Abstract
A series of novel ternary conductive composites have been prepared by incorporating functionalized multiwalled carbon nanotubes (FMWCNTs) into the binary composites of poly(amide-imide) (PAI) and poly(o-methoxyaniline) (POMA) to further improve the conductivity of PAI composites. With increasing the weight ratio of FMWCNTs, the conductivity of PAI/POMA/FMWCNTs ternary composites has been significantly enhanced from 10−3 S m−1 to 90 S m−1, much higher than that of the sum of PAI/POMA and PAI/FMWCNTs binary composites. The enhanced conductivity is mainly ascribed from a much more intensive conductive network formed in the PAI/POMA/FMWCNTs ternary composites. It is found that the N–H bending of amide group in the Fourier transform infrared spectrometry spectra is observed at 3298 cm−1 for PAI, 3296 cm−1 for PAI/POMA binary composites, and further blue shifted to 3292 cm−1 for PAI/POMA/FMWCNTs ternary composites. Moreover, the glass transition temperature has been increased from 82.5°C to 117.7°C. These results suggested that the hydrogen bonding interaction has been strengthened among PAI, POMA, and FMWCNTs, which is useful for dispersion of condcutive fillers into the polymer matrix.
Introduction
Conductive polymer composite (CPC) has gained more and more attention due to the rapid growth in the modern electronic industry. 1 -5 Compared with the traditional metallic counterparts, CPC exhibits much unique advantages, such as lightweight, excellent flexibility, and ease of processing. It is well known that most conventional polymers are poorly conductive and have been considered as an insulator, because valence electrons could not flow or jump in their backbones. For example, poly(amide-imide)s (PAIs) have been widely researched due to their excellent properties, including high thermal stability, suitable mechanical performances, and good processability. 6 -8 But like other conventional polymers, the application of PAIs in the conductive area is limited resulted from their low conductivity. It has been demonstrated that the transition from insulator to conductor can be fulfilled by adding conductive filler into the polymer matrixes. 9 -13 In general, conductive fillers used in the manufacture of CPC are mainly classified into two categories: (1) carbonaceous fillers (carbon black, 14 -17 graphene, 18 -20 and carbon nanotubes (CNTs)) 21 -28 and (2) intrinsically conductive polymer (ICP). 29 -32 Carbonaceous fillers have been considered as ideal conductive fillers because of their excellent mechanical and electrical properties. However, this kind of conductive filler is prone to aggregate in the polymer matrix, and the high cost also limits their use in commercial products. Compared with carbonaceous fillers, the cost of ICP is much lower, furthermore, the compatibility between the polymer matrix and the ICP was much better, suggesting that more conductive filler could be added into the polymer matrix without obvious aggregation. 33 But it is sadly the conductivity of ICP is lower compared with carbonaceous fillers.
Combining the advantages of carbonaceous fillers (high conductivity) and ICP (low cost and good compatibility), ternary conductive polymer composite (TCPC) has been paid more and more attention. 34 -40 For example, we have incorporated functionalized multiwalled carbon nanotubes (FMWCNTs) into the binary composites of PAI and polyaniline (PANI) to improve their conductive performance. 41 The conductivity of PAI/PANI/FMWCNTs ternary composites significantly increased from 10−3 S m−1 to 8.3 S m−1, much higher than that of the sum of PAI/PANI and PAI/FMWCNTs binary composites. This result suggested that utilizing the synergistic effect of ICP, carbonaceous filler was an effective method to improve inductivity of the polymer composite. Although the conductivity of PAI/PANI/FMWCNTs ternary composites has been improved, the conductivity of PAI composites could be further enhanced with a higher weight ratio of conductive fillers. Poly(o-methoxyaniline) (POMA) is one kind of PANI derivatives and possesses a polar group (methoxy unit) attached to its main chain, which can form much stronger hydrogen bonding interaction among the carbonaceous fillers, ICP, and polymer matrix to promote the uniform dispersion of conductive filler into the polymer matrix. 42 -44 Moreover, it has been reported that POMA and CNTs played a synergistic effect in conductivity, with incorporation 10 wt% of CNT into POMA, the conductivity of the hybrid conductive fillers POMA/CNT was improved by one order of magnitude. 45 Therefore, with good compatibility and synergistic effect of POMA and CNT, POMA and CNT would be an efficient combination to improve the conductivity of polymer composites.
