Abstract
Natural plant fiber-reinforced polymer composites have been in the limelight in the field of materials science for their mechanical properties, economy, and eco-friendliness. Properties of such novel composites depend on the adhesion and interaction between the fibers and the polymer matrix. Consequently, poor interaction can lead to declined mechanical properties, particularly strength. Surface modifications of fibers are carried out to enhance the bonding with the matrix by certain chemical treatments that remove hydroxyl groups in the amorphous cellulose region, making them hydrophobic and hence compatible with the matrix. Doing so also strengthens the composites, widening their scope of application. This review article provides comprehensive information about various surface modification techniques that include alkali, silane, acetylation, permanganate, peroxide, benzoylation, acrylation, acrylonitrile grafting, isocyanate, addition of maleated coupling agents, and fungal treatments. The working mechanisms and the effects of such treatments on mechanical strength are also elucidated. Furthermore, this review provides an overview of nanoclay inclusion in polymers, their addition techniques, and the augmentation of the mechanical properties of polymer matrix composites. The article concludes along with the field of applications, summary of pertinent challenges, and directions for future work.
Keywords
Introduction
Environmental consequences of practices have led to a search for biodegradable materials for day today applications. Industries now give importance to sustainability and energy costs besides considering the strength and processability of materials for the application. Not until two decades back when the recyclability of materials was burgeoning, the renewable raw natural plant fibers saw the limelight. 1 With the involvement of natural plant fiber-reinforced composites (NFRCs) in current research, extensive work has been carried out in implementing them in automotive and construction industries.2-5
The salient features of the NFRCs include low density, low cost, good specific strength properties, 6 and high biodegradability. 7 These composites have better mechanical properties than their pure polymer counterparts.8-11 In a comparative study with glass fibers, NFRCs have been verified as environmentally superior12,13 consuming 80% less energy for production. 14 Their low abrasive nature makes them a better choice than glass fibers from manufacturing and safety aspects. The major limitations of NFRCs include water retention and poor thermal stability. 15 These properties depend on the structure which, in turn, is influenced by the conditions of growth, such as land, climate, and age of plant.
Though the idea of creating biofriendly materials is captivating for industrialists and environmentalists, the NFRCs are far below the glass fiber composites when mechanical properties are compared. The need for improving the mechanical properties of NFRCs has created a foray into a new set of research in composite science, which has highlighted the main cause for lower values of mechanical properties as the inadequate bonding between the natural plant fibers and the polymer matrix. 16 Having provided a direction to work on, a major research effort is involved in treatments of fiber surfaces that make them compatible with the matrix to provide greater strength for the composite.
In the quest for identifying procedures to increase the composite strength, surface treatments of fibers have proved to be of great help.17-20 These treatments target the amorphous cellulose region to improve compatibility between the fiber and the polymer matrix. The amorphous portion of surface cellulose contains multiple hydroxyl groups, which render the fibers a polar nature and result in poor bonding with the predominantly nonpolar polymer resin. 21 Surface treatments help in enhancing the surface properties or reducing the polar nature by the removal of hydroxyl groups, thus improving the fiber–matrix adhesion and providing higher mechanical strength. 22 In recent years, the introduction of nanoclay dispersion in polymers has raised the attention due to their commendable property of strength enhancement through exfoliation. With 42% of the polymer global market focused on packaging, nanoclay/natural plant fiber hybrid composites are considered to be promising materials. 23
Components of natural plant fibers
Plant fibers are lignocellulosic in nature, containing cellulose, hemicellulose, lignin, pectin, and waxy substances, as shown in Figure 1(a) and (b). Their relative proportions dictate the mechanical properties of the NFRCs along with aspect ratio, morphology, hydrophilic tendency, and the dimensional stability of the fibers used. The major component is the cellulose, as shown in Figure 2, which provides strength, stiffness, and structural stability to the fiber, having multiple hydroxyl groups. 1 The cellulose in the fiber cell wall is present as crystalline regions and amorphous regions. The strong intramolecular hydrogen bonding in the crystalline region makes it impenetrable by other chemicals, including water. On the other hand, the amorphous regions easily absorb resins and dyes. The hydroxyl groups in this region easily combine with water molecules, making the fibers hydrophilic and polar in nature. 17

(a) Structure of a plant fiber and (b) structural organization of the constituents in the fiber cell wall. 17

Chemical structure of cellulose. 17
Hemicellulose is a group of polysaccharides (excluding pectin) that remains on removal of lignin, as shown in Figure 3. With low degree of polymerization, it exhibits high chain branching. Such branching strengthens the cell walls by interaction with cellulose and in some walls with lignin. Lignin has an aromatic structure and is amorphous in nature, as shown in Figure 4. While cellulose content increases from the primary to the secondary layers, lignin content decreases in this sequence. Lignin is less hydrophilic than the cellulose and hemicellulose. With an increase in lignin content, water absorption is reduced. Pectin comprises complex polysaccharides cross-linked with calcium ions and arabinose sugars. Lignin and pectin are coupled with the cellulose–hemicellulose network and provide adhesion between the molecules. Waxes, being minor elements of plant fibers, constitute different types of alcohols, which are insoluble in water and in several other acids. The chemical compositions of selected plants are presented in Table 1.

