the strength of the PCC materials increases slowly upon the addition of organoclay. This exclusive strength-mechanical behavior of the nanocomposite is mainly due to the agglomeration of organo-clay particles and intercalation between the organoMMT and the polymer matrix. 14 Moreover, the flexible alkyl groups in the clay help to orient the polymer chains properly. As a comparison between the prepared PCCs, the PMMA/organoMMT had the highest tensile strength due to its high elasticity while PVC/organoMMT having more crystalline structures had the least tensile strength. This enhanced property as observed in the tensile study along with supported SEM morphology and XRD studies suggests that the organoMMT is well dispersed in the PMMA matrix of PMMA/organoMMT composite. Even in the report by Lorenzo et al., 33 it was observed that the thermo-mechanical properties increased up to 10 wt% of clay in bioplastics and decreased as the clay content reached 30 wt%. Food packaging material prepared by organically modifying sodium montmorillonite by octadecylammonium-zinc oxide nanoparticles and incorporating into the chitosan matrix exhibited similar enhancement in thermo-mechanical properties 34 due to their compatibility. The PCCs for use in packaging industries reported in Idumah et al. 35 showed similar results wherein organically modifying the clay improves the thermo-mechanical properties of polymers. As a future scope, the clay can be imbedded in poly(N-vinyl caprolactam) a thermoresponsive polymer,36-39 and improve thermo-mechanical for various industrial applications.

Tensile properties of PCCs.
Degradation studies
Degradation of PMMA/organoMMT, CAB/organoMMT, PVC/organoMMT, and PVAc/organoMMT composites in the activated sludge was monitored for 20 days. After 20 days the PMMA/organoMMT, CAB/organoMMT, PVC/organoMMT, and PVAc/organoMMT underwent more degradation by 1%, 2.2%, 0.8% and 1.3%, respectively with respect to their pure polymers. This can be due to the hydration of organoclay in PCCs which further separates the layers and breaks the intramolecular bonds in the polymer. 31 The PCCs showed sufficient degradation due to hydrolytic degradation in activated sludge. 40 The problems such as to know the exact mechanism of degradation of the polymer upon addition of clay, organically modifying the clay using different surfactants, and synthesis of co-polymer-based clay composites discloses the scope for future work.
Conclusion
In this article, the structures of organically modified clay embedded in the synthetic and biodegradable polymers were characterized with a focus on the layered polymer dispersion. The changes in the thermo-mechanical properties were studied. In XRD the crystalline sharp peaks of clay became broader upon annealing with a polymer which reveals that the organoMMT has been successfully intercalated by the polymer chains. FTIR studies showed the existence of an interaction between organoMMT and respective polymers. The surface morphology studies revealed that the PVC and PVAc based PCCs had more voids, broken surfaces and aggregated compared to PMMA and CAB based PCC composites. Thermal studies show an increase in the segmental motion of PCCs at a temperature lower than that of their pure polymers. The tensile strength measurement indicated that the tensile strength of the polymers increased with an increase in clay content up to a critical point, beyond which it decreased. This demonstrates that the clay acts like a pillar in the polymer matrix. Comparatively, there was greater enhancement of degradation rate in PCCs than pure polymers. Hence, the inclusion of modified clay in the polymer properties such as tensile strength and biodegradation has improved thereby, the PCC is a potential candidate for use in packaging industrial applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