Herein, FMWCNTs were incorporated into PAI and POMA binary composites to construct PAI/POMA/FMWCNTs ternary composites for further improving the conductivity of PAI blends. As a result, the conductivity of PAI/POMA/FMWCNT ternary composites increased from 10−3 S m−1 to 90 S m−1. Moreover, the thermal stability, phase transition, and morphology of PAI/POMA/FMWCNT ternary composites were also researched by thermogravimetric analysis (TGA), differential scanning calorimetry (DSC), and field emission scanning electron microscopy (FESEM), respectively.
Experimental section
Materials
MWCNTs (outer diameter 8–15 nm, length 30–50 μm, and purity >90 wt%) were purchased from Chengdu Organic Chemicals Co. Ltd (Chengdu, China). Toluene-4-sulfonic acid (TSA) was purchased from Shanghai Lingfeng Chemicals Reagent Co. Ltd (Shanghai, China). Ammonium persulfate (APS), 1,4-diaminobenzene, pyridine (Py), anhydrous calcium chloride (CaCl2), N-methyl-2-pyrrolidone (NMP), 1,4-dihydroxybenzene, 4,4′-oxy-diphthalic anhydride (ODPA), 11-aminoundecanoic acid (AU), d-trifluoroacetic acid (TFA), lithium chloride (LiCl), and triphenyl phosphite (TPP) were supplied from Aladdin Industrial Corporation (Shanghai, China). Ammonium hydroxide (NH3·H2O), m-cresol, hydrogen peroxide aqueous solution (H2O2), nitric acid (HNO3), acetic acid (CH3COOH), and o-methoxyaniline (OMA) were obtained from Sinopharm Chemical Reagent Co. Ltd. (Shanghai, China) Other chemicals were supplied from Aladdin Industrial Corporation.
Preparation of FMWCNTs
Figure 1 shows the route of FMWCNTs. HNO3 (65%, 100 mL) and MWCNTs (4 g) were added to a 250-mL three-necked flask under mechanical stirring. H2O2 (50 mL) was added at a rate of 1–2 drops per second. The solution was stirred vigorously at 70°C for 2 h and then filtered under reduced pressure. The crude product was washed with deionized water to neutral. The final product FMWCNTs was dried under 80°C.

Synthetic route of FMWCNTs.
Preparation of conductive POMA