Chemical structure of hemicellulose. 17
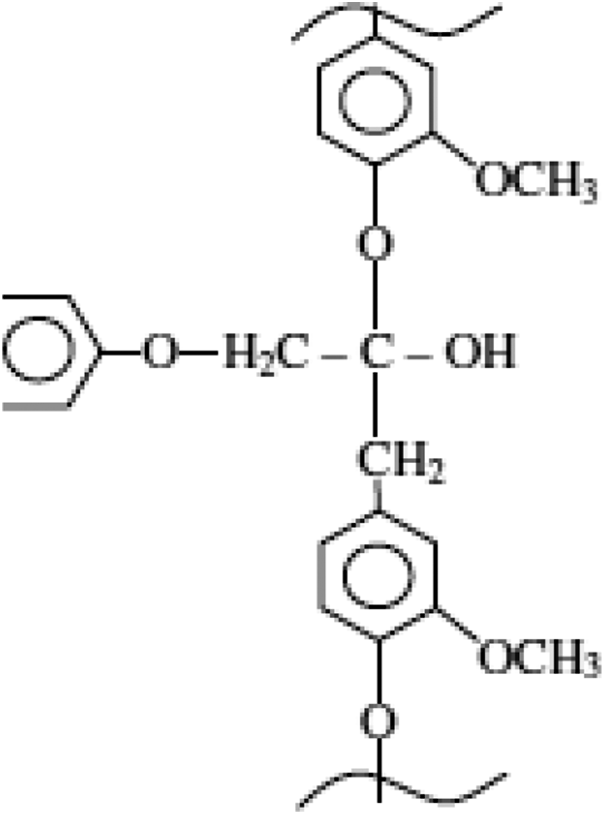
Chemical structure of lignin. 17
Higher concentrations of hydroxyl rich cellulose, hemicellulose, and lignin attribute to poor bonding with matrix materials. 27 The extent of interfacial interaction between the fiber and the matrix determines the stress transfer ability of the composite. 28 In addition to the fiber composition, the interfacial bonding depends on the surface morphology of the fiber, polarity of the matrix, and the presence of reactive probes in the matrix. Hence, chemical treatments that use reagents to alter the structure and composition of fiber surface have a great tendency to lower moisture absorption and improve stress transfer capabilities.
The fiber failure occurs due to fibril slip under the application of tensile stress. As a result, the total stress is shared by only a few cells. Further increase in the stress leads to fiber failure by cell wall rupturing and decohesion of cells. Surface modifications remove the amorphous components in the fibrillar structure, thus changing the deformation behavior of the fibers. The brittleness of the fiber is substantially reduced upon treatments. Table 2 presents the mechanical and physical properties of plant fibers.
Surface modifications by chemical treatments
The two primary components of the fiber polymer composites, viz., fibers and matrix influence the mechanical properties of the composite. 31 Fibers carry the imposed load while matrix transfers the load between the fibers. Furthermore, the fiber–matrix interface should have enough strength for loads to traverse the interface without inducing cracking and delamination. Tensile strength of the fiber is influenced by the refinement of the fiber surface. The voids and the amorphous portions of the fiber are responsible for about 10% of moisture content. 32 With regard to water absorption, refinements by chemical treatments enhance the hydrophobicity of the fibers. However, the chemically treated fiber polymer composites show higher water absorption when compared to pure polymer (without reinforcements) due to decreased fiber–matrix adhesion. 33 So, it is important for the user to define the required mechanical properties of the composite to find the “sweet spot” between the untreated NFRCs and pure polymers. These treatments are aimed at lowering the concentrations of voids and amorphous regions in the fibers.
Composites containing chemically treated fibers exhibit superior mechanical properties than untreated fiber-reinforced composites owing to better packaging of cellulose chains upon dissolution of lignin.34,35 Priya and Tiwari 36 showed that an improved interfacial bonding can be achieved through alkali, permanganate, benzoyl chloride, maleic anhydride (MA), and silane treatments, which improve the damping properties of the composite in comparison to those reinforced with untreated fibers. The commonly used surface modification treatments are described in the following sections.
Alkaline treatment
Alkaline treatment or mercerization involves the application of sodium hydroxide (NaOH) to the fibers. Alkali-sensitive hydroxyl groups (OH) present along the surface of fibers react with NaOH and release water molecules from the process. On reduction of the number of hydroxyl groups, the moisture absorption tendency is lowered. In addition, it also dissolves certain amounts of hemicellulose, lignin, pectin, and the waxy layer. The reaction mechanism between the fiber and alkali is shown in equation (1)

Alkali treatment results in the improvement of fiber surface adhesion characteristics by producing a rough surface topography, which enhances the mechanical interlocking at the interface. 28 This is achieved by removing natural and artificial impurities and disrupting hydrogen bonding in the network structure.26,37 The fiber is also made more thermodynamically stable.38,39 Alkali treatment eliminates microvoids, making the surface more even and improving the stress transfer capacity. A decrease in the fiber diameter that resulted an increase in aspect ratio can be noticed on NaOH treatment, which leads to good fiber–matrix interactions because of a larger effective surface area. 17 Being effective in removing the waxy layers and increasing the effective surface area, mercerization is often used as a pretreatment of fibers before other chemicals are used for strengthening.
Mechanical, thermal, and water retention behaviors are significantly improved with alkali/NaOH treatment. The concentration of NaOH solution and the soaking time required for obtaining the best mechanical properties should have optimum values. Increasing concentrations have been associated with the increase in mechanical properties. Further increase in concentration beyond the optimum value resulted in delignification of fibers and hence in damage.26,40 An optimal 5% NaOH-treated Tridax procumbens fiber gives increased wettability, crystallinity, reduced amorphous region, and fiber diameter. 41 Alkaline treatment also improves the tensile modulus and the flexural strength of the NFRCs. 42 Table 3 provides an overview of the acceptable concentrations of NaOH and the soaking time, which give an optimum strength, before delignification, which leads to wear and tear of the fiber.
Overview of acceptable concentrations of NaOH and its soaking time.

PF: phenol formaldehyde; PLA: polylactic acid; PP: polypropylene; LDPE: low-density polyethylene; HDPE: high-density polyethylene; WSF: wheat straw fiber; PALF: pineapple leaf fiber; MAPP: maleated polypropylene; MAPE: maleated polyethylene; FTIR: Fourier transform infrared; NaOH: sodium hydroxide; PSMA: polystyrene maleic anhydride.
Silane treatment
Silane (SiH4) being a multifunctional molecule is added as a coupling agent to enhance the fiber–matrix interaction. It undergoes several stages of hydrolysis, condensation, and bond formation during the treatment of fibers. In the presence of moisture, silanol is formed. The reaction mechanism for the same is shown in equation (2)