POMA was prepared by in-situ solution polymerization, as shown in Figure 2. OMA (0.1 mol, 12.3 g) was dissolved in 300 mL of 0.1 mol/L HCl, and the solution was stirred in an ice-water bath for 30 min. According to the molar ratio of APS to OMA of 1:4, 5.6 g of APS was dissolved in 100 mL of 1 mol/L HCI, and the solution was added dropwise by a constant pressure funnel. The reaction was carried out for 6 h in an ice-water bath. The mixture was filtered under reduced pressure and then washed with NH3·H2O, and deionized water in sequence until the filtrate became neutral. The obtained product was dried at 60°C for 12 h to obtain POMA.
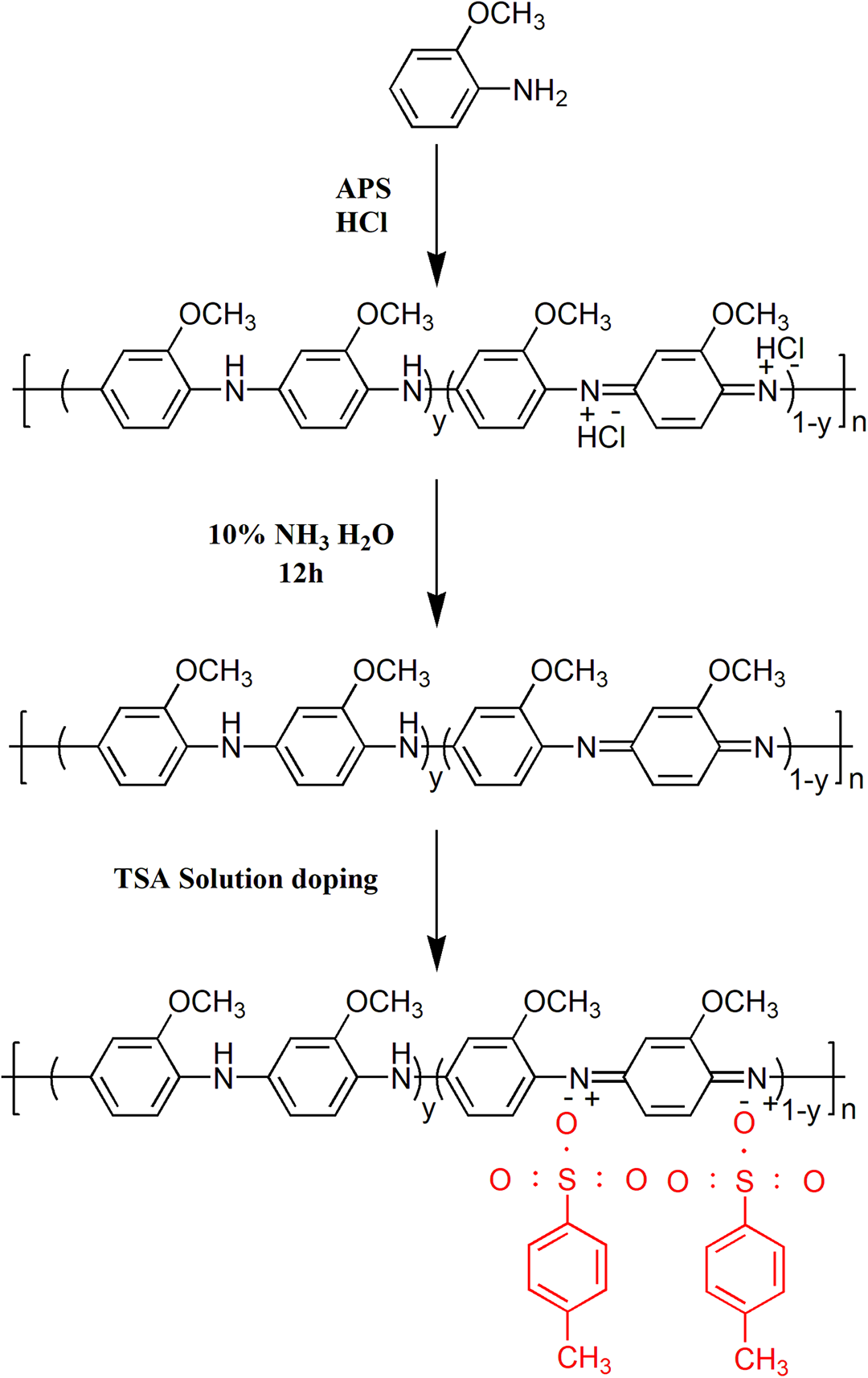
Synthetic route of conductive POMA.
Conductive POMA solution was prepared according to the literature. 41 First, 400 mg of POMA was dispersed in 12 mL of m-cresol, and then, p- TSA with a weight ratio of 120% (relative to POMA) was added. The conductive POMA dispersion was vigorously stirred for 12 h. Finally, the suspension was cast onto a Petri dish and placed in a vacuum oven at 90°C to evaporate the solvent.
Synthesis of PAI
PAI was prepared by Yamazaki–Higashi phosphorylation reaction, as reported previously, 46 -50 and the synthetic route is shown in Figure 3.

Synthesis of DIDA and PAI.
Synthesis of diimide diacid monomer
ODPA (6.20 g, 0.02 mmol), AU (8.04 g, 0.04 mmol), and CH3COOH (40 mL) were added to a 250-mL three-necked round-bottom flask. The mixture was stirred at room temperature overnight, and then gradually heated to 120°C under nitrogen for 8 h. The mixture was cooled to room temperature and then filtered. The precipitate was washed several times with deionized water and dried under vacuum at 80°C for 12 h (yield: 71%). 1H-NMR (δ/ppm, d-TFA): 11.50 (s, 2 H), 7.91 (d, 2 H), 7.52 (d, 2 H), 7.42 (dd, 2 H), 3.72 (t, 4 H), 2.43 (t, 4 H), 1.67 (m, 8 H), and 1.29 (m, 24 H). Fourier transform infrared (FTIR) (potassium bromide (KBr), ν, cm−1): 3467, 1771, 1698, 1396, 1367, 1081, 746, and 1263.