During condensation, one end of silanol reacts with the cellulose hydroxyl group of the fiber and the other end reacts with the functional group of the matrix, thereby creating a chemical bond between the two, 17 as shown in equation (3)
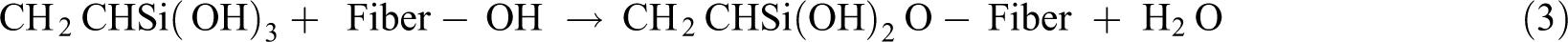
Generally, silane treatment starts with a derivative of silane (typically amine) with its solution in alcohol/acetone. Upon soaking the fibers in the silane solution, the modified fibers interact better with the matrix than alkaline-treated fibers, leading to higher tensile strength, tensile modulus, flexural rigidity, and thermal stability.26,42
Soaking of sisal fiber in 2% aminosilane containing 95% alcohol for 5 min at a pH value of 4.5–5.5 was done by Rong et al., 37 which led to improved fiber stiffness by enhancing crystallinity of the hard cellulose. Silane solutions in a water ethanol mixture with concentration of 0.033% and 1% were also used by Valadez et al. 45 and Agrawal et al. 62 to treat henequen fibers and oil palm fibers. Improved interfacial interactions and crystallinity percentage were observed. Zegaoui et al.63,64 used treated natural hemp and basalt fibers reinforced in cyanate ester and benzoxazine, respectively. They reported the improvement in microhardness, flexural strength, and thermal stability. Atiqah et al. 65 tested sugar palm fibers with silane, alkaline, and combination of silane and alkaline treatments. Doing so improved fiber roughness and thus, fiber–matrix bonding by removing the outer layers that contain impurities, less nodes, wax, and pectin. The works related to silane treatment on NFRC are summarized in Table 4.
The works related to silane treatment of fibers in NFRCs.

NFRC: natural plant fiber-reinforced composite; PF: polypropylene; PP: polypropylene; PS: polystyrene; HDPE: high-density polyethylene; LDPE: low-density polyethylene; PLA: polylactic acid; PALF: pineapple leaf fiber; NaOH: sodium hydroxide; APS: 3-amino propyltriethoxysilane.
Acetylation treatment
Acetylation treatment involves the addition of reagents with an acetyl group (CH3CO), such as acetic acid and acetic anhydride. The acetyl group reacts with the cellulose fibers, followed by the removal of hydroxyl groups, hence reducing the moisture absorption capacity. 71 While other chemical treatments leave more voids after the reaction, acetylation provides a rough surface topography with less number of voids, as a result of giving better interlocking capabilities. Acetylation could be performed with or without an acid catalyst, as shown in Figure 5(a) and (b), although the subsequent addition of acetic anhydride after soaking the fibers in acetic acid would respond faster. 17
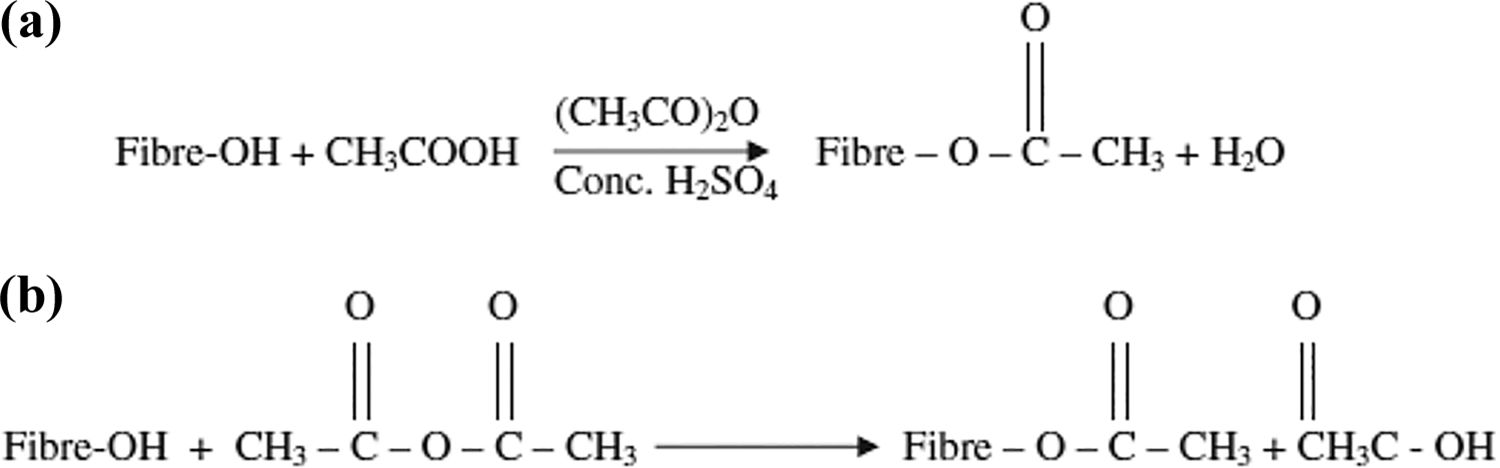
(a) Acetylation with acid catalyst and (b) acetylation without acid catalyst.
It is suggested that a limited amount of liquid acetic anhydride without a catalyst or cosolvent at 120–160°C can be used for acetylating the lignocellulosic mass. Acetic acid in small amounts could also be used to initiate the reaction by swelling the cell wall. 72
In tensile testing, the fibrillation is found to occur due to removal of binding materials and appearance of some micropores in the treated fibers. Furthermore, for higher degrees of acetylation, the fibrillation increases leading to fiber damage and cracks. 39 Moreover, improvement in dimensional stability, resistance to biological attack, and degradation due to ultraviolet energy can be observed in acetylated fiber composites. 73 Acetylation reduces water absorption and hence improves dimensional stability.72,74 Acetylation is also known to improve the tensile strength, stiffness, and the bond shear strength.42,75 Table 5 summarizes pertinent works on acetylation treatment of NFRC.
The works related to acetylation treatment of fibers in NFRCs.

NFRC: natural plant fiber-reinforced composite; PF: polypropylene; PP: polypropylene; PS: polystyrene; WSF: wheat straw fiber; LDPE: low-density polyethylene; H2SO4: sulfuric acid; NaOH: sodium hydroxide; MAH: maleic anhydride.
Permanganate treatment
Potassium permanganate (KMnO4) in acetone solution releases highly reactive Mn3+ ions. These ions react with the hydroxyl groups of the cellulose and form cellulose-manganate, as shown in equation (4). This initiates graft copolymerization, as shown in equation (5), providing high thermal stability to the fiber. In general, the experimentation is done using different concentrations of KMnO4 solution in acetone with soaking (1–3 min) after alkaline pretreatment
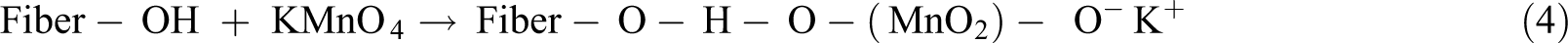
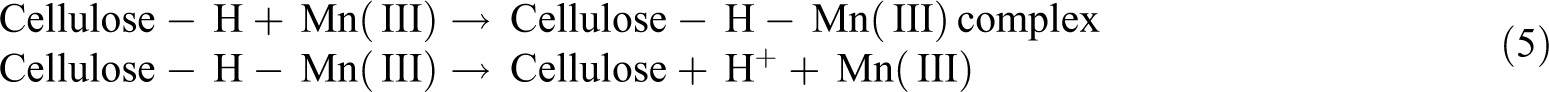
Apart from the removal of cellulose hydroxyl groups, it also reacts with lignin and separates it from the cell wall. As a result, high hydrophobicity is observed. The hydrophilic tendency of fiber decreases as the KMnO4 concentrations increase up to an optimum value. At a concentration of 1%, polar groups between fiber and matrix are formed leading to degradation of cellulosic fiber. 47 The fiber surface is etched by the oxidization of permanganate and becomes physically rougher. In this case, the interfacial adhesion is improved by the induced mechanical interlocking, and the contact area between the fiber and the matrix is also increased. 48 This mechanical bonding of the roughened fibers with the matrix due to oxidation can be contrasted with chemical bonding, as in the case of silane treatment. 79 The ductility, impact strength, flexural strength, flexural modulus, and thermal stability of NFRCs are improved by permanganate treatment.42,80-82 Using KMnO4 improves fiber roughness 83 and oxidizes the hydroxyl groups in cellulose to carboxyl and aldehyde groups. 80 Table 6 presents pertinent works on permanganate treatment of NFRCs.
The works related to permanganate treatment of fibers in NFRCs.

NFRC: natural plant fiber-reinforced composite; PF: polypropylene; PP: polypropylene; LDPE: low-density polyethylene; LLDPE: linear low-density polyethylene; HDPE: high-density polyethylene; NaOH: sodium hydroxide; KMnO4: potassium permanganate.
Peroxide treatment
Organic peroxides (RO–OR) are functional groups containing divalent O–O ion. These peroxides have a high tendency to decompose into free radicals (RO). These highly reactive free radicals react with the hydrogen in the hydroxyl groups, as shown in equation (6).
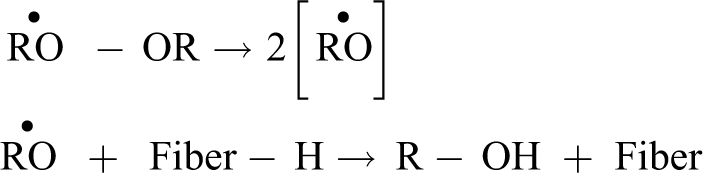
Additionally, when the free radical reacts with the matrix molecule, say polyethylene (PE-H), the reaction shown in equation (7) takes place to give polyethylene (PE) 47

The PE radicals could react with each other leading to cross-linking or grafting onto cellulose fibers, as shown in equation (8)

The commonly used organic peroxide treatments are benzoyl peroxide (BP, (C6H5CO)2O2) and dicumyl peroxide ((C6H3C(CH3)2O)2). The reaction of peroxide decomposition occurs during the time of curing of composites at the interface. Higher temperature is favored for the decomposition of the peroxides. 28 There exists a critical peroxide concentration, where the tensile strength of the composite reaches a maximum value for a given fiber content. At this stage, the fibers are covered with grafted polymer (matrix) molecules and further addition of peroxide contributes to some cross-linking of matrix molecules, which have negligible effect on the mechanical properties of the composite. 48 Therefore, peroxide treatment reduces water absorption and improves tensile strength, modulus of elasticity, thermal stability, and interfacial adhesion between the fiber and matrix.82,86-88 Table 7 summarizes pertinent works on some peroxide treatment of NFRCs.
The works related to peroxide treatment of fibers in NFRCs.

PALP: pineapple leaf powder; H2O2: hydrogen peroxide; NFRC: natural plant fiber-reinforced composite; PF: polypropylene; LDPE: low-density polyethylene; HDPE: high-density polyethylene; DCP: dicumyl peroxide; BP: benzoyl peroxide; NaOH: sodium hydroxide; PLA: polylactic acid.
Benzoylation treatment
Benzoylation is an effective method in enhancing the hydrophobicity and thermal stability of fibers. Benzoyl chloride, being the most used chemical in this treatment, includes the benzoyl (C6H5C=O) group, which is responsible for decreased hydrophilic nature of the treated fiber and improved interaction with the hydrophobic matrix.
During the process, the fibers are pretreated with alkali, as shown in Figure 6. On removal of the extractable materials like lignin, waxes, and oils covering the fiber, more reactive hydroxyl groups are exposed on the surface. These fibers are treated with BP. In this step, the benzoyl groups replace the hydroxyl groups on the fibers. This greatly enhances hydrophobicity and adhesion with the matrix.

Benzoylation.
Benzoylation roughens the fiber surface leading to fibrillation and provides better mechanical interlocking with the polymer matrix. For a tensile test, fiber breakage was observed rather than fiber debonding due to better fiber/matrix adhesion. 79 This technique improves the tensile strength and Young’s modulus, increasing the glass transition temperature and thermal stability while reducing moisture absorption.94-96 It also reduces voids and improves the hardness. 97 Table 8 lists pertinent works on benzoylation treatment in NFRCs.
The works related to benzoylation treatment of fibers in NFRCs.