Synthesis of PAI
CaCl2 (2 g, 0.018 mol), LiCl (1 g, 0.024 mol), 1, 4-diaminobenzene (0.599 g, 0.005 mol), and diimide diacid (DIDA) monomer (3.676 g, 0.005 mol) were added into a 100-mL three-necked flask and purged under nitrogen. TPP (6 mL), Py (5 mL), and NMP (15 mL) were added to the flask. The solution was gradually heated to 120°C for 8–12 h. The solution was slowly poured into a beaker containing magnetically stirred anhydrous ethanol and stirred at room temperature for 2 h. After removing the solvent, the crude product was further placed in warm water of 60°C for 1 h to remove CaCl2 and LiCl. The final product was dried in a vacuum oven at 80°C for 8 h (yield: 89%). 1H-NMR (δ/ppm, d-TFA): 7.88 (d, 2 H), 7.47 (s, 2 H), 7.41(s, 2 H), 7.38 (d, 4 H), 3.68 (t, 4 H), 2.67 (t, 4 H), 1.72 (m, 8 H), and 1.23 (m, 24 H). FTIR (KBr, ν, cm−1): 3298, 1654, 1515, and 1549.
Fabrication of PAI/POMA/FMWCNTs composites
PAI/POMA/FMWCNTs suspension with a blend ratio of PAI:POMA:FMWCNTs = 4:1:x mg mL−1 (0 ≤ x ≤ 1) was prepared using m-cresol as a solvent. Subsequently, the suspension was sonicated for 30 min at 30°C. The TCPC composites were prepared by drop casting the dispersion onto glass substrates and then, the solvent was allowed to evaporate in the fume hood at 90°C for overnight.
Characterization assessment
The FTIR spectrometry was obtained with an Avatar 370 spectrometer (Thermo Nicolet, Ramsey, Illinois, USA) and data were collected at a region of 4000–500 cm−1 with a spectral resolution of 0.1 cm−1. The thermal stability of the samples was determined by Perkin-Elmer TGA 4000 (Netzsch, Tarsus, Germany). The film sample (10 mg) was placed in ceramic crucible and heated from 30°C to 700°C at a heating rate of 10°C min−1 under nitrogen. The phase change behavior of the composite was measured by DSC (Perkin-Elmer DSC 8000) using a flow rate of 50 mL min−1. The film sample (5 mg) was placed in aluminum crucibles, and calibrate the instrument’s heat, temperature, and temperature using indium and zinc standards. The conductivity of PAI/POMA/FMWCNTs was studied on RTS-9 by standard four-point probe method at room temperature. Raman characterization was performed on a Thermo Fisher DXR Raman microscope with a 532-nm laser source. The microscopic morphology of the composite was evaluated using a SUPRA 12–1,000,000 in FESEM.
Results and discussion
Structure characterization
Figure 4 shows the Raman spectra of the pristine MWCNTs and FMWCNTs. As shown in the figure, the D-band at 1344 cm−1 and the G-band at 1588 cm−1 are observed in both samples, which correspond to various amorphous carbon impurities (or defects) and the ordered vibration of sp2-hybridized carbon atoms in a 2D hexagonal graphitic lattice, respectively. 51 It is well known that valence electrons could flow or jump in the 2D hexagonal graphitic lattice, in other words, the conductive structure of MWCNTs was maintained after the functionalized reaction. Furthermore, the relative intensity ratios (IG/ID) between the G-band and the D-band were estimated to be 0.84 for MWCNTs and 0.78 for FMWCNTs, respectively, the slightly lower IG/ID ratio of FMWCNTs indicates that the acidification treatment provides much more polar groups on the surface of the FMWCNTs.

Raman spectra of the pristine MWCNTs (black line) and FMWCNTs (red line).
Figure 5 shows FTIR spectra of PAI neat, PAI/POMA binary blend, PAI/POMA/FMWCNTs ternary blend composites, and their important characteristic peaks are marked. As shown in the figure, the bands at around 1083 cm− 1 are observed in the spectra of PAI, which is attributed to C–O stretching vibration. In the FTIR spectrum of PAI/POMA and PAI/POMA/FMWCNTs composites, the double peaks at 2925 and 2852 cm− 1 are the C–H stretching vibration of the aliphatic group, and the vibration peaks at 1769, 1710, and 1656 cm− 1 are attributed to the C=O of amide group. The vibration peaks at 1233 cm− 1 are attributed to C–O bond of the methoxy group of POMA, and the vibration peaks at 1035 cm− 1 are attributed to the S=O bond of dopant TSA for POMA. Moreover, the N–H bending of amide group is observed at 3298 cm− 1 for PAI and 3296 cm− 1 for PAI/POMA binary composites, and further blue shifted to 3292 cm− 1 for PAI/POMA/FMWCNT composites, which indicates that the hydrogen bonding interaction was strengthened with incorporation of FMWCNTs into PAI/POMA binary blend. 52

FTIR spectra of PAI (black line), PAI/POMA binary blend (red line), and PAI/POMA/FMWCNTs ternary blend films (blue line) with a weight ratio of 20 wt%.
Conductivity
Figure 6 shows the conductivity of PAI/FMWCNTs binary and PAI/POMA/FMWCNTs ternary composites with different weight ratio of FMWCNTs. The conductivity of these polymer composites was measured by four-point probe method at room temperature. For PAI/FMWCNTs binary conductive composites, the conductivity was improved compared with PAI pure film, and a conductivity of 3.7 S m−1 was achieved with adding 20 wt% FMWCNTs into the polymer matrix. With further increasing the weight ratio of FMWCNTs, the conductivity of PAI/FMWCNT composite nearly kept invariable, and the film became brittle. For PAI/POMA/FMWCNTs ternary composites, PAI/POMA binary composites exhibit a conductivity of 10−3 S m−1, with adding FMWCNTs into PAI/POMA binary blend, the conductivity has been enhanced hugely. In more detail, with incorporating 5 wt% FMWCNTs, the conductivity of PAI/POMA/FMWCNTs ternary composites increased by more than one order of magnitude. With further increasing the weight ratio of FMWCNTs, the conductivity increased correspondingly. When the weight ratio of FMWCNTs reaches 20 wt%, the conductivity increased to 90 S m−1, much higher than that of the sum of PAI/POMA and PAI/FMWCNTs binary composites. These results suggest that the POMA and FMWCNTs played an obvious synergistic effect in conductivity. 53 The impressive synergistic effect is probably because the much more intensive conductive network is formed in the PAI/POMA/FMWCNTs ternary composites.

The conductivity of PAI/FMWCNTs binary (black line) and PAI/POMA/FMWCNTs ternary composites with different weight ratio of FMWCNTs (red line).
Thermal stability
Figure 7 shows TGA curves of PAI/POMA binary and PAI/POMA/FMWCNTs ternary composites, and their corresponding data are collected in Table 1. As shown in the figure, both PAI/POMA binary composite and PAI/POMA/FMWCNTs ternary composites exhibit three distinct decomposition stages. The first stage observed in the temperature range of 126–156°C is attributed to the removal of dopant molecules (TSA), and the weight loss observed in the second stage between 466°C and 475°C is mainly attributed to the decomposition of side groups from the POMA. The third stage above 475°C is mainly attributed to the weight loss caused by the decomposition of backbones of PAI and POMA. It is found that with the addition of FMWCNTs, the initial decomposition temperature of PAI/POMA/FMWCNTs ternary composites increased, suggesting that the addition of FMWCNTs into PAI/POMA binary composite could improve the thermal stability of PAI composites.
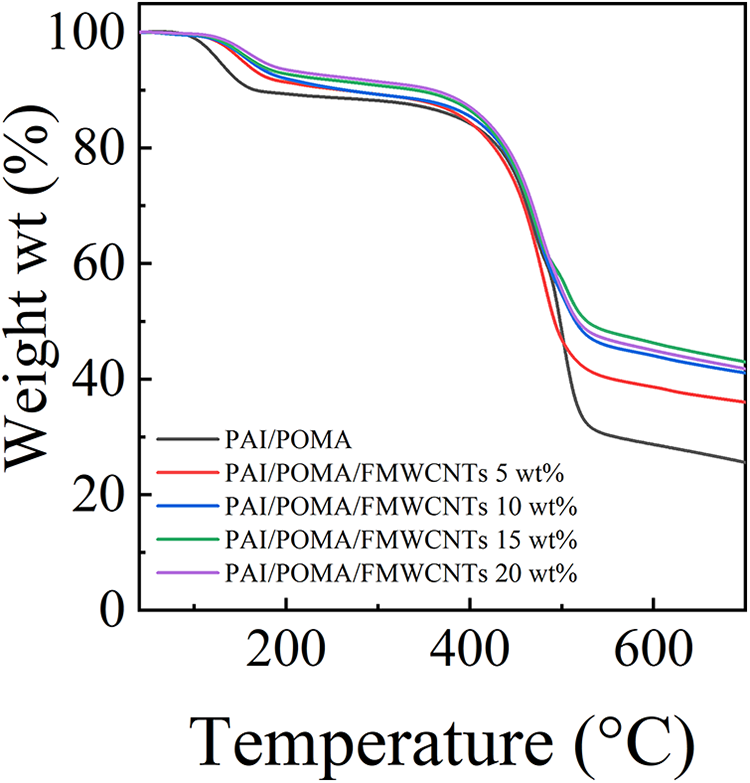
Thermogravimetric analysis curves of PAI/POMA binary and PAI/POMA/FMWCNTs ternary composites with different weight ratios of FMWCNTs.