NFRC: natural plant fiber-reinforced composite; PF: polypropylene; LDPE: low-density polyethylene; HDPE: high-density polyethylene; PP: polypropylene; PS: polystyrene; BP: benzoyl peroxide; NaOH: sodium hydroxide.
Acrylation treatment
Acrylic acid (CH2=CHCOOH) is used to enhance fiber–matrix (polypropylene) adhesion. The acrylic acid molecules react with the cellulose hydroxyl groups to provide cellulose radicals for the polymerization medium. The grafting of acrylic acid on the matrix is initiated by peroxide radicals. The O–O bond in peroxide removes hydrogen atoms from the cellulose hydroxyl groups. Hence, these acids form ester linkages with the hydroxyl groups, as shown in equation (9). This coupling mechanism between fiber and matrix enhances the stress transfer capacity at the interface and consequently improves composite properties. 98 Acrylation reduces the fibers hydrophilicity.99,100 Table 9 presents pertinent works on acrylation treatment in NFRCs.
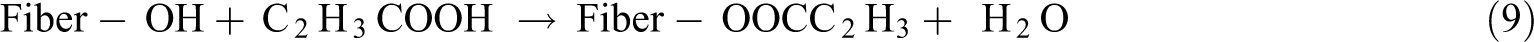
The works related to acrylation treatment of fibers in NFRCs.
NFRC: natural plant fiber-reinforced composite; PF: polypropylene; LLDPE: linear low-density polyethylene; NaOH: sodium hydroxide.
Acrylonitrile grafting
Acrylonitrile (AN) initiates free radicals, which react with cellulose molecules of the fiber by dehydrogenation and oxidation. The activated free-radical sites on the fiber surface interact with the monomer of the matrix. The generation of free radicals can be done in the reaction medium through a redox reaction and then transferred to the backbone or by direct oxidation of the backbone by transition metal ions (such as Ce4+, Cr6+, and V5+). 102 The overall reaction is shown in equation (10)

The lignin content in the fiber is very crucial in determining the extent of grafting. The presence of lignin generally retards the polymerization rate and acts as an inhibitor at higher lignin levels. 28 Properties, such as the swelling behavior in different solvents, solubility, moisture absorption, chemical and thermal resistance, can be improved by grafting. 102 Table 10 provides some works on AN treatment available in the literature.
The works related to acrylonitrile treatment of fibers in NFRCs.

NFRC: natural plant fiber-reinforced composite; PF: polypropylene; LDPE: low-density polyethylene; PALF: pineapple leaf fiber; PF: phenol formaldehyde; AN: acrylonitrile; KMnO4: potassium permanganate; H2SO4: sulfuric acid; HNO3: nitric acid.
Isocyanate treatment
Isocyanates are organic compounds that contain the isocyanate functional group R–N=C=O. These groups have high reactivity toward hydroxyl groups forming urethane linkages, as shown in equation (11). These are used to remove the hydroxyl groups on cellulose and lignin. In addition, isocyanate reacts with moisture on the fibers producing urea, which further reacts with hydroxyl groups in cellulose.

The major drawback of alkali treatment is that disrupts the hydrogen bonding in cellulosic hydroxyl groups, thereby making them more reactive than before. 48 Isocyanate treatment is seen as a promising alternative to alkali treatment as it is more effective in removing the free hydroxyl groups on the fiber surface. Therefore, isocyanate treatment improves mechanical properties, such as tensile strength, and reduces water absorption.105,106 Table 11 reports key works on isocyanate treatment.
The works related to isocyanate treatment of fibers in NFRCs.

DMF: dimethyl formamide; DIC: 1,6-diisocyanatohexane; NFRC: natural plant fiber-reinforced composite; PP: polypropylene; LDPE: low-density polyethylene; SEM: scanning electron microscope.
Maleated coupling agents
Addition of maleated coupling agents helps in increasing the interfacial bonding between the fiber and matrix. During grafting, MA reacts with the hydroxyl groups and removes them from the fiber cells. 109 This results in a long chain polymer attached to the fiber surface through covalent carbon–carbon bonds, 17 as shown in Figure 7. Before the treatment, the copolymer is heated to 170 C, followed by the esterification reaction. After this treatment, the surface energy of cellulose fibers is increased to a level much closer to the surface energy of the matrix. This ensures higher interfacial adhesion due to better wettability of the fiber. 26 The effectiveness of MAPP in enhancing the impact energy absorption, heat deflection temperature, and tensile and flexural strengths was reported by Daghigh et al. 110 for an optimal concentration of 2 wt% coupled with short Latania fiber. The effectiveness in enhancing the flexural strength and reducing the water absorption was given by Huang et al. 111 for wood–polypropylene composites. Maleated polyolefins (MaPOs) possess an excellent balance of properties to bridge the interface between polar and nonpolar species. This along with their ease of production makes MaPOs very successful. 112 Table 12 presents key works on maleated coupling agents.

The reaction of cellulose fibers with hot MAPP copolymer.
The works related to maleated coupling agent treatment of fibers in NFRCs.

NFRC: natural plant fiber-reinforced composite; PP: polypropylene; HDPE: high-density polyethylene; PALF-MA-g-PP: pineapple leaf fiber maleic anhydride grafted polypropylene; PVOH: polyvinyl alcohol; NaOH: sodium hydroxide; MAPP: maleated polypropylene; LLDPE: linear low-density polyethylene; MA: maleic anhydride; PP-g-MAH: polypropylene-grafted maleic anhydride.
Biological treatments: Fungal and enzymes treatment
Since chemical treatments pollute the environment, increase the production cost and time consuming, hence, the recent studies are focused on biological treatments.118-120 Being eco-friendly and a promising alternative for surface treatments, fungal treatment involves the production of enzymes. These enzymes act on the surface of the fibers to remove noncellulosic components, such as wax. White rot fungi produce extracellular oxidizing enzymes that react with lignin and assist in its removal, in addition to increasing hemicellulose solubility and reducing the hydrophobicity of the fiber. 121 Moreover, fungi produce hyphane that creates fine holes on the fiber surface and produces a rough surface for better interlocking with the matrix. Enzymes could directly be added to natural plant fibers without making use of fungi. Enzymes such as xylanase and laccase improve the hardness and tensile and flexural strengths by enhancing the cellulose content and removing lignin and hemicellulose.122,123 Table 13 enumerates fungal treatment mentioned in the literature.
The works related to fungal and enzymes treatment of fibers in NFRCs.

NFRC: natural plant fiber-reinforced composite; PP: polypropylene; PVOH: polyvinyl alcohol; EDTA: ethylenediaminetetraacetic acid; NaOH: sodium hydroxide; TGA: thermogravimetric analysis.
Nanoclay addition to polymer matrix: Nanocomposites
Nanoclays are inorganic nanoparticles (with size between 1 nm and 100 nm), which are used as reinforcements to enhance mechanical, thermal, or electrical properties. Nanoclay additives are widely used in industries, such as cable coatings, adhesives, inks, pharmaceutical, and automotives. 130 Among the various inorganic nanoparticles, layered clay minerals have received more attention due to their commercial availability, low cost, relatively simple processability, and significant enhancements in properties. 131 Montmorillonite (MMT) is one of the most common nanoclay forms and is classified as magnesium aluminum silicate having sheets morphology. These sheets or platelets have permanent negative charge and are held together by cations, such as Na+ and Ca2+. With a particle thickness of 1 nm and 70–100 nm crosswise silica platelets, 132 the total surface area of MMT can be as large as 750 m2 g−1 and high aspect ratio of 70–150. 133 Known for their availability and being inexpensive, minimal content (1–5 wt%) of nanoclay can improve the flexural modulus and lower the coefficient of linear thermal expansion of the polymer matrix. 134 Moreover, the heat resistant property of the nanoclay can increase the heat distortion temperature of the composites 135 by making them more dimensionally stable and flame retardant.130,136 Nanoclay, by virtue of its high aspect ratio, provides a tortuous path that makes it hard for gases and vapors to pass through, thereby improving the barrier properties. Alamri and Low 137 have summarized the advantages of polymer–clay nanocomposites as being excellent in physical (shrinkage, optical, dielectrics, and permeability), mechanical (toughness, strength, and modulus), and thermal (thermal expansion coefficient, flammability, decomposition, and thermal stability) properties.
Like natural plant fibers, the fiber–matrix interaction plays a major role in the properties of the composite. However, the properties of nanocomposites depend on the area of nanoclay exposed to the polymer matrix. Homogeneity in the nanoclay dispersion in polymer is linked with optimal properties. Sancaktar and Kuznicki 138 proposed three stages of dispersing nanoparticles in polymeric matrices. The formed nanostructure of the mixture could be conventional, intercalated, or exfoliated, in which exfoliated being the best, as shown in Figure 8. The final mixture is typically a combination of these three morphologies. Due to their high aspect ratio, a small percentage of nanoclay particles properly dispersed in the matrix can generate a very large surface area for polymer/filler interactions.