Thermal properties of PAI/POMA/FMWCNTs ternary blends with different compositions.
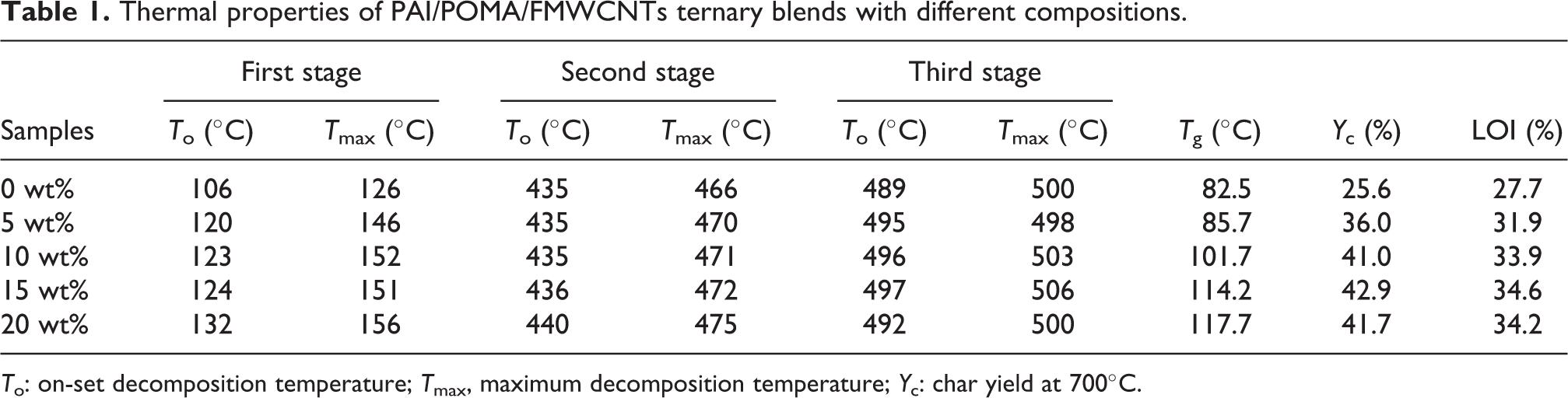
To: on-set decomposition temperature; Tmax, maximum decomposition temperature; Yc: char yield at 700°C.
On the other hand, the char yield (YC) is a decisive factor to calculate the limited oxygen indexes (LOIs), according to the Van Krevelen–Hoftyzer equation 54

where CR is the coke yield. As summarized in Table 1, the LOIs of all the ternary blend composites are much higher than 21%, which indicates that all the PAI/POMA/ FMWCNTs ternary composites can be considered as self-extinguishing materials.
Phase transition
The phase transition behavior of PAI/POMA/FMWCNTs ternary blend composites with different compositions was investigated by DSC at a heating rate of 10°C min−1 under nitrogen atmosphere. As shown in Figure 8, in general, the glass transition temperature (Tg) of PAI/POMA/FMWCNTs blend films increased with increasing the weight ratio of FMWCNTs, which is consistent with that we reported before. 41 When the weight ratio of FMWCNTs reached to 20%, the Tg increased from 82.5°C to 117.7°C. The change of Tg suggested the hydrogen bonding interaction between PAI/POMA and FMWCNTs was strengthened, 55 which could improve the dispersion of conductive fillers into the polymer matrix, as discussed below.