States of dispersion of nanoclay platelets. 138
The final properties of nanostructure will mainly depend on the choice of the mixing technique and the resulting degree of exfoliation of the nanoclay platelets. 139 The mixing techniques adopted are manual mixing, 133 thinky mixing, magnetic stirring, sonication, 140 three roll milling,140,141 high shear mixing, 139 high speed mixing via homogenizer, 114 direct melt compounding, and master batch dilution. 142 In the study of nanoclay–epoxy composites by Jumahat et al., 143 the presence of clusters of intercalated nanomers and nanovoids reduced the compressive strength of the epoxy system. At high nanoclay loadings, mechanical properties deteriorated due to the existence of agglomerates and air bubble voids in the epoxy matrix. Abdollahi et al. 131 investigated MMT as a filler (1, 3, and 5 wt%) in alginate matrix for food packaging applications. The addition of nanoclay was done by mechanical stirring and sonication. Among them, 5 wt% showed reduced water absorption from 99.55% (neat alginate film) to 77.49%, but the contact angle was also reduced from 40.7° to 36.18°, making it more hydrophilic. On the other hand, the tensile strength was decreased beyond 1 wt% loading. This was a result of the interaction of MMT layers by forming stacked clay themselves through the polymer matrix, high interfacial area and energy, which was a result of higher MMT content.
Ho et al. 133 added various amounts (0.5–8 wt%) of MMT in epoxy through mechanical stirring. The ultimate tensile strength was maximum at 5% with ductility dropping from 1%, and the higher weight concentrations were found to make it brittle. In Vickers hardness test, the 5% sample showed maximum value of 11.3 when compared to 9.8 for pure epoxy. As the wt% of nanoclay increased, “nanoribbon” structures of MMT interlocked with the epoxy network chains as a result of increasing the “grid lines” of the net. This would hinder the linking of epoxy chain networks making the composite brittle. Withers et al. 31 tested mechanical properties of unloaded, 2% and 4% loaded Cloisite B nanoclay, which is capable of bonding to epoxy resin compounds either noncovalently through hydrogen bonds or covalently by additional reaction. A loading of 2% showed a higher tensile strength with a 1000% higher fatigue life than the pristine composite when extrapolated to 109 cycles or a simulated 10-year life cycle. The properties with 4% loading were found to deteriorate due to intercalation, which did not support uniform stress transfer like exfoliated as in the case of 2% loading. Alamri and Low 137 investigated the effect of water absorption on the mechanical properties of nanoclay (modified MMT) recycled cellulose fiber-reinforced hybrid epoxy composites. They concluded that the presence of nanoclay platelet was found to slightly minimize the effect of moisture on the mechanical properties of the composites, as evidenced by improved flexural strength, modulus, and fracture toughness properties of the composites.
Shoumya and Kazi 144 compared the properties of polyester reinforced with graded and nongraded nanoclays. MMT-graded clay was surface modified with 15–35 wt% octadecylamine and 5 wt% aminopropyltriethoxysilane, while the nongraded clay was collected from local areas and nonprocessed. It was experimentally found that the graded nanoclay improved tensile strength, modulus, and flexural strength, whereas nongraded clay degraded the same properties. Alfred et al. 145 evaluated the properties of graded nanoclays (Nanomer I.28E and Cloisite 10A and 30B) and concluded that Cloisite 10A had the best dispersion due to highest intergallery spacing and viscosity.
Solyman et al. 146 fabricated nanoclay composites by assembling polymeric thiol surfactants on gold nanoparticles followed by impregnation into the clay matrix. The results showed that the polymeric thiol surfactants assembled on gold nanoparticles were located in the interlayer space of the clay mineral and affected the clay structure. Moreover, the assembled polymeric thiol surfactants on gold nanoparticles exfoliated and increased the activity of nanoclay. Pankil et al. 147 tested various surfactants (namely dodecyltrimethylammonium bromide, hexadecyltrimethylammonium bromide, tetradecyltrimethylammonium bromide, tetraphenylphosphonium bromide, and zinc stearate) on unmodified MMT. The surfactants were chosen on the basis of long hydrophobic carbon chains attached to the central atom that results in increasing surface area. This will lead to better intercalation of organic cation between the layers of unmodified MMT clay. Basal spacing is found to be the highest with modification of MMT with tetraphenylphosphonium ion. The nanoclays are found to be more thermally stable with modification using surfactants.
Haq et al. 148 observed that the fibers provided mainly the stiffness and strength to the composite while the nanoclay enhanced the hygrothermal and barrier properties. In their study on hemp-PP composites, the addition of Cloisite 30B nanoclay did not seem to alter the fiber–matrix adhesion. However, it increased the surface roughness and become tougher. The improvement in stiffness is attributed to the inorganic nanoclay fillers, which compensate for the low stiffness observed in organic fillers like the natural plant fibers in polymer matrix. 113 Behzad 149 used modified MMT in rice husk high-density polyethylene (HDPE) composite and observed an increase in dynamic mechanical properties with the addition of nanoclay. He has concluded that 2 per hundred compounds (phc) of nanoclay intercalated exhibited good crystalline properties. Extending his study to wood flour/glass reinforced PP hybrid composites and concluded that an increase in the percentage of glass fibers resulted in increased tensile strength and decreased in impact strength and water absorption for an optimum concentration of nanoclay as 4 phc. 150 The same author 151 studied the addition of nanoparticles in hemp-PP composites, which led to an increase in the tensile strength and modulus, decrease in the water absorption and thickness swelling, and a reduction in the impact strength.
Zhong et al. 152 observed that incorporation of organoclay in wood flour-HDPE composite reduced the thermal expansion and enhanced the mechanical properties while emphasizing that an ore compatibilizer is required in the presence of the nanoclay. A similar trend was observed by Najafi et al. 153 with optimum nanoclay loading at 4 phc. Essabir et al. 154 experimented with alfa, pine, bagasse, and coir fibers with nanoclay fillers and found that mechanical properties were optimal at 15 wt% of nanoclay loading. Although the addition of MMT in NFRCs increased the tensile strength and interfacial adhesion between the fibers and matrix, it also increased water absorption and decreased in biodegradability. 155
Arulmurugan and Venkateshwaran 156 exfoliated organically modified MMT in jute-PE composite using ultrasonication technique and concluded that best mechanical properties were observed at 5% clay loading. Ramakrishnan et al. 157 added nanoclay to jute-PE composite using magnetic stirring method and observed the enhancement in tensile, flexural, impact strengths and modulus of elasticity compared to pure, fiber-reinforced polyester, and nanoclay-filled resin. Shahroze et al. 158 added organomodified nanoclay to sugar palm fiber-reinforced unsaturated polyester resin composite. For an optimal value of 2%, the best performance for tensile properties was observed, while for 4%, the best performance of impact and flexural strengths was observed. Patel et al. 159 tested the effect of nanoclay on bamboo fiber-reinforced polyester composites for loadings of 0–4 wt%. The optimal loading for the best tensile strength (66.9 MPa) and flexural strength (127 MPa) was obtained at 1%. Hasan et al. 160 added MMT to jute–polyester resin composite in loadings of 1, 3, and 5% and tested by dynamic mechanical analysis and thermogravimetric analysis. The 5% and 1% sample showed better thermal stability and moisture resistance, respectively.
Applications of NFRCs
The previously discussed chemically treated NFRCs have applications in a wide array of industrial sectors, as shown in Figure 9. Strict rules on the recyclability of automobiles have raised the importance of NFRCs in the automotive industry in certain countries. In Europe, the EU “end of life vehicle” dictates that 95% (by weight) of the components must be recyclable. NFRCs the go-to material owing to their recyclability and light weight structural components used in automotive, which led to lower fuel consumption. A 25% reduction in vehicle weight is equivalent to a saving of 250 million barrels of crude oil and a reduction in carbon dioxide (CO2) emissions of 220 billion pounds per year. 161

Applications of natural plant fiber-reinforced composites.
NFRCs are used for various parts of an automobile, as shown in Figure 10. 162 This includes the Al-Qureshi designed MANACA—the first all-banana reinforced composite car panel 163 and the efforts of Johnson Controls Interiors GmbH and Co. (Westendhof 8 Essen, Germany) in producing plant fiber-reinforced thermoplastics for the interior of cars. A sustainable vehicle “Eco Car” was developed by automotive engineers to run on biofuels, having body panels made of NFRCs in a biodegradable resin matrix. 164 Table 14 presents the usage of NFRCs in automotive sector.165-168

Automobile components made of natural fiber-reinforced composites. 162
NFRCs have also led to two well-established products: Tufnol 6F/45 (cotton-epoxy composite), as shown in Figure 11(a), and Tespa (wood fiber-phenol) composites.
24
Several establishments use chemically treated NFRCs, including Tipco Industries (Valsad, Gujarat, India) TIPWOOD®50EX in furnishing, NEC Corp. (Minato, Japan) and UNITIKA’s “TERRAMAC” (Osaka, Japan) in cell phones due to improved moisture resistance and the fall impact durability
169
; EastarBio (Eastman Chemical Company, Kingsport, Tennessee, USA), BioPET or Biomax (DuPont, Deleware, USA), polylactic acid (Dow-Cargill, Nebraska, USA), Ecoflex (BASF), and poly (3-hydroxybutyrate-
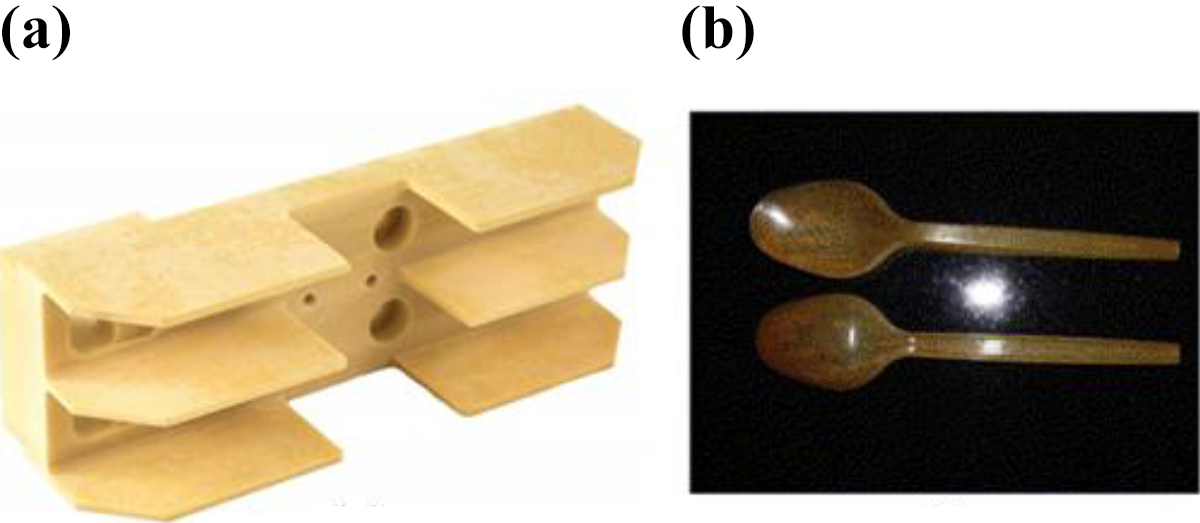
Jandas et al. 52 produced eco-friendly biodegradable cutlery using surface-treated banana fiber reinforced bio-nanocomposites, as shown in Figure 11(b). Similarly, Asim et al. 170 reported that PALF fibers can be employed in diversified applications, such as decoration, sports, furniture, aerospace, and fabric industries, as shown in Figure 12. Also, window frame, fencing, and railings of NFRCs are manufactured by commercial companies for household applications, as shown in Figure 13. 171 Sapuan et al.172,173 designed and fabricated a multipurpose table and telephone stand using banana fiber-reinforced composites, as shown in Figure 14(a) and (b). These biodegradable materials degrade in the natural condition leaving behind biomass and CO2. 161 Filler composites made from municipal solid waste (MSW) are very promising while considering the annual MSW generation in populous countries, like India, could go up to 68.8 million tons.174-176 Additionally, research on hybrid composites like MSW-banana and Bauhinia racemosa-glass fibers suggest good thermal and electrical insulation properties.177,178

Application of PALF fibers in various industries. 170 (a) Decoration, (b) sports industries, (c) furniture industries, and (d) aerospace industries.

Applications of NFRCs as construction materials. 171 (a) Railing, (b) window frame, and (c) fencing.

In the future, applications of NFRCs are expected to be seen majorly in the field of eco-friendly materials. The search for the “green materials” which are environmentally compatible is a growing social concern, increasing the rate of depletion of petroleum resources and new environmental regulations. With cost-effective and easy processing technologies, the eco-composites have the potential to compete with commodity plastics with a promising double-digit growth in the future market. 161 Table 15 clearly reveals the modern day usage of NFRCs in various sectors.
Challenges and future work
The techniques discussed are not perfect panaceas for attaining the best performance out of NFRCs. Each of these techniques is faced by unique challenges. For chemical treatments, these include delignification of fibers (alkaline treatment), degradation by UV radiation (acetylation treatment), and adverse effects on the pristine internal configurations (permanganate and isocyanate treatment). The challenges with nanoclay composites which manifest as a reduction in the properties of the polymer include poor dispersion and agglomeration of clay particles causing premature failure of the system, weak filler–matrix interface bonding, reducing the stress transfer capability and increasing resin viscosity with the proportion of nanoparticles, thus limiting processability.
Further research should be carried out in the following areas but are not limited to these: Employing multiple chemical treatments in succession to complement their individual effects, thereby bringing out the best properties of the NFRCs. Employing chemical treatments prior to dispersing nanoclay to improve fiber–matrix interactions. Testing new chemicals for their effectiveness in removing the hydroxyl group. Testing other biological species to achieve similar or better performance than fungal treatment. Employing alternative nanofillers to nanoclay that are hydrophobic in nature while providing appreciable mechanical properties. Combining chemical treatments with heat treatments to achieve superior properties. Employing bioresins to combine with chemically treated fibers to form superior and greener composites.
Advancement in the field of materials science will help to remove the mental block that prevents NFRCs from prevailing in the global market.
Conclusion
NFRCs, by virtue of their appreciable mechanical properties and environmental benefits, are promising materials for the future. Reducing the hydrophilicity of the plant fibers and improving their adhesion with the matrix, thereby achieving enhanced thermomechanical properties and commercializing the same is one of the aims of current research. This review propounds the removal of the hydroxyl groups as an effective way to achieve enhancement in these properties. This article delineated the common chemical treatments carried out to achieve superior properties along with their applications, effects, and working mechanisms. Another method to attain enhancement of properties is through the addition of nanoclay as reinforcement in the matrix. The addition may be selected to achieve conventional, intercalated, or exfoliated nanocomposites depending on the required properties and applications.
The presented challenges can be overcome to engender superior NFRCs, thus opening new vistas for greener products. Furthermore, the research at the intersection of nanocomposites and chemical treatment of plant fibers is in its early stages and is full of potential. We believe that this is the direction to head toward building a world with better materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.