DSC analysis curve of PAI/POMA binary and PAI/POMA/FMWCNTs ternary composites.
Interaction in the PAI/POMA/FMWCNT ternary composites
Figure 9 shows the possible interactions in the PAI/POMA/FMWCNTs ternary composites. It is well known that the higher weight ratio of conductive filler in polymer composites without obvious aggregation, the higher conductivity could be attained. Carbonaceous fillers are prone to aggregate in the polymer matrix because of (a) strong intertube van der Waals interaction and (b) lack of interfacial interaction between polymer and these carbonaceous fillers. Although there is π-π interaction and Coulomb force in the ternary composites, the forces are too weak to break the interaction between carbonaceous fillers. Therefore, much stronger interaction should be adopted to promote the dispersion of conductive filler in the polymer matrix. As discussed above, the Tg of PAI/POMA/FMWCNTs ternary composites increased with adding FMWCNTs into the binary blend composite of PAI/POMA, this is because there is hydrogen bonding interaction among the PAI, FMWCNTs, and POMA. 41 The hydrogen bonding interaction among PAI, POMA, and FMWCNTs could be confirmed again by the chemical shift of FTIR spectra in the PAI/POMA binary and PAI/POMA/FMWCNTs ternary blend composites. It is believed that the hydrogen bonding interaction in the PAI/POMA/FMWCNTs ternary blend composites is helpful in breaking the intermolecular force in the FMWCNTs, promoting the dispersion of conductive fillers.

The possible interactions in the PAI/POMA/FMWCNTs ternary composites.
Morphology
To investigate the dispersion of conductive filler in the polymer matrix, the microscopic morphology of PAI/ POMA binary and PAI/POMA/FMWCNTs ternary composites was observed by SEM, as shown in Figure 10. For the PAI/ POMA binary composite, POMA exhibits the morphology of node fibril, and some conductive networks (red circle) are isolated from each other, as shown in Figure 10(a). For the PAI/POMA/FMWCNTs ternary composites, with incorporation of FMWCNTs into the PAI/POMA binary composites, the discontinuous conductive network (red circle) is connected by FMWCNTs, as shown in Figure 10(c). Furthermore, with increasing the weight ratio of FMWCNTs, the conductive network became more intensive, and there is no large-scale aggregation even the weight ratio of FMWCNTs is as high as 20 wt%. It should be noted that FMWCNTs became aggregation in the PAI/FMWCNTs binary blend composites when the weight ratio of FMWCNTs reached 15 wt%. 46 For the PAI/POMA/FMWCNTs ternary blend composites, conductive fillers still disperse uniformly in the PAI matrix even the weight ratio of conductive fillers (POMA and FMWCNTs) is as high as 40 wt%. These results suggested that POMA played as compatibilizer between PAI and FMWCNTs, which could improve the dispersion of FMWCNTs into the PAI matrix. On the other hand, with the addition of FMWCNTs into the PAI/POMA binary blend, the hydrogen bonding interaction is strengthened, which is helpful in breaking the interaction force among FMWCNTs, as discussed above.

(a) SEM image of with PAI/POMA binary composites and PAI/POMA/FMWCNTs ternary composites with different weight ratios of FMWCNTs (b) 5 wt%, (c) 10 wt%, (d) 15 wt%, and (e) 20 wt%.
Conclusion
In summary, we have successfully demonstrated that the dispersion of conductive carbonaceous fillers in the polymer matrix could be improved using soluble PANI derivatives. Combining the advantages of CNTs (high conductivity) and POMA (good compatibility), a series of novel ternary conductive composites have been prepared through incorporating FMWCNTs into the PAI/POMA binary composites. With the enhanced hydrogen bonding interaction among PAI, POMA, and FMWCNTs, the conductive fillers (POMA and FMWCNTs) dispersed uniformly into the PAI matrix even the weight ratio of conductive fillers is as high as 40 wt%, and thus, the conductivity was increased from 10−3 S m−1 to 90 S m−1.
Footnotes
Acknowledgements
We appreciate the technical assistance from the Analysis and Test Center of 301 Changzhou University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Jiangsu Province (BK20160280), Double Plan of Jiangsu Province (2016), and Jiangsu Students’ platform for innovation, and 300 entrepreneurship training program.
